Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Neurite outgrowth stimulatory effects of mycosynthesized AuNPs from Hericium erinaceus (Bull.: Fr.) Pers. on pheochromocytoma (PC-12) cells
Authors Raman J , Lakshmanan H, John P, Zhijian C, Periasamy V, David P, Naidu M, Sabaratnam V
Received 11 May 2015
Accepted for publication 18 July 2015
Published 18 September 2015 Volume 2015:10(1) Pages 5853—5863
DOI https://doi.org/10.2147/IJN.S88371
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Jegadeesh Raman,1 Hariprasath Lakshmanan,1 Priscilla A John,1,2 Chan Zhijian,3 Vengadesh Periasamy,3 Pamela David,1,4 Murali Naidu,1,4 Vikineswary Sabaratnam1,2
1Mushroom Research Centre, 2Institute of Biological Sciences, Faculty of Science, University of Malaya, 3Low Dimensional Materials Research Center (LDMRC), Department of Physics, Faculty of Science, 4Department of Anatomy, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Background: Hericium erinaceus has been reported to have a wide range of medicinal properties such as stimulation of neurite outgrowth, promotion of functional recovery of axonotmetic peroneal nerve injury, antioxidant, antihypertensive, and antidiabetic properties. In recent years, the green synthesis of gold nanoparticles (AuNPs) has attracted intense interest due to the potential use in biomedical applications. The aim of this study was to investigate the effects of AuNPs from aqueous extract of H. erinaceus on neurite outgrowth of rat pheochromocytoma (PC-12) cells.
Methods: The formation of AuNPs was characterized by UV–visible spectrum, energy dispersive X-ray (EDX), field-emission scanning electron microscope (FESEM), transmission electron microscopy (TEM), particle size distribution, and Fourier transform-infrared spectroscopy (FTIR). Furthermore, the neurite extension study of synthesized AuNPs was evaluated by in vitro assay.
Results: The AuNPs exhibited maximum absorbance between 510 and 600 nm in UV–visible spectrum. FESEM and TEM images showed the existence of nanoparticles with sizes of 20–40 nm. FTIR measurements were carried out to identify the possible biomolecules responsible for capping and efficient stabilization of the nanoparticles. The purity and the crystalline properties were confirmed by EDX diffraction analysis, which showed strong signals with energy peaks in the range of 2–2.4 keV, indicating the existence of gold atoms. The synthesized AuNPs showed significant neurite extension on PC-12 cells. Nerve growth factor 50 ng/mL was used as a positive control. Treatment with different concentrations (nanograms) of AuNPs resulted in neuronal differentiation and neuronal elongation. AuNPs induced maximum neurite outgrowth of 13% at 600 ng/mL concentration.
Conclusion: In this study, the AuNPs synthesis was achieved by a simple, low-cost, and rapid bioreduction approach. AuNPs were shown to have potential neuronal differentiation and stimulated neurite outgrowth. The water-soluble bioconstituents could be responsible for the neuroactivity. This is the first report for the biosynthesis of AuNPs using the hot aqueous extract of H. erinaceus.
Keywords: AuNPs, nanoparticles, Hericium erinaceus, PC-12, neurite outgrowth
Introduction
Green chemistry approaches for the synthesis of gold nanoparticles (AuNPs) via biological methods using bacteria, fungi, plant extracts, or purified biomolecules have helped to offer reliable and environment-friendly alternatives to conventional chemical and physical synthesis approaches. These particles not only are ultrasmall in size, but are also biocompatible, have high surface area to mass ratio, and show considerable surface activity along with plasmon resonance bands.1 The AuNPs are ideally used in a number of biotechnology applications and in drug delivery systems.2
Hericium erinaceus (Bull.: Fr) Pers. is a spine fungus, which grows on the trunks, branches, and stumps of trees; it can be found throughout the northern hemisphere ranging from rare in central Europe to fairly common in southern England.3 Mushrooms are not only extremely good in taste, but have also been reported to show several medicinal properties. Previous studies have reported antimicrobial effects, antitumor activities, antioxidant properties, stimulation of neurite outgrowth, and nerve regeneration.4–8
Neurobiologists and neuroscientists are exploring the field of nerve regeneration, which has become one of the biggest and most funded areas in medical research. Many neurodegenerative diseases such as Alzheimer’s disease, dementia, Parkinson’s disease, and age-related macular degeneration occur in the central nervous system (CNS).9,10 Researchers have confirmed the neurotrophic activities of H. erinaceus such as neurite outgrowth, regeneration, and neuroprotection. Apart from therapeutics offered, natural products such as mushrooms have been proven to have regenerative properties in rat pheochromocytoma cells (PC-12) and peripheral nervous system retrospectively.7,11 Daily oral administration of aqueous extract of H. erinaceus enhanced recovery of damaged peripheral nerve in rats.7,8 On the basis of this scientific evidence, natural products such as mushrooms may trigger regeneration of axons in the aforementioned clinical condition, which is vital for establishing functional connection for recovery. Our previous findings showed that Hericenone E isolated from H. erinaceus increased the phosphorylation of extracellular-signal regulated kinases and protein kinase B, potentiating nerve growth factor (NGF)-induced neuritogenesis in PC-12 cells via the MEK/ERK and PI3K/Akt pathways.12 Similarly, Erinacine D isolated from the mycelium of H. erinaceus acted as a stimulator of NGF synthesis.13
Because of the poor regenerative capacity of CNS, other methods like drugs, gene therapy, stem cells therapy, and nanotechnology continue to be very active with significant clinical potential. However, many of these factors are high-molecular-weight polypeptides, which cannot cross the blood–brain barrier (BBB).14 Hence, small molecules may be employed to cross the BBB and trigger neuronal activity. A nanoparticle is one such small microscopic particle with at least one dimension less than 100 nm. So far, only few mushrooms have been used for the nanoparticle synthesis. It can be advantageous over other biological processes because it eliminates elaborate process of maintaining cell cultures and can be synthesized on a large scale. Edible mushrooms are considered safe for consumption and contain a vast array of bioactive components that have potential as functional foods. Nanoparticles can be synthesized using mushroom extracts at a faster rate due to the presence of higher water-soluble polysaccharide and protein contents. To date, there is no report on the synthesis of nanoparticles using commercially cultivated H. erinaceus. To our knowledge, this study is the first to demonstrate a green strategy for the preparation of AuNPs using the aqueous extract of H. erinaceus and the effects of the synthesized AuNPs neurite outgrowth on PC-12 cells.
Materials and methods
Materials and chemicals
Fresh fruiting bodies of H. erinaceus KLU-M 1232 basidiocarps were collected from Ganofarm Sdn Bhd, Tanjung Sepat, Selangor, Malaysia. Rat pheochromocytoma (PC-12) cell line was purchased from American Type Culture Collection (ATCC; Rockville, MD, USA; Catalog Number: CRL-1721.1TM). Permission to use this cell line was obtained from the Department of Veterinary Services, Ministry of Agriculture and Agro-Based Products, Malaysia. Gold (III) chloride hydrate, F-12 K medium (Kaighn’s Modification of Ham’s F-12 Medium), and NGF-7 S from murine submaxillary gland were obtained from Sigma Co. (St Louis, MO, USA). Fetal bovine serum (FBS) and horse serum (HS) were purchased from PAA Laboratories (Cölbe, Germany), and all other chemicals used for the studies were of analytical grade.
Preparation of hot aqueous extract of H. erinaceus
The fresh basidiocarps were sliced and boiled with triple-distilled water at a ratio of 1:10 (w/v) with agitation at 60°C±2°C for 30 minutes.15 The boiled mushroom was left covered for 30 minutes. Residues were then removed by filtration through absorbent cotton gauze cloth and centrifuged at 10,000 rpm for 30 minutes at 4°C. Supernatants were collected and filtered through Whatman Number1 filter paper. The sample was freeze dried (Christ Alpha 1-4 LD Freeze dryer) and the hot aqueous extract (HAE) powder was stored at 4°C±2°C until further use.
Mycosynthesis of AuNPs
AuNPs were synthesized according to the method described by Mohammed Fayaz et al.16 On the basis of the result of a preliminary trial (unpublished data), 1.0 mg/mL of aqueous extract of H. erinaceus was added to 5 mL of 1×10−3 M aqueous AuCl4 solution and kept at room temperature, and the mixed solution was continuously stirred and incubated in the dark after 36 hours. The light yellow color of Gold (III) chloride hydrate solution turned to vivid purple, indicating the formation of AuNPs.
Characterization of AuNPs
UV–visible spectroscopy measurement was carried out on a UV-160A (SHIMADZU) spectrophotometer operated at a resolution of 1 nm. The Fourier transform-infrared (FTIR) spectra were recorded using a PerkinElmer FTIR spectrophotometer at a resolution of 4 cm−1 to identify bioactive constituents present in the aqueous extract. For transmission electron microscopy (TEM) and field-emission scanning electron microscope (FESEM) imaging, the samples were prepared in a vacuum desiccator. TEM (JOEL JEM-2100F) and FESEM (FEG Quanta 450, EDX-OXFORD) were used to determine the size and morphology of AuNPs. Energy dispersive X-ray (EDX) analysis was performed using the QUANTA 450 FEG instrument equipped with an OXFORD EDX attachment. To know the average size of synthesized AuNPs, a particle size distribution (PSD) was performed by laser diffraction method in aqueous solution (Malvern-USA).
Estimation of soluble sugar and protein in aqueous extracts
Soluble sugar was estimated according to Dubois et al’s17 method, and protein was estimated by Bradford’s method.18
Determination of monosaccharide and protein composition in aqueous extract H. erinaceus
Monosaccharide composition was investigated by thin layer chromatography (TLC), according to the protocol of Thetsrimuang et al.19 Proteins were determined by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). The SDS-PAGE was executed with some modification of the method of Laemmli.20 Protein samples were from HAE of H. erinaceus with 10× sample buffer (1 M Tris–HCl, 30% glycerol, 0.06% SDS, 16% 2-mercaptoethanol, and 0.06% bromophenol blue) in 9:1 ratio followed by loading of 20 μL/well. The samples were then analyzed by using SDS-PAGE with acrylamide gel consisting 12% resolving gel and 4.5% stacking gel. Electrophoresis was run at 120 V for approximately 60 minutes in tris-glycine running buffer (30 mM Tris, 144 mM glycine, and 0.1% SDS). After electrophoresis, the gel was stained with Coomassie Brilliant Blue R-250 and destained with destaining buffer (10% methanol and 10% acetic acid).
PC-12 cell culture
The PC-12 cells (adherent variant, PC-12Adh) from ATCC were maintained in F-12 K medium (Sigma) supplemented with 2.5% (v/v) heat-inactivated FBS (PAA) and 15% (v/v) HS (PAA) with final pH 6.8–7.2. All incubations were performed at 37°C in a humidified environment of 5% CO2 and 95% air. The cells were maintained in the logarithmic phase of growth and were subcultured at 2–3-day intervals. For storage, the cells were frozen at −70°C liquid nitrogen in complete medium supplemented with 5% (v/v) dimethyl sulfoxide (Sigma) as a cryoprotectant.
Neurite outgrowth stimulation assay
Neurite outgrowth stimulation assay was performed according to Eik et al11 with some modifications. The cells were seeded in a 12-well plate at an initial density of 5,000 cells per well in 1 mL complete growth medium with different concentrations of biosynthesized AuNPs and aqueous extract of H. erinaceus (HAHE-50 μg/mL). For freeze-dried aqueous extract, a stock solution of 2 mg/mL (HAHE) and 10 μg/mL (AuNPs) (w/v) were prepared freshly each time prior to assay. In positive control experiments, cells were induced to differentiate by the addition of 50 ng/mL (w/v) NGF extracted from murine submaxillary gland (Sigma). Cells in complete growth medium only served as a negative control. All the cells were incubated for 5 days at 37°C, 95% air, and 5% CO2 to observe any neuronal differentiation activity.
Quantification of neurite-bearing cells
A cell was scored positive if it bore a thin neurite extension that was double or more the length of the cell body diameter.21 Ten fields per well were randomly examined under an inverted microscope (Nikon Eclipse TS100, Nikon Instruments Inc., Melville, NY, USA). The cells were photographed using a Nikon DS-Fi1 camera and processed with a Nikon’s Imaging Software, NIS-Elements D. The percentage of neurite-bearing cells were quantified by scoring the number of neurite-bearing cells over the total number of viable cells in ten microscopic fields, with average of randomly chosen 250–260 cells per well.
Immunofluorescence staining of neurofilament
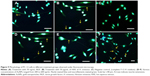
Immunofluorescence assay was carried out according to the method of Schimmelpfeng et al22 with some modifications. Approximately 5×103 cells per well were seeded in a 12-well microchamber (ibidi, Martinsried, Germany) containing complete F-12 K medium. After 1 hour, the cells were treated with 200, 400, and 600 ng/mL concentrations of AuNPs for 48 hours at 37°C±2°C in a 5% CO2-humidified incubator. Subsequently, the cells were fixed with 4% formalin (v/v) at room temperature for 20 minutes. After three washings with PBS, the cells were incubated with anti-NF-200 antibody produced in rabbit (1:80 dilution in blocking buffer) for 1 hour at room temperature. Then, the cells were incubated with fluorophore-conjugated secondary antibody, anti-rabbit IgG-FITC (fluorescein isothiocyanate) antibody produced in sheep (1:80 dilution in blocking buffer) for 1 hour at room temperature in the dark. Cells were mounted with aqueous mounting medium, ProLong® Gold Antifade Reagent (Life Technologies, Waltham, MA, USA) with 4′,6-diamidino-2-phenylindole (DAPI). Slides were observed under fluorescence illumination using fluorescein isothiocyanate (FITC) and DAPI filters and images were captured with Nikon’s Imaging Software, NIS Elements.
Statistical analysis
Results were expressed as percentage at each concentration level. A bar chart was used; each treatment level was treated as an independent category. Results were given as means ± standard deviation. Analysis of variance and Duncan’s multiple range analysis were carried out using Stat Graphics software to test the significance of the treatments.
Result and discussion
Extracellular mycosynthesis of AuNPs
Mycosynthesis of AuNPs was achieved by using the aqueous extract of H. erinaceus. The stacked UV–visible spectra of AuNPs formed from the reaction of aqueous AuCl4 with fresh basidiocarp hot aqueous extract at various time intervals is presented in Figure 1. Addition of extracts to the aqueous AuCl4 resulted in color change from light yellow to vivid purple, which indicated the formation of AuNPs in the reaction mixture. The color change is due to the excitation of surface plasmon vibration in metal nanoparticles, and the formation of AuNPs was confirmed by UV–visible spectroscopy.23,24 The absorbance peak falls between 520 and 560 nm at 36 hours incubation. Increasing the incubation time further to 40 hours and up to 1 month did not show any significant shift in the UV absorbance spectrum. The concentration of the mushroom extracts may affect the stability of the AuNPs formation. High concentration of the mushroom extracts contain high concentration of biomolecules (proteins/polysaccharide), which would act as strong capping agents as reported by previous studies.24,25 In an earlier study, the formation of the AuNPs was obtained under light conditions within 12 hours.26 This indicated that the formation of AuNPs depends on the condition, concentration of the mushroom extracts, and also the incubation time.
Electron microscopy study
The TEM image showed a very large density of AuNPs. At higher magnification, the morphology of the nanoparticles was clearly seen (Figure 2). The particles were predominantly spherical, with a small percentage being nanotriangles.27 The nascent nanocrystals devoid of protection were unstable, and gold nanotriangles might grow by a process involving rapid reduction, assembly, and room temperature sintering of spherical AuNPs.28 The presence of large quantity of extract causes strong interaction between protective biomolecules and surfaces of nanoparticles, preventing nascent gold nanocrystals from sintering. With larger quantities of the extract, the interaction is intensified, leading to size reduction of spherical nanoparticles.25 Interestingly, the nanoparticles appeared to have assembled into closed chain structures rather than a dense, closely packed assembly, as is normally the case in aqueous nanoparticle solutions. In this study, many of the AuNPs were spherical with sizes of 20–30 nm. The specific shapes and size are important for applications in biological and chemical sensing as well as for medical and electronic devices because the optical properties of AuNPs are dependent on the size and shape.29 Several studies have reported synthesis of various sizes of AuNPs using different fungi. Fusarium oxysporum produced spherical and triangular morphologies of particles with a size range of 20–40 nm.30 Alternatively, the fungi Neurospora crassa produced average AuNPs sizes of 3–100 nm with mean diameter of 32 nm.31 The FESEM analysis provided thinner probing beams at low- as well as high-electron energy. The three-dimensional images of AuNPs were dense and spherical in shape after 36 hours incubation, as seen in Figure 2. It was observed from the electron microscopic images that AuNPs mycosynthesized with aqueous extracts of H. erinaceus were irregular in shape, and this may be due to organic shell over the particles.26
Particle size analysis
The PSD uses laser diffraction technique to reveal the average hydrodynamic diameter of particles in a liquid suspension. These particle sizes are well within the range reported for photoluminescence of AuNPs.30 In this study, the PSD analysis of mushroom extract-mediated synthesis of AuNPs showed an average size of 28 nm (Figure 3). However, the nanoparticles such as gold and silver have also been shown to exhibit size-dependent optical properties. If the size of AuNP is smaller, the total extinction is contributed by absorption; however, an increase in the size in the AuNP would contribute to both absorption and scattering.32 In this study, we found that the absorption peak is at 520–560 nm, which corresponds to a particle size of 28.37 nm.
  | Figure 3 Size-distribution analysis by dynamic light scattering. |
EDX analysis
The EDX profile of AuNPs showed strong signals for gold atoms as shown in Figure 4. The EDX pattern clearly showed that the AuNPs were formed by the reduction of Aucl4− ions using aqueous extract of H. erinaceus. EDX results obtained in this study confirmed the presence of strong gold signal by the energy peaks for silver atoms in the range of 2–2.4 keV. The EDX spectrum of AuNPs from Inonotus obliquus showed a strong Au signal along with weak signals from O, C, Al, Ca, Cl, K, Mg, Si, and Na, which may have originated from the biomolecules bound to the surface of the nanoparticles.33
  | Figure 4 EDX image of AuNPs mycosynthesized by HAE of H. erinaceus. |
FTIR analysis
FTIR measurements were carried out to identify possible interactions between gold metal extracellular proteins and polysaccharides. Spectrums of aqueous extract of H. erinaceus and extracellular biosynthesized AuNPs are shown in Figure 5A and B. The band at 3,280 cm−1 indicates O–H stretching vibrations of hydroxyl group, H-bonded alcohols, phenols, or N–H stretching of 1° and 2° amines and amides.34 The weak band at 2,934.34 cm−1 wavenumber corresponds to C–H stretch alkene and O–H stretch carboxylic acids.35 Peaks at 2,163 and 2,102 cm−1 bends correspond to –C=C– stretch alkynes; primary amines results in a broad band in the ranges at 1,603–1,588 cm−1. The peak at 1,402–1,399.5 cm−1 is assigned to O–H bend and indicates the carbohydrate group.34 The peak at 1,075–1,035 cm−1 denotes the aliphatic amines (C–N stretch) open-chain carbon atoms. The presence of soluble proteins/polysaccharide might be responsible for bioreduction and for stabilizing and capping the newly formed nanostructures.30 The peak at 1,402 cm−1 could be assigned to the COO– symmetric stretch from intercellular mushroom proteins with carboxyl side groups in the amino acid residues. This strongly suggests that the gold ion binding sites (regardless of covalent or electrostatic origin) and the subsequent reduction of gold ions all involved oxygen-containing functional groups, most likely those from the carboxyl side groups in the intercellular mushroom proteins.
Quester et al36 had used fungal extracts for NP synthesis, which is a solution containing a high proportion of soluble fungal proteins (1.7 mg protein/mL N. crassa extract, as shown by the Bradford assay).18 The exact reduction mechanism remains unknown, but at a high temperature of 60°C, it is probable that all proteins are denatured and thereby lose their three-dimensional structures, which indicates that all functional groups necessary for bioreduction and stabilization are accessible. However, in this study, the H. erinaceus aqueous extract was prepared at 60°C, and the soluble protein was quantified (270 μg protein/mg).18 It is evidenced that the denaturing of proteins is not possible at this temperature, and the presence of highly soluble proteins might act as capping and bioreductants.
Quantification of bioactive constituents in the H. erinaceus extract
TLC plate showed the presence of soluble monosaccharides in aqueous extract of H. erinaceus (Figure 6). Meshram et al37 and Park et al38 reported that white sugar and polysaccharides are natural reservoir for the green synthesis of nanoparticles. Soluble proteins in the mushroom extract were first separated by dialysis according to molecular weight. SDS-PAGE analysis showed molecular weights were in the range of 14–116 kDa. The results revealed that the aqueous extract contains better protein profile, both in numbers and molecular weights (Figure 6). Preliminary gel electrophoresis indicated that the mushroom contained more than seven different proteins of molecular weight ranging between 12 and 116 kDa. The proteins act as a reducing and protecting agent in the synthesis of nanoparticles. Gold ions were trapped by the protein and formed the protein-capped nanoparticles. Functional groups of amino acid present on the periphery of the adsorbed protein layer served as anchoring points for the attachment of AuNPs.24
Neurite stimulation activity assay
Neurite outgrowth stimulation of AuNPs and aqueous extract of H. erinaceus were observed after 72 hours of incubation. The neurite outgrowth stimulation in treated PC-12 cells was dose dependent (Figure 7). The percentage of neurite-bearing cells of AuNPs at 400 ng/mL (11.8%±0.28%) was significantly higher (P<0.05) compared to the positive control (50 ng/mL NGF). Maximum stimulation of neuritogenesis by AuNPs of H. erinaceus was achieved at 600 ng/mL, with 13% of neurite-bearing cells. However, increasing the concentration showed minimal, or had a negative, effect on the number of neurite-bearing cells (Figure 7). Extracts of the tropical grown H. erinaceus had neuroprotective and neuroregenerative effects.39,40 A previous study showed that the aqueous extracts prepared from the fruiting bodies and mycelium of H. erinaceus stimulate neurite outgrowth from cultured cells of the neural hybrid clone NG108-15.6 The aqueous extract of H. erinaceus fresh basidiocarps was able to promote neurite outgrowth and nerve regeneration, and accelerate motor functional recovery of rodents in vivo after crash injury.7
The uniqueness of PC-12 cell line is that it responds to NGF, and NGF mimics extract of H. erinaceus and AuNPs with a change in its phenotype, followed by extension of neurite outgrowth (Figure 8). The percentage of neurite-bearing cells of AuNPs treated cells was significantly (P<0.05) increased in a concentration-dependent manner. There were significant differences (P<0.05) between the negative control and all concentrations of AuNPs and H. erinaceus extract tested. NGF (50 ng/mL)-treated cells served as positive controls. Neurite outgrowth can be qualitatively confirmed by immunofluorescence staining of the neurofilaments. Anti-neurofilament is a useful immunocytochemical marker for axons.41 Neuronal cells were treated with AuNPs at 200, 400, and 600 ng/mL (w/v) and incubated for 48 hours. Figure 9 shows immunofluorescence staining of PC-12 cells. After incubation with AuNPs, an enhancement of neurite outgrowth with extension that was double the length of cell body diameter was observed. Cells treated with 600 ng/mL AuNPs triggered outgrowth, which was significantly (P<0.05) higher compared to cells treated with 200 and 400 ng/mL of AuNPs. NGF is a signaling molecule that plays an important role in the differentiation and survival of peripheral sensory and sympathetic neurons.42 NGF activates ERK, and mitogen-activated protein (MAP) kinase pathway is responsible of neurotrophic responses in PC-12 cells.43
Neurotrophins like NGF are mostly mediated by the Trk family of receptor tyrosine kinase, TrKA. A previous study revealed that the compound hericenone isolated from H. erinaceus triggered ERK1/2 and PI3K/Akt signaling cascades.12 Similarly, α-Phenyl-N-tert-butylnitron was also found to induce neurite outgrowth in PC-12 independent of TrkA.44 Our previous study reported that Hericenone E is a neurotrophic compound that potentiates NGF-induced neurite outgrowth in PC-12 cells. In the presence of 5 ng/mL of NGF, hericenones significantly increased the percentage of neurite-bearing cells compared with control cell, which had either hericenones alone or 5 ng/mL of NGF.12 In contrast to this result, a polysaccharide purified from H. erinaceus mycelium enhanced neurite outgrowth of PC-12 cells, and its efficacy was significantly higher than the crude aqueous extract of NGF alone.45 It is thus predicted that based on the ability of mycosynthesized AuNPs from H. erinaceus to stimulate neurite outgrowth of PC-12 without NGF, AuNPs can mimic NGF and may activate Akt, which is required in combination with activated MAPK cascade proteins for neurite outgrowth. This finding suggests that neurite outgrowth potentiated by AuNPs in PC-12 cells is also regulating the cell signaling pathway.
Conclusion
To our knowledge, this is the first evidence of the effects of mycosynthesized AuNPs on neuronal differentiation and neurite outgrowth. The protein capped AuNPs mycosynthesized using HAE of H. erinaceus had neurotrophic activities. These nanosize particles may cross the BBB to trigger neurite outgrowths.
Acknowledgments
This research was supported by University of Malaya High Impact Research Grant UM.C/625/1/HIR/MOE/SC/02 from the Ministry of Education, Malaysia, and J-21001-7653 from the University of Malaya is gratefully acknowledged. The first and second authors thank the University of Malaya for the postdoctoral fellowships.
Disclosure
The authors report no conflicts of interest in this work.
References
Vithiya K, Sen S. Biosynthesis of nanoparticles. Int J Pharm Sci Res. 2011;2(11):2781–2785. | ||
Glomm WR. Functionalized gold nanoparticles for applications in bionanotechnology. J Disper Sci Technol. 2005;26(3):389–414. | ||
Pegler DN. Useful fungi of the world: the monkey head fungus. Mycologist. 2003;17(3):120–121. | ||
Mizuno T, Sation H, Nishitoba T, Kawagishi H. Antitumor active substances from mushroom. Food Rev Int. 1995;11(1):23–61. | ||
Wong KH, Vikineswary S, Noorlidah A, Kuppusamy UR, Naidu M. Effects of cultivation techniques and processing on antimicrobial and antioxidant activities of Hericium erinaceus (Bull.: Fr.) Pers. extracts. Food Technol Biotech. 2009;47(1):47–55. | ||
Wong KH, Sabaratnam V, Abdullah N, Naidu M, Keynes R. Activity of water extracts of lion’s mane mushrooms Hericium erinaceus (Bull.Fr) Pers (Aphyllohoromycetideae) on the neural cell line NG108-15. Int J Med Mushrooms. 2007;9(1):57–65. | ||
Wong KH, Naidu M, David RP, et al. Functional recovery enhancement following Injury to rodent peroneal nerve by lion’s mane mushroom, Hericium erinaceus (Bull.: Fr.) Pers. (Aphyllophoromycetideae). Int J Med Mushrooms. 2009;11(3):225–236. | ||
Wong KH, Naidu M, David RP, et al. Peripheral nerve regeneration following crush injury to rat peroneal nerve by aqueous extract of medicinal mushroom Hericium erinaceus (Bull.: Fr) Pers. (Aphyllophoromycetideae). Evid Based Complement Alternat Med. 2011;2011:580752. doi:10.1093/ecam/neq062. | ||
Emard JF, Thouez JP, Gauvreau D. Neurodegenerative diseases and risk factors: a literature review. Soc Sci Med. 1995;40(6):847–858. | ||
Bunce C, Wormald R. Leading causes of certification for blindness and partial sight in England and Wales. BMC Public Health. 2006;6:58. | ||
Eik LF, Naidu M, David P, Wong KH, Tan YS, Sabaratnam V. Lignosus rhinocerus (Cooke) Ryvarden: a medicinal Mushroom that stimulates neurite outgrowth in PC-12 cells. Evid Based Complement Alternat Med. 2012;2012:320308. doi:10.1155/2012/320308. | ||
Phan CW, Lee GS, Hong SL, et al. Hericium erinaceus (Bull.: Fr) Pers. cultivated under tropical conditions: isolation of hericenones and demonstration of NGF-mediated neurite outgrowth in PC12 cells via MEK/ERK and PI3K-Akt signaling pathways. Food Funct. 2014;5(12):3160–3169. | ||
Kawagishi H, Simada A, Shizuki K, et al. Erinacine D, a stimulator of NGF-synthesis, from the mycelia of Hericium erinaceum. Heterocycl Commun. 1996;2(1):51–54. | ||
Obara Y, Hganuma A, Murakami S, et al. 5,19-cyclo-9beta,10xi-androstane-3,17-dione promotes neurotrophic factor biosynthesis in 1321N1 human astrocytoma cells and improves passive avoidance learning impairment. Brain Res. 2007;1184:57–64. | ||
Jegadeesh R, Rajasekar Reddy G, Hariprasath L, et al. Mycosynthesis and characterization of silver nanoparticles from Pleurotus djamor var. roseus and their in vitro cytotoxicity effect on PC3 cells. Process Biochem. 2015;50(1):140–147. | ||
Mohammed Fayaz A, Girilal M, Venkatesan R, Kalaichelvan P. Biosynthesis of anisotropic gold nanoparticles using Maduca longifolia extract and their potential in infrared absorption. Colloids Surf B Biointerfaces. 2011;88(1):287–291. | ||
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem. 1956;28(3):350–356. | ||
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72(1–2):248–254. | ||
Thetsrimuang C, Khammuang S, Sarnthima R. Antioxidant activity of crude polysaccharides from edible fresh and dry mushroom fruiting bodies of Lentinus sp. strain RJ-2. Int J Pharm. 2011;7(1):58–65. | ||
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680–685. | ||
Carmeliet P. Blood vessels and nerves: common signals, pathways and diseases. Nat Rev Genet. 2003;4(9):710–720. | ||
Schimmelpfeng J, Weibezahn KF, Dertinger H. Quantification of NGF dependent neuronal differentiation of PC-12 cells by means of neurofilament-L mRNA expression and neuronal outgrowth. J Neurosci Methods. 2004;139(2):299–306. | ||
Suresh AK, Pelletier DA, Wang W, et al. Biofabrication of discrete spherical gold nanoparticles using the metal-reducing bacterium Shewanella oneidensis. Acta Biomater. 2011;7(5):2148–2152. | ||
Gurunathan S, Han J, Park JH, Kim JH. A green chemistry approach for synthesizing biocompatible gold nanoparticles. Nanoscale Res Lett. 2014;9(1):248. | ||
Philip D. Biosynthesis of Au, Ag and Au–Ag nanoparticles using edible mushroom extract. Spectrochim Acta A Mol Biomol Spectrosc. 2009;73(2):374–381. | ||
Bhat R, Sharanabasava VG, Deshpande R, Shetti U, Sanjeev G, Venkataraman A. Photo-bio-synthesis of irregular shaped functionalized gold nanoparticles using edible mushroom Pleurotus florida and its anticancer evaluation. J Photochem Photobiol B. 2013;125:63–69. | ||
Absar A, Satyajyoti S, Khan MI, Rajiv K, Sastry M. Extra-/intracellular biosynthesis of gold nanoparticles by an alkalotolerant fungus, Trichothecium sp. J Biomed Nanotechnol. 2005;1(1):47–53. | ||
Huang J, Li Q, Sun D, Lu Y, et al. Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology. 2007;18(10):105104. doi:10.1088/0957-4484/18/10/105104. | ||
Rao CNR, Cheetham AK. Science and technology of nanomaterials: current status and future prospects. J Mater Chem. 2001;11:2887–2894. | ||
Mukherjee P, Senapati S, Mandal D, et al. Extracellular synthesis of gold nanoparticles by the fungus Fusarium oxysporum. Chembiochem. 2002;3(5):461–463. | ||
Castro-Longoria E, Vilchis-Nestor AR, Avalos-Borja M. Biosynthesis of silver, gold and bimetallic nanoparticles using the filamentous fungus Neurospora crassa. Colloids Surf B Biointerfaces. 2011;83(1):42–48. | ||
Husseiny MI, El-Aziz MA, Badr Y, Mahmoud MA. Biosynthesis of gold nanoparticles using Pseudomonas aeruginosa. Spectrochim Acta A Mol Biomol Spectrosc. 2007;67(3–4):1003–1006. | ||
Punuri JB, Sharma P, Sibyala S, Tamuli R, Bora U. Piper betle-mediated green synthesis of biocompatible gold nanoparticles. Int Nano Lett. 2012;2(18):18–32. | ||
Rastogi L, Arunachalam J. Sunlight based irradiation strategy for rapid green synthesis of highly stable silver nanoparticles using aqueous garlic (Allium sativum) extract and their antibacterial potential. Mater Chem Phys. 2011;129(1–2):558–563. | ||
Rajeshkumar S, Kannan C, Annadurai G. Synthesis and characterization of antimicrobial silver nanoparticles using marine brown seaweed Padina tetrastromatica. Drug Invention Today. 2012;4(10):511–513. | ||
Quester K, Avalos-Borja M, Vilchis-Nestor AR, Camacho-López MA, Castro-Longoria E. SERS properties of different sized and shaped gold nanoparticles biosynthesized under different environmental conditions by Neurospora crassa extract. PLoS One. 2013:8(10):e77486. doi:10.1371/journal.pone.0077486. | ||
Meshram SM, Bonde SR, Gupta IR, Gade AK, Rai MK. Green synthesis of silver nanoparticles using white sugar. IET Nanobiotechnol. 2013;7(1):28–32. | ||
Park Y, Hong YN, Weyers A, Kim YS, Linhardt RJ. Polysaccharides and phytochemicals: a natural reservoir for the green synthesis of gold and silver nanoparticles. IET Nanobiotechol. 2011;5(3):69–78. | ||
Lai PL, Naidu M, Sabaratnam V, et al. Neurotrophic properties of the lion’s mane medicinal mushroom, Hericium erinaceus (higher Basidiomycetes) from Malaysia. Int J Med Mushrooms. 2013;15(6):539–554. | ||
Wong KH, Naidu M, David RP, Bakar R, Sabaratnam V. Neuroregenerative potential of lion’s mane mushroom, Hericium erinaceus (Bull.: Fr.) Pers. (higher Basidiomycetes), in the treatment of peripheral nerve injury (review). Int J Med Mushrooms. 2012;14(5):427–446. | ||
Tojima T, Yamane Y, Takahashi M, Ito E. Acquisition of neuronal proteins during differentiation of NG108-15 cells. Neurosci Res. 2000;37(2):153–161. | ||
Fiore M, Chaldakov GN, Aloe L. Nerve growth factor as a signaling molecule for nerve cells and also for the neuroendocrine-immune systems. Rev Neurosci. 2009;20(2):133–145. | ||
Xing J, Kornhauser JM, Xia Z, Thiele EA, Greenberg ME. New growth factor activates extracellular signal-regulated kinase and p38 mitogen-activated protein kinase pathways to stimulate CREB serine 133 phosphorylation. Mol Cell Biol. 1998;18(4):1946–1955. | ||
Tsuji M, Inanami O, Kuwabara M. Induction of neurite outgrowth in PC12 Cells by α-phenyl-N-tert-butylnitron through activation of protein kinase C and the Ras-extracellular signal-regulated kinase pathway. J Biol Chem. 2001;276(35):32779–32785. | ||
Park YS, Lee HS, Won MH, Lee JH, Lee SY, Lee HY. Effect of an exo-polysaccharide from the culture broth of Hericium erinaceus on enhancement of growth and differentiation of rat adrenal nerve cells. Cytotechnol. 2002;39:155–162. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.