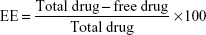Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Novel piperine-loaded Tween-integrated monoolein cubosomes as brain-targeted oral nanomedicine in Alzheimer’s disease: pharmaceutical, biological, and toxicological studies
Authors Elnaggar Y , Etman S, Abdelmonsif D , Y. Abdallah O
Received 25 April 2015
Accepted for publication 13 June 2015
Published 27 August 2015 Volume 2015:10(1) Pages 5459—5473
DOI https://doi.org/10.2147/IJN.S87336
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Yosra SR Elnaggar,1 Samar M Etman,1 Doaa A Abdelmonsif,2 Ossama Y Abdallah1
1Department of Pharmaceutics, Faculty of Pharmacy, Alexandria University, Alexandria, Egypt; 2Department of Medical Biochemistry, Faculty of Medicine, Alexandria University, Alexandria, Egypt
Abstract: Alzheimer’s disease (AD) is one of the most patient devastating central nervous system diseases with no curative therapy. An effective oral therapy with brain-targeting potential is required that is hampered by blood–brain barrier. Piperine (PIP) is a natural alkaloid with memory enhancing potentials. Oral PIP delivery suffers from its hydrophobicity and first-pass metabolism. In this study, novel Tween-modified monoolein cubosomes (T-cubs) were elaborated as bioactive nanocarriers for brain-targeted oral delivery of PIP. Seven liquid crystalline nanoparticles (cubosomes) were prepared testing different bioactive surfactants (Tween 80, poloxamer, and Cremophor). Full in vitro characterization was carried out based on particle size, zeta potential, polydispersity index, entrapment efficiency, and in vitro release. Morphological examination and structure elucidation were performed using transmission and polarizing microscopes. Sporadic dementia of Alzheimer’s type was induced in 42 male Wistar rats on which full behavioral and biochemical testing was conducted. Brain toxicity was assessed based on Caspase-3 assay for apoptosis and tumor necrosis factor-α for inflammation. Liver and kidney toxicity studies were conducted as well. Among others, T-cubs exhibited optimum particle size (167.00±10.49 nm), polydispersity index (0.18±0.01), and zeta potential (-34.60±0.47 mv) with high entrapment efficiency (86.67%±0.62%). Cubs could significantly sustain PIP in vitro release. In vivo studies revealed T-cubs potential to significantly enhance PIP cognitive effect and even restore cognitive function to the normal level. Superiority of T-cubs over others suggested brain-targeting effect of Tween. Toxicological studies contended safety of cubs on kidney, liver, and even brain. T-cubs exhibited potential anti-inflammatory and anti-apoptotic activity of loaded PIP, indicating potential to stop AD progression that was first suggested in this article. Novel oral nanoparticles elaborated possess promising in vitro and in vivo characteristics with high safety for effective chronic treatment of AD.
Keywords: piperine, monoolein, cubosome, nanoparticles, Alzheimer’s disease, liquid crystalline
Introduction
Alzheimer’s disease (AD) is the most common form of dementia with no curative treatment so far.1 This neuropathological condition presents two established pathophysiological hallmarks in the brain. These include extracellular accumulations of amyloid-β peptide and intracellular neurofibrillary tangles of hyperphosphorylated tau protein.2 The estimated number of patients suffering from dementia worldwide is 35.6 million in 2010, with expectation of double this number every 20 years due to aging of human population, which will cause heavy economic and social burden.3
Over the past years, the goal of medical research was to better understand AD causes and pathophysiology and to develop an effective treatment for the disease.4 However, the proposed treatments so far showed only symptomatic relief and were not able to stop the disease progression. Moreover, they also have various side effects.5
The use of herbal medicines has been increasing in the last few years. Recent pharmaceutical research had focused on phytomedicine.6,7 Numerous medicinal plants possessing profound central nervous system (CNS) effects and antioxidant activity have recently received attention to improve cognitive function in AD. Piperine (PIP) (1-piperoylpiperidine) is a nitrogenous pungent alkaloid present in the fruits of black pepper (Piper nigrum), long pepper (Piper longum), and other piper species (family: Piperaceae). Recently, there was a focus on the beneficial physiological effects of PIP as a drug. The effect of PIP on memory performance and neurodegeneration in the animal model of AD has been investigated.8
Nevertheless, PIP oral delivery is hampered by many pharmaceutical challenges. The drug is highly lipophilic with log P=2.259 and low water solubility. Furthermore, the drug suffers extensive first-pass metabolism and the absolute oral bioavailability in Wistar rats is 24%.10 The drug also has pH-mediated metabolism to piperidine and photoisomerization.11 Therefore, oral delivery of PIP to the brain is challenging. Tailoring of brain-targeted, hydrophobic nanocarrier integrated with bioactive ingredients (BEs) to inhibit PIP metabolism is therefore required. Among BEs, Tween 80 is known for its ability to target the brain via low density lipoprotein receptors.12 Furthermore, Cremophor RH 40 along with Tween 80 were recognized as inhibitors for liver metabolizing enzymes,13–15 which may protect PIP from hepatic first-pass effect. In a consequence, incorporation of these two BEs into the nanosystem is anticipated to enhance PIP oral delivery to the brain.
Recently, lipid-based nanocarriers have been extensively exploited by our work group to improve delivery characteristics of challenging lipophilic actives.6,14–22 Lipid-based liquid crystalline nanoparticles (LCN) have recently captured the obvious interest of pharmaceutical research to enhance the bioavailability of lipophilic drugs.23 LCN enjoy characteristics of biocompatibility, bioadhesivity, and biodegradability offering various advantages for brain delivery. These systems are formed mainly of glyceryl monooleate (GMO) dispersed in excess water. The system requires the presence of steric stabilizer to allow stabilization of the formed crystals. Self-assembled property of GMO necessitates the need for relatively lower surfactant concentration compared with other nanocarriers, which is an advantage in oral delivery. The most commonly used stabilizer is poloxamer 407 (usually in a GMO:surfactant ratio of 9:1).24,25 Nevertheless, Tween-integrated cubosomes have not been so far investigated for brain delivery.
This study is the first one to elaborate novel brain-targeted GMO cubosomes with bioactive excipients for oral PIP delivery. The potential of the cubosomes to improve oral PIP efficacy in Alzheimer’s therapy was investigated. Full in vitro characterization was carried out. Biochemical, behavioral, and toxicological appraisal was investigated in adult male Wistar rats sporadic dementia of Alzheimer’s type (SDAT) model.
Materials and methods
Materials
Piperine (molecular weight =285.34 Da, purity 98%) was purchased from Alpha Aesar (Ward Hill, MA, USA); Peceol® (GMO, hydrophilic-lipophilic balance (HLB) =3) was a kind gift from Gattefosse Co. (Lyon, France); Cremophor RH 40® (Polyoxy 40 hydrogenated castor oil, HLB =14–16) and Poloxamer 407 were obtained from BASF Co. (Ludwigshafen, Germany); Tween 80 (HLB =15) was obtained from El-Nasr Pharmaceutical Co. (Abu Zaabal, Egypt). Colchicine, donepezil hydrochloride, thiobarbituric acid (TBA), trichloracetic acid, nitroblue tetrazolium (NBT), acetylthiocholine iodide, dithiobisnitrobenzoic acid, Folin phenol reagent, protease inhibitor cocktail, and reagents of in vivo study were obtained from Sigma-Aldrich (St Louis, MO, USA). Caspase-3 colorimetric assay kit and tumor necrosis factor-α (TNF-α) Immunoassay kit were purchased from R&D Systems Inc., Wiesbaden-Nordenstadt, Germany. alanine aminotransferase (ALT) and aspartate aminotransferase (AST) assay kits were purchased from Spectrum (Hannover, Germany). Urea assay kit was purchased from Diamond Diagnostics (Hannover, Germany). Creatinine assay kit was purchased from Spectrum. All other chemicals and reagents used were of analytical grade.
Preparation of cubosomal formulations
The preparation of cubosomal dispersions followed the procedures described by Gustafsson et al26 with slight modifications. GMO and poloxamer 407 were molten at 70°C in a hot water bath until homogeneous mixture was formed. The molten mixture was then added dropwise to water at 70°C, and the prepared coarse dispersions were left standing at room temperature for 48 hours to attain equilibrium. The prepared dispersions were then subjected to homogenization at 10,000 rpm for 2 hours.
Optimization and drug loading
Prepared cubs were optimized for stabilizer concentration and type. Different ratios of poloxamer (0.4%, 0.5%, and 0.6% w/w) were added and physicochemical properties were investigated. Each sample was prepared in triplicate and the dispersions were stored at room temperature and kept away from light for 1 week. During this week, the stability of the prepared dispersions was assessed. Visual inspection of color, homogeneity, the presence of macroscopic particles, and deposition of material on the glass surface of the vial at the air-dispersion-glass contact line (referred to as “ring formation”) was carried out. The dispersions showing poor stability were excluded and the optimum concentration of stabilizer needed for the preparation of stable cubs was determined.
After the determination of the optimum concentration of stabilizer needed for the preparation of stable cubs, integration of different types of bioactive surfactants (Tween 80 and Cremophor RH 40) was investigated using the optimum ratio either alone or in combination with poloxamer 407.
Different formulations were prepared and assessed for physicochemical stability by visual inspection, measurement of particle size (PS) and polydispersity index (PDI), and visualization under cross-polarizing microscope. All formulations were prepared in triplicates. Drug-loaded samples were prepared using the same procedure except that PIP was dissolved with GMO and poloxamer 407 by magnetic stirring before addition to water.
Physicochemical characterization of cubs
PS, zeta potential, and PDI
PS, PDI, and zeta potential (ZP) were determined by dynamic light scattering technique using Zetasizer Nano ZS (Malvern Instruments Ltd., Malvern, UK). Samples were diluted with water and the dilution factor was 1:100. Samples were sonicated for 10 minutes before measurement. All samples were measured in triplicates and results were represented as mean value ± standard deviation.
Determination of entrapment efficiency
The entrapment efficiency (%EE) of the prepared cubs was determined by ultrafiltration27 using Vivaspin ultra filter tubes (Vivaspin® 6 Centrifugal Concentrator, molecular weight cutoff 100,000; Vivaproducts, Inc., Littleton, MA, USA).
Cubosomal dispersion loaded with drug (3 mL) was placed in the centrifuge tube and subjected to centrifugation against placebo cubs at 3,000 rpm for 15 minutes at 4°C (Cooling centrifuge, Sigma 3-30K, Sigma-Aldrich). The separated supernatant was used for determination of un-entrapped drug by measuring absorbance at 342 nm using UV-VIS spectrophotometer (UV-160A; Shimadzu, Kyoto, Japan).
The entrapped drug was calculated by the following equation:
|
In vitro drug release
The in vitro release profile of PIP from cubs was determined using dialysis bag method16 (VISKING® dialysis tubing molecular weight cutoff 12,000–14,000). Samples investigated encompassed PIP-loaded cubs (poloxamer stabilized); Tween-integrated cubosomes (T-cubs), and surfactant mixture cubosomes (S mix cubs) in a PIP concentration of 0.5 mg/mL. Release from cubs formulations was compared with drug solution (0.5 mg/mL) and placebo cubs. Samples were filled into dialysis tubes that were immersed in 140 mL of phosphate buffer (pH 7.4), and then placed in shaking water bath at 37°C and 50 rpm. At predetermined time intervals, samples (2 mL) were withdrawn and replaced with the same volume of phosphate buffer. The samples were analyzed in triplicates using UV spectrophotometer at maximum wavelength 342 nm, against the placebo samples.
Structural elucidation
To identify the nature of the formed liquid crystals either isotropic or anisotropic, a drop of the coarse cubosomal dispersion was placed on a glass slide and examined under the cross-polarizing microscope (Model CX 31, Olympus Corporation, Tokyo, Japan).19,28
Transmission electron microscopy
The morphology of the prepared cubs was determined by transmission electron microscopy (TEM). Cubs were diluted with water and the dilution factor was 1:50.
A drop of the diluted dispersion was placed on a paraffin sheet and carbon-coated grid was placed on the sample and left for 1 minute to allow the adherence of cubs on the carbon substrate. The remaining suspension was removed by adsorbing the drop with the corner of a piece of filter paper. Samples were stained with uranyl acetate for 30 seconds before microscopic investigation.
Short-term stability testing
The prepared cubosomal dispersions were kept at room temperature away from light for a period of 3 months and were analyzed periodically by measuring mean PS, PDI, ZP, and %EE. Samples were measured in triplicates and the results obtained were the mean of three measurements.
In vivo study
Animals and dose administration
Adult male Wistar rats (180–220 g, 8 weeks old, a total of 42) were included in the current study because of their advantage in spatial learning.29 Upon arrival, they were housed in standard metal cages at 21°C±1°C and 65% relative humidity with a 10-hour light/14-hour dark cycle. In addition, they were given standard chow and water ad libitum for the duration of the study and they were given 2 weeks to adapt to their environment before the experiment. The animals were kept at the animal house of the Alexandria faculty of medicine, Egypt. Alexandria University ethical committee approval was obtained and animals were treated according to the ethical guidelines of Alexandria University. Also, attempts have been made to minimize animal suffering in accordance with the international principles for laboratory use and care of the European Community (EEC Directive of 1986; 86/609/EEC). SDAT was induced in rats by a single dose of intracerebroventricular (ICV) injection of colchicine.30 Rats were anesthetized with sodium pentobarbital (45 mg/kg, intraperitoneally). Each rat then received 15 μg of colchicine dissolved in artificial cerebrospinal fluid (ACSF), 7.5 μg in 10 μL/site, (ACSF: 147 mM NaCl, 2.9 mM KCl, 1.6 mM MgCl2, 1.7 mM CaCl2, and 2.2 mM dextrose). The injection was 0.8 mM posterior to bregma, 1.8 mM lateral to sagittal suture, and 3.6 mM beneath the cortical surface using Hamilton microsyringe. To promote diffusion, the microsyringe was left in place for a period of 2 minutes after injection. After ICV injection, all rats received gentamicin (5 mg/kg, intraperitoneally) to prevent sepsis. The same procedures were done in the control group with ICV injection of ACSF (10 μL/site). Special care was taken during the postoperative period and free access to food and water.30,31
The rats were randomly assigned into seven groups (six animals per group): Negative control (ACSF injected), positive control (untreated-colchicine injected), blank cubs – colchicine injected, PIP-cubs – colchicine injected (2.5 mg equivalent/kg [0.2 mL]), PIP-T-cubs – colchicine injected (2.5 mg equivalent/kg [0.2 mL]), PIP-S mix cubs – colchicine injected (2.5 mg equivalent/kg [0.2 mL]), PIP suspension – colchicine injected (2.5 mg equivalent/kg [0.2 mL]).
Care was taken to conduct each experiment at the same period of the day to avoid deviations in animal’s behavior. After 3 days of surgery, experimental sessions were started and continued routinely until the animals were sacrificed. Alzheimer-treating drugs were given as a single oral daily dose for 21 days. The learning and memory behavior was assessed using passive avoidance test on days 20 and 21 of the experiment.
Behavioral passive avoidance test
Impaired memory in SDAT model was evaluated through passive avoidance apparatus. The apparatus consists of two equal sized light and dark compartments (30×20×30 cm). A 40 W lamp was fixed 30 cm above the apparatus floor in the center of the light compartment. The floor of the dark compartment consisted of a metal grid connected to shock scrambler. The two compartments were separated by a trap door that could be raised up to 10 cm.32
On the 20th day after colchicine injection, training trial was done. In the pre-acquisition trial, rats were allowed to explore the light compartment of the apparatus. Thirty seconds later, the door between the two compartments was opened where the animal was allowed to explore the dark compartment. After 15 minutes, the acquisition trial was started by placing the rats in the light compartment of the apparatus. After 30 seconds of exploration, the door between the compartments was opened. Training latency was measured in seconds and represented the time needed by each rat to enter the dark compartment with all four paws inside. Immediately after the rat entered the dark chamber with all the four paws inside, the trap door was closed and an electric foot shock (0.8 mA) was delivered for 3 seconds. The rat was removed from the dark chamber after 5 seconds and returned to its cage. In order to avoid any confounding olfactory cues, light and dark compartments were well cleaned between each training session. A retention trial was performed 24 hours after the acquisition trial without any foot shock. Retention latency was recorded as a measure of the memory to painful stimuli. It represented the time needed by the animal to move from light to dark compartment with its four paws in. If the animal did not enter into the dark compartment within 300 seconds, then it was returned to its cage, and a maximum latency of 300 seconds was recorded.
Biochemical tests
The animals were sacrificed by decapitation 24 hours after the last behavioral test, ie, on day 22 of the experiment. Brains were removed, washed with ice-cold saline, and hippocampi were dissected. Next, hippocampal samples were homogenized with ten times (w/v) ice-cold phosphate-buffered saline solution (pH 7.2) containing a protease inhibitor cocktail. The homogenates were centrifuged at 10,000× g for 15 minutes at 4°C and aliquots of the supernatant were separated for measurement of the parameters of oxidative stress (OS) ([ie, malondialdehyde (MDA), total antioxidant capacity [TAC], and superoxide dismutase activity [SOD]) and acetylcholine esterase (AChE) activity. Besides, hippocampal caspase-3 activity and TNF-α level were assayed as indicators for brain toxicity. Moreover, blood samples were taken and allowed to clot. Next, blood samples were centrifuged at 3,000× g for 15 minutes. The recovered serum was stored at -20°C for determination of liver functions (ALT and AST) and kidney functions (urea, creatinine).
Oxidative stress parameters
Malondialdehyde
The quantitative measurement of lipid peroxidation marker, MDA, in brain was performed using colorimetric assay according to the method of Wills33 using TBA reagent. Five hundred microliters of hippocampal homogenate supernatant was well mixed with 1 mL trichloracetic acid. Next, the mixture was centrifuged at 3,000 rpm for 10 minutes. In all, 1 mL of the latter mixture was then added to 0.5 mL of TBA and boiled for 10 minutes, then cooled. The absorbance of samples was read at 532 nm against blank. The results were normalized to tissue weight to be expressed as ng/g tissue.
Total antioxidant capacity
TAC was determined using colorimetric assay according to the method of Koracevic et al.34 This method is used to measure the reductive power of a sample. It is based on suppression of the production of TBA reactive substances in the presence of antioxidants. The reactive mixture contained 0.5 mL of Na-Benzoate (10 mMol/L), 0.2 mL of H2O2 (10 mMol/L), 0.49 mL of phosphate buffer (100 mMol/L, pH=7.4), and 0.2 mL of Fe-EDTA complex (2 mMol/L). In all, 10 μL of the supernatant was added to the latter reactive mixture and was incubated at 37°C for 60 minutes. Finally, 1 mL glacial acetic acid (20 mMol/L) and 1 mL TBA (0.8% w/v in 100 mL of 50 mMol/L NaOH) were added and the absorbance was measured spectrophotometrically at 532 nm after incubation at 100°C for 10 minutes. The results were normalized to total tissue proteins to be expressed as mM/mg protein.
Superoxide dismutase activity
Superoxide dismutase activity was determined using colorimetric assay according to the method of Kono.35 This assay relies on the ability of the enzyme to inhibit the phenazine methosulfate-mediated reduction of NBT dye. For SOD activity assay, a mixture of 0.1 mM EDTA, 50 mM sodium carbonate, and 96 mM of NBT was prepared. In the cuvette, 2 mL of previous solution was mixed with 0.05 mL of the hippocampal supernatant and 0.05 mL of hydroxylamine hydrochloride (adjusted to pH 6.0 with NaOH). The auto-oxidation of hydroxylamine was detected by measuring the change in optical density at 560 nm for 2 minutes at 30/60 second intervals. SOD activity was normalized to tissue weight to be expressed as U/g tissue.
Acetyl cholinesterase activity
As AChE activity is a marker of the extensive cholinergic loss in the brain, it was measured in the hippocampus according to the method of Ellman et al.36 The hippocampal supernatant was pre-incubated with 1 mL of the assay solution (100 mM phosphate buffer, pH 8, 75 mM acetylthiocholine iodide) for 15 minutes at 37°C. Next, 0.32 mM dithiobisnitrobenzoic acid was added as a second substrate to initiate the reaction. The increase in absorbance at 412 nm was recorded for 5 minutes at 37°C at 5 second intervals, and the results were normalized to total tissue proteins to be expressed as moles of substrate hydrolyzed/min/mg protein.
Clinical observations and toxicity studies
As a part of toxicity assessment, animals’ body weight was measured at the start of the experiment and once weekly from there on using an electronic balance (Precisa Instrument, Dietikon, Switzerland). The amounts of food (g/day) and water (mL/day) consumption were also determined. Food and water consumption during 24 hours was measured twice weekly and the mean of them used for that week. Additionally, clinical signs of toxicity and mortalities were continuously checked during the first 24 hours of treatment and daily afterwards. The animals that died before the end of study have been excluded from the experiment.
After the animals were sacrificed, controls and treated animals’ organs (liver, kidneys, spleen, brain, etc) were subjected to gross examination (macroscopic analysis) to check for any significant change in texture and shape.
Serum ALT activity
Serum level of ALT activity was measured by spectrophotometric method using Spectrum assay kit. ALT catalyses the transfer of an amino group from l-alanine to 2-oxoglutarate to form pyruvate and l-glutamate. The pyruvate concentration is determined spectrophotometrically at 546 nm in the form of hydrazine formed with 2,4-dinitrophenylhydrazine in an alkaline medium. Enzyme activity was determined from pyruvate standard calibration curve.37
Serum AST activity
Serum level of AST activity was measured by spectrophotometric method using Spectrum assay kit. AST catalyses the transfer of an amino group from L-aspartate to 2-oxoglutarate to form oxaloacetate and L-glutamate. The oxaloacetate concentration is determined spectrophotometrically at 546 nm in the form of hydrazone formed with 2,4-dinitrophenylhydrazine in an alkaline medium. Enzyme activity was determined from pyruvate standard calibration curve.37
Serum urea
Serum level of urea was measured by enzymatic colorimetric method using Diamond Diagnostics assay kit. Urea in sample is hydrolyzed enzymatically into ammonia and carbon dioxide. The formed ammonia ion reacts with salicylate and hypochlorite in the presence of the catalyst nitroprusside to form a green indophenol. The intensity of the color formed is measured spectrophotometrically at 578 nm.38
Serum creatinine
Serum level of creatinine was measured by buffered kinetic Jaffe reaction using Spectrum assay kit. Creatinine reacts with picric acid under alkaline conditions to form a yellow–red complex. The intensity of the color formed is measured spectrophotometrically at 492 nm.39
Brain toxicity study
Brain caspase-3 assay
Caspase-3 is an intracellular cysteine protease pro-enzyme. It is activated during the cascade of apoptosis. Hippocampal caspase-3 enzymatic activity was measured by colorimetric reaction provided by R&D Systems. Caspase activity was determined by the addition of a caspase-specific peptide that is conjugated to the color reporter molecule p-nitroaniline. Chromophore p-nitroaniline is released after cleavage of the peptide by the caspase. It can then be quantitated spectrophotometrically at a wavelength of 405 nm. The level of caspase enzymatic activity in the tissue lysate is directly proportional to the color intensity. Caspase-3 activity was expressed as a percent of the baseline (control) levels.40
Brain TNF-α ELISA
The quantification of TNF-α was done with the help and instructions provided by R&D Systems Quantikine rat TNF-α immunoassay kit. The assay is based on sandwich enzyme immunoassay technique. A rat TNF-α-specific monoclonal antibody has been precoated in the microplate. Rat-TNF-α present in standards or samples is captured by the immobilized antibody. After removing unbound substances by washing, a rat TNF-α-specific enzyme-linked polyclonal antibody is added to the wells. After a second wash, an enzyme-specific substrate solution is added. The enzyme reaction yields a blue colored product that turns yellow when the stop solution is added. The intensity of the color is proportional to the amount of the bound rat TNF-α and values are interpreted from TNF-α standard curve.41
Estimation of total protein
Aliquots of hippocampal homogenate supernatant were analyzed in duplicate for total protein by the Lowry’s method using Folin phenol reagent with bovine serum albumin as a standard.42
Statistical analysis
Data of the in vivo study were computerized and analyzed using IBM SPSS software package version 20. The data were described using mean and standard deviation. The distributions of quantitative variables were tested for normality using Kolmogorov–Smirnov test, Shapiro–Wilk test, and D’Agstino test, also histogram and QQ plot. As the data were normally distributed, comparison between the studied groups was done using F-test (ANOVA) and post hoc test (Scheffe) for pair-wise comparisons. Significance of the obtained results was judged at the 5% level.43,44 P=0.05 means that the chance that the null hypothesis (numbers are not statistically significant) is true is only 5%. P=0.001 means that the chance that the null hypothesis (numbers are not statistically significant) is true is only 0.1%.
Results and discussion
Optimization of cubs
Self-assembly of GMO-based LCN prerequisites the presence of steric stabilizer to stabilize the formed crystals in excess water. The gold standard steric stabilizer for the preparation of LCN is poloxamer 407.24,45 Poloxamer 407 is composed of polyethylene oxide (PEO)-polypropylene oxide-PEO block copolymer.
The role of this triblock co-polymer in stabilizing the formed LCN can be explained by understanding its structure. Poloxamer 407 contains both hydrophilic (PEO) part and hydrophobic (polypropylene oxide) part; the hydrophobic part is proposed to interact with the lipid bilayer, while the hydrophilic part will face the aqueous portion. The stabilizing effect of poloxamer is a result of the balance between hydrophilic and hydrophobic parts.46 Optimization of stabilizer concentration and type was carried out based on physicochemical properties (PS, PDI and ZP) and visual observations of instability signs for 1 week.
Impact of stabilizer concentration
The concentration of poloxamer 407 is critical for formation of stable cubs. In the current work, different concentrations of poloxamer 407 were investigated (0.4%, 0.5%, and 0.6% w/w, Table 1). The choice of the proper ratio was based on visual inspection of the prepared dispersion for aggregation or precipitation. Results inferred that all ratios of the stabilizer showed uniform opaque white mixtures with no visible signs of aggregates at the time of preparation.
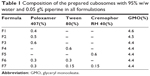  | Table 1 Composition of the prepared cubosomes with 95% w/w water and 0.05 g% piperine in all formulations |
Nevertheless, upon standing, the formula containing 0.4% poloxamer 407 (F1, Table 1) started to show aggregations. Visible microparticles were observed by the third day and the ring formation was clear by the end of the week indicating that the amount of poloxamer was not enough for steric stabilization of cubs. Formulations containing 0.5% poloxamer (F2) and 0.6% poloxamer (F3) showed excellent stability at room temperature. Among the three formulations, F3 exhibited the lowest PS, PDI and sufficient negative ZP (Table 2). Consequently, formula F3 was chosen for further studies.
Impact of stabilizer type
As bioactive surfactants, Tween 80 and Cremophor RH 40 were investigated for their ability to act as stabilizers for cubs. Rational choice of these surfactants was based on their required bioactivity to protect PIP from premature metabolism by liver microsomal enzymes. Furthermore, reported brain-targeting potential of Tween 80 would be exploited to deliver PIP to its target organ. Besides, these polyoxyethylated non-ionic surfactants do have structural properties close to that of poloxamer 407. Both have HLB of 15 and possess the balance between hydrophilic and hydrophobic parts required for cubosomal stabilization. Both of them contain PEO as a hydrophilic moiety and the hydrophobic part stearate for Cremophor RH 40 and monooleate for Tween 80.
Cremophor RH 40 was previously proposed as a stabilizer for cubs but the concentration used was as high as 50% of GMO concentration.47 Cremophor RH 40 was also reported to have the ability to stabilize LCN at a concentration of 0.5% w/w.46 On the other hand, Tween 80 has not been investigated so far for stabilization of GMO-based cubs.
The dispersions prepared by 0.6% Tween 80 (F4) and 0.6% Cremophor RH 40 (F5, Table 1) were left standing at room temperature and visually inspected to detect stability. On preparation, the dispersions were milky white with no visible aggregates but their mean PS and PDI were significantly larger than that seen with poloxamer 407-stabilized cubs. These results suggested that the use of surfactant mixture to combine the excellent stabilizing effect of poloxamer 407 with the bioactivity of Tween 80 and Cremophor RH 40 as PS is a critical factor to ensure a better brain delivery.48
Two surfactant mixtures were assessed; Tween 80 (0.3%) with poloxamer 407 (0.3%) in (F6) formulation referred as T-cubs, and Tween 80 (0.15%)/Cremophor RH 40 (0.15%)/poloxamer 407 (0.3%) in (F7) formulation referred to as S mix cubs.
Both mixtures showed good stability at room temperature; no aggregates were formed and no ring formation was noted; the prepared dispersions remained uniform milky white. The results of mean PS and PDI were suitable for brain delivery for both formulations (F6 and F7).
Physicochemical characterization of cubs
PS, PDI, and ZP
PS, PDI, and ZP of the prepared formulations are depicted in Table 2. Generally, the PS of the prepared cubs should be lower than 200 nm to be able to cross the blood–brain barrier (BBB) in acceptable concentration. Suitable PDI values are below 0.3 and a ZP value of ±30 mv, which is considered as an approximate stability threshold.
In this work, the results of PS and ZP came in accordance with that of visual inspection. The optimized formulations F2, F3, F6, and F7 showed a PS less than 200 nm, they had a low PDI, and their ZP values indicated a formation of stable dispersions.
On the other hand, F1 prepared with a low stabilizer concentration showed large PS and low ZP indicating instability. F5 containing only Cremophor RH 40 as a stabilizer showed higher PS (245.30 nm) and a PDI of 0.41. The ZP value was -21.29 mv confirming the potential instability of the cubosomal dispersion, which formed aggregates (ring formation) upon standing. In a consequence, this work suggested that Cremophor RH 40 cannot withstand alone as a stabilizer for cubs in a concentration of 0.6% w/w.
Determination of entrapment efficiency
The %EE was calculated for the optimized formulation F3, F6, and F7 and the results showed good entrapment of PIP. Cubs (F3) showed 89.38%±1.24% entrapment, T-cubs (F6) showed 86.67%±0.62% entrapment, and %EE for S mix cubs was 85.13%±0.43%. The decrease in %EE between different formulations may be explained by the increased solubility of PIP in aqueous solution when Tween 80 and Cremophor RH 40 were used as stabilizers.
In vitro drug release
As shown in Figure 1, the release of PIP for all cubs formulations followed a biphasic pattern with initial burst release of approximately 21% of the drug released in the first hour then the drug release was sustained with only 45% released after 6 hours. This is compared with nearly 80% release from the PIP suspension in the first 2 hours.
The initial burst release may be attributed to the drug weakly adsorbed on the surface of cubs, then the drug release became relatively slow as the drug encapsulated within the structure of cubs was released. PIP as a hydrophobic drug has a strong interaction with the hydrophobic part of LCN leading to the sustained release. These results were in accordance with that obtained by Zeng et al49 who stated that the release pattern of cubs was also biphasic with initial burst release followed by sustained release.
Structural elucidation
Polarizing microscope was reported as a method to detect the presence of crystals in colloidal systems.22 Crystals are birefringence and have the ability to split the cross-polarized light.
Upon examination of different cubosomal coarse dispersions under the microscope, anisotropic nature of the formed crystals was confirmed. Cubs stabilized with Poloxamer 407 (Figure 2A), Tween 80 and Poloxamer 407 (Figure 2B), or the S mix (Figure 2C) showed crystals with uniform PS that were abundant in the mount. On the other hand, cubs stabilized with Cremophor RH 40 (Figure 2D) alone showed less crystals in the mount with the presence of visible aggregates. The latter result confirms unsuitability of Cremophor alone for stabilization of cubs in the concentration investigated (0.5%).
Transmission electron microscopy
The TEM images (Figure 3A–C) showed the cubic structure of the formed LCN. The size of the particles was between 100 and 200 nm as shown by dynamic light scattering technique with no visible aggregations.
Short-term stability testing
The prepared cubosomal dispersions were kept at room temperature away from light to check their shelf stability. The values of mean PS, PDI, ZP, and %EE are depicted in Table 3. There was an insignificant increase in PS of all tested formulations that did not affect %EE, indicating good stability of the nanocarriers.
In vivo study
In this study, hydrophobic nanocarriers were tested as an oral drug delivery system for PIP. LCN (cubs) were proposed as an anisotropic drug delivery system having various advantages. The goal of this study was to evaluate the role of bioactive excipients in improving PIP bioavailability and brain delivery. PIP is a drug that suffers hepatic first-pass effect leading to low oral bioavailability. Besides, PIP was intended for treating AD that requires brain delivery in sufficient concentrations.
Two bioactive excipients (Tween 80 and Cremophor RH 40) were tested. Both excipients are reported to have the ability to prevent premature liver metabolism13 with Tween 80 reported brain-targeting ability. The current in vivo animal study was used to evaluate the biological effect of bioactive excipients on PIP delivery and the potential toxicological effect of the prepared formulations.
Behavioral test
Passive avoidance test
The chosen PIP oral dose was based on the previous reported dose, pharmacokinetic profile, and toxicity studies. It was found that PIP at dosage range between 2.5 and 86 mg/kg produced effects on CNS such as anti-stress50 and anti-convulsion effects.51 Consequently, the dose of 2.5mg/kg was the minimal reported effective to be enrolled in the current study.
In the present study, cognitive function was evaluated by passive avoidance test where an animal learned to avoid a specific place associated with an unpleasant event. Increased retention latency reflected learning ability and adequate cognitive function. During the training session (day 1), there were no significant differences between any of the studied groups (F=1.369, P=0.227) (Table 4) as expected during training.
However, there was a significant difference between groups in the retention test (day 2) (F=189.177, P=<0.001). In the present study, ICV administration of colchicine impaired the cognition by decreasing the retention latencies in the passive avoidance test.
The effect of colchicine was partially reversed by treatment with free PIP (2.5 mg/kg/day) and PIP-loaded cubs (2.5 mg equivalent/kg/day) formulations (Table 4). This was shown by increased retention latencies (significant difference from positive control).
In comparison with free PIP, all lipid-based formulations produced significantly better effect on cognitive functions. Nevertheless, no formulation could restore cognition to base line except T-cubs (insignificant difference from negative control) (Table 4) showing superior effect over free drug and all other studied formulations.
On the other hand, blank cubs formulations had no significant effect on the retention latency (Table 4). Results obtained from behavioral test were further confirmed by biochemical analysis.
Biochemical tests
OS parameters
In the current study, centrally administered colchicine caused a significant increase in the oxidative damage as evidenced by increased MDA, and depleted TAC and SOD compared with the negative control group (Table 5).
However, free PIP (2.5 mg/kg/day) and PIP-loaded cub (2.5 mg equivalent/kg/day) formulations significantly attenuated levels of OS parameters compared with the positive controls (significant difference) (Table 5).
In comparison with free PIP, cubs formulations had a superior effect on the OS parameters (significant difference with free PIP). Compared with other formulations, T-cubs were again the only formulation comparable to the negative controls (insignificant difference) regarding its effect on MDA and TAC. On the other hand, blank cubs formulations had no significant effect on the OS parameters (Table 5).
In view of the above-mentioned literature and current study results, the antioxidant effect of PIP may be one of the important factors responsible for the beneficial effects in the used SDAT rat model. This is in accordance with the earlier studies reporting the OS lowering effects of PIP.8,52 53
AChE activity
In the current study, the development of AD was further confirmed by the significantly increased AChE activity leading to memory deficits. This activity was significantly decreased by only PIP-loaded cubs (2.5 mg equivalent/kg/day) formulations where free PIP did not show significant difference from the positive control (Table 5).
Compared with other formulations, T-cubs showed superior AChE activity suppressing effect (Table 5).
T-cubs formulation was the only one capable of decreasing the activity of AChE to a level that is insignificantly different from the negative control. On the other hand, blank cubs formulations had no significant effect on the AChE activity (Table 5).
Based on the aforementioned results (behavioral and biochemical analysis), it could be concluded that novel T-cubs formulation was superior over all other lipid-based formulations.
Generally cubs containing GMO are proposed to be suitable candidates for lymphatic absorption54 by-passing the hepatic first-pass effect.
Compared with other cubs, T-cubs may have a better ability to deliver PIP to the brain combining the effects of both Tween 80 (brain-targeting surfactant) and Poloxamer 407 (P-gp inhibitor55) present in sufficient concentrations.
Assessment of the potential nanoparticles toxicity
Clinical observation and organ functions tests
The applications of nanoparticles in technology, research, and medicine have gained increasing interest. The use of nanoparticles in the biomedical field might carry the risk of increasing access to tissues and organs and, consequently, potential toxic effects.
“Nanotoxicity” is a relatively new concept to science. It refers to the potential adverse effect of nanoparticles on the normal structure and function of animals’ organs and tissues. Nanoparticles toxicity is mostly related to the physiochemical characteristics of the nanoparticles such as PS, shape, surface charge, composition, and stability.
Oral nanoparticles administration has a more rapid systemic effect compared with the transdermal route. After entering the circulation, most substances are subjected to first-pass metabolism within the liver where they may accumulate or distribute to the end organs.
In a trial to assess the potential toxic effect of the studied formulations, clinical signs of toxicity were monitored during the study period. Physical observation showed no signs of toxicity (eg, skin, fur, eyes, mucus membrane changes, tremors, sleep, and coma) within the treated animals. After animal sacrificing, no organomegaly or other gross organ changes were observed.
Besides, animals’ nutritional state was assessed. It revealed gradual increases in body weight of the control, free-PIP, and cubs formulations-taking animals. At the end of the study, the percentages increase in body weight of treated rats were 14.18%±1.65%, 15.10%±2.97%, 16.10%±1.51%, and 14.11%±1.03% for free PIP, blank cubs, T-cubs, and S mix cubs formulations, respectively (Table 6).
The progressive increase in body weights of the control, free PIP, and cubs formulations treated rats during the study period highlights the improved nutritional state of the animals. Such improved nutritional state could be due to increased food and water intake, reflecting good health and denying incidence of toxicity.
To further confirm or exclude their potential systemic toxicity, biochemical parameters of liver and kidney functions were assessed in the studied groups.
Hepatotoxicity refers to liver dysfunction or liver damage that is associated with an overload of drugs or xenobiotics.56 Liver injury can be diagnosed by certain biochemical markers like ALT, AST, alkaline phosphatase, and bilirubin. Elevations in serum enzyme levels are taken as the relevant indicators of liver toxicity.57
The current results showed no significant hepatotoxic effect of the studied formulations as shown by the insignificant difference of ALT and AST between the control and treated groups (Table 7).
On the other hand, kidney is one of the main sites of xenobiotic-induced toxicity, which represents an important cause of renal failure.58 Such toxicity is partly due to the high renal blood flow rate, which leads to delivery of high concentrations of xenobiotics to the kidneys.59
Regarding kidney functions, serum urea and creatinine, the traditional kidney function tests, were assessed. Both blank and PIP-loaded cubs formulations treated groups showed no nephrotoxic effect indicating safety of the prepared formulations (Table 7). The abovementioned results denied any liver or kidney toxicity of the prepared cubs.
Brain toxicity study
Brain is a vital organ with limited regenerative capacity. Consequently, it must be protected from exogenous injurious agents. This is achieved by the BBB that separates the Cerebrospinal fluid (CSF) surrounding the brain from the systemic circulation via tight junctions around the capillaries.60
Targeted drug delivery carries the advantage of the use of smaller drug doses with consequent reduced systemic adverse effects. On the other hand, it might carry the risk of organ toxicity.60 In context to brain targeting, an insult to BBB might elicit OS-mediated neural injury, inflammatory reaction, and consequently apoptosis.61,62 In the current study, we tried to assess or exclude the possible neurotoxic effect of the used formulations. As a result, the degree of neural apoptosis was assessed through caspase-3 assay and the level of neural inflammatory reaction was assessed by measuring TNF-α level in the hippocampus. The present results report no brain toxic effect to any of the studied formulations. In contrast, some of the used formulations showed significant anti-apoptotic and anti-inflammatory effects (Table 8).
In the current study, ICV injection of colchicine induced apoptosis and inflammatory reaction as shown by the significantly higher caspase-3 and TNF-α value of the positive control compared with the negative control. Previous studies reported that ICV colchicine caused apoptosis through activation of caspase-3 pathway.63 Moreover, colchicine could elicit a direct inflammatory response in the CNS.64
An interesting finding was the improvement of these colchicine-induced brain effects by the treatment with cubs. Besides, free-PIP showed an inferior effect on apoptosis and inflammation suppression than the cub formulations used. Further, the current study reports a superior effect of T-cubs formulations over all other cubs (Table 8). Finally, blank cubs showed no significant effect on the apoptosis and inflammation (Table 8).
To the best of our knowledge, the present study is the first to reveal apoptosis and inflammation suppression as other possible mechanisms for the neuroprotective effect of PIP in colchicine induced SDAT model as confirmed in this article and Elnaggar et al.65
Conclusion
This work aimed at formulation of Tween-stabilized cubs as brain-targeted non-invasive PIP delivery system for treatment of AD. The elaborated T-cubs had suitable PS and PDI for brain delivery. PIP-loaded cubs had superior effect over free drug and were able to restore the cognitive functions of the treated animals. Results of toxicological studies highlighted the safety of the prepared formulations on liver and kidneys. Cubosomal formulations were safe on the brain cells and T-cubs revealed the ability of PIP to act as anti-inflammatory and anti-apoptotic agent.
Disclosure
The authors report no conflicts of interest in this work.
References
Brambilla D, Le Droumaguet B, Nicolas J, et al. Nanotechnologies for Alzheimer’s disease: diagnosis, therapy, and safety issues. Nanomedicine. 2011;7(5):521–540. | ||
Querfurth HW, LaFerla FM. Alzheimer’s disease. N Engl J Med. 2010;362(4):329–344. | ||
Prince M, Bryce R, Albanese E, Wimo A, Ribeiro W, Ferri CP. The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement. 2013;9(1):63–75.e2. | ||
Sahni JK, Doggui S, Ali J, Baboota S, Dao L, Ramassamy C. Neurotherapeutic applications of nanoparticles in Alzheimer’s disease. J Control Release. 2011;152(2):208–231. | ||
Bullock R, Dengiz A. Cognitive performance in patients with Alzheimer’s disease receiving cholinesterase inhibitors for up to 5 years. Int J Clin Pract. 2005;59(7):817–822. | ||
Freag MS, Elnaggar YS, Abdallah OY. Lyophilized phytosomal nanocarriers as platforms for enhanced diosmin delivery: optimization and ex vivo permeation. Int J Nanomedicine. 2013;8:2385–2397. | ||
Peng S, Hung WL, Peng YS, Chu IM. Oligoalanine-modified Pluronic-F127 nanocarriers for the delivery of curcumin with enhanced entrapment efficiency. J Biomater Sci Polym Ed. 2014;25(12):1225–1239. | ||
Chonpathompikunlert P, Wattanathorn J, Muchimapura S. Piperine, the main alkaloid of Thai black pepper, protects against neurodegeneration and cognitive impairment in animal model of cognitive deficit like condition of Alzheimer’s disease. Food Chem Toxicol. 2010;48(3): 798–802. | ||
Wu Z-J, Xia X-J, Huang X-S. Determination of equilibrium solubility and apparent oil/water partition coefficient of piperine. J Jinan Uni Natl Sci Med. 2012;5(33):473–476. | ||
Sahu PK, Sharma A, Rayees S, et al. Pharmacokinetic study of piperine in Wistar rats after oral and intravenous administration. Int J Drug Deliv. 2014;6(1):82–88. | ||
Hashimoto K, Yaoi T, Koshiba H, et al. Photochemical isomerization of piperine, a pungent constituent in pepper. Food Sci Technol Int. 1996;2(1):24–29. | ||
Prabhakar K, Afzal SM, Surender G, Kishan V. Tween 80 containing lipid nanoemulsions for delivery of indinavir to brain. Acta Pharma Sinica B. 2013;3(5):345–353. | ||
Rao Z, Si L, Guan Y, Pan H, Qiu J, Li G. Inhibitive effect of cremophor RH40 or tween 80-based self-microemulsiflying drug delivery system on cytochrome P450 3A enzymes in murine hepatocytes. J Huazhong Univ Sci Technolog Med Sci. 2010;30(5):562–568. | ||
Elnaggar YS, El-Massik MA, Abdallah OY. Self-nanoemulsifying drug delivery systems of tamoxifen citrate: design and optimization. Int J Pharm. 2009;380(1–2):133–141. | ||
Elsheikh MA, Elnaggar YS, Gohar EY, Abdallah OY. Nanoemulsion liquid preconcentrates for raloxifene hydrochloride: optimization and in vivo appraisal. Int J Nanomedicine. 2012;7:3787–3802. | ||
Elnaggar YS, El-Massik MA, Abdallah OY. Sildenafil citrate nanoemulsion vs. self-nanoemulsifying delivery systems: rational development and transdermal permeation. Int J Nanotechnol. 2011;8(8): 749–763. | ||
Elnaggar YS, El-Massik MA, Abdallah OY. Fabrication, appraisal, and transdermal permeation of sildenafil citrate-loaded nanostructured lipid carriers versus solid lipid nanoparticles. Int J Nanomed. 2011;6:3195–3205. | ||
Elnaggar YS, El-Refaie WM, El-Massik MA, Abdallah OY. Lecithin-based nanostructured gels for skin delivery: an update on state of art and recent applications. J Control Release. 2014;180:10–24. | ||
El-Refaie WM, Elnaggar YS, El-Massik MA, Abdallah OY. Novel self-assembled, gel-core hyaluosomes for non-invasive management of osteoarthritis: in-vitro optimization, ex-vivo and in-vivo permeation. Pharm Res. 2015;32(9):2901–2911. | ||
Elsheikh MA, Elnaggar YS, Abdallah OY. Rationale employment of cell culture versus conventional techniques in pharmaceutical appraisal of nanocarriers. J Control Release. 2014;194:92–102. | ||
Freag MS, Elnaggar, Abdallah OY. Development of novel polymer-stabilized diosmin nanosuspensions: in vitro appraisal and ex vivo permeation. Int J Pharm. 2013;454(1):462–471. | ||
El-Refaie WM, Elnaggar YS, El-Massik MA, Abdallah OY. Novel curcumin-loaded gel-core hyaluosomes with promising burn-wound healing potential: development, in-vitro appraisal and in-vivo studies. Int J Pharm. 2015;486(1–2):88–98. | ||
Yang D, Armitage B, Marder SR. Cubic liquid-crystalline nanoparticles. Angew Chem Int Ed Engl. 2004;43(34):4402–4409. | ||
Worle G, Drechsler M, Koch MH, Siekmann B, Westesen K, Bunjes H. Influence of composition and preparation parameters on the properties of aqueous monoolein dispersions. Int J Pharm. 2007;329(1–2):150–157. | ||
Lai J, Lu Y, Yin Z, Hu F, Wu W. Pharmacokinetics and enhanced oral bioavailability in beagle dogs of cyclosporine A encapsulated in glyceryl monooleate/poloxamer 407 cubic nanoparticles. Int J Nanomedicine. 2010;5:13–23. | ||
Gustafsson J, Ljusberg-Wahren H, Almgren M, Larsson K. Submicron particles of reversed lipid phases in water stabilized by a nonionic amphiphilic polymer. Langmuir. 1997;13:6964–6971. | ||
Chung H, Kim J, Um JY, Kwon IC, Jeong SY. Self-assembled “nanocubicle” as a carrier for peroral insulin delivery. Diabetologia. 2002;45(3):448–451. | ||
Zeng N, Gao X, Hu Q, et al. Lipid-based liquid crystalline nanoparticles as oral drug delivery vehicles for poorly water-soluble drugs: cellular interaction and in vivo absorption. Int J Nanomedicine. 2012;7:3703–3718. | ||
D’Hooge R, De Deyn PP. Applications of the Morris water maze in the study of learning and memory. Brain Res Brain Res Rev. 2001;36(1):60–90. | ||
Ganguly R, Guha D. Alteration of brain monoamines & EEG wave pattern in rat model of Alzheimer’s disease and protection by Moringa oleifera. Indian J Med Res. 2008;128(6):744–751. | ||
Veerendra Kumar MH, Gupta YK. Intracerebroventricular administration of colchicine produces cognitive impairment associated with oxidative stress in rats. Pharmacol Biochem Behav. 2002;73(3):565–571. | ||
Monleon S, Urquiza A, Carmen Arenas M, Vinader-Caerols C, Parra A. Chronic administration of fluoxetine impairs inhibitory avoidance in male but not female mice. Behav Brain Res. 2002;136(2):483–488. | ||
Wills ED. Mechanisms of lipid peroxide formation in animal tissues. Biochem J. 1966;99(3):667–676. | ||
Koracevic D, Koracevic G, Djordjevic V, Andrejevic S, Cosic V. Method for the measurement of antioxidant activity in human fluids. J Clin Pathol. 2001;54(5):356–361. | ||
Kono Y. Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys. 1978;186(1):189–195. | ||
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7:88–95. | ||
Reitman S, Frankel S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol. 1957;28(1):56–63. | ||
Tabacco A, Meiattini F, Moda E, Tarli P. Simplified enzymic/colorimetric serum urea nitrogen determination. Clin Chem. 1979;25(2):336–337. | ||
Murray RL. Creatinine. In: Kaplan A, Peace AL, editors. Clinical Chemistry. St Louis. Toronto. Princeton: The C.V. Mosby Co; 1984:1261–1266. | ||
Casciola-Rosen L, Nicholson DW, Chong T, et al. Apopain/CPP32 cleaves proteins that are essential for cellular repair: a fundamental principle of apoptotic death. J Exp Med. 1996;183(5):1957–1964. | ||
Tiwari V, Chopra K. Resveratrol prevents alcohol-induced cognitive deficits and brain damage by blocking inflammatory signaling and cell death cascade in neonatal rat brain. J Neurochem. 2011;117(4):678–690. | ||
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265–275. | ||
Chan YH. Biostatistics 102: quantitative data – parametric and non-parametric tests. Singapore Med J. 2003;44(8):391–396. | ||
Kotz S, Balakrishnan N, Read CB, Vidakovic B, editors. Encyclopedia of Statistical Sciences. Vol 2. Hobojen (NJ): John Wiley & Sons; 2006. | ||
Nasr M, Ghorab MK, Abdelazem A. In vitro and in vivo evaluation of cubosomes containing 5-fluorouracil for liver targeting. Acta Pharma Sin B. 2015;5(1):79–88. | ||
Madheswaran T, Baskaran R, Yong CS, Yoo BK. Enhanced topical delivery of finasteride using glyceryl monooleate-based liquid crystalline nanoparticles stabilized by cremophor surfactants. AAPS Pharm Sci Tech. 2014;15(1):44–51. | ||
Baskaran R, Madheswaran T, Sundaramoorthy P, Kim HM, Yoo BK. Entrapment of curcumin into monoolein-based liquid crystalline nanoparticle dispersion for enhancement of stability and anticancer activity. Int J Nanomedicine. 2014;9:3119–3130. | ||
Kulkarni SA, Feng SS. Effects of particle size and surface modification on cellular uptake and biodistribution of polymeric nanoparticles for drug delivery. Pharm Res. 2013;30(10):2512–2522. | ||
Zeng N, Gao X, Hu Q, et al. Lipid-based liquid crystalline nanoparticles as oral drug delivery vehicles for poorly water-soluble drugs: cellular interaction and in vivo absorption. Int J Nanomedicine. 2012;7:3703–3718. | ||
Li S, Wang C, Wang M, Li W, Matsumoto K, Tang Y. Antidepressant like effects of piperine in chronic mild stress treated mice and its possible mechanisms. Life Sci. 2007;80(15):1373–1381. | ||
D’Hooge R, Pei YQ, Raes A, Lebrun P, van Bogaert PP, de Deyn PP. Anticonvulsant activity of piperine on seizures induced by excitatory amino acid receptor agonists. Arzneimittelforschung. 1996;46(6):557–560. | ||
Wattanathorn J, Chonpathompikunlert P, Muchimapura S, Priprem A, Tankamnerdthai O. Piperine, the potential functional food for mood and cognitive disorders. Food Chem Toxicol. 2008;46(9):3106–3110. | ||
Yusuf M, Khan M, Khan RA, Ahmed B. Preparation, characterization, in vivo and biochemical evaluation of brain targeted Piperine solid lipid nanoparticles in an experimentally induced Alzheimer’s disease model. J Drug Target. 2013;3(21):300–311. | ||
Vistisen B, Mu H, Hoy CE. Lymphatic recovery of exogenous oleic acid in rats on long chain or specific structured triacylglycerol diets. Lipids. 2006;41(9):827–834. | ||
Saxena V, Hussain MD. Poloxamer 407/TPGS mixed micelles for delivery of gambogic acid to breast and multidrug-resistant cancer. Int J Nanomedicine. 2012;7:713–721. | ||
Navarro VJ, Senior JR. Drug-related hepatotoxicity. N Engl J Med. 2006;354(7):731–739. | ||
Singh A, Bhat TK, Sharma OP. Clinical biochemistry of hepatotoxicity. J Clin Toxicol. 2011. | ||
Susan Emeigh H, Lewis BK. Assessing Renal Effects of Toxicants In Vivo, in Toxicology of the Kidney, 3rd ed. Informa Healthcare; 2004: 81–147. | ||
Rached E, Hoffmann D, Blumbach K, Weber K, Dekant W, Mally A. Evaluation of putative biomarkers of nephrotoxicity after exposure to ochratoxin a in vivo and in vitro. Toxicol Sci. 2008;103(2): 371–381. | ||
Yildirimer L, Thanh NT, Loizidou M, Seifalian AM. Toxicology and clinical potential of nanoparticles. Nano Today. 2011;6(6):585–607. | ||
Oberdorster G, Elder A, Rinderknecht A. Nanoparticles and the brain: cause for concern? J Nanosci Nanotechnol. 2009;9(8):4996–5007. | ||
Trickler WJ, Lantz SM, Murdock RC, et al. Silver nanoparticle induced blood-brain barrier inflammation and increased permeability in primary rat brain microvessel endothelial cells. Toxicol Sci. 2010;118(1): 160–170. | ||
Liu Z, Gastard M, Verina T, Bora S, Mouton PR, Koliatsos VE. Estrogens modulate experimentally induced apoptosis of granule cells in the adult hippocampus. J Comp Neurol. 2001;441(1):1–8. | ||
Kumar A, Seghal N, Padi SV, Naidu PS. Differential effects of cyclooxygenase inhibitors on intracerebroventricular colchicine-induced dysfunction and oxidative stress in rats. Eur J Pharmacol. 2006;551(1):58–66. | ||
Elnaggar YSR, Etman SM, Abdelmonsif DA, Abdallah OY. Optimization and in-vivo appraisal of piperine-loaded intranasal chitosan nanoparticles as brain-targeted dual acting therapy in Alzheimer’s disease. Journal of Pharmaceutical Sciences. Available: http://www.researchgate.net/publication/279446825_Optimization_and_In-vivo_Appraisal_of_Piperine-loaded_Intranasal_Chitosan_Nanoparticles_as_Brain-Targeted_Dual_Acting_Therapy_in_Alzheimer’s_disease. Accessed July 3, 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.