Back to Journals » Clinical Ophthalmology » Volume 14
Ocular Neuropathic Pain: An Overview Focusing on Ocular Surface Pains
Authors Ebrahimiadib N , Yousefshahi F , Abdi P , Ghahari M, Modjtahedi BS
Received 10 May 2020
Accepted for publication 24 August 2020
Published 25 September 2020 Volume 2020:14 Pages 2843—2854
DOI https://doi.org/10.2147/OPTH.S262060
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Nazanin Ebrahimiadib,1 Fardin Yousefshahi,2 Parisa Abdi,1 Mohammadreza Ghahari,1 Bobeck S Modjtahedi3– 5
1Farabi Eye Hospital, Tehran University of Medical Sciences, Tehran, Iran; 2Department of Anesthesiology, Pain and Critical Care, Tehran University of Medical Sciences, Tehran, Iran; 3Department of Ophthalmology, Southern California Permanent Medical Group, Baldwin Park, CA, USA; 4Eye Monitoring Center, Kaiser Permanent Southern California, Baldwin Park, CA, USA; 5Department of Research and Evaluation, Southern California Permanente Medical Group, Pasadena, CA
Correspondence: Parisa Abdi
Farabi Eye Hospital, Tehran University of Medical Sciences, Tehran, Iran
Email [email protected]
Objective: This paper reviews ocular pain with the main focus on ocular surface discomfort and dry eye pain. Anatomy, physiology, epidemiology, assessment, and treatment are discussed in this paper.
Methods: A PubMed search was conducted for studies published from 2000 to 2019 on the anatomy, pathophysiology, epidemiology, assessment, and treatment of ocular pain. Reviews, meta-analyses, and randomized clinical trials were included. Inclusion criteria focused on ocular surface discomfort, dry eye pain and neuropathic pain.
Results: A total of 112 articles were found through searches, 45 of which were selected and studied in this review.
Discussion: Pain in general can be acute or chronic. Acute pain is usually a physiologic response to a serious damage to the tissues and alleviates with pain relief treatments. Chronic pain is defined as the persistence of pain for more than three months. From another point of view, pain has been classified into either nociceptive or neuropathic. Nociceptive pain is a physiologic response to a noxious stimulus. Both central and peripheral nervous systems can be involved in the development of a neuropathic pain, which is characterized by positive or negative sensory signs, a pain perceived disproportionate to a noxious stimulus, and/or not responsive to analgesics. Chronic pain usually has a neuropathic component. Ocular surface pain is a well-known complaint after any corneal surgery. This is mainly due to abnormal regeneration of damaged corneal nerve endings and abnormal connections with adjacent nerve endings which produce spontaneous activity. Tear hyperosmolarity and the resultant ocular surface inflammation can also trigger voluntary activity of corneal nerve endings. Referral pain to the first and second division of the trigeminal nerve has been reported. Interference with vision and even sleep, which is out of proportion to the examination are among patients’ complaints. All of these elements proposed the new concept of ocular neuropathic pain syndrome. The first step in conventional evaluation of ocular discomfort is search for tear insufficiency. Pathologies of lid and blinking as well as conjunctival irregularities should be addressed. Anti-inflammatory agents and, in resistant cases, systemic neuromodulators are shown to be helpful. Education on behavioral changes and reassurance are essential steps. Considering the neuropathic origin for the ocular pain, treatment modalities used for such pain in other parts of the body can be considered for this syndrome.
Keywords: ocular pain, dry eye, neuropathic pain
Key Messages
- Acute pain is usually a physiologic response to a damage to the tissues and alleviates with pain relief treatments. Persistent pain for more than three months is known as chronic pain. Chronic pain usually has a neuropathic component, and needs a multimodal approach including antineuropathic pain treatment.
- Inflammation plays an important role as well as peripheral and central nervous system mechanisms.
- A multidisciplinary approach with the collaborating of ophthalmologists and pain specialists can be helpful.
Introduction
There are few things more ubiquitous in the patient experience than a fear of pain. From the time point of view, pain can be acute or chronic. Duration of pain for more than three months is considered chronic. In another categorization, pain can either be nociceptive, which is a physiologic response to a noxious stimulus, or neuropathic which is the result of malfunction of the nervous system.1 Nociceptive neurons in the dorsal root ganglion are responsible for physiologic perception of acute pain which is proportional to the severity of the insult, and can subside with resolution of stimulation. Conversely, neuropathic pain is out of proportion or even can occur in the absence of active noxious stimuli and can result in debilitating pain which is not serving a physiologic purpose. In these circumstances, patients may experience pain to normally benign stimuli (allodynia) or an exaggerated response to painful stimuli (hyperalgesia).2
Acute painful stimuli can evolve to create chronic pain disorders. For example, aberrant activation of sensory nerves during regeneration following surgery, can result in chronic pain.3
Neuroplasticity can occur through central or peripheral nervous systems which results in desensitization and adaptation to a painful stimulus with the central or peripheral nervous system. In chronic pain, abnormal plasticity of central nervous system (CNS) can occur in synaptic connections and lead to hyperalgesia or allodynia and altered descending pain modulation, which are clinical presentations of neuropathic pain.2
In neuropathic pain, the same stimuli can cause varying degrees of discomfort and functional impairment depending on individual patient factors. Neuropathic pain has been proposed to have a genetic component via influences on neurotransmission, immune response, and metabolism. Variations in catechol-O-methyltransferase (COMT) can result in variable susceptibility to pain.4 Animal studies have demonstrated that gene regulation influenced by sex results in different types of pain following nerve injury.5 In ocular surface discomfort or ocular neuropathic pain syndrome, symptoms have also been found to vary based on gender with women expressing more severe symptoms.6 Corneal sensitivity decreases with age as well as during pregnancy and has been found to fluctuate during menstrual cycles.7
The perception of ocular surface discomfort or ocular neuropathic pain can be highly influenced by psychological factors such as stress, depression, and anxiety as well as sociocultural factors.8 In addition, sleep deprivation and the presence of chronic pain in other parts of the body can play a role in the development of neuropathic pain.9
The neural pathways responsible for ocular discomfort are varied and complex with contributions from both central and peripheral nervous systems. Ocular surface discomfort can often be difficult to categorize because it can lack some of the classic features of acute inflammation. Patient complaints of ocular surface discomfort can often seem out of proportion to objective exam findings leading some clinicians to dismiss or trivialize these complaints. Corneal pain may in part be neurotrophic in nature, such as in patients with a history of herpetic eye disease, or have a nociceptive origin, such as corneal surgeries. Tear hyperosmolarity is a trigger of ocular surface inflammation which in turn stimulates corneal nerve endings which are at the level of corneal epithelium and results in the release of inflammatory neuropeptides from corneal nerves. Inflammation lowers the threshold of neuron activation and as a result, duration and intensity of responses are increased with time. This is peripheral sensitization. Continued local inflammation can result in spontaneous activation of corneal nerves which can subsequently provoke a neuropathic pain which can be perceived as hyperalgesia and burning sensation.10,11 Interestingly, in this setting, even local anesthesia cannot relieve the pain as there is not only sensitization of related peripheral nervous system, but also dysregulation of CNS.12
It has been shown that a component of corneal somatosensory dysfunction contributes to symptoms of dry eye in a subgroup of patients with dry eye.2 Confocal microscopy has shown the morphologic changes in subbasal nerve plexus as well as its abnormal sprouting and thickening, low density, tortuosity, and beading in the cornea of some patients with dry eye compared to normal subjects.13 In addition, loss of healthy nerves in the cornea is associated with more inflammatory cells and formation of new vessels.14 This is related to somatosensory dysfunction that will eventually alter the CNS of these patients and lead to a neuropathic pain (allodynia and hyperalgesia) in the absence of ocular surface pathology.
Initially, stimulation of peripheral nerves such as ocular surface surgery, air pollution or dry weather causes corneal afferent neuronal injury and inflammation. As a result, neuronal nuclear, cytosolic and membrane signaling mechanism alters and leads to increased sensitivity of central neurons. At this stage, the correlation between symptoms and signs of ocular surface may be lost.13
In this paper, we aim to review different aspects of ocular pain with the main orientation on ocular surface discomfort and dry eye syndrome and the recent hypothesis of ocular neuropathic pain syndrome.
Methods
Data pertaining to ocular pain were extracted from reviews, meta-analyses, and randomized trials. A search of literature published from January 2000 to May 2019 on the anatomy, pathophysiology, epidemiology, assessment, and treatment of ocular pain was conducted using PubMed. The search was performed using the following terms and associations between them: “ocular pain”, “ocular discomfort”, “neuropathic pain”, “dry eye syndrome”, “ocular pain AND quality of life”. Articles not published in English were excluded. Moreover, despite being published in the period other than specified, some relevant studies with important results were also included. In addition, articles from the references of selected studies were added if they had useful information for the purpose of this review.
Results
By using the keywords mentioned, 112 articles were found through searches. Forty articles were selected based on the inclusion criteria. Besides, some relevant references contained in the selected manuscripts and other studies that were relevant and important, were included. Thus, 45 articles were studied in this review.
Anatomy and Physiology
Neural pathway of ocular discomfort begins at ocular surface by corneal nociceptors which make the first division of the trigeminal nerve and travel to the trigeminal ganglion and nucleus. On their way to the somatosensory cortex in the cerebrum, these neurons synapse in thalamus (limbic).15 Also, some axons from trigeminal nucleus travel to regions in the brain to adjust lacrimation (superior salivatory nucleus) and blinking (facial motor nucleus). In this way, feedback will be sent to lacrimal gland, goblet cells and meibomian glands to modify basal and reflex tear secretion.16 Parasympathetic efferent fibers from CN VII innervate lacrimal glands and regulate its secretion. Conjunctival and corneal epithelial cells can also modify tear composition through the secretion or absorption of electrolytes and water.17
Figure 1 denotes the sensory pathway for the ocular surface ascending to the somatosensory cortex. In addition, descending pathways originating from CNS exist that regulate the afferent pain signals. Due to convergences from skin and viscera with corneal projections in trigeminal nuclei and thalamus, referral pain can be perceived.15 The brain stem has a role in the process of nociception, limbic system has an effect on emotional state, parietal lobe has memories of pain and the frontal lobe has a role in the level of attention given to the pain,18–20 therefore, interaction in multiple levels can downregulate or upregulate the process. Therefore, discrepancy between sign and symptoms is a common finding.
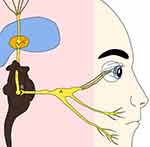 |
Figure 1 Sensory pathway for ocular surface. |
Eyes and periocular tissue (except lens and retina) have a rich innervation.7 The cornea has the highest density of sensory neurons in the body and has the largest cortical representation among sensory organs.21 Free corneal nerve endings, after losing their myelin sheath about 1 mm from the limbus, enter the cornea from the periphery in a radial fashion (mostly at three and nine oʹclock) and make the compact subbasal nerve plexus in the anterior stroma which then penetrates into the epithelium, very near to the ocular surface, making them susceptible to environmental exposures and stimuli.7,22
The naked terminal ends (nociceptors) go to medium or small neurons with thin myelinated (A-delta) or unmyelinated (C) axons. The similar nociceptors existing for the corneal surface, are present for the bulbar conjunctiva and even uvea and sclera.7
The cornea possesses sensory nerve fibers responsive to polymodal, mechanical and cold stimuli.
Corneal mechanonociceptors (A-delta type) comprise 15% corneal fibers and transmit acute pain; for example, sensing a foreign body. They are the fastest conducting neuron and have a low threshold compared to their skin counterparts. They only respond to mechanical stimuli and as they adapt very quickly, they mostly serve to identify the stimulus and the velocity of change in it.7
The majority (approximately 70%) of corneal sensory fibers are of polymodal type which are activated by heat (>39°C) or extreme cold (below 29°C), chemicals (exogenous irritants or endogenous inflammatory mediators). These polymodal nociceptors are mostly from slow-conducting C type axons but some of them belong to the thin myelinated (A delta type) group. Therefore, fast immediate and dull long-lasting pain, both can be perceived by this type of nociceptor. They remain active with the continuity of the mechanical, thermal or chemical stimuli and perceive the intensity of the stimulus as well. The stimulation by polymodals spread not only centripetally (towards the CNS) but also peripherally (antidromic propagation) with the aid of neuropeptides which depolarize distant branches of the same axon neuron that were not stimulated directly by the initial stimulus. Some studies suggest that these processes have a role in trophic maintenance of the cornea. They also play a role in reflex tearing and neuropathic pain.7
The remaining 15% are cold receptors which are A-delta and C nerve fibers that send impulses at rest and when the temperature drops (below 33°C which is normal temperature of ocular surface). Recently, the cold nociceptors and their role in ocular surface integrity have been explained. These receptors identify evaporation (eg, in the absence of blinking) and contribute to basal tear secretion as well. As evaporation occurs faster in dry eyes, an exaggeratedly cold sensation is reported by these patients.7,23 Perception of cold stimuli characterizes ocular neuropathic pain in contrast to other neuropathic pain syndromes.24
As examples in daily life, cold receptor fibers activate when applying cold eye drops or exposing to the cold air. Cold stimuli are sometimes perceived as dryness and evoke tearing without pain. Corneal cold receptors are unique in that they possess spontaneous activity at room temperature as well.25
When a moving object touches the cornea, polymodal nociceptors and mainly mechanonociceptors respond. Firstly, a sharp pain and after that the sense of irritation is conceived by polymodal nerve fibers. If the foreign body remains in conjunctiva or cornea, polymodal nociceptors continue to work and give a feeling of discomfort.26
Pain and irritation are the only sensations that are perceived upon corneal nerve stimulation. Mechanical and polymodal nociceptors can induce the sensation of irritation.16 Ocular surface dryness is considered a mechanical stimulation.
Upon mechanical, hot or cold injuries to the cornea or irritation from chemical substances, different population of ocular nociceptors may activate and lead to neuroma formation of sensory nerve fibers that show abnormal response such as hypesthesia and dysesthesias which are assumed as clinical signs of peripheral neuropathic pain.7 Corneal pain can be elicited from central neuropathic networks as well as those caused by damage to the pain processing neuronal groups (by ischemia, hemorrhage, mechanical compression, infection, degenerative process). Free corneal nerve endings are located between the superficial epithelial cells, very close to the ocular surface, making them susceptible to environmental exposures and stimuli. Dryness is the most common trigger for neuropathic corneal pain. Other triggers including trauma, infection, chemical, or thermal irritants or dysregulated metabolism can result in inflammatory nerve damage and aberrant activation of sensory nerves of the eye and develop a peripheral neuropathic pain.2
From another perspective, different types of pain can originate from injury to different parts of the eye including (1) conjunctiva and muscles, (2) globe (such as eye perforation or corneal ulcer), (3) optic nerve, and finally, (4) autonomic or sympathetic nerves which results in retinal ischemia.27
Epidemiology and Different Etiologies of Ocular Pain
Ocular discomfort secondary to dry eye affects 5–30% of the population aged 50 and above.13 Evaluating pain score in different types of acute eye diseases, herpes zoster ophthalmicus, ultraviolet keratitis and thermal burn were the top three conditions associated with the most severe ocular pain. Blepharitis was in the middle and dry eye appeared at the bottom of this list.28 However, the chronicity, and not the severity, may be the bothersome issue for the majority of patients with blepharitis or dry eye.
In the early post refractive surgery period, up to 95% of patients showed to have dry eye symptoms. These symptoms improve in 60% of patients in one month after surgery and majority relieve within 6–12 months.22 In a study, the prevalence of chronic dry eye after LASIK was reported to be 28% among Asians, while 5% of Caucasians showed such symptoms.29
Second to ocular surface pathologies which are the main source of ocular pain, other pathologies such as uveitis, acute angle closure glaucoma, and endophthalmitis can cause intense ocular pain. The type of sensory nerve fibers involved in transferring the pain of the posterior segment is not fully understood; however, polymodal nociceptors are involved in pain related to the uveal tract.16
Scleritis is a rare cause of periocular and facial pain which exacerbates with ocular movement and interferes with sleep. Conjunctival disorders showed a lower scale of pain.28
Ocular motor cranial nerve palsies (most commonly CN III) due to ischemia is another source of pain which is of similar intensity among diabetic and nondiabetic patients. Frontal headache and brow pain in addition to ocular pain was reported by the patients, suggesting the involvement of the first division of trigeminal nerve. Hyperalgesia in the territory of second division of CN V has also been reported. The nature of the pain reported is “dull” and “achy”, lasting from several days to several weeks. Theoretically, concomitant involvement of the CN V with epineural sheath of CN III in the cavernous sinus has been suggested.30
The iris has some nociceptive sensory fibers that can be activated upon pupillary dilation.31
The pain of an anophthalmic socket is a kind of neuropathic pain (damage to central or peripheral neurons) mediated by the trigeminal nerve. The pain is described as shooting, stabbing, cramps, pinch, or burning. Sometimes there is visual hallucination and occasionally the sensations of the missing lid or itching of nonexisting tissues.32 Patients with an amputated eye (enucleation, evisceration or exenteration) reported pain in about 22.5%. They reported chilliness as a trigger. It may be due to decreased blood flow to the orbit due to tissue deprivation which causes pain in eyelid and conjunctiva. In addition to cold weather, windy weather and psychiatric causes were risk factors for experiencing pain.32
Another proposed mechanism of ocular pain after injury is complex regional pain syndrome (CRPS), which is a neuropathic pain syndrome well described in extremities. CPRS is rare in orofacial regions. Postganglionic sympathetic fibers along the carotid plexus enter the orbit following ophthalmic artery and CN V (ophthalmic and maxillary divisions). These sympathetic nerves supply eyelid muscles, forehead skin and those along the ophthalmic nerve regulate blood flow to ocular and orbital regions. A substantial trauma to the craniofacial region may provoke CPRS signs and symptoms which include deep persistent pain, burning sensation and edema along with moist skin.31
In one study assessing painful blind eye, removing the globe (enucleation and evisceration) was reported to be effective in relieving pain within three months after surgery. Complications of surgery or orbital implant can cause recurrent pain.33
Other causes of unilateral headaches and eye pathologies include cluster headache, Tolosa–Hunt syndrome and cavernous carotid fistula.34
Chronic fatigue, fibromyalgia, urological disorders, carpal tunnel syndrome and sleep disorders have neuropathic central sensitization and are associated with orofacial pain.35
Ocular Surface Mechanisms Triggering Pain
Peripheral ocular fibers are usually affected by two main pathologic processes: inflammation and/or direct peripheral or central neuronal injury. Inflammation activates polymodal nociceptor fibers (inflammatory pain) whereas peripheral neuronal injury causes ectopic continuous activity (neuropathic pain). Nerve injury affects both low and high-threshold cold thermoreceptors; the low threshold ones provoke a sense of dryness with a cooling component while high threshold cold thermoreceptors activation illicit a sense of painful dryness. But inflammation depresses the activation of both cold thermoreceptors with the aid of inflammatory mediators.16
In dry eye disease, both cooling and hyperosmolarity stimulate cold thermoreceptors. Dryness damages the surface epithelium which results in inflammation and nerve damage with subsequent activation of the polymodal nociceptors.
Allergic keratoconjunctivitis is another example of ocular surface inflammation which is accompanied by increased activity of mechano- and polymodalnociceptors (lower threshold) and decreased activity of cold thermoreceptors. This pattern is compatible with the clinical symptoms of grittiness and itching.16
Refractive surgery affects polymodal and mechanosensory fibers to the way that they become less responsive to the stimuli but the cold thermoreceptors show increased background activity.16 More than 40% of patients report neuropathic pain which does not wake them up. Corneal neuropathy was observed in the absence of objective dry eye. Although dry eye was previously blamed for postoperative symptoms, transected corneal nerves through peripheral and central sensitization was found to induce neuropathic pain.36 If exam or laser scanning confocal microscopy does not show any corneal pathology, then the central mechanism has a role in pain. Topical anesthesia relieves pain only in those with peripheral pathology.28,37
LASIK with the creation of flap and laser ablation, disrupts both the dense subbasal nerve plexus and stromal corneal nerves. The deeper the stromal ablation, the increased severity of dry eye symptoms will be after surgery. Conjunctival sensation and goblet cells may also be damaged during LASIK (both femtosecond and microkeratome assisted). After LASIK, inflammatory cytokines exist in the tear film and conjunctival epithelium. Alteration in corneal shape may change the relationship between the eyelids and ocular surface and result in tear instability during blinking. Moreover, surgical ablation of nerve fibers interrupts the cornea-trigeminal nerve-brainstem-facial nerve-lacrimal gland reflex arc that leads to decreased basal and reflex tear secretion and blinking.22,38 Also, there is evidence confirming the presence of cornea-nervous system-lacrimal gland regulatory loops which leads to the expression of nerve factors, growth factors, and cytokines that improve healing of the corneal epithelium. Damage to the nerves of the cornea can destroy these mechanisms and weakens the neurotrophic effect on corneal epithelial cells which make the epithelial apoptosis more likely.39,40
During refractive surgery, the injured axon and its nerve endings degenerate while the sprouts of nerve ends invade from the periphery to cover the injured area. With an incomplete regrowth of the damaged axon, a tangled mass of aborted sprouts and end bulbs form microneuroma. These neuromas themselves give rise to new sprouts and the older sprouts form neighbourhood nerves degenerate. These ectopic neurons with altered ion channels of corneal membranes show spontaneous activity while irresponsive to the natural stimuli.7 Belmonte described the dry eye sensation after refractive surgery as a “phantom” cornea.41
Similarly, grooved incisions during cataract surgery can trigger dry eye symptoms early postoperatively, even in the absence of dry eye before surgery.37
Contact lens and lens solutions can cause hypoxia and inflammation that interact with ocular surface and lid margins, which can subsequently lead to vasodilation, reflex blinking, tearing and pain.16 Asian ethnicity is among predisposing risk factors for dry eye in contact lens wearers.42
Dry Eye Syndrome and Ocular Neuropathic Pain Syndrome
Clinicians may minimize the severity of dry eye symptoms; however, moderate or severe symptoms of dry eye have been reported to be equal to moderate to severe angina or hospital dialysis and mild dry eye symptoms have been reported to be equivalent to severe migraines.23 Patients with dry eye may suffer from allodynia, hyperalgesia, hypesthesia (decreased sensitivity to pain), and hyperesthesia.13 Objective findings and patients’ subjective complaints can often be difficult to reconcile.36 Dry eye is a complex multifactorial disease that can broadly be categorized as the result of either insufficient tear production or increased evaporation. Understanding the underlying cause of a patient’s dry eye symptoms can help to direct initial treatment. For example, lid hygiene is an important measure to address evaporative tear loss and frequent use of artificial tears and punctal occlusion are important interventions to manage insufficient tear production. Treatment with anti-inflammatory agents are helpful for both conditions.3 Lid position, the presence of meibomian gland dysfunction, reduced tear production from lacrimal dysfunction, poor or infrequent blinking, conjunctival irregularities (such as conjunctival chalasis), abnormal tear osmolarity, and activation of ocular surface inflammatory cascades, all can play a role.36,43 Inflammation plays a significant role in the pathogenesis of dry eye. T cells are recruited to the conjunctiva of dry eye patients and neutrophil infiltration has been observed after nerve injury (eg following corneal surgery). In addition, tear levels of MMP-9 (matrix metalloproteinase-9) and reactive oxygen species are increased. Various inflammatory mediators including interleukins (2, 4, 6, 8, 10, and 17) as well as TNF-alpha show increased concentration in the tear. The level of anti-inflammatory mediators (such as those derived from omega 3 and 6 fatty acids) are elevated as well and the balance between pro- and anti-inflammatory arms determine the sign and symptoms in dry eye.2
Older age and female gender are known risk factors for dry eye syndrome. Low estrogen levels and lacrimal gland dysfunction in older age play an important role. The number of corneal nerves decreases with age which results in a corresponding reduction in tactile sensation; however, hyperalgesia to evaporation by the remaining nerve increases. In addition, there may be an enhancement in central pain regulation.35
Atopic dermatitis, obesity, indoor occupation and lack of regular exercise are other predisposing factors for dry eye.44 Dry eye syndrome is also associated with psychiatric conditions such as depression, anxiety, post-traumatic stress disorder, and sleep disruption.9
Dry eye is an important component of several medical conditions including diabetes mellitus, rheumatoid arthritis, Sjorgen's syndrome, thyroid disease, allergy, and hyperlipidemia. Additionally, patients who suffer from other pain control conditions such as irritable bowel syndrome, chronic pain syndrome, and pelvic pain are likely to suffer from dry eye. Taking anticholinergic medications can contribute to dry eye symptoms. Vitamin D deficiency is commonly encountered in many of these same systemic disorders and can affect the immune system. Therefore, taking supplement or sunlight exposure is encouraged.44
In dry eye, inflammation can disturb somatosensory processing to that degree that affects the function of plasticity.
Peripheral sensitization (increased responsiveness of peripheral nociceptors) can lead to central sensitization (neuroplastic changes to CNS) which eventually cause hyperexcitability of nociceptors in the CNS. This can result in patients have symptoms in the absence of ocular surface abnormalities.23 Inhibition of pain occurs in the trigeminal nucleus and in the process of central sensitization. Allodynia is the result of central sensitization with involvement of Aβ and C fibers phonotypic switch from harmless stimuli to a pain transmission. In these circumstances, patients may experience pain from normally benign stimuli. Several therapies including opioids, serotonergic, alpha-2 adrenergic agonists, and gabapentin, control neuropathic pain by effecting the trigeminal nucleus with diminishing excitation or increasing inhibition pathways.23 Neuropathic pain is an autonomous irritation which does not respond to local anesthetics or tear dysfunction therapy because the pain originates in CNS sensitization. This is why patients can have pain in the absence of clinical findings consistent with tear dysfunction. Environmental exposure, fatigue and other factors may influence patient’s perception of pain and awareness of symptoms.
Dry eye patients can experience hyperalgesia when they experience pain along the distribution of the first and second trigeminal nerve beyond the ocular/periocular regions with extension to the face, ears, cheek, temple, teeth, and temporomandibular joint.15 The pain can be spontaneous and described as burning hot, pins, cutting, pounding, pressure/aching, tingling, and electric pain. Blepharospasm can be seen in orofacial pain and dry eye.
Migraine and dry eye can coexist due to the common pathways within the trigeminal system with photophobia being a common shared symptom. Migraine patients have a 1.4-fold higher chances of having dry eye.2 Exposure to light can play a role in both conditions with light exposure being known to trigger migraine headaches and bright lights causing reflex blinking via trigeminal. Thermal or painful stimuli can exacerbate photophobia in patients with migraine.15 In patients with centralized orofacial pain, touch of the skin in the territory of ophthalmic branch of CN V has been reported to be painful. Hypersensitivity to sound (hyperacusis), tinnitus, vertigo and changes in smell and taste were also reported.35
Clinical Assessment of Ocular Discomfort
The clinical evaluation of patients with ocular discomfort necessitates careful history taking and examination. Of paramount importance is ruling out signs of intraocular inflammation or infection which can be sight threatening. Detailed slit lamp examination is necessary to examine the ocular surface to assess for evidence of ocular surface compromise including irregular tear films and corneal epithelial changes such as punctate epithelial erosion. Dry eye workup including tear breakup time, Schirmer's test, and vital stains are necessary in all patients.
Corneal sensitivity can be measured using a wisp of cotton from a cotton tip applicator or a Cochet–Bonnet anesthesiometer, although the utility and reproducibility of these measures has been debated. Most patients’ response to corneal stimuli can vary based on underlying disease status. Most of the patients with primary dry eye syndrome show a lower threshold for pain and mechanical corneal stimuli. Patients with dry eye secondary to Sjogren's syndrome were to have a negative correlation between dry eye symptoms and corneal sensitivity. These patients show low corneal sensitivity and ocular symptoms despite the presence of ocular surface disease.13,45
Hypersensitivity to other stimuli and systemic pain status should be checked in patients to rule out central sensitization. For example, patients with fibromyalgia have a twofold increased rate of dry eye signs and symptoms. These patients have been shown to have increased corneal thresholds to heat and chemical stimuli but normal corneal mechanical threshold. Patients with Sjogren's syndrome show abnormal mechanical threshold but normal chemical threshold compared to normal controls.13 Discrepancy between mechanical and chemical sensitivity has been shown in several studies but its mechanism remained to be elucidated. It might be related to the abnormal responsiveness of polymodal nociceptors. Conversion of CO2 to H2CO3 is inhibited in aqueous deficiency causing a diminished local H+ concentration. Alternatively, degeneration or regeneration of corneal nerve fibers may cause an abnormal responsiveness to natural stimuli.46
Patients’ emotional and psychological status is also important to assess. Association of comorbidities like depression and anxiety with ocular pain disorders have been described.
Alterations in confocal microscopy can be helpful in assessment of dry eye patients; pattern and density of subbasal nerves can help in diagnosis and other pathologies of the cornea can be ruled out.47
After all, neuropathic pain is resistant to usual treatments. It is of paramount importance to diagnose the neuropathic features in dry eye patients, because this condition often requires alternative measures. An accurate history taking with the aid of questionnaires is an available approach for this purpose. However, most accessible questionnaires such as the Ocular Surface Disease Index (OSDI) and Dry Eye Questionnaire 5 (DEQ5) are not reliable ones to reach to the diagnosis of neuropathic components in dry eye and more sophisticated and comprehensive questionnaires with modifications are required for this goal.
Treatment
The treatment of ocular surface discomfort or ocular neuropathic pain syndrome requires a multimodal approach that addresses several underlying mechanisms for this condition. Evidently, majority of neuropathic pain treatment modalities have not been well studied and adapted in ocular neuropathic pain syndrome. Studies show that conventional treatments for dry eye syndrome, as a localized disease process, are of little value in ocular neuropathic pain syndrome.
Anti-inflammatory agents can be helpful to address ocular surface inflammation. Cyclosporine, corticosteroids, and tetracycline can reduce the risk of neuroplastic peripheral corneal or lid wiper sensitization by decreasing ocular surface inflammation.48,49 Topical cyclosporine 0.05% has been used to increase tear production in patients with inflammatory conditions and should be considered in the perioperative management of dry eye in patients undergoing procedures that will require corneal manipulation.50 Lid hygiene with warm compresses and lid scrubs as well as topical or oral antibiotics can reduce inflammation.51 Stabilizing the tear film before any corneal procedures can be helpful in preventing pain. NSAIDs have been used with success for postoperative pain.52 Autologous serum contains neurotrophic elements, growth factors, and anti-inflammatory substances that can aid in treating ocular surface disease and corneal nerve dysfunction. Further, autologous serum prevents apoptosis in corneal and conjunctival epithelium.22,53
In cases where high intraocular pressure is causing pain, ocular hypotensive drugs are helpful.
Addressing the underling lid abnormalities such as ectropion can be helpful in dry eye symptoms. Punctal plugging can allow for prolonged retention of a patient’s tears. Scleral gas permeable and bandage contact lenses are useful for dry eye patients.22 Special glasses are shown to be helpful in preventing evaporation.54
Education plays a key role in managing patient’s expectations and controlling symptoms. Patients need to be aware to avoid environmental triggers and behavioral changes should also be addressed. Basal tear production is under the control of the hypothalamus and limbic system so managing stress or psychosocial imbalances can be helpful.55,56
A number of new drugs have also emerged in recent years for treatment of ocular pain. Oxervate™ (cenegermin-bkbj) is an FDA approved ophthalmic solution that contains cenegermin, a recombinant form of human nerve growth factor and has an important role in the maintenance, development and survival of neurons.57 Lifitegrast is a novel drug that is approved by FDA for treatment of dry eye. It is a lymphocyte function-associated antigen intercellular adhesion molecule 1 antagonist that inhibits the inflammatory pathways in dry eye disease.58 Nexagon is an oligonucleotide that downregulates expression of the key gap junction protein Cx43, and is in trial for treatment of persistent corneal epithelial defects.59
After all these evaluations and treatments, patients with neuropathic pain and central sensitization may continue to have symptoms. In this situation, a multimodal approach including antineuropathic pain treatment should be considered, although its best management is controversial and depends on factors such as severity of pain, underlying pathophysiology and patient’s comorbidities.
Centrally acting neuromodulators have been successfully used for neuropathic pain in the absence of ocular surface pathology.36 Antiepileptics (Gabapentin and Pregabalin) can be considered as the first line treatment; Serotonin–norepinephrine reuptake inhibitors (duloxetine, venlafaxine) as the second-line (or as first-line agents in concomitant musculoskeletal pain or depression); and tricyclic antidepressants (nortriptyline, amitriptyline) as third-line agents. Combination therapy or weak opioids (tramadol) can also be used in the case of resistance to treatment in the setting of a broad neurologic pain. Diazepam and even injection of BTX-A in the location of pain has been reported to be helpful.2,54,60 However, none of these modalities are studied in ocular neuropathic pain syndrome.
Gabapentin regulates central sensitization. In addition, it is helpful in decreasing the discharges from damaged peripheral neurons. Evidence support effect of this category of neuromodulators in a wide range of ocular pains such as immediate post refractive surgery pain, eyelid surgery and postherpetic neuralgia.23,61
Cognitive Behavioral Therapy (CBT) in chronic eye pain has been shown to be effective.54
More aggressive nonpharmacologic interventions have also been employed in the treatment of ocular pain. Matsuura et al treated 35 patients with intractable ocular pain long after eye surgery and unresponsive to anti-inflammatory medications with a stellate ganglion block and found this was effective for both nociceptive and neuropathic, with greater success in addressing nociceptive pains.62 Blockade of sphenopalatine ganglion (SPG) and stellate ganglion were also suggested for neuropathic ocular pain management. Extreme pain was controlled with alcohol injection into the orbit. The effect of gasserian ganglion block or neurolysis remained to be elucidated in the treatment of refractory ocular neuropathic pain syndrome or oculofacial pain syndrome.63,64
The most important steps and treatment measures for the management of nociceptive and neuropathic ocular pain are shown in Figure 2.
 |
Figure 2 The most important steps in the management of nociceptive and neuropathic ocular pain. |
Conclusion
Neuropathic pain in ophthalmology has been overlooked for the past years. Ocular neuropathic pain syndrome, can be easily misdiagnosed as dry eye syndrome or other localised or regional pains.
Ocular neuropathic pain syndrome is a chronic pain that interferes with daily activities. It presents with hyperalgesia to cold, heat and mechanical stimuli and can even be perceived in the absence of any peripheral stimuli. Even a nonpainful stimulus (such as wind or draught) can provoke an ocular pain (allodynia). Inflammation plays an important role as well as peripheral and CNS mechanisms. Any mechanical or chemical injury to the corneal nerve endings can result in ectopic sprouts and neuroma formation which show spontaneous activity. Ocular irritation such as burning, photophobia, pain, and grittiness are reported by these patients while objective tests may not establish the diagnosis of dry eye. Pain may be referral in the territory of trigeminal nerve. In case assessments show sufficient amount of tear, anti-inflammatory agents and centrally acting neuromodulators will be the treatment of choice. A multidisciplinary approach with the collaborating of ophthalmologists and pain specialists can be helpful. Preventive measures in susceptible patients should be considered to prohibit an ongoing signal which may transform a peripheral pain to a central and chronic one. Cognitive and behavioral therapy as well as reassurance should be considered as well.
Acknowledgment
We want to acknowledge Kimia Karkheiran for the contribution she made for illustration.
Funding
There is no funding to report.
Disclosure
Bobeck S Modjtahedi reports grants from Genentech, outside the submitted work. The author report no other conflicts of interest in this work.
References
1. Treede RD, Rief W, Barke A, et al. Chronic pain as a symptom or a disease: the IASP classification of chronic pain for the international classification of diseases (ICD-11). Pain. 2019;160(1):19–27. doi:10.1097/j.pain.0000000000001384
2. Galor A, Levitt RC, Felix ER, Martin ER, Sarantopoulos CD. Neuropathic ocular pain: an important yet underevaluated feature of dry eye. Eye (Lond). 2015;29(3):301–312.
3. Lalisse S, Hua J, Lenoir M, Linck N, Rassendren F, Ulmann L. Sensory neuronal P2RX4 receptors controls BDNF signaling in inflammatory pain. Sci Rep. 2018;8(1):964. doi:10.1038/s41598-018-19301-5
4. Veluchamy A, Hebert HL, Meng W, Palmer CNA, Smith BH. Systematic review and meta-analysis of genetic risk factors for neuropathic pain. Pain. 2018;159(5):825–848. doi:10.1097/j.pain.0000000000001164
5. Korczeniewska OA, Husain S, Khan J, Eliav E, Soteropoulos P, Benoliel R. Differential gene expression in trigeminal ganglia of male and female rats following chronic constriction of the infraorbital nerve. Eur J Pain. 2018;22(5):875–888. doi:10.1002/ejp.1174
6. Vehof J, Sillevis Smitt-Kamminga N, Nibourg SA, Hammond CJ. Sex differences in clinical characteristics of dry eye disease. Ocul Surf. 2018;16(2):242–248. doi:10.1016/j.jtos.2018.01.001
7. Belmonte C, Aracil A, Acosta MC, Luna C, Gallar J. Nerves and sensations from the eye surface. Ocul Surf. 2004;2(4):248–253. doi:10.1016/S1542-0124(12)70112-X
8. Li W, Graham AD, Lin MC. Understanding ocular discomfort and dryness using the pain sensitivity questionnaire. PLoS One. 2016;11(5):e0154753. doi:10.1371/journal.pone.0154753
9. Galor A, Covington D, Levitt AE, et al. Neuropathic ocular pain due to dry eye is associated with multiple comorbid chronic pain syndromes. J Pain. 2016;17(3):310–318. doi:10.1016/j.jpain.2015.10.019
10. Brooks J, Tracey I. From nociception to pain perception: imaging the spinal and supraspinal pathways. J Anat. 2005;207(1):19–33. doi:10.1111/j.1469-7580.2005.00428.x
11. Reichling DB, Levine JD. Critical role of nociceptor plasticity in chronic pain. Trends Neurosci. 2009;32(12):611–618. doi:10.1016/j.tins.2009.07.007
12. Goyal S, Hamrah P. Understanding neuropathic corneal pain–gaps and current therapeutic approaches. Semin Ophthalmol. 2016;31(1–2):59–70. doi:10.3109/08820538.2015.1114853
13. Spierer O, Felix ER, McClellan AL, et al. Corneal mechanical thresholds negatively associate with dry eye and ocular pain symptoms. Invest Ophthalmol Vis Sci. 2016;57(2):617–625. doi:10.1167/iovs.15-18133
14. Ferrari G, Hajrasouliha AR, Sadrai Z, Ueno H, Chauhan SK, Dana R. Nerves and neovessels inhibit each other in the cornea. Invest Ophthalmol Vis Sci. 2013;54(1):813–820. doi:10.1167/iovs.11-8379
15. Rosenthal P, Borsook D, Moulton EA. Oculofacial pain: corneal nerve damage leading to pain beyond the eye. Invest Ophthalmol Vis Sci. 2016;57(13):5285–5287. doi:10.1167/iovs.16-20557
16. Belmonte C, Acosta MC, Merayo-Lloves J, Gallar J. What causes eye pain? Curr Ophthalmol Rep. 2015;3(2):111–121. doi:10.1007/s40135-015-0073-9
17. Meng ID, Kurose M. The role of corneal afferent neurons in regulating tears under normal and dry eye conditions. Exp Eye Res. 2013;117:79–87. doi:10.1016/j.exer.2013.08.011
18. Bushnell MC, Ceko M, Low LA. Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci. 2013;14(7):502–511. doi:10.1038/nrn3516
19. Hofbauer RK, Rainville P, Duncan GH, Bushnell MC. Cortical representation of the sensory dimension of pain. J Neurophysiol. 2001;86(1):402–411. doi:10.1152/jn.2001.86.1.402
20. Foltz EL, White LE. Pain “relief” by frontal cingulumotomy. J Neurosurg. 1962;19:89–100. doi:10.3171/jns.1962.19.2.0089
21. Andreotti AM, Goiato MC, Pellizzer EP, et al. Phantom eye syndrome: a review of the literature. ScientificWorldJournal. 2014;2014:686493. doi:10.1155/2014/686493
22. Shtein RM. Post-LASIK dry eye. Expert Rev Ophthalmol. 2011;6(5):575–582. doi:10.1586/eop.11.56
23. Levitt AE, Galor A, Weiss JS, et al. Chronic dry eye symptoms after LASIK: parallels and lessons to be learned from other persistent post-operative pain disorders. Mol Pain. 2015;11:21. doi:10.1186/s12990-015-0020-7
24. Hirata H, Meng ID. Cold-sensitive corneal afferents respond to a variety of ocular stimuli central to tear production: implications for dry eye disease. Invest Ophthalmol Vis Sci. 2010;51(8):3969–3976. doi:10.1167/iovs.09-4744
25. Kovacs I, Luna C, Quirce S, et al. Abnormal activity of corneal cold thermoreceptors underlies the unpleasant sensations in dry eye disease. Pain. 2016;157(2):399–417. doi:10.1097/j.pain.0000000000000455
26. Belmonte C, Garcia-Hirschfeld J, Gallar J. Neurobiology of ocular pain. Prog Retin Eye Res. 1997;16(1):117–156. doi:10.1016/S1350-9462(96)00027-4
27.
28. Roberts CJ, MacLeod JD, Elkington AR. Ocular pain: a casualty study. The spectrum and prevalence of pain in acute eye disease. Eye (Lond). 1997;11(Pt 3):342–344. doi:10.1038/eye.1997.72
29. Albietz JM, Lenton LM, McLennan SG. Dry eye after LASIK: comparison of outcomes for Asian and Caucasian eyes. Clin Exp Optom. 2005;88(2):89–96. doi:10.1111/j.1444-0938.2005.tb06673.x
30. Wilker SC, Rucker JC, Newman NJ, Biousse V, Tomsak RL. Pain in ischaemic ocular motor cranial nerve palsies. Br J Ophthalmol. 2009;93(12):1657–1659. doi:10.1136/bjo.2008.155150
31. Thapa D, Ahuja V. Complex regional pain syndrome - I following eye injury. Indian J Anaesth. 2012;56(6):596–597. doi:10.4103/0019-5049.104596
32. Rasmussen ML, Prause JU, Toft PB. Phantom pain after eye amputation. Acta Ophthalmol. 2011;89(1):10–16. doi:10.1111/j.1755-3768.2010.02058.x
33. Shah-Desai SD, Tyers AG, Manners RM. Painful blind eye: efficacy of enucleation and evisceration in resolving ocular pain. Br J Ophthalmol. 2000;84(4):437–438. doi:10.1136/bjo.84.4.437
34. Park T, Choi G. Unilateral fronto-temporal headache with ocular pain caused by lens subluxation due to spontaneous zonulysis. Clin Exp Emerg Med. 2015;2(2):133–136. doi:10.15441/ceem.15.031
35. Rosenthal P, Borsook D. Ocular neuropathic pain. Br J Ophthalmol. 2016;100(1):128–134. doi:10.1136/bjophthalmol-2014-306280
36. McMonnies CW. The potential role of neuropathic mechanisms in dry eye syndromes. J Optom. 2017;10(1):5–13. doi:10.1016/j.optom.2016.06.002
37. Cho YK, Kim MS. Dry eye after cataract surgery and associated intraoperative risk factors. Korean J Ophthalmol. 2009;23(2):65–73. doi:10.3341/kjo.2009.23.2.65
38. Ambrósio R, Tervo T, Wilson SE. LASIK-associated dry eye and neurotrophic epitheliopathy: pathophysiology and strategies for prevention and treatment. J Refract Surg. 2008;24(4):396–407. doi:10.3928/1081597X-20080401-14
39. Wilson SE. Laser in situ keratomileusis-induced (presumed) neurotrophic epitheliopathy. Ophthalmology. 2001;108(6):1082–1087. doi:10.1016/S0161-6420(01)00587-5
40. Wilson SE, Liang Q, Kim WJ. Lacrimal gland HGF, KGF, and EGF mRNA levels increase after corneal epithelial wounding. Invest Ophthalmol Vis Sci. 1999;40(10):2185–2190.
41. Belmonte C. Eye dryness sensations after refractive surgery: impaired tear secretion or “phantom” cornea? J Refract Surg. 2007;23(6):598–602. doi:10.3928/1081-597X-20070601-11
42. Tran N, Graham AD, Lin MC. Ethnic differences in dry eye symptoms: effects of corneal staining and length of contact lens wear. Cont Lens Anterior Eye. 2013;36(6):281–288. doi:10.1016/j.clae.2013.06.001
43. Baudouin C, Irkec M, Messmer EM, et al. Clinical impact of inflammation in dry eye disease: proceedings of the ODISSEY group meeting. Acta Ophthalmol. 2018;96(2):111–119. doi:10.1111/aos.13436
44. Yoon SY, Bae SH, Shin YJ, et al. Low serum 25-hydroxyvitamin D levels are associated with dry eye syndrome. PLoS One. 2016;11(1):e0147847. doi:10.1371/journal.pone.0147847
45. Adatia FA, Michaeli-Cohen A, Naor J, Caffery B, Bookman A, Slomovic A. Correlation between corneal sensitivity, subjective dry eye symptoms and corneal staining in Sjogren’s syndrome. Can J Ophthalmol. 2004;39(7):767–771. doi:10.1016/S0008-4182(04)80071-1
46. Tuisku IS, Konttinen YT, Konttinen LM, Tervo TM. Alterations in corneal sensitivity and nerve morphology in patients with primary Sjogren’s syndrome. Exp Eye Res. 2008;86(6):879–885. doi:10.1016/j.exer.2008.03.002
47. Alhatem A, Cavalcanti B, Hamrah P. In vivo confocal microscopy in dry eye disease and related conditions. Semin Ophthalmol. 2012;27(5–6):138–148. doi:10.3109/08820538.2012.711416
48. Pflugfelder SC, Kinoshita S. Management and therapy of dry eye disease: report of the management and therapy subcommittee of the international dry eye workShop (2007). Ocul Surf. 2007;5(2):163–178.
49. Kim YJ, Ryu JS, Park SY, et al. Comparison of topical application of TSG-6, cyclosporine, and prednisolone for treating dry eye. Cornea. 2016;35(4):536–542. doi:10.1097/ICO.0000000000000756
50. Kanellopoulos AJ. Incidence and management of symptomatic dry eye related to LASIK for myopia, with topical cyclosporine A. Clin Ophthalmol. 2019;13:545–552. doi:10.2147/OPTH.S188521
51. Nichols KK, Foulks GN, Bron AJ, et al. The international workshop on meibomian gland dysfunction: executive summary. Invest Ophthalmol Vis Sci. 2011;52(4):1922–1929. doi:10.1167/iovs.10-6997a
52. Colin J. The role of NSAIDs in the management of postoperative ophthalmic inflammation. Drugs. 2007;67(9):1291–1308. doi:10.2165/00003495-200767090-00004
53. Higuchi A. Autologous serum and serum components. Invest Ophthalmol Vis Sci. 2018;59(14):DES121–DES129. doi:10.1167/iovs.17-23760
54. Jacobs DS. Diagnosis and treatment of ocular pain: the ophthalmologist’s perspective. Curr Ophthalmol Rep. 2017;5(4):271–275. doi:10.1007/s40135-017-0152-1
55. Apkarian AV, Bushnell MC, Treede RD, Zubieta JK. Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain. 2005;9(4):463–484.
56. Kawashima M, Uchino M, Yokoi N, et al. Associations between subjective happiness and dry eye disease: a new perspective from the Osaka study. PLoS One. 2015;10(4):e0123299. doi:10.1371/journal.pone.0123299
57. Sheha H, Tighe S, Hashem O, Hayashida Y. Update on cenegermin eye drops in the treatment of neurotrophic keratitis. Clin Ophthalmol. 2019;13:1973–1980. doi:10.2147/OPTH.S185184
58. Abidi A, Shukla P, Ahmad A. Lifitegrast: a novel drug for treatment of dry eye disease. J Pharmacol Pharmacother. 2016;7(4):194–198. doi:10.4103/0976-500X.195920
59. Ormonde S, Chou CY, Goold L, et al. Regulation of cennexin43 gap junction protein triggers vascular recovery and healing in human ocular persistent epithelial defect wounds. J Membrane Biol. 2012;245(7):381–388. doi:10.1007/s00232-012-9460-4
60. Nam KE, Kim JS, Hong BY, et al. Botulinum toxin type A injection for neuropathic pain in a patient with a brain tumor: a case report. Ann Rehabil Med. 2017;41(6):1088–1092. doi:10.5535/arm.2017.41.6.1088
61. Kuhnle MD, Ryan DS, Coe CD, et al. Oral gabapentin for photorefractive keratectomy pain. J Cataract Refract Surg. 2011;37(2):364–369. doi:10.1016/j.jcrs.2010.08.041
62. Matsuura M, Matsuura M, Ando F, Sahashi K, Torii Y, Hirose H. [The effect of stellate ganglion block on prolonged post-operative ocular pain]. Nippon Ganka Gakkai Zasshi. 2003;107(10):607–612.
63. Xavier TV, de Oliveira TR, Mendes TC. Treatment of patients with painful blind eye using stellate ganglion block. Braz J Anesthesiol. 2016;66(1):75–77. doi:10.1016/j.bjan.2012.12.009
64. Tolba R, Weiss AL, Denis DJ. Sphenopalatine ganglion block and radiofrequency ablation: technical notes and efficacy. Ochsner J. 2019;19(1):32–37. doi:10.31486/toj.18.0163
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
