Back to Journals » Drug Design, Development and Therapy » Volume 10
Neuroprotective effect of β-asarone against Alzheimer’s disease: regulation of synaptic plasticity by increased expression of SYP and GluR1
Authors Liu S, Yang C, Zhang Y, Su R, Chen J, Jiao M, Chen H, Zheng N, Luo S, Chen Y, Quan S, Wang Q
Received 1 August 2015
Accepted for publication 16 December 2015
Published 15 April 2016 Volume 2016:10 Pages 1461—1469
DOI https://doi.org/10.2147/DDDT.S93559
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Wei Duan
Si-jun Liu,1,* Cong Yang,2,* Yue Zhang,2,* Ru-yu Su,2 Jun-li Chen,2 Meng-meng Jiao,2 Hui-fang Chen,2 Na Zheng,2 Si Luo,2 Yun-bo Chen,2 Shi-jian Quan,1 Qi Wang2
1School of Chinese Materia Medica, 2Institute of Clinical Pharmacology, Guangzhou University of Chinese Medicine, Guangzhou, People’s Republic of China
*These authors contributed equally to this work
Aim: β-asarone, an active component of Acori graminei rhizome, has been reported to have neuroprotective effects in Alzheimer’s disease. As the underlying mechanism is not known, we investigated the neuroprotective effects of β-asarone in an APP/PS1 double transgenic mouse model and in NG108 cells.
Materials and methods: APPswe/PS1dE9 double transgenic male mice were randomly assigned to a model group, β-asarone treatment groups (21.2, 42.4, or 84.8 mg/kg/d), or donepezil treatment group (2 mg/kg/d). Donepezil treatment was a positive control, and background- and age-matched wild-type B6 mice were an external control group. β-asarone (95.6% purity) was dissolved in 0.8% Tween 80 and administered by gavage once daily for 2.5 months. Control and model animals received an equal volume of vehicle. After 2.5 months of treatment, behavior of all animals was evaluated in a Morris water maze. Expression of synaptophysin (SYP) and glutamatergic receptor 1 (G1uR1) in the hippocampus and cortex of the double transgenic mice was assayed by Western blotting. The antagonistic effects of β-asarone against amyloid-β peptide (Aβ) were investigated in vitro in the NG108-15 cell line. After 24 hours of incubation, cells were treated with 10 µm Aβ with or without β-asarone at different concentrations (6.25, 12.5, or 25 µM) for an additional 36 hours. The cytotoxicity of β-asarone was evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay of cell viability, and cell morphology was evaluated by bright-field microscopy after 24 hours of treatment. The expression of SYP and GluR1 in cells was detected by Western blot assay in the hippocampus and brain cortex tissues of mice.
Results: β-asarone at a high dose reduced escape latency and upregulated SYP and GluR1 expression at both medium and high doses. Cell morphology evaluation showed that β-asarone treatment did not result in obvious cell surface spots and cytoplasmic granularity. β-asarone had a dose-dependent effect on cell proliferation.
Conclusion: β-asarone antagonized the Aβ neurotoxicity in vivo, improved the learning and memory ability of APP/PS1 mice, and increased the expression of SYP and GluR1 both in vivo and in vitro. Thus, β-asarone may be a potential drug for the treatment of Alzheimer’s disease.
Keywords: neuroprotective effect, β-asarone, synaptic plasticity, synaptophysin, glutamatergic receptor 1
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disease that is the most common type of dementia, and it gradually worsens with age. The potential mechanism of AD may involve synaptic dysfunction due to a decrease of synaptic proteins and interruption of axonal transport caused by disruption of the cytoskeleton.1 The pathology of behavioral changes, such as those in learning and memory, are related to plasticity in the function and structure of synapses. The dry rhizome of Acori graminei (Shi Changpu in Chinese) has been used in People’s Republic of China to induce resuscitation and improve intelligence. β-asarone is the major active agent of A. tatarinowii Schott,2 which is reported to have neuroprotective effects both in vivo and in vitro in AD models.3–6 β-asarone has also been reported to prevent synaptic loss and improve cognitive function in a senescence-accelerated prone 8 (SAMP8) mouse model.3
The transfer of information by synaptic transmission is the key modulator of neural plasticity.7–9 Synapse-associated proteins, primarily synaptophysin (SYP) and SYP1, are phosphoproteins present in synaptic vesicles and are closely associated with cognitive function.10–12 As a specific marker of synaptic vesicles, SYP reflects the density and distribution of synapses.13,14 Previous studies indicated that loss of synapses in the cerebrum and hippocampus was much more obvious in older people with cognitive impairment than in those who were unimpaired.15 Gene chip assays showed that expression of SYP1 messenger RNA declined early in the progression of AD.16 Expression of SYP1 also decreased in transgenic mice with cognitive impairment and mutations in amyloid precursor protein (APP).16 Hence, SYP is thought to contribute to neural plasticity by affecting the structure of synapses and mediating neurotransmitter release by phosphorylation.
Glutamate receptors (GluRs) not only mediate mammalian central nervous system fast excitatory synaptic transmission, but also play an important role in neural development, neural cell death, and synaptic plasticity and are associated with learning and memory.17–19 As a subunit of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, GluR1 plays a vital role in maintaining plasticity and spatial memory. Evidence from clinical pathology indicates that the expression of GluR1 in the entorhinal cortex is ~43% lower in AD patients than in healthy controls.20
In this study, we evaluated the effects of β-asarone both in vivo and in vitro and found that it could antagonize amyloid-β peptide (Aβ) neurotoxicity in vivo and improve the learning and memory ability in an APP/PS1 mouse model. β-asarone also increased SYP and GluR1 expression in vivo and in vitro. Our data suggest that β-asarone might be a potential drug for AD treatment.
Materials and methods
Animals
APPswe/PS1dE9 double transgenic male mice were purchased from the Animal Model Research Center of Nanjing University. Mice were housed in a controlled environment (22°C–25°C, 40%–60% relative humidity, and a 12-hour light/dark cycle) with distilled water and a standard diet. All experimental procedures followed the health care guidelines of the People’s Republic of China (1998) and the ethical standards for laboratory animals of Guangzhou Traditional Chinese Medicine University. This study was approved by the Animal Ethics Committee of Guangzhou University of Chinese Medicine. All animals were raised in the specific pathogen-free experimental laboratory animal center at the Guangzhou University of Traditional Chinese Medicine. At 4.5 months of age, APP/PS1 male mice were randomly assigned to five groups of eleven animals each. These were a model group (MG), β-asarone treatment group (21.2 mg/kg/d, low-dose group [LDG]; 42.4 mg/kg/d, medium-dose group [MDG]; or 84.8 mg/kg/d, high-dose group [HDG]), donepezil treatment group (2 mg/kg/d), donepezil treatment positive control group (PG), and 13 wild-type background- and age-matched B6 control mice group (CG). Animals were treated for 2.5 months; body weight and general health of the mice were recorded daily. Donepezil hydrochloride (091221A; Eisai Co, Ltd, Jiangsu, People’s Republic of China) was dissolved in distilled water. β-asarone was obtained from Sigma-Aldrich Co (5273-86-9; St Louis, MO, USA) and dissolved in absolute ethanol (GL Biochem Shanghai Ltd, Minhang, Shanghai, People’s Republic of China). The chemical structure of β-asarone is shown in Figure 1.
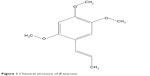  | Figure 1 Chemical structure of β-asarone. |
Morris water maze
After 2.5 months of oral administration, the performance of all mice was assessed in the Morris water maze, which was comprised of a circular 100 cm diameter, 32 cm deep, water-filled pool (23°C±1°C). Different geometric shapes were positioned around the pool to provide spatial orientation. The objective was for the mice to discover a 10 cm diameter translucent acrylic plastic platform located 1 cm below the water surface in the middle of a quadrant of the pool 20 cm away from the wall. Mice were tested three times daily at 15-minute intervals for 5 days. The learning and memory of the mice were assessed on the last day of the trial using a probe trial test. The platform was located in a different quadrant to determine whether the mice were able to generalize the task by learning a new position. At the start of each trial, a mouse was carefully placed in the water in any of the three quadrants without the platform and with its head toward the pool wall. The time required for the mouse to find the platform hidden below the water surface was used to assess dynamic changes of learning and memory. If a mouse found the platform within 60 seconds, it was able to stay on the platform for 10 seconds. Mice that did not find the platform were carefully assisted to the platform by the experimenter. Between trials, mice were returned to their home cages using a spoon-net to avoid direct contact with the experimenter. Escape latency, indicated as the time to arrive at the platform and the number of times that mice passed by the platform, was monitored by an automated system that recorded and analyzed movements (Number GLBehv-STG-4; Guangzhou Feidi Ltd, Guangzhou, People’s Republic of China).
Western blot analysis of SYP and GluR1 in the hippocampus and cortex of double transgenic mice
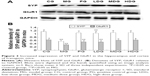
After the Morris water maze trial, the mice were fasted for 12 hours and then anesthetized in the supine position by intraperitoneal injection with 10% chloral hydrate (400 mg/kg). The limbs of anesthetized mice were fixed to the operating table, and an incision was made from the xiphoid to the diaphragm with scissors. The ribs were lifted and fixed with a hemostat to fully expose the heart and liver. A blunt needle was inserted into the left ventricle, passed into the aorta, and fixed with the hemostatic forceps. The right atrial appendage was cut to expose venous blood. The heart was clipped and perfused with 150 mL normal saline until the liver was completely white and clear liquid flowed from the right atrial appendage. The brains of mice were rapidly dissected and the hippocampus and cortex were removed on an ice-table. The hippocampus and cortex tissues were homogenized at 0°C in 1 mL working liquid. The homogenate was transferred to an ice-cold 1.5 mL centrifuge tube, centrifuged at 12,000 rpm for 15 minutes at 4°C, and the proteins were collected in the supernatant. Radioimmunoprecipitation assay lysate (30 μL), 0.6 μL 100 mM phenylmethylsulfonyl fluoride, and 0.3 μL phosphatase inhibitor were combined with the protein extracts. Protein concentration was evaluated using a bicinchoninic acid (BCA) assay kit (CW0014; Kang Century Biological Ltd, San Carlos, CA, USA). Protein samples were heated at 95°C–100°C for 3–5 minutes and prepared for electrophoresis by adding 1/5 volume of 5× loading buffer and 1/20 volume of 20× reducing agent (R0891; FERMENTAS Company, Thermo Fisher Scientific, Waltham, MA, USA) before applying to gels (1.9 mL ddH2O, 0.33 mL 30% acrylamide, 0.25 mL Tris-HCL, 0.02 mL 10% sodium dodecyl sulfate, 0.02 mL 10% APS, 0.002 mL tetramethylethylenediamine). Following blotting, polyvinylidene fluoride membranes were incubated with primary antibodies in TBST (Tris-buffered saline Tween 20) at 4°C overnight. Polyvinylidene fluoride membranes were washed three times and incubated with secondary antibodies at 25°C for 2 hours. Membranes were then developed and read by enhanced chemiluminescence (Super ECL Plus; Number 32109, Thermo Scientific). The primary antibodies and the secondary antibodies used in this study were from Cell Signaling Technology, Danvers, MA, USA (GluR1 was diluted 1:200, SYP was diluted 1:1,000). Protein expression was assayed using the Image Lab: (http://www.bio-rad.com/en-us/product/image-lab-software) gel image processing system to measure net optical densities and estimate molecular weights. GAPDH monoclonal antibody (bsm-0978M, diluted 1:1,000) was also probed and used as a sample loading control for normalization, and the data were expressed as the ratio to GAPDH.
Cell culture and drug treatment
NG108-15 cells were obtained from Professor Zhenguo Zhong (Guangxi University of Traditional Chinese Medicine, Nanning, Guangxi, People’s Republic of China) and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (No 180231; GL Biochem Shanghai Ltd) with 10% heat-inactivated fetal bovine serum and 100 U/mL penicillin in a humidified atmosphere of 95% air and 5% carbon dioxide at 37°C (Heraeus type B 5060EK/C02; Heraeus, Hanau, Germany). Cells in logarithmic phase of growth (3×103 cells/mL) were seeded in 96-well plates (100 μL/well), and after culture for 24 hours, the plates were treated with 10 μmol/L Aβ1-42 for 12 hours to set up the AD model. The culture plates were then allocated to one of the four treatments groups: 1) a MG, 2) 6.25 μmol/L β-asarone, 3) 12.5 μmol/L β-asarone, and 4) the 25 μmol/L β-asarone. Two additional 96-well plates were used as a solvent control group treated with absolute ethanol. Each group included two 96-well plates which were observed at 12 and 24 hours.
Microscopic examination of NG108-15 cells after 24 hours
NG108-15 cell growth was monitored after 24 hours of treatment. Cells were then grown in 55×15 mm2 culture flasks at 37°C in a humidified atmosphere of 95% air and 5% carbon dioxide. After appropriate incubation periods, the culture supernatants were aspirated, washed three times with phosphate-buffered saline to remove nonadherent cells, fixed with 1 mL methanol, air-dried, and observed without staining with a modular, bright-field inverted microscope (Leica DM IL LED; Leica Microsystems, Wetzlar, Germany) with bright-field settings at magnification of 40×.
Assessment of cytotoxicity
A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide colorimetric assay was used to assess the cytotoxicity of β-asarone following the manufacturer’s instructions (BioTek, Winooski, VT, USA). Briefly, 96-well plates were seeded with 1×104 cells/well in quadruplicate and cultured with 6.25–25 μmol/L β-asarone for 12 or 24 hours. Absorbance was recorded at 450 nm (Spectrafluor Fluorometer; Bio-Rad, Hercules, Shinagawa-ku, Tokyo, Japan).
Western blot assay of SYP and GluR1 in NG108-15 cells
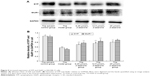
After drug intervention, the medium was decanted and the culture flasks were inverted to drain on absorbent paper. The cells were washed twice in ice-cold phosphate-buffered saline. After removing the phosphate-buffered saline, flasks were placed on ice, 35 μL sodium dodecyl sulfate cell lysate solution was added to the flask, and the cells were scraped to one side of the flask. A pipettor was used to transfer the cell debris to cold, 1.5 mL centrifuge tubes. The cold centrifuge tubes were placed in boiling water for 10 minutes, cooled to room temperature, centrifuged at 12,000 rpm for 5 minutes, and the supernatant was aliquoted and either stored at −20°C or the protein concentration was assayed immediately. The BCA working solution was prepared by combining 50 parts BCA reagent A and 1 part BCA reagent B (50:1 by volume) and then added to the microtiter plate wells. After blots were incubated with primary antibodies and the polyvinylidene fluoride membranes were washed, they were incubated with secondary antibodies and developed to assay protein expression as previously described. The primary and secondary antibodies were from Cell Signaling Technology (GluR1 1:200, SYP 1:1,000). GAPDH monoclonal antibody (bsm-0978M, diluted 1:1,000; Cell Signaling Technology) was also probed and used as a sample loading control for normalization, and the data were expressed as the ratio to GAPDH.
Statistical analysis
Data were exported to SPSS 18.0 statistics software (SPSS Inc, Chicago, IL, USA). Results of the Morris water maze test were analyzed by two-way analysis of variance (ANOVA) with multivariate repeated measures. Fisher’s least significant difference t-test was used for paired, between-group comparisons. The Kruskal–Wallis test was used for analysis of variance of nonparametric variables (N=3), and the results were expressed as mean ± standard error. ANOVA with post hoc Tukey’s test was used for multiple comparisons among study groups, with calculation of 95% confidence intervals. P-values ≤0.05 were considered as statistically significant, and P-values ≤0.01 were considered as highly statistically significant.
Results
Spatial learning performance in Morris water maze
The Morris water maze test was used to evaluate the hippocampus-dependent spatial memory and learning ability by monitoring the latency to reach the platform. Escape latency was also assessed by the Morris water maze test (Figure 2A). Two-way ANOVA with repeated measures was used to analyze differences in escape latency over 5 days, and a significant group effect (F=42.191, P=0.000) and significant interaction between factors, ie, group × day (F=3.151, P=0.000), was found. The differences between dose groups MG and PG, LDG, MDG, and HDG were significant (P<0.05, P=0.000, 0.001, 0.000, 0.000, 0.000). Animals in the control group had shorter escape latencies than those in the MG, PG, LDG, and MDG (P<0.05, P=0.000), but not in the HDG (P>0.05, P=0.217). These results indicated that increased β-asarone dose (PG, LDG, MDG, and HDG) significantly reduced escape latency, and HDG reduced the escape latency very significantly.
Learning and memory were also evaluated by a probe trial test on the last day of the research trial. There was a statistically significant difference between the MG and all other groups (P<0.05) in the number of crossings of the original platform. Animals in the MG had a lower frequency of target-zone crossing. This indicated that β-asarone increased the number of crossings of the target quadrant in all the active treatment groups (Figure 2B). Moreover, animals in the PG and all β-asarone groups spent more time in the target quadrant than animals in the MG (P<0.05, Figure 2C). The results indicated that β-asarone may have a potent memory-enhancing effect on this AD model.
The expression of SYP and G1uR1 in mice
We assayed SYP and G1uR1 expression in hippocampus and cortex of double transgenic mice by Western blotting. As shown in Figure 3A and B, expression of SYP and G1uR1 was significantly higher in the PG and in all groups treated with β-asarone than in the MG (P<0.05). SYP expression was significantly higher in the LDG than in the CG (P<0.05), and G1uR1 expression in the LDG and PG was significantly higher than in the CG (P<0.05). However, expression of SYP and G1uR1 in the MDG, HDG, and CG were not significantly different (P>0.05). MDG and HDG were thus more able to upregulate the expression of SYP and GluR1.
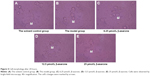
Observation of cell morphology
Figure 4 shows cell morphology viewed at 40× by bright-field microscopy after 24 hours of incubation. The control group cells were uniform in size, with translucent cytoplasm surrounded by villous or short processes, and had no obvious spots and sediment. The cytoplasm of cells from MG animals was less translucent and contained vacuoles; some cells were black, formed aggregates, or were dead. Cell axons were thin and some were broken, leaving only the cell fragments. The cells from animals treated with 6.25, 12.5, and 25 μmol/L β-asarone were relatively large, with uniform cytoplasm. Most cells had no obvious surface spots or sediment.
Cell proliferation
The cytotoxic effect of 10 μmol/L Aβ1-42 on NG108-15 cells after 12 and 24 hours of treatment was significant compared with the solvent controls (P<0.05, Table 1). After 12 hours of incubation, cell proliferation was not affected at any dose of β-asarone, and was not statistically significant from the MG (P>0.05). However, incubation of the cells with 25 μmol/L β-asarone for 24 hours increased the proliferation rate compared with the 10 μmol/L Aβ1-42 group (P<0.05). β-asarone thus had a dose-dependent effect on cell proliferation.
SYP and GluR1 expression in NG108-15 cells
As shown in Figure 5, the expression of SYP in the 10 μmol/L Aβ1-42 group was significantly different from that in the other four groups (P<0.05), indicating that it decreased SYP expression. All doses of β-asarone had significant effects on upregulation of SYP expression compared with the MG. In addition, all doses of β-asarone significantly upregulated GluR1 expression (P<0.05). In conclusion, β-asarone upregulated SYP and GluR1 expression and had a dose-dependent effect.
Discussion
Research in AD has been limited by a lack of an ideal animal model due to the incomplete understanding of its etiology and mechanism. Recently, transgenic mouse models have shown an advantage in investigating the relationship between pathological changes, exogenous genes, and molecular mechanisms. These models had been used extensively to investigate new, potential medicines for AD.21–23 In transgenic animal models, foreign genes, such as wild-type or mutant type APP genes, are stably integrated, expressed, and inherited by offspring. The APP gene and its mutants can be expressed excessively in animals and cause Aβ deposition and its related pathological damage or clinical symptoms. AD-related genes that have been screened include the APP gene, presenilin-1, presenilin-2, apolipoprotein E, and tau protein. Most transgenic animal models can reproduce various pathological features of AD patients, such as extracellular Aβ deposition, inflammatory constituents of the neurotrophic barrier, and glial proliferation.24 The APP transgenic mouse model is an ideal animal model for the simulation of pathological changes of AD and the screening of dementia drugs.
Early damage of memory and progressive neurodegeneration are characteristics of AD, which is pathologically related to numerous amyloid plaques and neurofibrillary tangles. Synaptic and neuronal losses are known to be important changes in the nervous system of AD patients. Disruption of the processes of synaptic plasticity that underlie learning and memory partly contributes to this pathophysiology. SYP is a specific marker protein of synaptic vesicles, and its density and distribution indirectly reflects the number and distribution of synapses in the brain. It is generally believed that SYP can influence synaptic structure and play a role in synaptic plasticity through phosphorylation and release of neurotransmitters. A previous study found that reduction in SYP was the major contributor,25 and synaptic degeneration played a critical role in the progression of AD.4,26
In the central nervous system, GluRs are considered to be the primary excitatory neurotransmitter receptors. They not only regulate fast excitatory synaptic transmission, but also play an important role in a number of functions including neural development, neural cell death, and synaptic plasticity (ie, learning and memory). The AMPA receptors are found in many brain regions, including the hippocampus and the cerebellum, and are clustered on postsynaptic membranes. AMPA receptors are also found within cells, where they form intracellular receptor repertoires. The N-methyl-d-aspartate (NMDA) receptor is an ion channel protein that is widely distributed in the hippocampus, cortex, and other brain regions that are related to learning and memory. It is regulated not only by voltage but also by neurotransmitters (primarily glutamate), which is a key influence in learning and plasticity in the nervous system. The function of NMDA ionotropic GluR is impaired in AD and generally decreases with aging.27
The active ingredients in the volatile oil of A. tatarinowii Schott are β-asarone (63.2%–81.2%) and α-asarone (3.4%–13.7%). Studies have shown that the main central nervous system effect of α-asarone was inhibitory, and it has been used to treat epilepsy, asthma, and depression.28,29 β-asarone has been used in the treatment of epilepsy, schizophrenia, constipation, cough, and asthma, as well as in the treatment of memory weakness. It has also been reported to have carcinogenic properties, and possesses immunosuppressive, central nervous system inhibitory, sedative, and hypothermic properties.30,31 However, the neuroprotective effects of β-asarone have been supported in recent studies, especially in studies of AD.32–35 It was reported that 100 mg/kg/d of β-asarone improved learning and memory ability in a rat model of AD induced by G-gal and Alcl3, and β-asarone was also shown to be safe.1,36 β-asarone easily penetrates the blood–brain barrier and demonstrates a variety of neuroprotective effects, such as protecting neurons from apoptosis, significantly reducing upregulation of ROCK expression and autophagy in the hippocampus of SAMP8 mice, preventing synaptic loss, and improving cognitive function. Although the signaling pathway involved in increased expression of SYP and GluR1 in response to β-asarone had not been evaluated, our study demonstrated the value of β-asarone in regulating synaptic plasticity. β-asarone has potential as a preventive drug for AD, following chemical modification to achieve high effectiveness and low toxicity.
Conclusion
β-asarone could antagonize the Aβ neurotoxicity in vivo and improved the learning and memory ability of APP/PS1 double transgenic mouse model. It also increased in vivo and in vitro SYP and GluR1 expression. The data suggest that β-asarone has potential for AD treatment.
Acknowledgments
This work was supported by the Doctoral Fund of Education Ministry of China (Number 20114425110007), PhD Start-up Fund of Natural Science Foundation of Guangdong Province of China (Number 2015A030310414), Excellent Young Scholars Research Fund of Guangzhou University of Chinese Medicine (Number 201303), Guangdong Provincial Major Science and Technology for Special Program of China (Number 2012A080202017), the Characteristic Key Discipline Construction Fund of Chinese Internal Medicine of Guangzhou University of Chinese Medicine, and South China Chinese Medicine Collaborative Innovation Center (Number A1-AFD01514A05).
Disclosure
The authors report no conflicts of interest in this work.
References
Querfurth HW, LaFerla FM. Alzheimer’s disease. N Engl J Med. 2010;362(4):329–344. | ||
Jaiswal Y, Liang Z, Ho A, et al. Metabolite profiling of tissues of Acorus calamus and Acorus tatarinowii Rhizomes by using LMD, UHPLC-QTOF MS, and GC-MS. Planta Med. 2015;81(4):333–341. | ||
Chen Y, Wei G, Nie H, et al. β-Asarone prevents autophagy and synaptic loss by reducing ROCK expression in senescence-accelerated prone 8 mice. Brain Res. 2014;1552:41–54. | ||
Zeng Y, Zhang J, Zhu Y, et al. Tripchlorolide improves cognitive deficits by reducing amyloid β and upregulating synapse-related proteins in a transgenic model of Alzheimer’s disease. J Neurochem. 2015;133(1):38–52. | ||
Irie Y, Keung WM. Rhizoma acori graminei and its active principles protect PC-12 cells from the toxic effect of amyloid-β peptide. Brain Res. 2003;963(1–2):282–289. | ||
Xue Z, Guo Y, Zhang S, et al. β-Asarone attenuates amyloid β-induced autophagy via Akt/mTOR pathway in PC12 cells. Eur J Pharmacol. 2014;741:195–204. | ||
Harrill JA, Chen H, Streifel KM, Yang D, Mundy WR, Lein PJ. Ontogeny of biochemical, morphological and functional parameters of synaptogenesis in primary cultures of rat hippocampal and cortical neurons. Mol Brain. 2015;8:10. | ||
García-Morales V, Montero F, González-Forero D, et al. Membrane-derived phospholipids control synaptic neurotransmission and plasticity. PLoS Biol. 2015;13(5):e1002153. | ||
Xiao Y, Fu H, Han X, et al. Role of synaptic structural plasticity in impairments of spatial learning and memory induced by developmental lead exposure in Wistar rats. PLoS One. 2014;9(12):e115556. | ||
Sarnat HB, Born DE. Synaptophysin immunocytochemistry with thermal intensification: a marker of terminal axonal maturation in the human fetal nervous system. Brain Dev. 1999;21:41–50. | ||
Tarsa L, Goda Y. Synaptophysin regulates activity-dependent synapse formation in cultured hippocampal neurons. Proc Natl Acad Sci U S A. 2002;99:1012–1016. | ||
Wiedenmann B, Franke WW. Identification and localization of synaptophysin, an integral membrane glycoprotein of Mr 38,000 characteristic of presynaptic vesicles. Cell. 1985;41:1017–1028. | ||
Calhoun ME, Kurth D, Phinney AL, et al. Hippocampal neuron and synaptophysin positive bouton number in aging C57BL/6 mice. Neurobiol Aging. 1998;19(6):599–606. | ||
Kwon KB, Kim JS, Chang BJ. Translocational changes of localization of synapsin in axonal sprouts of regenerating rat sciatic nerves after ligation crush injury. J Vet Sci. 2000;1(1):1–9. | ||
Baldelli P, Fassio A, Valtorta F, Benfenati F. Lack of synapsin I reduces the readily releasable pool of synaptic vesicles at central inhibitory synapses. J Neurosci. 2007;27(49):13520–13531. | ||
Chan SL, Furukawa K, Mattson MP. Presenilins and APP in neuritic and synaptic plasticity: implications for the pathogenesis of Alzheimer’s disease. Neuromolecular Med. 2002;2(2):167–196. | ||
Wang N, Xu J. Functions of kinesin superfamily proteins in neuroreceptor trafficking. Biomed Res Int. 2015;2015:639301. | ||
Gundersen V, Storm-Mathisen J, Bergersen LH. Neuroglial transmission. Physiol Rev. 2015;95(3):695–726. | ||
Fuchigami T, Nakayama M, Yoshida S. Development of PET and SPECT probes for glutamate receptors. ScientificWorldJournal. 2015;2015:716514. | ||
Yasuda RP, Ikonomovic MD, Sheffield R, Rubin RT, Wolfe BB, Armstrong DM. Reduction of AMPA-selective glutamate receptor subunits in the entorhinal cortex of patients with Alzheimer’s disease pathology: a biochemical study. Brain Res. 1995;678(1–2):161–167. | ||
Li Z, Li H, Zhao C, et al. Protective effect of notoginsenoside R1 on an APP/PS1 mouse model of Alzheimer’s disease by up-regulating insulin degrading enzyme and inhibiting Aβ accumulation. CNS Neurol Disord Drug Targets. 2015;14(3):360–369. | ||
Gao Y, Hu YZ, Li RS, et al. Cattle encephalon glycoside and ignotin injection improves cognitive impairment in APPswe/PS1dE9 mice used as multitarget anti-Alzheimer’s drug candidates. Neuropsychiatr Dis Treat. 2015;11:537–548. | ||
Dionísio PA, Amaral JD, Ribeiro MF, Lo AC, D’Hooge R, Rodrigues CM. Amyloid-β pathology is attenuated by tauroursodeoxycholic acid treatment in APP/PS1 mice after disease onset. Neurobiol Aging. 2015;36(1):228–240. | ||
Yu Y, He J, Zhang Y, et al. Increased hippocampal neurogenesis in the progressive stage of Alzheimer’s disease phenotype in an APP/PS1double transgenic mouse model. Hippocampus. 2009;19(12):1247–1253. | ||
Robinson JL, Molina-Porcel L, Corrada MM, et al. Perforant path synaptic loss correlates with cognitive impairment and Alzheimer’s disease in the oldest-old. Brain. 2014;137(Pt 9):2578–2587. | ||
Wuwongse S, Cheng SS, Wong GT, et al. Effects of corticosterone and amyloid-β on proteins essential for synaptic function: implications for depression and Alzheimer’s disease. Biochim Biophys Acta. 2013;1832(12):2245–2256. | ||
Ménard C, Herzog H, Schwarzer C, Quirion R. Possible role of dynorphins in Alzheimer’s disease and age-related cognitive deficits. Neurodegener Dis. 2014;(2–3):82–85. | ||
Huang C, Li WG, Zhang XB, et al. α-Asarone from Acorus gramineus alleviates epilepsy by modulating A-type GABA receptors. Neuropharmacology. 2013;65:1–11. | ||
Lee B, Sur B, Yeom M, Shim I, Lee H, Hahm DH. α-Asarone, a major component of Acorus gramineus, attenuates corticosterone-induced anxiety-like behaviours via modulating TrkB signaling process. Korean J Physiol Pharmacol. 2014;18(3):191–200. | ||
Kevekordes S, Spielberger J, Burghaus CM, et al. Micronucleus formation in human lymphocytes and in the metabolically competent human hepatoma cell line Hep-G2: results with 15 naturally occurring substances. Anticancer Res. 2001;21(1A):461–469. | ||
Hasheminejad G, Caldwell J. Genotoxicity of the alkenylbenzenes α- and β-asarone, myristicin and elimicin as determined by the UDS assay in cultured rat hepatocytes. Food Chem Toxicol. 1994;32(3):223–231. | ||
Geng Y, Li C, Liu J, et al. β-Asarone improves cognitive function by suppressing neuronal apoptosis in the β-amyloid hippocampus injection rats. Biol Pharm Bull. 2010;33(5):836–843. | ||
Liu J, Li C, Xing G, et al. β-Asarone attenuates neuronal apoptosis induced by β amyloid in rat hippocampus. Yakugaku Zasshi. 2010;130(5):737–746. | ||
Wei G, Chen YB, Chen DF, et al. β-Asarone inhibits neuronal apoptosis via the CaMKII/CREB/Bcl-2 signaling pathway in an in vitro model and AβPP/PS1 mice. J Alzheimers Dis. 2013;33(3):863–880. | ||
Zou DJ, Wang G, Liu JC, et al. β-Asarone attenuates β-amyloid-induced apoptosis through the inhibition of the activation of apoptosis signal-regulating kinase 1 in SH-SY5Y cells. Pharmazie. 2011;66(1):44–51. | ||
Yan SS, Chen D, Yan S, Guo L, Du H, Chen JX. RAGE is a key cellular target for Aβ-induced perturbation in Alzheimer’s disease. Front Biosci (Schol Ed). 2012;4:240–250. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.