Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 16
New Insights into the Mutual Promotion of Rosacea, Anxiety, and Depression from Neuroendocrine Immune Aspects
Received 19 March 2023
Accepted for publication 22 May 2023
Published 29 May 2023 Volume 2023:16 Pages 1363—1371
DOI https://doi.org/10.2147/CCID.S413237
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
Xiaoting Yang, Mei Cai
Department of Dermatology and Venereology, the Second Affiliated Hospital of Kunming Medical University, Kunming, Yunnan, 650101, People’s Republic of China
Correspondence: Mei Cai, Department of Dermatology and Venereology, the Second Affiliated Hospital of Kunming Medical University, No. 374, Yunnan-Myanmar Avenue, Kunming, Yunnan, 650101, People’s Republic of China, Tel +86 13888532488, Email [email protected]
Abstract: Rosacea is a common chronic inflammatory skin disease with a complex etiology and undefined pathogenesis, and there is still a lack of targeted clinical treatment. Patients with rosacea are at a higher risk of anxiety and depression compared to the healthy population. Compared to skin conditions such as acne and psoriasis, rosacea has been much less studied in relation to multiple-etiology psychiatric disorders such as anxiety and depression. In contrast to the mainstream belief that the causal association between rosacea and psychiatric disorders is that rosacea increases the psychological burden of patients and thus triggers psychiatric disorders simply by altering their facial appearance, this review outlines the possible common mechanisms between rosacea and anxiety and depression disorders, starting from the pathophysiological mechanisms of transient receptor potential family cation channels, HPA axis, and Th1/Th17 cell polarization. It envisages the possibility of the neuroendocrine-immune interplay between rosacea and anxiety and depression, and new ideas on the complex causal relationship between rosacea and psychiatric disorders, offering more orientations to open up new therapeutic approaches for rosacea.
Keywords: rosacea, anxiety disorders, depressive disorders, neurological factors, endocrine factors, immunological factors
Introduction
Rosacea is a chronic, recurrent inflammatory skin disease that occurs in the middle of the face and mainly involves the blood vessels, nerves, and sebaceous units of the hair follicles.1 According to the latest international meta-analysis of epidemiological data across populations, the average global prevalence rate is 5.46%.2 The prevalence of rosacea in China has reached a maximum of 4.64% in the last decade.3 With increased economic income, improved living conditions, and mental stress, more people are concerned about this type of cosmetic skin disease and are actively seeking medical care, which is likely the main reason for the significantly increased number of consultations in recent years. The disease usually occurs in women between the ages of 20 and 50 and is more likely to occur during menopause, but it can also occur in children and the elderly.4,5 Clinical manifestations typically include paroxysmal flushing of the facial skin, persistent erythema or papules, pustules, and capillary dilation, with hyperplastic hypertrophy and ocular changes in some patients. The exact pathogenesis of rosacea is still not fully understood, but the clinical manifestations can be explained by multiple factors, including genetic susceptibility, impaired epidermal barrier, congenital and acquired immune system dysregulation, neuroinflammation, and neurovascular regulatory imbalance.6 The complex pathophysiology suggests that rosacea is not limited to the skin but may also be associated with various systemic diseases such as gastrointestinal, cardiovascular, psychiatric, and neurological disorders. To date, several epidemiological studies have also identified a possible link between rosacea and various co-morbidities, such as psychiatric disorders.7 In addition, depression and anxiety disorders are also more prevalent in women, with similar time periods of onset. The prevalence of anxiety disorders is highest in the 45–55 age group, whereas the prevalence of depression is generally increasing in women after perimenopause, in addition to being more prevalent during the reproductive years.8,9
Previous studies have demonstrated that rosacea can increase the risk of depression and anxiety, while psychological factors such as stress, anxiety, an immature personality with excessive shame and guilt, and social anxiety secondary to easy blushing can, in turn, exacerbate flushing and participate in a vicious cycle of rosacea progression.10,11 Rosacea seriously impacts patients’ appearance, normal social interaction, and life, and most patients have recurrent attacks over several years or decades, or even several times a year, which requires repeated treatment.1 However, the complex etiology of anxiety and depression and the skin as a neuroendocrine-immune trinity organ suggest that the interplay between rosacea and psychiatric disorders should not be driven solely by cosmetic stress factors.
Compared to studies of acne, psoriasis, eczema, and other disorders associated with anxiety and depression, rosacea has been relatively little studied in this regard. However, whether it is rosacea or other more extensively studied dermatological disorders, most of the relevant studies involving psychiatric disorders such as anxiety and depression have focused on risk factors in terms of morbidity and psychosocial perspectives, and no clear study has ever delved into the psychiatric co-morbidity of the skin in terms of pathophysiological mechanisms. Dermatologists are not sure who exactly is the initiating factor between skin disorders and mental disorder disorders, or what the specific mechanisms are in their mutual advancement. Therefore, this paper uses rosacea as an entry point to outline the possible common pathogenic pathways between anxiety and depression and rosacea in terms of neurological, endocrine, and immunological aspects, and to propose specific possible mechanisms by which they contribute to each other.
TRP in Neurological Factors
Studies have shown that sensory neuron density is significantly increased in rosacea lesions.12 Physical stimuli, such as temperature changes, and chemical stimuli, such as fragrances, can activate sensory nerves via transient receptor potential(TRP) family cation channels.13 TRP cation channels are activated, and vasoactive neuropeptides such as substance P(SP), calcitonin gene-related peptide(CGRP), and vasoactive intestinal peptide(VIP) are released, leading to rosacea neurogenic inflammation.14 SP and CGRP are classical neuropeptides that act directly on vascular endothelial cells and smooth muscle cells, resulting in vascular effects. SP increases vascular permeability, leading to subsequent plasma extravasation and edema.14,15 SP release increases intercellular adhesion molecules and vascular cell adhesion molecules on vascular epithelial cells and induces the release of vascular endothelial growth factor from mast cells, thereby promoting vascular neovascularization and inflammatory cell infiltration.16,17 CGRP is a powerful microvascular dilator involved in much of the neurogenic vasodilation with the recruitment of inflammatory cells.12,18 VIP enhances the presentation of Th17 cell differentiation, thereby shifting Th cells to Th17 responses.19
Extensive literature has confirmed that TRP channels, especially transient receptor potential ankyrin 1(TRPA1), as cation channels activated by active compounds, are the gatekeepers of cytokine signals that mediate skin inflammation and activate sensory nerves.14 TRPA1 can be activated by fragrances such as cinnamaldehyde and mustard oil, as well as by thermal stimulation. In mouse experiments, topical cinnamaldehyde was found to activate TRPA1-dependent signaling pathways to induce vasodilation, which may be associated with the flushing phenomenon in rosacea patients.20 TRPA1 also senses oxidants and supports the role of reactive oxygen species (ROS) in the progression of rosacea.21 In rat neurons, TRPA1 co-localizes with protease-activated receptor 2, which can be activated by proteases, thereby inducing inflammation in human skin.22 The antagonist or gene deletion of TRPA1 reduces the scratching behavior induced by intradermal injection of hydrogen peroxide in mice.23 Additionally, blocking TRPA1 or TRPA1 gene deletion was found to reduce depression and anxiety-like behavior in juvenile mice, and TRPA1 expressed in the central nervous system (CNS) was found to exert a stress control on the emergence of depression and anxiety-like symptoms in mice.24 Therefore, TRPA1 may participate in the occurrence of anxiety and depression disorder after the activation of predisposing factors in rose acne. More studies are needed to confirm the promoting effect and specific mechanism of TRPA1 in anxiety and depression-like behaviors in rose acne.
In addition to TRPA1, transient receptor potential vanilloid (TRPV)1–4 has also been shown to be involved in the pathogenesis of rosacea. Different subtypes of TRPV have different functions: TRPV1 plays a role in vascular regulation and nociception and is activated by capsaicin, heat, and inflammation; TRPV2 plays a role in innate immunity, nociception, inflammation, vascular regulation, and thermosensitivity; TRPV3 and TRPV4 have a role in thermal sensitivity,25–27 and TRPV 1 highly expresses in skin sensory nerves like TRPA 1.23 Whereas TRPV1 is an injurious cation channel that responds to high temperatures (>43°C) and acts as a sensor of pain and chemical stimuli, capsaicin is its natural agonist.28 In endothelial cells and smooth muscle cells, TRPV1-mediated calcium inward flow releases nitric oxide to induce vasodilation. Animal experiments have also demonstrated that genetic deletion or pharmacological inhibition of TRPV1 reduces histamine-induced scratching behavior in mice.23,28 In psychiatric disorders, TRPV1 has been shown to play a role in the regulation of anxiety and depression.29,30 Roet et al31 found that TRPV1 is abundantly expressed in astrocytes of the human medial frontal and cingulate gyrus and speculated that through these cells, neuromodulation of mood and other neuropsychiatric disorders may occur. Compared to wild-type mice, TRPV1-deficient mice exhibit less anxiety-related behavior, and anxiety relief is also achieved after TRPV1 inhibition in the medial prefrontal cortex.32 Immobility is an indicator of depressive behavior. Blocking, desensitizing, or inhibiting TRPV1 reduces immobility in the forced swim test, and enhancing TRPV1 activity promotes immobility. This result may be due to changes in 5-hydroxytryptamine (5-HT)1A, GABAA, and NMDA receptor expression, which can be prevented by antidepressant drugs.33 However, the mechanism by which TRPV1 aggregation regulates the whole process remains unclear, and TRPV1 is associated with other neurotransmitter systems associated with neuropsychiatric disorders, especially endogenous cannabinoids,34 which makes its neurobiological function more challenging to understand. Moreover, TRPV4 is likewise abundantly expressed in astrocytes. TRPV4-mediated calcium influx in astrocytes has been reported to contribute to neuronal activation and enhance concomitant vasodilation.35 Dexmedetomidine administration ameliorates anxiety-like behavior induced by cerebral hemorrhage in mice, and the mechanism may be related to attenuated neuronal activation regulated by the conversion of astrocytes from type A1 to type A2 through inhibition of TRPV4 channel opening.36
The HPA Axis in Endocrine Factors
The hypothalamus-pituitary-adrenal axis(HPA) plays a vital role in the course of rosacea. Traditional activation of the conventional HPA axis begins with the secretion of corticotrophin-releasing factor (CRF) from the hypothalamus, which stimulates the paraventricular nucleus (PVN) to produce corticotropin-releasing hormone (CRH). CRH reaches the pituitary gland and activates type 1 CRH receptors in the anterior pituitary gland, inducing the cleavage of proopiomelanocortin(POMC)into adrenocorticotropic hormone (ACTH), melanocyte-stimulating hormone (MSH), and endorphin (END). ACTH stimulates the secretion of glucocorticoids (GCs), mainly cortisol, from the adrenal cortex.37 GCs, as end products of the HPA axis, are necessary for skin homeostasis and can downregulate inflammation in response to stressors. Under physiological conditions, GCs levels are regulated by a negative feedback loop in the HPA axis, but chronic stress alters HPA activity, disrupts the negative feedback loop, and leads to glucocorticoid resistance.38 Numerous studies in recent years have highlighted the importance of skin involvement in corticogenesis, confirming the presence of the cutaneous HPA axis.39 The sebaceous glands are similar to the “brain of the skin”, providing an equivalent to the conventional HPA axis.40 Phan et al41 demonstrated the importance of the dynamic homeostatic role of keratinocytes (KCs)derived GCs and the whole skin equivalent of the HPA axis through a mouse model study.
Like many other inflammatory skin diseases, rosacea begins with the perception of external physical, chemical, and biological stimuli by the skin’s nervous and immune systems, such as ultraviolet radiation, which is the primary initial stressor triggering the skin’s inflammatory response.12 When UV light irradiates the skin, KCs produce hormone products similar to those produced during systemic stress events, such as CRH, POMC, END, ACTH, and MSH, promoting cortisol production and downregulating glucocorticoid receptors, advancing glucocorticoid resistance, especially ACTH which directly stimulates cortisol production in hair follicles.42 CRH, as a central coordinator of neuroendocrine-immune responses, in addition to participating in HPA axis activity, also promotes mast cell degranulation and increases vascular permeability, regulates interleukin (IL)-18 secretion by KCs and IL-6 and IL-8 production by sebaceous cells, and mediates MAPK and NF-kB pathways, leading to facial erythema and inflammation in rosacea patients.12 In addition, CRH may synergize with cortisol to increase the expression of Toll-like receptors (TLRs), and activation of TLR2 may lead to abnormally high levels of expression of antimicrobial peptides, which induce CXCL8 to promote leukocyte migration and angiogenesis. CXCL8 expression in KCs is thought to be a key event in neutrophil recruitment for pustule formation; CRH activates the cannabinoid and vanilloid pathways, advancing the disease progression of rosacea.43,44
A large body of previous evidence suggests that dysfunction of the HPA axis is closely associated with the development of anxiety and depression disorders. Both anxiety disorders and depressive disorders are associated with abnormalities in the stress response system, where inhibition of the subgenual prefrontal cortex and activation of the amygdala stimulates the autonomic sympathetic axis and the HPA axis when the organism is exposed to a physical or psychological stressor.45 Both anxiety disorders and depressive disorders are associated with abnormalities in the stress response system, where inhibition of the subgenual prefrontal cortex and activation of the amygdala stimulates the autonomic sympathetic axis and the HPA axis when the organism is exposed to a physical or psychological stressor.46 Under repeated uncontrollable stress, the HPA axis negative feedback mechanism is abolished, leading to increased cortisol, increased amygdala reactivity, and reduced hippocampus activity.47 While structural-functional changes in the hippocampus play a crucial role in the pathogenesis of anxiety and depression, and reduced functional connectivity of bidirectional connections between the subgenual prefrontal cortex and hypothalamus of the brain is observed in patients with depressive disorders. This cortex is responsive to cortisol, with significantly higher cortisol levels in patients than in healthy controls, suggesting that these changes in structural-functional connectivity may be related to dysregulation of the HPA axis.48,49 Meanwhile, high glucocorticoid levels may induce neurotoxicity and subsequent atrophy of the hippocampus.38 Results of previous studies also indicate that glucocorticoid resistance is closely related to the severity of anxiety-depressive symptoms.50 In addition, GCs upregulate CRH in the amygdala, frontal cortex, and bed nucleus of the stria terminalis, while downregulating CRH in the hypothalamus, leading to differential expression of cortisol and CRH and consequently to different anxiety symptoms.51 CRH has been shown to be associated with the encoding of anxiety and emotional memory,52,53 emphasizing the critical role of CRH in the stress response, while neuronal circuits of CRH in the central amygdala can interact with the 5-HT and noradrenergic systems, both of which are closely associated with anxiety and depression disorders.52
Th1/Th17 Cell Polarization in Immune Factors
Systemic immune cells act on the skin through chemotaxis and homing, and in a stable dynamic equilibrium, T cells patrol surrounding tissues, such as the skin, to promote rapid immune response to pathogens. Once homeostasis is imbalanced in the body, chemokines are expressed in the immune response and bind to chemokine receptors on T cells, resulting in chemotaxis and homing.12 Chen et al54 found significant Th1/Th17 pathway activation in an LL-37-induced inflammatory model of rosacea in mice. Buhl et al55 proposed that T cells are actively involved in the development of different symptoms of rosacea and confirmed that the T cell response is dominated by an immune response of Th1/Th17 cell polarization by transcriptome analysis and staining for signature cytokines, including interferon (IFN)-γ and IL-17. Gene expression profiles of skin subtypes showed a general elevation of the associated genes in Th1 and Th17 cells. Extrinsic and internal triggers induce primary proinflammatory cytokines, such as tumor necrosis factor (TNF)-α and IL-1 family members, through TLR 2 signaling or other mechanisms. The first wave of chemokines is subsequently produced, resulting in concentrated aggregation of T cells into the surrounding hair follicle space.44 Conversely, T cell-derived cytokines, such as IL-17 or IL-22, may, together with UV radiation, activate KCs to produce CCL20, CXCL1, and CXCL8. CCL20 attracts additional Th17, while CXCL1 and CXCL8 absorb large numbers of neutrophils, leading to the formation of pustules. Although Th17 cells preferentially express the chemokine receptor CCR6, their specific ligand, CCL20, upregulates in keratinocytes stimulated by UVB radiation or TNF-α/IL-1.44 IL-17 not only directly promotes angiogenesis and expansion but also stimulates the production of TNF-α, IL-1β, and IL-6, and coordinates with IL-1β and TNF-α to upregulate other angiogenic factors, such as vascular endothelial growth factor and IL-8.56,57 Previous studies have suggested that inflammation and angiogenesis may be mutually regulated through a positive feedback loop. There is evidence that activated T cells that produce vascular endothelial growth factor increase angiogenesis at sites of inflammation, while vascular endothelial growth factor promotes Th1 cell differentiation through a positive feedback loop.58 IL-8-mediated neutrophil chemotaxis and TNF-mediated amplification of inflammation are present in pustule formation, papule formation, and downstream vascular effects, respectively; other TNF-mediated effects include heat generation, activation of excess NF-kB-responsive genes in the IL-1β activation pathway, angiogenesis, and chemokines required to upregulate local adhesion molecules and for additional immune cell migration and activation, further prolonging the inflammatory process.59 Whereas the induction of the epidermal antimicrobial peptide LL-37 by IL-17a and IL-22 has been demonstrated, LL-37 can, in turn, induce the production of Th1/Th17 polarization-related genes and enhance the induction of IL-8 by Th1 and Th17 cytokines.60 Furthermore, IFN-γ is a potent activator of macrophages, suggesting the possibility of an interconnection between the CD4+T cell population and the natural immune network in rosacea.59
A growing number of studies have demonstrated that Th1/Th17 cells play a key role in the pathogenesis of anxiety and depression disorders.61 Increased differentiation of Th1 and Th17 cells may lead to the recruitment of other immune cells and hyperactivation of microglia, promote the secretion of IFN-γ and IL-17a, and enhance activation of medial prefrontal cortical neurons, leading to the onset of anxiety and depression.62 Meanwhile, Th1 and Th17 cells were found to promote anxious and depressive behavior in mice in animal models and were significantly increased in the blood of depressed patients with concomitant activation of mononuclear macrophages or lymphocytes.63 In the current study, increased production of IFN-γ was identified as a feature of major depressive disorder, highlighting the critical involvement of the Th1 phenotype and cellular immunity in the disorder.64 TNF-α produced by Th1 cells activates the HPA axis and indoleamine 2,3-dioxygenase, leading to tryptophan depletion, which triggers anxiety and depression; IL-17A, IFN-γ, IL-1, IL-6, and IL-8 activate the kynurenine pathway and decrease 5-HT levels, leading to anxiety and depressive symptoms.65
Previous literature suggests that Th17 cells are recruited into the CNS beginning in the initial stages of chronic stress, propagating from the peripheral immune response to the CNS and progressively exacerbating depression-like symptoms.66 Stress first causes the release of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α into the circulation, which then accumulate in the brain with chemokines and attack the blood-brain barrier (BBB). Once the BBB function is impaired, pro-inflammatory factors penetrate the brain and activate the natural immune system of the CNS. The dorsal striatum showed the most significant BBB disruption in the brain regions tested, and its abnormal activity was associated with core symptoms of depression, such as lack of excitatory pleasure and delayed psychomotor development. Highly inflammatory Th17 cells in the CNS can convert their surface features from CCR6 to CCR2.67 CCR2/CCL2 drives the infiltration of peripherally activated Th17 cells into the brain, and Th17-produced IL-17 is considered a key cytokine in the pathophysiology of neurological and psychiatric disorders that can further compromise the integrity of the blood-brain barrier, allowing more immune cells and cytokines to enter the CNS more easily. Astrocytes, in turn, can respond to IL-17 by releasing mediators that promote tissue damage.66 In addition, Th17 cells can also induce anxiety and depressive symptoms by promoting stress responses through the gut-brain axis.62
Conclusion
Although rosacea can be treated with good results with topical medications and laser therapy, it mostly does not guarantee control the recurrence of the disease. For patients with more severe symptoms, the clinical efficacy of topical treatment alone is often poor, so more research is needed to treat the categories and symptoms of rosacea systematically. The complex pathophysiology of rosacea, anxiety disorder, and depression disorder suggests that these three are multisystem involvement diseases, and there overlap between sex, age, and pathogenic and progression mechanisms in nerve-endocrine-immune aspects (Figure 1).
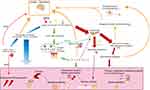 |
Figure 1 Schematic diagram of possible overlap mechanisms between rosacea and anxiety and depression. |
This demonstrates that neuropsychiatric disorders and skin disorders are not simply cosmetic stressors as understood by the general public and that there can be a connection in treatment. For example, the clinically potent and highly selective 5-HT reuptake inhibitor anxiolytic-depressant paroxetine may treat hot flashes by modulating vascular function and may be anti-inflammatory through various mechanisms.68 These same TRP cation channels targets between anxiety, depression and rosacea suggest that the cutaneous nervous system is closely linked to the CNS. Further investigation of target mechanisms of action could lead to the targeted use of selective antagonists or corresponding knockout treatments to alleviate recurrent rosacea and reduce the risk of psychiatric disorders. The HPA axis has an important influence on the pathogenesis and progression of all three, and anxiety, depression symptoms can, in turn, increase the stress load. The hormonal products produced by KCs further disrupt normal HPA axis function and co-promote glucocorticoid resistance. Therefore, the HPA axis, including the cutaneous HPA axis, deserves more research to clarify the specific mechanisms of its co-occurring action in rosacea and psychiatric disorders in order to alleviate organismal stress and morbidity. In addition, abnormally active immune inflammatory response systems are present in rosacea, anxiety, and depression disorders. Th1/Th17 cell polarization responses play an essential role in all three disorders, suggesting that suppression of T cell differentiation and function may have some potential in alleviating the symptoms of rosacea, anxiety, and depression. The overlap of inflammatory immune disorders in psychiatric disorders and rosacea also suggests a complex of co-morbidities and homeostatic imbalance in the organism. Thus early selective blockade of TRP cation channels, reduction of glucocorticoid resistance, and targeting of Th1/Th17-specific cytokines such as IL-1β and IL-17 target therapy may be some effective treatments for rosacea and remission of anxiety and depressive symptoms to prevent relapse.
It is necessary for the research team to study the common pathways between rosacea and anxiety and depression disorders at a deeper level, explore co-morbid risk factors, clarify the pathogenesis of rosacea from multiple perspectives, and provide a theoretical basis for the development of targeted treatments for rosacea as soon as possible.
In addition, dermatologists can collaborate with physicians in other departments to study whether the overlapping mechanisms of anxiety, depression, and rosacea in terms of neuroendocrine immunity are equally applicable to the rest of the dermatological disorders, and deepen local and systematic understanding of skin organs. This will not only provide new ideas and a basis for the standardized treatment and control of the recurrence of rosacea but also seek more profound research on other skin diseases with complex etiologies and diverse manifestations.
Funding
This review was supported by the Investigator Initiated Trail Projects of the Second Affiliated Hospital of Kunming Medical University (grant NO.ynIIT2022001).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Li J, Hongfu X. Guidelines for the diagnosis and treatment of rosacea in China (2021 edition). Chin J Dermatol. 2021;54(4):279–281. doi:10.35541/cjd.20201078
2. Gether L, Overgaard LK, Egeberg A, et al. Incidence and prevalence of rosacea: a systematic review and meta-analysis. Br J Dermatol. 2018;179(2):282–289. doi:10.1111/bjd.16481
3. Hongzuo G, Yuhui Z, Yan S, et al. The prevalence of rosacea in China: a meta-analysis. JEBM. 2019;19(5):543–549. doi:10.7507/1672-2531.201809112
4. Choi D, Choi S, Choi S, et al. Association of Rosacea With Cardiovascular Disease: a Retrospective Cohort Study. J Am Heart Assoc. 2021;10(19):e020671. doi:10.1161/JAHA.120.020671
5. Li J, Lin H, Hongfu X, et al. Recognition of classifying rosacea. J Clin Med Pract. 2015;1(2):1–2. doi:10.3969/j.issn.1672-6170.2015.02.001
6. Woo YR, Lim JH, Cho D, et al. Rosacea: molecular Mechanisms and Management of a Chronic Cutaneous Inflammatory Condition. Int J Mol Sci. 2016;17(9):1562. doi:10.3390/ijms17091562
7. Chang HC, Huang YC, Lien YJ, et al. Association of rosacea with depression and anxiety: a systematic review and meta-analysis. J Affect Disorders. 2022;299:239–245. doi:10.1016/j.jad.2021.12.008
8. Lu J, Xu X, Huang Y, et al. Prevalence of depressive disorders and treatment in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2021;8(11):981–990. doi:10.1016/S2215-0366(21)00251-0
9. Huang Y, Wang Y, Wang H, et al. Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2019;6(3):211–224. doi:10.1016/S2215-0366(18)30511-X
10. Woo YR, Han YJ, Kim HS, et al. Updates on the Risk of Neuropsychiatric and Gastrointestinal Comorbidities in Rosacea and Its Possible Relationship with the Gut-Brain-Skin Axis. Int J Mol Sci. 2020;21(22):8427. doi:10.3390/ijms21228427
11. Heisig M, Reich A. Psychosocial aspects of rosacea with a focus on anxiety and depression. Clin Cosmet Investig Dermatol. 2018;11:103–107. doi:10.2147/CCID.S126850
12. Jin R, Luo L, Zheng J. The Trinity of Skin: skin Homeostasis as a Neuro-Endocrine-Immune Organ. Life. 2022;12(5). doi:10.3390/life12050725
13. Rainer BM, Kang S, Chien AL. Rosacea:Epidemiology, pathogenesis, and treatment. Dermatoendocrinol. 2017;9(1):e1361574. doi:10.1080/19381980.2017.1361574
14. Choi JE, DiNardo A. Skin neurogenic inflammation. Semin Immunopathol. 2018;40(3):249–259. doi:10.1007/s00281-018-0675-z
15. Vidal Yucha SE, Tamamoto KA, Kaplan DL. The importance of the neuro-immuno-cutaneous system on human skin equivalent design. Cell Proliferat. 2019;52(6):e12677. doi:10.1111/cpr.12677
16. Dantzer R. Neuroimmune Interactions: from the Brain to the Immune System and Vice Versa. Physiol Rev. 2018;98(1):477–504. doi:10.1152/physrev.00039.2016
17. Russo AF. Overview of Neuropeptides: awakening the Senses? Headache. 2017;57(2):37–46. doi:10.1111/head.13084
18. Theoharides TC. The impact of psychological stress on mast cells. Ann Allerg Asthma Im. 2020;125(4):388–392. doi:10.1016/j.anai.2020.07.007
19. Ding W, Manni M, Stohl LL, et al. Pituitary adenylate cyclase-activating peptide and vasoactive intestinal polypeptide bias Langerhans cell Ag presentation toward Th17 cells. Eur J Immunol. 2012;42(4):901–911. doi:10.1002/eji.201141958
20. Wei Y, Cai J, Zhu R, et al. Function and therapeutic potential of transient receptor potential ankyrin 1 in fibrosis. Front Pharmacol. 2022;13:1014041. doi:10.3389/fphar.2022.1014041
21. Alvarado MG, Thakore P, Earley S. Transient Receptor Potential Channel Ankyrin 1: a Unique Regulator of Vascular Function. Cells. 2021;10(5):1167. doi:10.3390/cells10051167
22. Pyo HJ, An X, Cho H. The role of free fatty acid receptor pathways in a selective regulation of TRPA1 and TRPV1 by resolvins in primary sensory neurons. J Cell Physiol. 2022;237(9):3651–3660. doi:10.1002/jcp.30826
23. Kaneko Y, Szallasi A. Transient receptor potential (TRP) channels: a clinical perspective. Brit J Pharmacol. 2014;171(10):2474–2507. doi:10.1111/bph.12414
24. De Moura JC, Noroes MM, Rachetti Vde P, et al. The blockade of transient receptor potential ankirin 1 (TRPA1) signalling mediates antidepressant- and anxiolytic-like actions in mice. Brit J Pharmacol. 2014;171(18):4289–4299. doi:10.1111/bph.12786
25. Rodrigues-Braz D, Zhao M, Yesilirmak N, et al. Cutaneous and ocular rosacea: common and specific physiopathogenic mechanisms and study models. Mol Vis. 2021;27:323–353.
26. Jung SW, Park GH, Kim E, et al. Rosmarinic Acid, as an NHE1 Activator, Decreases Skin Surface pH and Improves the Skin Barrier Function. Int J Mol Sci. 2022;23(7):3910. doi:10.3390/ijms23073910
27. Son JH, Chung BY, Jung MJ, et al. The Risk of Rosacea According to Chronic Diseases and Medications: a 5-Year Retrospective, Multi-Institutional Case-Control Study. Ann Dermatol. 2018;30(6):676–687. doi:10.5021/ad.2018.30.6.676
28. Boillat A, Alijevic O, Kellenberger S. Calcium entry via TRPV1 but not ASICs induces neuropeptide release from sensory neurons. Mol Cell Neurosci. 2014;61:13–22. doi:10.1016/j.mcn.2014.04.007
29. Sartim AG, Brito BM, Gobira PH, et al. Attenuation of glutamatergic and nitrergic system contributes to the antidepressant-like effect induced by capsazepine in the forced swimming test. Behav Pharmacol. 2019;30(1):59–66. doi:10.1097/FBP.0000000000000416
30. Reyes-Mendez ME, Castro-Sánchez LA, Dagnino-Acosta A, et al. Capsaicin produces antidepressant-like effects in the forced swimming test and enhances the response of a sub-effective dose of amitriptyline in rats. Physiol Behav. 2018;195:158–166. doi:10.1016/j.physbeh.2018.08.006
31. Roet M, Jansen A, Hoogland G, et al. Endogenous TRPV1 expression in the human cingulate- and medial frontal gyrus. Brain Res Bull. 2019;152:184–190. doi:10.1016/j.brainresbull.2019.07.018
32. Martins D, Tavares I, Morgado C. ”Hotheaded”: the role OF TRPV1 in brain functions. Neuropharmacology. 2014;85:151–157. doi:10.1016/j.neuropharm.2014.05.034
33. Ma K, Zhang H, Wang S, et al. The molecular mechanism underlying GABAergic dysfunction in nucleus accumbens of depression-like behaviours in mice. J Cell Mol Med. 2019;23(10):7021–7028. doi:10.1111/jcmm.14596
34. Lazarini-Lopes W, Silva-Cardoso GK, Leite-Panissi CRA, et al. Increased TRPV1 Channels and FosB Protein Expression Are Associated with Chronic Epileptic Seizures and Anxiogenic-like Behaviors in a Preclinical Model of Temporal Lobe Epilepsy. Biomedicines. 2022;10(2):416. doi:10.3390/biomedicines10020416
35. Ye L, Xu M, Hu M, et al. TRPV4 is involved in irisin-induced endothelium-dependent vasodilation. Biochem Bioph Res Co. 2018;495(1):41–45. doi:10.1016/j.bbrc.2017.10.160
36. An P, Zhao XC, Liu MJ, et al. Dexmedetomidine Alleviates Intracerebral Hemorrhage-Induced Anxiety-Like Behaviors in Mice Through the Inhibition of TRPV4 Opening. Front Pharmacol. 2022;13:852401. doi:10.3389/fphar.2022.852401
37. Spencer RL, Deak T. A users guide to HPA axis research. Physiol Behav. 2017;178:43–65. doi:10.1016/j.physbeh.2016.11.014
38. Cernackova A, Durackova Z, Trebaticka J, et al. Neuroinflammation and depressive disorder: the role of the hypothalamus. J Clin Neurosci. 2020;75:5–10. doi:10.1016/j.jocn.2020.03.005
39. Nikolakis G, Stratakis CA, Kanaki T, et al. Skin steroidogenesis in health and disease. Rev Endocr Metab Disord. 2016;17(3):247–258. doi:10.1007/s11154-016-9390-z
40. Briganti S, Truglio M, Angiolillo A, et al. Application of Sebum Lipidomics to Biomarkers Discovery in Neurodegenerative Diseases. Metabolites. 2021;11(12):819. doi:10.3390/metabo11120819
41. Phan TS, Schink L, Mann J, et al. Keratinocytes control skin immune homeostasis through de novo-synthesized glucocorticoids. Sci Adv. 2021;7(5). doi:10.1126/sciadv.abe0337
42. Rao A, Douglas SC, Hall JM. Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: a Connecting Hypothesis. Cells. 2021;10(6):1439. doi:10.3390/cells10061439
43. Jamieson BB, Kim JS, Iremonger KJ. Cannabinoid and vanilloid pathways mediate opposing forms of synaptic plasticity in corticotropin-releasing hormone neurons. J Neuroendocrinol. 2022;34(4):e13084. doi:10.1111/jne.13084
44. Kim HB, Na EY, Yun SJ, et al. The Effect of Capsaicin on Neuroinflammatory Mediators of Rosacea. Ann Dermatol. 2022;34(4):261–269. doi:10.5021/ad.21.223
45. Wiebenga JXM, Heering HD, Eikelenboom M, et al. Associations of three major physiological stress systems with suicidal ideation and suicide attempts in patients with a depressive and/or anxiety disorder. Brain Behav Immun. 2022;102:195–205. doi:10.1016/j.bbi.2022.02.021
46. Chen X, Jiang F, Yang Q, et al. Bilateral repetitive transcranial magnetic stimulation ameliorated sleep disorder and hypothalamic-pituitary-adrenal axis dysfunction in subjects with major depression. Front Psychiatry. 2022;13. doi:10.3389/fpsyt.2022.951595
47. Govic A, Nasser H, Levay EA, et al. Long-Term Calorie Restriction Alters Anxiety-like Behaviour and the Brain and Adrenal Gland Transcriptomes of the Ageing Male Rat. Nutrients. 2022;14(21):4670. doi:10.3390/nu14214670
48. Dolotov OV, Inozemtseva LS, Myasoedov NF, et al. Stress-Induced Depression and Alzheimer’s Disease: focus on Astrocytes. Int J Mol Sci. 2022;23(9):4999. doi:10.3390/ijms23094999
49. Bertollo A, Grolli E, Plissari E, et al. Stress and serum cortisol levels in major depressive disorder: a cross-sectional study. AIMS Neurosci. 2020;7(4):459–469. doi:10.3934/Neuroscience.2020028
50. Mikulska J, Juszczyk G, Gawrońska-Grzywacz M, et al. HPA Axis in the Pathomechanism of Depression and Schizophrenia: new Therapeutic Strategies Based on Its Participation. Brain Sci. 2021;11(10):1298. doi:10.3390/brainsci11101298
51. Barron AM, Higuchi M, Hattori S, et al. Regulation of Anxiety and Depression by Mitochondrial Translocator Protein-Mediated Steroidogenesis: the Role of Neurons. MOL NEUROBIOL. 2021;58(2):550–563. doi:10.1007/s12035-020-02136-5
52. Corredor K, Duran JM, Herrera-Isaza L, et al. Behavioral effects of environmental enrichment on male and female wistar rats with early life stress experiences. Front Physiol. 2022;13:837661. doi:10.3389/fphys.2022.837661
53. Alizamini MM, Fattahi M, Sayehmiri F, et al. Regulatory Role of PFC Corticotropin-Releasing Factor System in Stress-Associated Depression Disorders: a Systematic Review. Cell Mol Neurobiol. 2022;10. doi:10.1007/s10571-022-01289-2
54. Chen M, Xie H, Chen Z, et al. Thalidomide ameliorates rosacea-like skin inflammation and suppresses NF-κB activation in keratinocytes. Biomed Pharmacother. 2019;116:109011. doi:10.1016/j.biopha.2019.109011
55. Buhl T, Sulk M, Nowak P, et al. Molecular and Morphological Characterization of Inflammatory Infiltrate in Rosacea Reveals Activation of Th1/Th17 Pathways. J Invest Dermatol. 2015;135(9):2198–2208. doi:10.1038/jid.2015.141
56. Lee HJ, Hong YJ, Kim M. Angiogenesis in Chronic Inflammatory Skin Disorders. Int J Mol Sci. 2021;22(21):345.
57. Zhengni Z, Jie Z, Xiaoling C, et al. Immune pathogenesis of rosacea. Chin J Allergy Clin Immunol. 2022;16(2):178–182. doi:10.3969/j.issn.1673-8705.2022.02.012
58. Chen Z, Zhang M, Liu Y, et al. VEGF-A enhances the cytotoxic function of CD4+ cytotoxic T cells via the VEGF-receptor 1/VEGF-receptor 2/AKT/mTOR pathway. J Transl Med. 2023;21(1):74. doi:10.1186/s12967-023-03926-w
59. Holmes AD, Steinhoff M. Integrative concepts of rosacea pathophysiology, clinical presentation and new therapeutics. Exp Dermatol. 2017;26(8):659–667. doi:10.1111/exd.13143
60. El-Serafi AT, El-Serafi I, Steinvall I, et al. A Systematic Review of Keratinocyte Secretions: a Regenerative Perspective. Int J Mol Sci. 2022;23(14):7934. doi:10.3390/ijms23147934
61. Medina-Rodriguez EM, Rice KC, Jope RS, et al. Comparison of inflammatory and behavioral responses to chronic stress in female and male mice. Brain Behav Immun. 2022;106:180–197. doi:10.1016/j.bbi.2022.08.017
62. Zhou Y, Yu K. Th1, Th2, and Th17 cells and their corresponding cytokines are associated with anxiety, depression, and cognitive impairment in elderly gastric cancer patients. Front Surg. 2022;9:996680. doi:10.3389/fsurg.2022.996680
63. Ross RE, VanDerwerker CJ, Saladin ME, et al. The role of exercise in the treatment of depression: biological underpinnings and clinical outcomes. Mol Psychiatr. 2022. doi:10.1038/s41380-022-01819-w
64. Maes M, Rachayon M, Jirakran K, et al. The Immune Profile of Major Dysmood Disorder: proof of Concept and Mechanism Using the Precision Nomothetic Psychiatry Approach. Cells. 2022;11(7):1183. doi:10.3390/cells11071183
65. Tong N, Zhang Y, Yang A, et al. The potency of common proinflammatory cytokines measurement for revealing the risk and severity of anxiety and depression in psoriasis patients. J Clin Lab Anal. 2022;36(9):e24643. doi:10.1002/jcla.24643
66. Peng Z, Peng S, Lin K, et al. Chronic stress-induced depression requires the recruitment of peripheral Th17 cells into the brain. J Neuroinflammation. 2022;19(1):186. doi:10.1186/s12974-022-02543-6
67. Kara EE, McKenzie DR, Bastow CR, et al. CCR2 defines in vivo development and homing of IL-23-driven GM-CSF-producing Th17 cells. Nat Commun. 2015;6:8644. doi:10.1038/ncomms9644
68. Orleans RJ, Li L, Kim MJ, et al. FDA approval of paroxetine for menopausal hot flushes. New Engl J Med. 2014;370(19):1777–1779. doi:10.1056/NEJMp1402080
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
