Back to Journals » International Journal of Nanomedicine » Volume 13
Natural product-based nanomedicines for wound healing purposes: therapeutic targets and drug delivery systems
Authors Hajialyani M , Tewari D, Sobarzo-Sánchez E, Nabavi SM, Farzaei MH , Abdollahi M
Received 14 May 2018
Accepted for publication 3 July 2018
Published 3 September 2018 Volume 2018:13 Pages 5023—5043
DOI https://doi.org/10.2147/IJN.S174072
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Marziyeh Hajialyani,1 Devesh Tewari,2 Eduardo Sobarzo-Sánchez,3,4 Seyed Mohammad Nabavi,5 Mohammad Hosein Farzaei,1 Mohammad Abdollahi6
1Pharmaceutical Sciences Research Center, Kermanshah University of Medical Sciences, Kermanshah, Iran; 2Department of Pharmaceutical Sciences, Faculty of Technology, Kumaun University, Nainital, India; 3Laboratory of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, Spain; 4Instituto de Investigación e Innovación en Salud, Facultad de Ciencias de la Salud, Universidad Central de Chile, Chile; 5Applied Biotechnology Research Center, Baqiyatallah University of Medical Sciences, Tehran, Iran; 6Toxicology and Diseases Group, The Institute of Pharmaceutical Sciences (TIPS) and Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
Abstract: Wound healing process is an intricate sequence of well-orchestrated biochemical and cellular phenomena to restore the integrity of the skin and subcutaneous tissue. Several plant extracts and their phytoconstituents are known as a promising alternative for wound healing agents due to the presence of diverse active components, ease of access, and their limited side effects. The development of nanotechnological methods can help to improve the efficacy of different therapeutics as well as herbal-based products. Here, we present a review of the efficacy of the plant based-nanomaterials in the management of wounds and discuss the involved therapeutic targets. For this purpose, a profound search has been conducted on in vitro, in vivo, and/or clinical evidences evaluating the efficacy and pharmacological mechanisms of natural product-based nanostructures on different types of wounds. Different pharmacological targets are involved in the wound healing effects of herbal-based nanostructures, including suppressing the production of inflammatory cytokines and inflammatory transduction cascades, reducing oxidative factors and enhancing antioxidative enzymes, and promoting neovascularization and angiogenic pathways through increasing the expression of vascular endothelial growth factor, fibroblast growth factor, and platelet-derived growth factor. Moreover, nanostructure of plant extracts and their phytochemicals can enhance their bioavailability, control their release in the form of sustained delivery systems to the wound site, and enhance the permeability of these therapeutics to the underlying skin layers, which are all necessary for the healing process. Overall, various plant extracts and their natural compounds, used in nanoformulations, have demonstrated high activity in the management of wounds and thus can be assumed as future pharmaceutical drugs.
Keywords: nanomedicine, nanoparticle, nanofiber, natural product, medicinal plants, phytochemicals, herbal products, hydrogels, nanoemulsion, electrospinning, wound healing, wound dressing, nanostructure
Introduction
Wound healing process is an intricate and essential regulated sequence of several well-orchestrated biochemical and cellular phenomena to restore the integrity of the skin. During the wound healing process, wound progresses within three differentiated, though overlapping stages: inflammation, proliferation (neo-angiogenesis, granulation, re-epithelialization), and maturation (extracellular matrix [ECM] remodeling).1–3 Wound management and the efficacy of wound healing in occlusion of the injured tissue can considerably depend on the materials used in the wound dressing.4 Traditional wound healing therapies have been investigated experimentally and clinically, and a wealth of information about the role of traditional therapies in alleviating the underlying causes of nonhealing wounds is found in several studies.5–7 Medicinal plants can be taken into account as the potent and promising therapeutics for improvement of wound healing processes based on the variety of the active and effective components such as flavonoids, essential oils, alkaloids, phenolic compounds, terpenoids, fatty acids, and so on.7 These traditional medicines can be preferred over modern therapy due to the low cost, limited adverse effects, bioavailability, and efficacy.8,9
Beside the advantages of medicinal plants for wound management, one of the promising ways to promote their efficacy is to subject them to nanosizing process or incorporate them into nanostructures. Nanomaterials possess unique characteristics due to their nanoscale size and the high surface area to volume ratio, and nanosizing the medicinal plants can occur in association with modification in their physical and chemical characteristics.10,11
Natural product-based compounds can be used directly as medicaments for alleviating the wound or as drug carriers for delivery of other therapeutics.12 The advantageous efficacy of the nanostructured medicinal plants stimulated the authors to provide a comprehensive review of the plant-based nanomaterials obtained by different methods and their therapeutic targets in regulating the wound healing process.
Search strategy and study design
A comprehensive literature review was carried out by the authors in the electronic databases of Scopus, PubMed, ScienceDirect, and Cochrane Central Register of Controlled Trials. The search was conducted without time restriction and using the following search strings: “Wound” in the title, and “Herb” OR “Plant” OR “Phytochemical” in the title, abstract, and keywords. On PubMed, all these words were searched in “title, abstract”. The reference lists of the former review articles and the retrieved papers were manually reviewed for additional applicable studies. The initial search results, including 6,622 reports, were recorded for investigating whether they used herbal-based nanomaterials. Two individual authors initially assessed the papers based on their title and abstract. In this step, regarding the titles and abstracts, the duplicate articles, review papers, non-English papers, and the papers which were irrelevant to the topic or were not in nanoscale range (totally 6,446 papers) were excluded. The full text of the retrieved articles (176 reports) was carefully examined by the authors to examine the potential of inclusion in the current review, and 96 papers were excluded in this step based on the full text. The exclusion criteria were as follows: the papers that included plant extracts and phytochemicals which have not undergone nanosizing process; the papers that reported nanoformulations containing biomaterials which were not of plant-based origin; and the papers that included plant-derived nanoformulations which were not directly evaluated for wound healing effect and the involved mechanisms. Finally, 80 original articles, which have reported nanoscale wound healing process based on herbal substances, were extracted from the search results to be used as the main source of study in this paper. The diagram of the search study procedure is illustrated in Figure 1.
  | Figure 1 Search diagram of study selection. |
Foremost methods used for producing natural product-based nanomedicine for wound healing
Different nanostructured formulations have been successfully produced to help in natural wound healing (Figure 2). Here we discuss the foremost methods in detail.
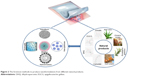  | Figure 2 The foremost methods to produce nanoformulations from different natural products. |
Electrospinning
The porous structure and excellent pore interconnectivity of nanofibers make them desirable for wound dressing and wound healing due to their oxygen permeability, the ability of keeping the moisture at the desired level, their inhibitory effect on the exogenous microorganism invasions, their conformity to skin at the wound site, and their ability to alleviate scars.13–15 Incorporating the herbal extracts and phytochemicals in the nanofibrous membranes has been carried out in several studies, which superposed the advantages of these structures and the benefits of herbal compounds for ameliorating different wounds.16–22 Emodin, 3,8-trihydroxy-6-methyl-anthraquinone, an extract of some medicinal plants (such as Polygonum and Aloe vera), has been frequently used for treating the wounds. It has several advantages such as anti-inflammatory and antibacterial activity, ability to increase the rate of migration of fibroblasts into the wounded region, and ability to enhance the nucleotide excision repair of DNA damage in human cells.23,24 The incorporation of this compound into the polyvinylpyrrolidone nanofibrous nonwoven membrane produced a promising wound healing structure for treating acute full-thickness skin wound, and the drug was well distributed on the porous membrane structure.23 Electrospinning of polyvinylpyrrolidone/emodin gave the favorable, nontoxic, non-allergenic, and highly biocompatible nanofibrous membrane with a considerably higher dissolution rate of emodin in comparison to the pure drug. The effect of this nanostructure on the full-thickness skin wound in rats promoted fluid retainment and continuity of re-epithelialization with shrinkage of the wound area, in comparison to the free drug. It was also successful in accelerating the wound healing process.25 Incorporation of emodin in a nanostructure of cellulose acetate (CA) fiber exhibited the potential to enhance the synthesis of collagen from human dermal fibroblast adults cells >100%.26 CA is a highly hydrophilic derivative of cellulose with a high potential to absorb water. The electrospun CA nanofibers provided a biocompatible environment for attachment and proliferation of L929 skin fibroblasts.27 In spite of its advantages, its low breaking stress, strain, and poor resistance are the limitations in clinical use of this biopolymer, and it should be electrospun in combination with other biomedical materials for wound healing applications.28–30 Nanostructured wound dressings based on CA have been prepared by co-electrospinning of this biopolymer with polyester urethane,31,32 gelatin,33,34 poly (ε-caprolactone) (PCL)/polyurethane and dextran,35–37 polyurethane and zein,28 and polylactic acid (PLA).38,39 The polyhexamethylene biguanide-loaded nanofibrous membrane of CA and polyether urethane showed strong antibacterial activity, good moisture retention and air permeability, good physical and mechanical properties, and accelerated the wound healing process. The presence of CA in nanofibrous membrane increased the water uptake of wound dressing and prepared a moist environment for the wound, increased adhesion to the rat skin fibroblast, and supported the rapid regeneration of epidermal layer.31 Co-electrospun CA and gelatin membranes can successfully simulate fibrous ECM, which is a complex combination of proteins and polysaccharides. In addition to their ability to mimic the fibrous structure of the native dermis, they can increase the proliferation of human dermal fibroblast, which is necessary for the regeneration stage and healing of any wound; they also possess high fibroblast affinity and collagen secretion, which make them appropriate for healing different types of skin injuries.33 Higher affinity for the proliferation of fibroblasts and bioactivity was also observed in the presence of CA in PLA nanofibrous membranes.38 This could be due to the high hydrophilicity of CA, which promotes cellular interaction. The CA/PLA nanofibrous membranes were also potent in accelerating the re-epithelialization of wounds in mice and increasing the rate of wound closure in comparison with the control.38 Incorporating the herbal therapeutic agents in CA nanofibers leads to combining the advantages of these biomaterials as an interactive wound dressing material. Asiaticoside (one of the major phytoconstituents of Centella asiatica), a trisaccharide triterpene, was loaded on CA nanofibers and provided advantageous antioxidative effect at the initial stage of wound healing.40 Asiaticoside showed a significant effect in the proliferation and production of types I and III procollagen mRNAs and also increased the levels of corresponding proteins of skin fibroblasts.41,42 The other herbal compound loaded on the CA was curcumin, which enhanced the attachment and proliferation of fibroblasts, increased the amount of collagen synthesis, and protected the normal human dermal fibroblast cells against H2O2-induced oxidative stress.43 Curcumin (1,7-bis (4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione), an active ingredient of turmeric, is a polyphenolic compound obtained from Curcuma longa L. Curcumin is an active ingredient possessing a broad range of innate biological activities such as anti-inflammatory, antibacterial, antioxidant, anticancer, and angiogenic effects,44 which make it a valuable agent for treating wounds. Curcumin has long been used in clinical studies and different in vivo animal models for accelerating cutaneous wound healing.45 In the in vivo studies on animals treated with curcumin, this phytochemical showed its activity in early re-epithelialization through increasing the rate of collagen synthesis due to upregulating the level of transforming growth factor (TGF)-β1 growth factors,46 increasing the granulation tissue and blood vessels,47 enhancing neovascularization, increasing the fibroblast and vascular densities,48 and accelerating the migration of cells.49 The ethanolic extract of curcumin caused the tissue debris and hemorrhages disappear and formed keratin layer on the epidermal surface of the wound in Black Bengal goats.45
However, the low insolubility of curcumin, poor absorption, and instability cause inherent limitations impeding the use of curcumin alone.50 Incorporating curcumin into hydrophilic nanoformulations is a useful way to circumvent unwanted properties of this herbal compound.51,52 The incorporation of curcumin into gelatin biomimetic nanofibrous mats was studied in acute wound in rats.53
Green-synthesized metal nanoparticles using plants
Metal-based nanoparticles are extensively used in diverse fields such as engineering, chemistry, biology, and medicine.54–56 There is growing interest on the biological applications of metal nanoparticles in medicine and pharmacy. Tremendous growth in these expanding applications has opened applied frontiers and novel methods for synthesis of these nanoparticles, including physical and chemical methods. Most of these methods are expensive and environmentally hazardous due to the application of toxic and perilous chemicals with high biological risks.57–59 The biologically inspired experimental processes have been evolved to overcome these drawbacks and are more acceptable in medical applications due to high biocompatibility, biodegradability, nontoxicity, and the green nature of the agents, and also their cost-effectiveness. The most common metallic nanoparticles in biomedical and medicinal applications are silver- and gold-based nanostructures (nanoparticles, nanocomposites, and nanocoating), which have drawn the attention of researchers. Silver nanoparticles (AgNPs) and different silver salts have recently intrigued medical scientists in the fields of clinical and fundamental researches due to their excellent antibacterial and antimicrobial activities,60 which could be attributed to their large surface area to volume ratio, and could be remarkably interesting due to growing microbial resistance against metal ions, antibiotics, and the development of resistant strains.61 In addition to these characteristics, these nanoparticles possess the advantages of high chemical stability, antiviral, antifungal, and anti-inflammatory activities, and possibility to be incorporated into different composite structures, cosmetic products, food industry, and so on.57,62,63 These plant-mediated biosynthesized nanoparticles exhibit potential in wound healing and in efficiently retarding and preventing bacterial infections.64,65 The biological methods for preparation of AgNPs are based on the administration of reducing agents such as bacteria, fungi, and plant extracts to interact with the Ag ions and reduce them into AgNPs. A large variety of herbal extracts have been used to develop the green-synthesized AgNPs and prepare proper medications for wound. Some of the prominent examples of these plants are Cassia roxburghii,66 Drosera binata Labill.,67 Indigofera aspalathoides DC.,68 Azadirachta indica A. Juss.,69 Arnebia nobilis Rech.f.,70 Melia dubia Cav.,71 Terminalia chebula Retz.,72 Lansium domesticum Corrêa,73 Orchidantha chinensis T.L.Wu,74 A. vera (L.) Burm.f.,75 glucuronoxylan,76 Momordica charantia L.,77,78 Carica papaya L.,79 Cymbopogon citratus (DC.) Stapf,80 Nyctanthes arbor-tristis L.,81 Naringi crenulata (Roxb.) Nicolson,82 Phytophthora infestans,83 Biophytum sensitivum,84 Propolis,85 and Bryonia laciniosa L.86
Most of these extracts possess inherent antibacterial and antimicrobial activity, which makes them appropriate for wound care. The importance of the presence of antimicrobial agents in wound dressing is their substantial role in controlling the microorganism colonization and subsequent proliferation, which helps accelerating wound healing process.67,87–89 In vivo treatment of mice with AgNPs, synthesized with Catharanthus roseus leaf extract, could successfully control bacterial and fungal growth, prompt the closing of wound, and considerably reduce the wound site.90 In addition to the extracts applied for green synthesis of AgNPs, the antibacterial endophytic fungus of O. chinensis was also advantageous in biosynthesis of AgNPs and treated the infected wounds developed on the Sprague Dawley rats.74 It produced well-stabilized AgNPs and inhibited different bacterial strains by metabolizing special proteins as well. Additionally, it successfully accelerated wound healing process, enhanced the wound contraction rate, completely re-epithelialized the epidermis, minimized the scars after treatment period, minimized the bacterial count in the infected wound site, and downregulated the level of proinflammatory cytokines tumor necrosis factor alpha (TNF-α), interleukin (IL)-1β, and IL-6.
The other parameter which can be controlled to accelerate the wound healing process is the rate of synthesis of collagen.73 The first step in synthesis of collagen is hydroxylation of proline to form hydroxyproline.91 Thus, the hydroxyproline content is a good marker for collagen deposition, which has been determined in several studies on the application of green-synthesized AgNPs. Phytosynthesized AgNPs by L. domesticum fruit peel extract were potent in the enhancement of hydroxyproline content, a marker of collagen deposition, as well as the wound closure time. Complete epithelialization with keratinization as well as fibrous connective tissue proliferation were also the results of administration of nanoparticles.73 The level of hydroxyproline and total proteins at the initial phase of wound healing was also increased in the presence of biosynthesized AgNPs, which confirmed the effect of these biosynthesized nanoparticles on cellular hyperplasia and deposition of matrix proteins in granulation tissues.74 The linseed hydrogel was another choice for the green synthesis of AgNPs, possessing the advantages of increasing the wound closure percentage and also the collagen content at the wound site.60
Cassia auriculata L.-mediated AgNPs were effective on both incision and excision wound models in Wistar albino rats. Although the wound healing activity of Cas. auriculata extract alone has been established in the literature, the Cas. auriculata AgNPs exhibited better performance in wound healing process rather than the extract and also Povidone Iodine ointment. Nanoparticles were also more effective on enhancement of excision wound contraction.92
Wound tensile strength is a key parameter governing neocollagen production, and the quality and speed of tissue regeneration is directly related to the collagen content of wounds.93 Biosynthesized AgNPs can promisingly be applied to enhance the collagen content and tensile strength of wounds. The administration of L. domesticum-mediated and linseed-mediated AgNPs to the animals increased the tensile strength of wound due to organization of collagen fibers.60,73 Guar gum/AgNPs were other phytosynthesized medications possessing significant effect on the tensile strength and modulation of collagen deposition, in addition to regulation of keratinocytes and accelerating the essential re-epithelialization process.94
In addition to the abovementioned nanostructures, green-synthesized AgNPs can be included into/or coated on the common dressings, electrospun nanofibers, and nanofibrous membranes. This integrates the advantages of electrospun nanostructures, AgNPs, and the medicinal plants. M. charantia fruit extract was used to prepare AgNPs by biological reduction, and then, addition of PLA to AgNPs caused stabilization of the Ag particles and made them electrospinnable. The electrospun nanofibers were found to be capable of wound healing and were highly efficient against bacteria and highly cytocompatible. Although AgNPs alone possessed antibacterial activity, the presence of M. charantia extract caused potentiation of antimicrobial activity as well as reduction of cytotoxic activity against fibroblasts.77 AgNPs prepared using Piper nigrum leaf extracts were included in PCL membrane and this nanostructure was found highly promising in inhibiting bacterial colonization in wounds.95
Green-synthesized titanium dioxide, gold, and copper oxide nanoparticles are other wound healing enhancers, which cause rapid wound healing and prevent/decrease infections and posttreatment side effects. Gold nanoparticles synthesized by Coleus forskohlii root extract remarkably accelerated re-epithelialization of excision wound created on rats, increased connective tissue formation, and promoted the rate of proliferation and migration of epidermal cells.96 The synthesis of titanium dioxide nanoparticles (TiNPs) in the presence of Moringa oleifera leaf extract enhanced wound contraction and reduced the excision wound site in Albino rats.97 Similar results were observed in Albino rats after treating the excision wound with biosynthesized copper oxide nanoparticles using Ficus religiosa leaf extract. These nanostructures also possessed persistent inhibitory activity against human pathogenic bacteria, and effectively increased the formation of macrophages, fibroblast, and collagen fibers.98
Incorporation of natural products in different forms of nanoparticles to achieve a controlled delivery system to the wound site
Another common technique for utilizing the medicinal plants for wound healing involves their incorporation into different forms of nanoparticles. In drug delivery systems, control over the release of drug at the target organ is critically important. Several forms of biocompatible drug carriers are used in controlled drug delivery systems, including nanoparticles, nanoemulsion, nanohydrogels, nanofilms, and nanoliposomes. These systems are capable of diminishing the side effects and increasing the efficacy of different therapeutic agents by providing a sustained and controlled delivery system, and they increase the dissolution rate of the drugs based on their surface characteristics.99–105 Besides the advantages of the herbal-based compounds in wound healing process, nanosizing these therapeutics or incorporation of these materials in nanoparticles provides a chance of controlling their delivery to the injured side and can increase their chemical activity.106 For instance, nanosizing the curcumin particles provided a well-regulated and sustained delivery system and enhanced their wound healing activity by increasing the antimicrobial effect and accelerating the formation of granular tissues and collagen synthesis.99,107
Nanoemulsions are nanostructures that provide the possibility to nanosize different essential oils. Upon application of nanoemulsions on the injured sites, the droplets form a film on the injured sites after evaporation of their water content. The plant essential oils have attracted considerable attention because of their high content of bioactive components.108 The essential oil of Eucalyptus globulus was nanosized by nanoemulsification method, which promoted its antibacterial activity. The high activity and accelerated wound healing could be attributed to the existence of 45.4% 1,8-cineole (eucalyptol), which is known to be an active compound facilitating the penetration in transdermal and topical drug delivery systems.109 Similar results were obtained in the in vitro and in vivo studies on application of tragacanth gum nanoemulsions impregnated with A. vera extract, which was attributed to the potential of A. vera in modulation of proteases.110 The encapsulation of active components of medicinal plants into nanoemulsions has provided a new approach for controlled drug delivery. A vital issue in the topical administration of lipophilic drugs for wounds is the localization of these ingredients in the superficial skin layers. Encapsulation of these drugs into nanoemulsions facilitates their penetration into the skin layers and provides a dispersed oil droplet phase to improve their solubility.111–113
Nanohydrogels are a group of nanostructures for wound dressing that offer discrete advantages of high flexibility, high hydrophilicity, high mechanical strength, tunable structure, and the ability to absorb wound exudates as well as permeate oxygen and prevent wound dehydration.114,115 Due to their porous structure, they can be considered as another promising nanostructure for providing a sustained and controlled delivery system to the wound.116 Li et al prepared a sticky micro/nanohydrogel from alginate-gum arabic. Adhesive nanohydrogels possess the ability to bind to the injured tissues, and can successfully act as hemostat and provide a microenvironment to facilitate the proliferation, differentiation, and migration of cells.117 As mentioned, curcumin as an active herbal compound suffers from the drawback of low water solubility and low bioavailability, which restrict its therapeutic applications. One promising method to overcome this drawback of curcumin and other hydrophobic compounds is to incorporate them in the aqueous nanostructures such as nanohydrogels. Incorporation of curcumin in polyethylene glycol (PEG)-PCL and PEG-PCL-PEG hydrogels improved the solubility and bioavailability of the drug, accelerated the re-epithelialization of wound, and regulated the granulation tissues.118
In addition to the mentioned formulations, nanoliposomes can be considered as the nanostructures which could be utilized to improve the solubility and efficacy of poorly soluble herbal-based therapeutics. Nanoliposomes (liposomes in nanometric scales) are colloidal structures composed of lipids and/or phospholipid bilayers encapsulating aqueous compartment(s), and they possess the ability to improve bioavailability, cause sustained transdermal delivery of different medicinal compounds, and overcome the possible drug overdose and the toxicity.119,120 Modification of nanoliposomes with polymers for dermal delivery of therapeutics causes the promotion of skin permeability and prolongs the retention time. PEG is one of the polymers successfully used in association with liposomes to enhance the dermal delivery of natural compounds. Entrapment of curcumin in PEG-nanoliposomes promisingly prolonged its anti-inflammatory activity, promoted the permeation rate into the dermal layers, and accelerated the wound closure.120,121
Cellular and molecular mechanisms involved in the wound healing potential of herbal-based nanostructures
Antioxidative stress
Oxygen is an important local factor which is critical for the wound healing process due to its vital role in different stages of wound healing by mediating angiogenesis, enhancing re-epithelialization, and increasing collagen synthesis and fibroblast proliferation.122,123 Reactive oxygen species, such as hydrogen peroxide (H2O2) and superoxide (O2-), by-products of oxygen metabolism in the human body, are important regulators of wound healing.124,125 For maintaining the levels of free radicals at the desired level, medicinal plants are promising choices due to their inherent antioxidant potential, which are potent in regulating enzymes SOD, glutathione peroxidase, and catalase. The herbal-based compounds, plant extracts and essential oils, such as curcumin, genistein, cellulose (extracted from Citrus reticulate), Asiaticoside, Chromola enaodorata extract, Co. forskohlii root extract, black seed oil, and wheat germ oil, in wound dressings inhibit the oxidative stress and exert antioxidant activity.40,89,96,120,121 Curcumin is known as a potent antioxidant compound, and nanosizing this herbal-based compound can be achieved without any significant alteration in its intrinsic antioxidant activity. This has been recognized by Li et al who observed <1% reduction in the antioxidant activity of nanocurcumin incorporated into chitosan/alginate hydrogels, in comparison to the unmodified curcumin (native form).119 The inclusion of curcumin in wound dressings significantly decreased the wound oxidative stress by diminishing the SOD level.118 In another study, bioconjugation of AgNPs by Cat. roseus leaf extract caused strong antioxidant activity, rather than the conventional AgNPs, due to adherence of the functional groups of Cat. roseus extract to the nanoparticles.90 Fraxinus angustifolia bark and leaf extracts showed antioxidant activity against H2O2-induced oxidative stress in both in vitro and in vivo studies, which was due to the presence of a high content of quercetin and tannic acid, two of the most potent antioxidants. Apart from these results, the ability of these extracts was proved in chelating ferrous ions and this inhibited their activity to convert peroxides to free radicals. Their high chelating activity could be another reason for their antioxidative ability in the wound tissue. The higher cellular uptake of nanoformulated samples led to higher antioxidant activity and better wound healing function of these structures.126
Anti-inflammatory activity
Inflammation is involved in the first stage of wound healing process, which is characterized by the migration of leukocytes into the wound, and mainly starts by the aggregation of platelets followed by infiltration of leukocytes. Leukocytes are indispensable cellular components involved in the inflammatory response, which affect the pathogens, tissue degradation, and tissue formation, as well.127 The inflammatory response is crucial for the healing process, which orchestrates the cellular cascades associated with wound healing by supplying the growth factor as well as cytokine signals.3 On the other hand, in the physiological inflammatory response, inflammatory cells can cause preventive and inhibitory effects against bacterial invasion and debris degradation, as well.128
The infiltration of cells at the wound site occurs after the invasion of monocytes into the wound tissue and their differentiation into macrophages. Macrophages can provide several growth factors and proinflammatory cytokines such as IL-1α and IL-1β, TNF-α, platelet derived growth factor (PDGF), TGF-α, keratinocyte growth factor, and vascular endothelial growth factor (VEGF).129 It is worth noting that inflammation can cause either advantageous (attraction of the immune system elements to the injury site, facilitating the repair of damage) or destructive (tissue dysfunction within prolonged inflammatory phase) effects during treatment of different disorders,130 and thus, control over inflammation is highly crucial. Based on the various growth factors and cytokines involved in normal and acute wound healing, the mechanism of modulation of inflammatory processes in wound repair should be cleared for different types of medications. Polyphenolic compounds are the promising candidates for regulating and modifying inflammatory responses. They have been found to be potent in regulating the levels of TNF-α, interferon-γ, and different types of ILs.131,132 Curcumin, one of the most important polyphenols, has long been used due to its high radical scavenging activity and its ability to reduce the inflammatory response, inhibit NF-kB, and downregulate TNF-α, COX2, LOX, NOS, and MMP-9, all of which play a significant role in the inflammatory phase.46,133,134 In a study, curcumin showed efficient pro-healing activity against excision wounds in rats, which was due to decreasing the level of TNF-α, increasing the level of IL-10, and up-regulating the production of TGF-β1.46 The anti-inflammatory activity of other polyphenol-rich plants, such as Fraxinus angustifolia,126 confirmed the effectiveness of this group of herbal-based products on early stages of wound healing.
Biosynthesized AgNPs and plant-loaded AgNPs are the other nanostructures possessing intrinsic anti-inflammatory activity. Bamboo cellulose nanocrystals impregnated with AgNPs were administered to mice, where they caused decreased inflammation through downregulating the levels of proinflammatory cytokines IL-6 (which is responsible for stimulating fibroblast proliferation) and TNF. The high levels of these two cytokines cause hyperinflammation and delay the wound healing process.135 The AgNPs formulated using Co. forskohlii root extract caused decrease in the number of neutrophils, macrophages, and other inflammatory cells.96 Application of B. laciniosa leaf extract in preparing AgNPs resulted in a significant decrease in the levels of proinflammatory cytokines IL-6 and IL-10 and consequently diminished the inflammation and provided scarless wound healing.86
Neovascularization and angiogenesis
After controlling inflammation, re-epithelialization, and collagen synthesis, capillary tissues, called granulation tissues, are formed due to growth of capillaries and lymphatic vessels from the pre-existing vessels at the wound site.2 Two of the most important stimuli for angiogenesis are fibroblast growth factor (FGF) and VEGF.127 VEGF induces healing by aiding in vascular permeability and prevents inflammatory cells to reach the injured site, and accelerates the proliferation and migration of endothelial cells.136
The tragacanth gum scaffold incorporated with curcumin showed high efficacy in enhancing angiogenesis and forming new blood vessels. Curcumin could be responsible for the well-increased angiogenesis.137 The angiogenesis can be triggered by green-synthesized metal nanoparticles.138 Apart from other effects of curcumin on wound healing stages, nanosizing curcumin causes enhancement in neovascularization and angiogenesis, which could be due to its effects on nitric oxide synthase.107 Gold nanoparticles are an important group that successfully triggered angiogenesis by differential regulation of growth factors such as VEGF and related protein expression.36,139,140 The green-synthesized AuNP-deposited hydrocolloid membranes showed accelerating effect on angiogenesis-related proteins by upregulating the expressions of VEGF, Ang-1, and Ang-2.121 Biosynthesized CuO nanoparticles (synthesized with F. religiosa leaf extract) and AgNPs (synthesized with guar gum and L. domesticum fruit peel extract) were also found potent in enhancing the capillary blood formation and facilitating angiogenesis and regulating proteins.73,94,98
PDGF is another growth factor possessing inhibitory effect in the wound microenvironment and regulating wound vascularization.141 Impregnation of bamboo cellulose nanocrystals by AgNPs vascular network formation induced angiogenesis by intensifying the production of VEGF and FGF (P<0.05) and also increased the PDGF level.135 The production and accumulation of these growth factors could be due to the high capability of the prepared nanostructure to absorb water and regulate the moisture content of the wound microenvironment well, which enhances the proteolytic activity and induces wound repair.142
Re-epithelialization and wound regeneration
The synthesis of collagen, from fibroblast cells, is a key factor in homeostasis, re-epithelialization, and regeneration of wound healing process. Based on this fact, the probable effective role of different plant-based nanostructures on promoting collagen synthesis has been investigated. Emodin acts as a promoter of collagen synthesis and fibroblast proliferation, after encapsulating in fibrous mats.25,26 Fenugreek, Trigonella foenum-graecum, is an herb capable of modifying the physicochemical characteristics of collagen and has been used for preparation of collagen-based materials.89 Incorporation of fenugreek in silk fibroin nanofibrous mats was another way to improve the collagen synthesis and deposition, due to the high amounts of saponins and flavonoids present in it.143
Nanosized curcumin could successfully increase collagen deposition in the granulation tissue, increase myofibroblasts and blood vessels in the granulation tissue and capillary formation, and enhance the contraction of wounds in a mouse model. This could be due to the fact that the nanostructure enhanced the bioavailability of curcumin at the wound site and was potent in absorption of growth factors and cytokines.119
Tragacanth gum causes quicker regeneration due to the faster signaling pathway, which simulates the natural ECM and causes absorption of fibroblast cells to the derma layer.137 This biopolymer comprises mineral constitutes, namely, calcium (a key factor for epidermal cell migration and regeneration), acts as promoter in normal homeostasis of the dermal cells, and successfully modulates the proliferation of keratinocytes.144 On the other hand, magnesium is effective in the motility of fibroblast cells as well as keratinocytes.145
Concluding remarks
Wounds are severe disorders affecting the quality of life of people all over the word. An efficient and fast healing process can reduce the costs and hospitalization, but achieving an ideal medicament is still an issue due to the complexity of skin tissue structure. In spite of the recent advances in the field of wound management and novel medications for wound healing and skin regeneration, traditional methods based on herbal and natural therapeutics are still known as promising alternative medications due to the diversity of active components, ease of access, and their limited side effects and low costs. There are several review studies in the literature about the role of natural products as wound dressing. Some of them have analyzed the role of biomaterials from animal origin146–148 and some others have dedicated their focus of interest on plant-based biomaterials, but not in nanosize scales.2,149,150 The potential of these natural occurring products has been well presented in these studies. In a recent review, Andreu et al100 elucidated the efficacy of natural origin compounds in combination with the common wound dressings. They have examined the current methods for preparation of nanostructured wound dressing from both natural and synthetic product polymers. Furthermore, the role of herbal medicine in management of different wounds has been reviewed in their study, while the studied compounds were not in the nanoscale. This study, for the first time, addressed the nanosizing method considerations for herbal-based natural compounds, as well as the mechanistic evaluation of the present herbal-based nanoformulations for wound dressing. In fact, this review presented a comprehensive study on the nanoformulations of herbal origin for wound management from both engineering and mechanistic points of view, while there is currently no similar review article available in the literature. Tables S1 and S2 give a summary of the plants and plant-derived phytochemicals that help in alleviation and healing process successfully. At this time, in spite of the advantages of the natural products in wound dressing and wound healing, development of these medicaments is continuously required to be improved. The appearance and development of nanoscience and technology could help to improve the efficacy of different therapeutics as well as herbal-based products. In recent years, nanostructures and nanoformulations have promisingly overcome the drawbacks of common medicaments, provide a smart healing process, regulate the release of therapeutics, reduce the doses required for healing, and provide a unique opportunity to facilitate healing even for chronic wounds. Different nanoformulation methods have long been used for producing plant-based nanostructures in the presence or absence of synthetic materials. Electrospining method successfully provides wound dressings which includes herbal compounds as the base material. The well-regulated porosity and the similarity of electrospun nanofibers to the skin tissue, in association with several other benefits such as the possibility of incorporation of various types of materials, make them the ideal dressing for wound management. According to the literature review, it can be concluded that, at this time, the plant-based electrospun nanofibers mainly facilitate the adhesion, proliferation, and differentiation of fibroblasts and keratinocytes. This could be attributed to their ability to provide ideal microenvironments due to the porous structure and promising permeability and moisture retention of these herbal-based nanostructures. Another advantage of these nanostructures is the ability for co-electrospinning of the herbal-based compounds with other biomaterials, combining the advantages of all the constituents. Application of medicinal plants and their extracts in forming metal-based biocompatible nanoparticles in a green and cost-effective method has produced a group of nanostructures with excellent antibacterial and antimicrobial activities. Different plant-mediated metal nanoparticles possess the ability of preventing the bacterial colonization as well as prolonging the anti-inflammatory effects. According to the literature, impregnation of herbal-based compounds in nanoparticles, nanoemulsion, nanoliposomes, hydrogels, and so on is one of the most effective methods for enhancing their availability, controlling their release in the form of sustained delivery system to the wound site, and enhancing the permeability of these therapeutics even to the underlying skin layers, which all are necessary to the healing process. The water-insoluble herbal-based compounds are commonly used in this method to improve their solubility and wound healing efficacy.
The most important cellular and molecular mechanisms corresponding to the herbal-based nanostructures are antioxidative stress, anti-inflammatory activity, angiogenesis, neovascularization, and re-epithelialization (Figure 3). Medicinal plants are the richest sources of antioxidants such as polyphenols and flavonoids. Nanostructures including herbal products such as curcumin, Asiaticoside, and Cat. roseus possessed radical scavenging activity and regulated the oxygen level in the wound site. Controlling the inflammatory phase could be achieved by reducing the inflammatory responses, inhibiting NF-kB, targeting inflammation pathways (intracellular transcription and transduction), and downregulating the proinflammatory cytokines. Curcumin-based nanostructures were found to be the most potent herbal-based nanostructures in inflammatory phase, regulating the levels of TNF-α, IL-10, and TGF-β1. Biosynthesized AgNPs were another group of plant-based nanostructures playing a vital role in controlling the wound inflammation. Different herbal-based compounds and extracts (such as Co. forskohlii, B. laciniosa, O. chinensis) used in the preparation of AgNPs caused a considerable anti-inflammatory activity attributed to the downregulated levels of TNF-α, IL-6, IL-1β, and IL-10. Improving angiogenesis and vascularization during the wound healing process depends on the stimulation of FGF, VEGF, and PDGF. Curcumin and plant-mediated metal nanoparticles were found to be potent in triggering angiogenesis via regulating these growth factors. Another key factor in wound healing process is the rate of re-epithelialization. Nanostructures including natural products such as emodin, fenugreek, curcumin, and tragacanth gum are some of the examples with the ability of prompting collagen synthesis and proliferation of fibroblasts, resulting in accelerated re-epithelialization. It is worth noting that although all of the prepared herbal-based nanostructures were successful in ameliorating wounds, curcumin-based nanostructures were found to be the most potent nanostructures, which play a significant role in controlling most of the wound healing stages. Among different methods used for preparing nanoformulations of phytochemicals, metal-based nanoparticles (especially AgNPs) involve the most pharmacological targets in the wound healing process, indicating their best therapeutic properties. The results of the current review article mainly confirmed the significance of natural compounds as alternative choices for healing different wounds and corroborated the success of nanotechnology in enhancing the efficacy of different medicaments. The impact of nanostructure approaches for natural wound healing agents has gained wider attention because of improvement in targeted therapy and bioavailability, as well as development of stability. Further pharmacological experiments are mandatory to evaluate the intracellular targets involved in wound healing effects of natural nanomedicine. Also, conducting well-designed clinical trials is necessary to confirm the safety and efficacy of natural product-based nanoformulations in treating wounds.
Disclosure
The authors report no conflicts of interest in this work.
References
Groeber F, Holeiter M, Hampel M, Hinderer S, Schenke-Layland K. Skin tissue engineering – in vivo and in vitro applications. Adv Drug Deliv Rev. 2011;63(4–5):352–366. | ||
Pereira RF, Bártolo PJ. Traditional therapies for skin wound healing. Adv Wound Care. 2016;5(5):208–229. | ||
Reinke JM, Sorg H. Wound repair and regeneration. Eur Surg Res. 2012;49(1):35–43. | ||
V Singh A, As A, N Gade W, et al. Nanomaterials: new generation therapeutics in wound healing and tissue repair. Curr Nanosci. 2010;6(6):577–586. | ||
Devi K, Santhi M, Umadevi U. Phytochemical analysis of selected wound healing medicinal plants. Int J Pharm Sci Res. 2017;8(2):852. | ||
Bahmani M, Asadi-Samani M. A short look to the most important medicplants effective on wound healing. J Inj Inflamm. 2016;1(1):e07. | ||
Bahramsoltani R, Farzaei MH, Rahimi R. Medicinal plants and their natural components as future drugs for the treatment of burn wounds: an integrative review. Arch Dermatol Res. 2014;306(7):601–617. | ||
Gamit R, Nariya M, Acharya R, Shukla VJ. Wound healing potential of some medicinal plants with their screening models: a review. Pharma Sci Monit. 2017;8(1):208–227. | ||
Hosein FM, Abbasabadi Z, Reza S-AM, Abdollahi M, Rahimi R. A comprehensive review of plants and their active constituents with wound healing activity in traditional Iranian medicine. Wounds. 2014;26(7):197–206. | ||
Alvarez-Román R, Naik A, Kalia YN, Guy RH, Fessi H. Skin penetration and distribution of polymeric nanoparticles. J Control Release. 2004;99(1):53–62. | ||
Mordorski B, Prow T. Nanomaterials for wound healing. Curr Dermatol Rep. 2016;5(4):278–286. | ||
Li S, Zhang T, Xu W, et al. Sarcoma-targeting peptide-decorated polypeptide nanogel intracellularly delivers shikonin for upregulated osteosarcoma necroptosis and diminished pulmonary metastasis. Theranostics. 2018;8(5):1361–1375. | ||
Ehterami A, Salehi M, Farzamfar S, et al. In vitro and in vivo study of PCL/COLL wound dressing loaded with insulin-chitosan nanoparticles on cutaneous wound healing in rats model. Int J Biol Macromol. 2018;117:601–609. | ||
Zhang J, Zheng T, Alarçin E, et al. Porous electrospun fibers with self-sealing functionality: an enabling strategy for trapping biomacromolecules. Small. 2017;13(47):1701949. | ||
Naseri-Nosar M, Farzamfar S, Sahrapeyma H, et al. Cerium oxide nanoparticle-containing poly (ε-caprolactone)/gelatin electrospun film as a potential wound dressing material: in vitro and in vivo evaluation. Mater Sci Eng C Mater Biol Appl. 2017;81:366–372. | ||
Cerchiara T, Abruzzo A, Ñahui Palomino RA, et al. Spanish Broom (Spartium junceum L.) fibers impregnated with vancomycin-loaded chitosan nanoparticles as new antibacterial wound dressing: preparation, characterization and antibacterial activity. Eur J Pharm Sci. 2017;99:105–112. | ||
Lin L, Perets A, Har-El YE, et al. Alimentary ‘green’ proteins as electrospun scaffolds for skin regenerative engineering. J Tissue Eng Regen Med. 2013;7(12):994–1008. | ||
Selvaraj S, Fathima NN. Fenugreek incorporated silk fibroin nanofibers – a potential antioxidant scaffold for enhanced wound healing. ACS Appl Mater Interfaces. 2017;9(7):5916–5926. | ||
Suganya S, Senthil Ram T, Lakshmi BS, Giridev VR. Herbal drug incorporated antibacterial nanofibrous mat fabricated by electrospinning: an excellent matrix for wound dressings. J Appl Polym Sci. 2011;121(5):2893–2899. | ||
Vargas EAT, do Vale Baracho NC, de Brito J, de Queiroz AAA. Hyperbranched polyglycerol electrospun nanofibers for wound dressing applications. Acta Biomater. 2010;6(3):1069–1078. | ||
Yang Y, Wang F, Yin D, Fang Z, Huang L. Astragalus polysaccharide-loaded fibrous mats promote the restoration of microcirculation in/around skin wounds to accelerate wound healing in a diabetic rat model. Colloids Surf B Biointerfaces. 2015;136:111–118. | ||
Yousefi I, Pakravan M, Rahimi H, Bahador A, Farshadzadeh Z, Haririan I. An investigation of electrospun henna leaves extract-loaded chitosan based nanofibrous mats for skin tissue engineering. Mater Sci Eng C Mater Biol Appl. 2017;75:433–444. | ||
Tang T, Yin L, Yang J, Shan G, Emodin SG. Emodin, an anthraquinone derivative from Rheum officinale Baill, enhances cutaneous wound healing in rats. Eur J Pharmacol. 2007;567(3):177–185. | ||
Lu HM, Ni WD, Liang YZ, Man RL. Supercritical CO2 extraction of emodin and physcion from Polygonum cuspidatum and subsequent isolation by semipreparative chromatography. J Sep Sci. 2006;29(14):2136–2142. | ||
Dai X-Y, Nie W, Wang Y-C, Shen Y, Li Y, Gan S-J. Electrospun emodin polyvinylpyrrolidone blended nanofibrous membrane: a novel medicated biomaterial for drug delivery and accelerated wound healing. J Mater Sci Mater Med. 2012;23(11):2709–2716. | ||
Panichpakdee J, Pavasant P, Supaphol P. Electrospun cellulose acetate fiber mats containing emodin with potential for use as wound dressing. Chiang Mai J Sci. 2016;43(1):1249–1259. | ||
Nosar MN, Salehi M, Ghorbani S, Beiranvand SP, Goodarzi A, Azami M. Characterization of wet-electrospun cellulose acetate based 3-dimensional scaffolds for skin tissue engineering applications: influence of cellulose acetate concentration. Cellulose. 2016;23(5):3239–3248. | ||
Unnithan AR, Gnanasekaran G, Sathishkumar Y, Lee YS, Kim CS. Electrospun antibacterial polyurethane–cellulose acetate–zein composite mats for wound dressing. Carbohydr Polym. 2014;102:884–892. | ||
Li J, Feng X, Liu B, et al. Polymer materials for prevention of postoperative adhesion. Acta Biomater. 2017;61:21–40. | ||
Farzamfar S, Naseri-Nosar M, Vaez A, et al. Neural tissue regeneration by a gabapentin-loaded cellulose acetate/gelatin wet-electrospun scaffold. Cellulose. 2018;25(2):1229–1238. | ||
Liu X, Lin T, Gao Y, et al. Antimicrobial electrospun nanofibers of cellulose acetate and polyester urethane composite for wound dressing. J Biomed Mater Res B Appl Biomater. 2012;100B(6):1556–1565. | ||
Liu X, Lin T, Fang J, et al. In vivo wound healing and antibacterial performances of electrospun nanofibre membranes. J Biomed Mater Res A. 2010;94(2):499–508. | ||
Vatankhah E, Prabhakaran MP, Jin G, Mobarakeh LG, Ramakrishna S. Development of nanofibrous cellulose acetate/gelatin skin substitutes for variety wound treatment applications. J Biomater Appl. 2014;28(6):909–921. | ||
Samadian H, Salehi M, Farzamfar S, et al. In vitro and in vivo evaluation of electrospun cellulose acetate/gelatin/hydroxyapatite nanocomposite mats for wound dressing applications. Artif Cells Nanomed Biotechnol. 2018;44:1–11. | ||
Liao N, Unnithan AR, Joshi MK, et al. Electrospun bioactive poly (ε-caprolactone)–cellulose acetate–dextran antibacterial composite mats for wound dressing applications. Colloids Surf A Physicochem Eng Asp. 2015;469:194–201. | ||
Lee J, Kim HY, Zhou H, et al. Green synthesis of phytochemical-stabilized Au nanoparticles under ambient conditions and their biocompatibility and antioxidative activity. J Mater Chem. 2011;21(35):13316–13326. | ||
Unnithan AR, Barakat NA, Pichiah PB, et al. Wound-dressing materials with antibacterial activity from electrospun polyurethane-dextran nanofiber mats containing ciprofloxacin HCl. Carbohydr Polym. 2012;90(4):1786–1793. | ||
Gomaa SF, Madkour TM, Moghannem S, El-Sherbiny IM. New polylactic acid/cellulose acetate-based antimicrobial interactive single dose nanofibrous wound dressing mats. Int J Biol Macromol. 2017;105:1148–1160. | ||
Khalifa SF. Fabrication and characterization of antibacterial herbal drug-loaded polylactic acid/cellulose acetate composite nanofiberous for wound dressing application[dissertation]. The American University in Cairo; 2016. | ||
Suwantong O, Ruktanonchai U, Supaphol P. In vitro biological evaluation of electrospun cellulose acetate fiber mats containing asiaticoside or curcumin. J Biomed Mater Res A. 2010;94(4):1216–1225. | ||
Maquart F-X, Bellon G, Gillery P, Wegrowski Y, Borel J-P. Stimulation of collagen synthesis in fibroblast cultures by a triterpene extracted from Centella Asiatica. Connect Tissue Res. 1990;24(2):107–120. | ||
Panichpakdee J, Pavasant P, Supaphol P. Electrospinning of asiaticoside/2-hydroxypropyl-β-cyclodextrin inclusion complex-loaded cellulose acetate fiber mats: release characteristics and potential for use as wound dressing. Polym Kor. 2014;38(3):338–350. | ||
Suwantong O, Opanasopit P, Ruktanonchai U, Supaphol P. Electrospun cellulose acetate fiber mats containing curcumin and release characteristic of the herbal substance. Polymer. 2007;48(26):7546–7557. | ||
Momtazi-Borojeni AA, Haftcheshmeh SM, Esmaeili S-A, Johnston TP, Abdollahi E, Sahebkar A. Curcumin: a natural modulator of immune cells in systemic lupus erythematosus. Autoimmun Rev. 2017;17(2):125–135. | ||
Miah M, Hasan M, Sarker Y, Alam M, Juyena N. Clinical evaluation of ethanolic extract of curcumin (Curcuma longa) on wound healing in Black Bengal goats. J Adv Vet Anim Res. 2017;4(2):1. | ||
Prasad R, Kumar D, Kant V, Tandan SK, Kumar D. Curcumin enhanced cutaneous wound healing by modulating cytokines and transforming growth factor in excision wound model in rats. Int J Curr Microbiol Appl Sci. 2017;6(7):2263–2273. | ||
Sidhu GS, Singh AK, Thaloor D, et al. Enhancement of wound healing by curcumin in animals. Wound Repair Regen. 1998;6(2):167–177. | ||
Jagetia GC, Rajanikant GK. Curcumin treatment enhances the repair and regeneration of wounds in mice exposed to hemibody gamma-irradiation. Plast Reconstr Surg. 2005;115(2):515–528. | ||
Goel A, Kunnumakkara AB, Aggarwal BB. Curcumin as “Curecumin”: from kitchen to clinic. Biochem Pharmacol. 2008;75(4):787–809. | ||
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007;4(6):807–818. | ||
Bui HT, Chung OH, dela Cruz J, Park JS. Fabrication and characterization of electrospun curcumin-loaded polycaprolactone-polyethylene glycol nanofibers for enhanced wound healing. Macromol Res. 2014;22(12):1288–1296. | ||
Lin Y-H, Lin J-H, Hong Y-S. Development of chitosan/poly-γ-glutamic acid/pluronic/curcumin nanoparticles in chitosan dressings for wound regeneration. J Biomed Mater Res B Appl Biomater. 2017;105(1):81–90. | ||
Dai X, Liu J, Zheng H, et al. Nano-formulated curcumin accelerates acute wound healing through Dkk-1-mediated fibroblast mobilization and MCP-1-mediated anti-inflammation. NPG Asia Mater. 2017;9(3):e368. | ||
Schrand AM, Rahman MF, Hussain SM, Schlager JJ, Smith DA, Syed AF. Metal-based nanoparticles and their toxicity assessment. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2010;2(5):544–568. | ||
Chekman I, Ulberg Z, Gorchakova N, et al. The prospects of medical application of metal-based nanoparticles and nanomaterials. Lik Sprava. 2011;1(2):3–21. | ||
Bahadar H, Maqbool F, Niaz K, Abdollahi M. Toxicity of nanoparticles and an overview of current experimental models. Iran Biomed J. 2016;20:(1):1. | ||
Ahmad A, Mukherjee P, Senapati S, et al. Extracellular biosynthesis of silver nanoparticles using the fungus Fusarium oxysporum. Colloids Surf B Biointerfaces. 2003;28(4):313–318. | ||
Huang J, Li Q, Sun D, et al. Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology. 2007;18(10):105104. | ||
Sumi Maria B, Devadiga A, Shetty Kodialbail V, Saidutta MB, Maria BS, Kodialbail VS. Synthesis of silver nanoparticles using medicinal Zizyphus xylopyrus bark extract. Appl Nanosci. 2015;5(6):755–762. | ||
Haseeb MT, Hussain MA, Abbas K, et al. Linseed hydrogel-mediated green synthesis of silver nanoparticles for antimicrobial and wound-dressing applications. Int J Nanomedicine. 2017;12:2845–2855. | ||
Kaviya S, Santhanalakshmi J, Viswanathan B. Green synthesis of silver nanoparticles using Polyalthia longifolia leaf extract along with D-sorbitol: study of antibacterial activity. J Nanotechnol. 2011;2011(4):1–5. | ||
Choudhary MK, Kataria J, Cameotra SS, Singh J. A facile biomimetic preparation of highly stabilized silver nanoparticles derived from seed extract of Vigna radiata and evaluation of their antibacterial activity. Appl Nanosci. 2016;6(1):105–111. | ||
Ahmed S, Ahmad M, Swami BL, Ikram S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: a green expertise. J Adv Res. 2016;7(1):17–28. | ||
Fong J, Wood F. Nanocrystalline silver dressings in wound management: a review. Int J Nanomedicine. 2006;1(4):441–449. | ||
Thomas R, Soumya KR, Mathew J, Radhakrishnan EK. Electrospun polycaprolactone membrane incorporated with biosynthesized silver nanoparticles as effective wound dressing material. Appl Biochem Biotechnol. 2015;176(8):2213–2224. | ||
Pannerselvam B, Dharmalingam Jothinathan MK, Rajenderan M, et al. An in vitro study on the burn wound healing activity of cotton fabrics incorporated with phytosynthesized silver nanoparticles in male Wistar albino rats. Eur J Pharm Sci. 2017;100(Supplement C):187–196. | ||
Krychowiak M, Grinholc M, Banasiuk R, et al. Combination of silver nanoparticles and Drosera binata extract as a possible alternative for antibiotic treatment of burn wound infections caused by resistant Staphylococcus aureus. PLoS One. 2014;9(12):e115727. | ||
Arunachalam KD, Annamalai SK, Arunachalam AM, Kennedy S. Green synthesis of crystalline silver nanoparticles using Indigofera aspalathoides-medicinal plant extract for wound healing applications. Asian J Chem. 2013;25(Supplementary Issue):S311. | ||
Bansod SD, Bawaskar MS, Gade AK, Rai MK. Development of shampoo, soap and ointment formulated by green synthesised silver nanoparticles functionalised with antimicrobial plants oils in veterinary dermatology: treatment and prevention strategies. IET Nanobiotechnol. 2015;9(4):165–171. | ||
Garg S, Chandra A, Mazumder A, Mazumder R. Green synthesis of silver nanoparticles using Arnebia nobilis root extract and wound healing potential of its hydrogel. Asian J Pharm. 2014;8(2):95. | ||
Kathiravan V, Ravi S, Ashokkumar S. Synthesis of silver nanoparticles from Melia dubia leaf extract and their in vitro anticancer activity. Spectrochim Acta A Mol Biomol Spectrosc. 2014;130(Supplement C):116–121. | ||
Mahadevi R. Green synthesis of silver nanoparticles using Terminalia chebula and its bactericidal activity against wound causing S. aureus. World J Pharm Pharm Sci. 2017;6(6):1228–1238. | ||
Shankar S, Jaiswal L, Aparna RSL, Prasad RGV, Kumar GP, Manohara CM. Wound healing potential of green synthesized silver nanoparticles prepared from Lansium domesticum fruit peel extract. Mater Express. 2015;5(2):159–164. | ||
Wen L, Zeng P, Zhang L, Huang W, Wang H, Chen G. Symbiosis theory-directed green synthesis of silver nanoparticles and their application in infected wound healing. Int J Nanomedicine. 2016;11:2757. | ||
Chandran SP, Chaudhary M, Pasricha R, Ahmad A, Sastry M. Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol Prog. 2006;22(2):577–583. | ||
Muhammad G, Hussain MA, Amin M, et al. Glucuronoxylan-mediated silver nanoparticles: green synthesis, antimicrobial and wound healing applications. RSC Adv. 2017;7(68):42900–42908. | ||
Alippilakkotte S, Kumar S, Sreejith L. Fabrication of PLA/Ag nanofibers by green synthesis method using Momordica charantia fruit extract for wound dressing applications. Colloids Surf A Physicochem Eng Asp. 2017;529:771–782. | ||
Dhivya G, Rajasimman M. Synthesis of silver nanoparticles using Momordica charantia and its applications. J Chem Pharm Res. 2015;7:107–113. | ||
Jain D, Kumar Daima H, Kachhwaha S, Kothari SL. Synthesis of plant-mediated silver nanoparticles using papaya fruit extract and evaluation of their anti microbial activities. Dig J Nanomater Biostruct. 2009;4(3):557–563. | ||
Geetha N, Geetha TS, Manonmani P, Thiyagarajan M. Green synthesis of silver nanoparticles using Cymbopogan Citratus (Dc) Stapf. extract and its antibacterial activity. Aust J Basic Appl Sci. 2014;8(3):324–331. | ||
Rout A, Jena PK, Sahoo D, Parida UK, Bindhani BK. Green synthesis and characterization of silver nanoparticles for antimicrobial activity against burn wounds contaminating bacteria. Int J Nanosci. 2014;13(2):1450010. | ||
Bhuvaneswari T, Thiyagarajan M, Geetha N, Venkatachalam P. Bioactive compound loaded stable silver nanoparticle synthesis from microwave irradiated aqueous extracellular leaf extracts of Naringi crenulata and its wound healing activity in experimental rat model. Acta Trop. 2014;135:55–61. | ||
Thirumurugan G, Veni VS, Ramachandran S, Rao JV, Dhanaraju MD. Superior wound healing effect of topically delivered silver nanoparticle formulation using eco-friendly potato plant pathogenic fungus: synthesis and characterization. J Biomed Nanotechnol. 2011;7(5):659–666. | ||
Augustine R, Augustine A, Kalarikkal N, Thomas S. Fabrication and characterization of biosilver nanoparticles loaded calcium pectinate nano-micro dual-porous antibacterial wound dressings. Prog Biomater. 2016;5(3–4):223–235. | ||
Patil S, Desai N, Mahadik K, Paradkar A. Can green synthesized propolis loaded silver nanoparticulate gel enhance wound healing caused by burns? Eur J Integr Med. 2015;7(3):243–250. | ||
Dhapte V, Kadam S, Moghe A, Pokharkar V. Probing the wound healing potential of biogenic silver nanoparticles. J Wound Care. 2014;23(9):431–441. | ||
Bhide B, Ashok B, Acharya R, Ravishankar B. Anti-microbial and wound healing activities of Cordia macleodii Hook. f & Thoms. leaves. Indian J Nat Prod Resour. 2011;1(2):198–203. | ||
Tian J, Wong KK, Ho CM, et al. Topical delivery of silver nanoparticles promotes wound healing. ChemMedChem. 2007;2(1):129–136. | ||
Ali A, Haq UII, Akhtar J, et al. Synthesis of silver nanoparticles impregnated cellulose composite material: its possible role in wound healing and photocatalysis. IET Nanobiotechnol. 2017;11(4):477–484. | ||
Al-Shmgani HSA, Mohammed WH, Sulaiman GM, Saadoon AH. Biosynthesis of silver nanoparticles from Catharanthus roseus leaf extract and assessing their antioxidant, antimicrobial, and wound-healing activities. Artif Cells Nanomed Biotechnol. 2017;45(6):1234–1240. | ||
De Bruin E, Werten M, De Wolf F. Method for production of hydroxylated collagen-like compounds. United States patent US 10/340,780. 2003 Sep 4. | ||
Kumarasamyraja D, Swamivelmanickam M. Evaluation of wound healing activity of biosynthesized silver nanoparticles from aqueous extract of Cassia Auriculata L. Int J Phytopharm. 2014;5:201–209. | ||
Ziv-Polat O, Topaz M, Brosh T, Margel S. Enhancement of incisional wound healing by thrombin conjugated iron oxide nanoparticles. Biomaterials. 2010;31(4):741–747. | ||
Ghosh Auddy R, Abdullah MF, Das S, Roy P, Datta S, Mukherjee A. New guar biopolymer silver nanocomposites for wound healing applications. Biomed Res Int. 2013;2013(5):1–8. | ||
Augustine R, Kalarikkal N, Thomas S. Electrospun PCL membranes incorporated with biosynthesized silver nanoparticles as antibacterial wound dressings. Appl Nanosci. 2016;6(3):337–344. | ||
Naraginti S, Kumari PL, Das RK, Sivakumar A, Patil SH, Andhalkar VV. Amelioration of excision wounds by topical application of green synthesized, formulated silver and gold nanoparticles in albino Wistar rats. Mater Sci Eng C Mater Biol Appl. 2016;62:293–300. | ||
Sivaranjani V, Philominathan P. Synthesize of titanium dioxide nanoparticles using Moringa oleifera leaves and evaluation of wound healing activity. Wound Medicine. 2016;12:1–5. | ||
Sankar R, Baskaran A, Shivashangari KS, Ravikumar V. Inhibition of pathogenic bacterial growth on excision wound by green synthesized copper oxide nanoparticles leads to accelerated wound healing activity in Wistar Albino rats. J Mater Sci Mater Med. 2015;26(7):1–7. | ||
El-Refaie WM, Elnaggar YSR, El-Massik MA, Abdallah OY. Novel curcumin-loaded gel-core hyaluosomes with promising burn-wound healing potential: development, in-vitro appraisal and in-vivo studies. Int J Pharm. 2015;486(1–2):88–98. | ||
Andreu V, Mendoza G, Arruebo M, Irusta S. Smart dressings based on nanostructured fibers containing natural origin antimicrobial, anti-inflammatory, and regenerative compounds. Materials. 2015;8(8):5154–5193. | ||
Singla R, Soni S, Kulurkar PM, et al. In situ functionalized nanobiocomposites dressings of bamboo cellulose nanocrystals and silver nanoparticles for accelerated wound healing. Carbohydr Polym. 2017;155:152–162. | ||
Cui M-D, Pan Z-H, Pan L-Q. Danggui Buxue extract-loaded liposomes in thermosensitive gel enhance in vivo dermal wound healing via activation of the VEGF/PI3K/Akt and TGF-β/Smads signaling pathway. Evid Based Complement Alternat Med. 2017;2017(10):8407249–13. | ||
Buranasukhon W, Athikomkulchai S, Tadtong S, Chittasupho C. Wound healing activity of Pluchea indica leaf extract in oral mucosal cell line and oral spray formulation containing nanoparticles of the extract. Pharm Biol. 2017;55(1):1767–1774. | ||
Faezizadeh Z, Gharib A, Godarzee M. In-vitro and in-vivo evaluation of silymarin nanoliposomes against isolated methicillin-resistant Staphylococcus aureus. Iran J Pharm Res. 2015;14(2):627. | ||
Ghayempour S, Montazer M, Mahmoudi Rad M. Encapsulation of Aloe vera extract into natural Tragacanth Gum as a novel green wound healing product. Int J Biol Macromol. 2016;93:344–349. | ||
Arana L, Salado C, Vega S, et al. Solid lipid nanoparticles for delivery of Calendula officinalis extract. Colloids Surf B Biointerfaces. 2015;135:18–26. | ||
Krausz AE, Adler BL, Cabral V, et al. Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent. Nanomedicine. 2015;11(1):195–206. | ||
Burt S. Essential oils: their antibacterial properties and potential applications in foods – a review. Int J Food Microbiol. 2004;94(3):223–253. | ||
Sugumar S, Ghosh V, Nirmala MJ, Mukherjee A, Chandrasekaran N. Ultrasonic emulsification of eucalyptus oil nanoemulsion: antibacterial activity against Staphylococcus aureus and wound healing activity in Wistar rats. Ultrason Sonochem. 2014;21(3):1044–1049. | ||
Kianvash N, Bahador A, Pourhajibagher M, et al. Evaluation of propylene glycol nanoliposomes containing curcumin on burn wound model in rat: biocompatibility, wound healing, and anti-bacterial effects. Drug Deliv Transl Res. 2017;7(5):654–663. | ||
Perumal G, Pappuru S, Chakraborty D, Maya Nandkumar A, Chand DK, Doble M. Synthesis and characterization of curcumin loaded PLA-hyperbranched polyglycerol electrospun blend for wound dressing applications. Mater Sci Eng C Mater Biol Appl. 2017;76:1196–1204. | ||
Gharib A, Faezizadeh Z, Godarzee M. Therapeutic efficacy of epigallocatechin gallate-loaded nanoliposomes against burn wound infection by methicillin-resistant Staphylococcus aureus. Skin Pharmacol Physiol. 2013;26(2):68–75. | ||
Naumov AA, Shatalin YV, Potselueva MM. Effects of a nanocomplex containing antioxidant, lipid, and amino acid on thermal burn wound surface. Bull Exp Biol Med. 2010;149(1):62–66. | ||
Engel H, Kao SW, Larson J, et al. Investigation of dermis-derived hydrogels for wound healing applications. Biomed J. 2015;38(1):58–64. | ||
Li S, Dong S, Xu W, et al. Antibacterial hydrogels. Adv Sci (Weinh). 2018;5(5):1700527. | ||
Anumolu SS, Menjoge AR, Deshmukh M, et al. Doxycycline hydrogels with reversible disulfide crosslinks for dermal wound healing of mustard injuries. Biomaterials. 2011;32(4):1204–1217. | ||
Li M, Li H, Li X, et al. A bioinspired alginate-gum arabic hydrogel with micro-/nanoscale structures for controlled drug release in chronic wound healing. ACS Appl Mater Interfaces. 2017;9(27):22160–22175. | ||
Gong C, Wu Q, Wang Y, et al. A biodegradable hydrogel system containing curcumin encapsulated in micelles for cutaneous wound healing. Biomaterials. 2013;34(27):6377–6387. | ||
Li X, Chen S, Zhang B, et al. In situ injectable nano-composite hydrogel composed of curcumin, N, O-carboxymethyl chitosan and oxidized alginate for wound healing application. Int J Pharm. 2012;437(1–2):110–119. | ||
Gumus ZP, Guler E, Demir B, et al. Herbal infusions of black seed and wheat germ oil: their chemical profiles, in vitro bio-investigations and effective formulations as phyto-nanoemulsions. Colloids Surf B Biointerfaces. 2015;133:73–80. | ||
Kim JE, Lee J, Jang M, et al. Accelerated healing of cutaneous wounds using phytochemically stabilized gold nanoparticle deposited hydrocolloid membranes. Biomater Sci. 2015;3(3):509–519. | ||
Bishop A. Role of oxygen in wound healing. J Wound Care. 2008;17(9):399–402. | ||
Guo S, Dipietro LA. Factors affecting wound healing. J Dent Res. 2010;89(3):219–229. | ||
Soneja A, Drews M, Malinski T. Role of nitric oxide, nitroxidative and oxidative stress in wound healing. Pharmacol Rep. 2005;57 Suppl:108. | ||
Saeidnia S, Abdollahi M. Toxicological and pharmacological concerns on oxidative stress and related diseases. Toxicol Appl Pharmacol. 2013;273(3):442–455. | ||
Moulaoui K, Caddeo C, Manca ML, et al. Identification and nanoentrapment of polyphenolic phytocomplex from Fraxinus angustifolia: in vitro and in vivo wound healing potential. Eur J Med Chem. 2015;89:179–188. | ||
Hosgood G. Stages of wound healing and their clinical relevance. Vet Clin North Am Small Anim Pract. 2006;36(4):667–685. | ||
Zheng Y, Watanabe M, Kuraishi T, Hattori S, Kai C, Shibuya M. Chimeric VEGF-ENZ7/PlGF specifically binding to VEGFR-2 accelerates skin wound healing via enhancement of neovascularization. Arterioscler Thromb Vasc Biol. 2007;27(3):503–511. | ||
Hübner G, Brauchle M, Smola H, Madlener M, Fässler R, Werner S. Differential regulation of pro-inflammatory cytokines during wound healing in normal and glucocorticoid-treated mice. Cytokine. 1996;8(7):548–556. | ||
Ribatti D, Levi-Schaffer F, Kovanen PT. Inflammatory angiogenesis in atherogenesis – a double-edged sword. Ann Med. 2008;40(8):606–621. | ||
Xu XH, Shah PK, Faure E, et al. Toll-like receptor-4 is expressed by macrophages in murine and human lipid-rich atherosclerotic plaques and upregulated by oxidized LDL. Circulation. 2001;104(25):3103–3108. | ||
Davatgaran-Taghipour Y, Masoomzadeh S, Farzaei MH, et al. Polyphenol nanoformulations for cancer therapy: experimental evidence and clinical perspective. Int J Nanomedicine. 2017;12:2689–2702. | ||
Bharti AC, Donato N, Singh S, Aggarwal BB. Curcumin (diferuloylmethane) down-regulates the constitutive activation of nuclear factor-kappa B and IkappaBalpha kinase in human multiple myeloma cells, leading to suppression of proliferation and induction of apoptosis. Blood. 2003;101(3):1053–1062. | ||
Aggarwal BB, Shishodia S. Suppression of the nuclear factor-kappaB activation pathway by spice-derived phytochemicals: reasoning for seasoning. Ann N Y Acad Sci. 2004;1030(1):434–441. | ||
Singla R, Soni S, Patial V, et al. In vivo diabetic wound healing potential of nanobiocomposites containing bamboo cellulose nanocrystals impregnated with silver nanoparticles. Int J Biol Macromol. 2017;105(Pt 1):45–55. | ||
Losi P, Briganti E, Errico C, et al. Fibrin-based scaffold incorporating VEGF- and bFGF-loaded nanoparticles stimulates wound healing in diabetic mice. Acta Biomater. 2013;9(8):7814–7821. | ||
Ranjbar-Mohammadi M, Rabbani S, Bahrami SH, Joghataei MT, Moayer F. Antibacterial performance and in vivo diabetic wound healing of curcumin loaded gum tragacanth/poly(ε-caprolactone) electrospun nanofibers. Mater Sci Eng C Mater Biol Appl. 2016;69:1183–1191. | ||
Bajpai SK, Ahuja S, Chand N, Bajpai M. Nano cellulose dispersed chitosan film with Ag NPs/curcumin: an in vivo study on albino rats for wound dressing. Int J Biol Macromol. 2017;104(Pt A):1012–1019. | ||
Leu J-G, Chen S-A, Chen H-M, et al. The effects of gold nanoparticles in wound healing with antioxidant epigallocatechin gallate and α-lipoic acid. Nanomedicine. 2012;8(5):767–775. | ||
Chen SA, Chen HM, Yao YD, Hung CF, Tu CS, Liang YJ. Topical treatment with anti-oxidants and Au nanoparticles promote healing of diabetic wound through receptor for advance glycation end-products. Eur J Pharm Sci. 2012;47(5):875–883. | ||
Kipanyula MJ, Seke Etet PF, Vecchio L, Farahna M, Nukenine EN, Nwabo Kamdje AH. Signaling pathways bridging microbial-triggered inflammation and cancer. Cell Signal. 2013;25(2):403–416. | ||
Czaja WK, Young DJ, Kawecki M, Brown RM. The future prospects of microbial cellulose in biomedical applications. Biomacromolecules. 2007;8(1):1–12. | ||
Mitra T, Manna PJ, Raja STK, Gnanamani A, Kundu PP. Curcumin loaded nano graphene oxide reinforced fish scale collagen – a 3D scaffold biomaterial for wound healing applications. RSC Adv. 2015;5(119):98653–98665. | ||
Ponnanikajamideen M, Devi Priya S, Vanaja M, Paulkumar K, Rajeshkumar S, Annadurai G. In-vivo wound healing efficiency of curcumin loaded on chitosan polyvinyl propylene nanofilm. Adv Sci Eng Med. 2016;8(10):763–770. | ||
Thomas L, Zakir F, Mirza MA, Anwer MK, Ahmad FJ, Iqbal Z. Development of curcumin loaded chitosan polymer based nanoemulsion gel: In vitro, ex vivo evaluation and in vivo wound healing studies. Int J Biol Macromol. 2017;101:569–579. | ||
Kalashnikova I, Das S, Seal S. Nanomaterials for wound healing: scope and advancement. Nanomedicine (Lond). 2015;10(16):2593–2612. | ||
Deepachitra R, Lakshmi RP, Sivaranjani K, Chandra JH, Sastry T. Nanoparticles embedded biomaterials in wound treatment: a review. J Chem Pharm Sci. 2015;8:324–328. | ||
Bajpai J, Bajpai A, Chhatri A, Lawrance S, Dsouza A. Polymer biomaterials in wound dressing: a review. Chem Sci Rev Lett. 2015;4(16):1124–1147. | ||
Ghosh PK, Gaba A. Phyto-extracts in wound healing. J Pharm Pharm Sci. 2013;16(5):760–820. | ||
Mittal S, Dixit PK. Natural remedies for wound healing: a literary review. Pharmacie Globale (IJCP). 2013;4(3):1. |
Supplementary materials
  | Table S1 Herbal-based nanoformulations used for wound management |
References
Yang Y, Wang F, Yin D, Fang Z, Huang L. Astragalus polysaccharide-loaded fibrous mats promote the restoration of microcirculation in/around skin wounds to accelerate wound healing in a diabetic rat model. Colloids Surf B Biointerfaces. 2015;136:111–118. | ||
Ranjbar-Mohammadi M, Rabbani S, Bahrami SH, Joghataei MT, Moayer F. Antibacterial performance and in vivo diabetic wound healing of curcumin loaded gum tragacanth/poly(ε-caprolactone) electrospun nanofibers. Mater Sci Eng C Mater Biol Appl. 2016;69:1183–1191. | ||
Vargas EAT, do Vale Baracho NC, de Brito J, de Queiroz AAA. Hyperbranched polyglycerol electrospun nanofibers for wound dressing applications. Acta Biomater. 2010;6(3):1069–1078. | ||
Yousefi I, Pakravan M, Rahimi H, Bahador A, Farshadzadeh Z, Haririan I. An investigation of electrospun henna leaves extract-loaded chitosan based nanofibrous mats for skin tissue engineering. Mater Sci Eng C Mater Biol Appl. 2017;75:433–444. | ||
Selvaraj S, Fathima NN. Fenugreek incorporated silk fibroin nanofibers – a potential antioxidant scaffold for enhanced wound healing. ACS Appl Mater Interfaces. 2017;9(7):5916–5926. | ||
Lin L, Perets A, Har-El YE, et al. Alimentary ‘green’ proteins as electrospun scaffolds for skin regenerative engineering. J Tissue Eng Regen Med. 2013;7(12):994–1008. | ||
Cerchiara T, Abruzzo A, Ñahui Palomino RA, et al. Spanish Broom (Spartium junceum L.) fibers impregnated with vancomycin-loaded chitosan nanoparticles as new antibacterial wound dressing: prepara characterization and antibacterial activity. Eur J Pharm Sci. 2017;99:105–112. | ||
Suganya S, Senthil Ram T, Lakshmi BS, Giridev VR. Herbal drug incorporated antibacterial nanofibrous mat fabricated by electrospinning: an excellent matrix for wound dressings. J Appl Polym Sci. 2011;121(5):2893–2899. | ||
Suwantong O, Opanasopit P, Ruktanonchai U, Supaphol P. Electrospun cellulose acetate fiber mats containing curcumin and release characteristic of the herbal substance. Polymer. 2007;48(26):7546–7557. | ||
Pannerselvam B, Dharmalingam Jothinathan MK, Rajenderan M, et al. An in vitro study on the burn wound healing activity of cotton fabrics incorporated with phytosynthesized silver nanoparticles in male Wistar albino rats. Eur J Pharm Sci. 2017;100(Supplement C):187–196. | ||
Garg S, Chandra A, Mazumder A, Mazumder R. Green synthesis of silver nanoparticles using Arnebia nobilis root extract and wound healing potential of its hydrogel. Asian J Pharm. 2014;8(2):95. | ||
Augustine R, Augustine A, Kalarikkal N, Thomas S. Fabrication and characterization of biosilver nanoparticles loaded calcium pectinate nano-micro dual-porous antibacterial wound dressings. Prog Biomater. 2016;5(3–4):223–235. | ||
Kim JE, Lee J, Jang M, et al. Accelerated healing of cutaneous wounds using phytochemically stabilized gold nanoparticle deposited hydrocolloid membranes. Biomater Sci. 2015;3(3):509–519. | ||
Naraginti S, Kumari PL, Das RK, Sivakumar A, Patil SH, Andhalkar VV. Amelioration of excision wounds by topical application of green synthesized, formulated silver and gold nanoparticles in albino Wistar rats. Mater Sci Eng C Mater Biol Appl. 2016;62:293–300. | ||
Krychowiak M, Grinholc M, Banasiuk R, et al. Combination of silver nanoparticles and Drosera binata extract as a possible alternative for antibiotic treatment of burn wound infections caused by resistant Staphylococcus aureus. PLoS One. 2014;9(12):e115727. | ||
Sankar R, Baskaran A, Shivashangari KS, Ravikumar V. Inhibition of pathogenic bacterial growth on excision wound by green synthesized copper oxide nanoparticles leads to accelerated wound healing activity in Wistar Albino rats. J Mater Sci Mater Med. 2015;26(7):1–7. | ||
Bhuvaneswari T, Thiyagarajan M, Geetha N, Venkatachalam P. Bioactive compound loaded stable silver nanoparticle synthesis from microwave irradiated aqueous extracellular leaf extracts of Naringi crenulata and its wound healing activity in experimental rat model. Acta Trop. 2014;135:55–61. | ||
Ali A, Haq UII, Akhtar J, Sher M, Ahmed N, Zia M. Synthesis of silver nanoparticles impregnated cellulose composite material: its possible role in wound healing and photocatalysis. | ||
Sivaranjani V, Philominathan P. Synthesize of titanium dioxide nanoparticles using Moringa oleifera leaves and evaluation of wound healing activity. Wound Medicine. 2016;12:1–5. | ||
Thirumurugan G, Veni VS, Ramachandran S, Seshagiri Rao JVLN, Dhanaraju MD. Superior wound healing effect of topically delivered silver nanoparticle formulation using eco-friendly potato plant pathogenic fungus: synthesis and characterization. J Biomed Nanotechnol. 2011;7(5):659–666. | ||
Ghosh Auddy R, Abdullah MF, das S, Roy P, Datta S, Mukherjee A. New guar biopolymer silver nanocomposites for wound healing applications. Biomed Res Int. 2013;2013(5):1–8. | ||
Rout A, Jena PK, Sahoo D, Parida UK, Bindhani BK. Green synthesis and characterization of silver nanoparticles for antimicrobial activity against burn wounds contaminating bacteria. Int J Nanosci. 2014;13(02):1450010. | ||
Bajpai SK, Ahuja S, Chand N, Bajpai M. Nano cellulose dispersed chitosan film with Ag NPs/curcumin: an in vivo study on albino rats for wound dressing. Int J Biol Macromol. 2017;104(Pt A):1012–1019. | ||
Dhapte V, Kadam S, Moghe A, Pokharkar V. Probing the wound healing potential of biogenic silver nanoparticles. J Wound Care. 2014;23(9):431–441. | ||
Kumarasamyraja D, Swamivelmanickam M. Evaluation of wound healing activity of biosynthesized silver nanoparticles from aqueous extract of Cassia Auriculata L. Int J Phytopharm. 2014;5:201–209. | ||
Shankar S, Jaiswal L, Aparna RSL, Prasad RGV, Kumar GP, Manohara CM. Wound healing potential of green synthesized silver nanoparticles prepared from Lansium domesticum fruit peel extract. Mater Express. 2015;5(2):159–164. | ||
Bansod SD, Bawaskar MS, Gade AK, Rai MK. Development of shampoo, soap and ointment formulated by green synthesised silver nanoparticles functionalised with antimicrobial plants oils in veterinary dermatology: treatment and prevention strategies. IET Nanobiotechnol. 2015;9(4):165–171. | ||
Augustine R, Kalarikkal N, Thomas S. Electrospun PCL membranes incorporated with biosynthesized silver nanoparticles as antibacterial wound dressings. Appl Nanosci. 2016;6(3):337–344. | ||
Al-Shmgani HSA, Mohammed WH, Sulaiman GM, Saadoon AH. Biosynthesis of silver nanoparticles from Catharanthus roseus leaf extract and assessing their antioxidant, antimicrobial, and wound-healing activities. Artif Cells Nanomed Biotechnol. 2017;45(6):1234–1240. | ||
Patil S, Desai N, Mahadik K, Paradkar A. Can green synthesized propolis loaded silver nanoparticulate gel enhance wound healing caused by burns? Eur J Integr Med. 2015;7(3):243–250. | ||
Wen L, Zeng P, Zhang L, Huang W, Wang H, Chen G. Symbiosis theory-directed green synthesis of silver nanoparticles and their application in infected wound healing. Int J Nanomedicine. 2016;11:2757. | ||
Haseeb MT, Hussain MA, Abbas K, et al. Linseed hydrogel-mediated green synthesis of silver nanoparticles for antimicrobial and wound-dressing applications. Int J Nanomedicine. 2017;12:2845–2855. | ||
Alippilakkotte S, Kumar S, Sreejith L. Fabrication of PLA/Ag nanofibers by green synthesis method using Momordica charantia fruit extract for wound dressing applications. Colloids Surf A Physicochem Eng Asp. 2017;529:771–782. | ||
Sugumar S, Ghosh V, Nirmala MJ, Mukherjee A, Chandrasekaran N. Ultrasonic emulsification of eucalyptus oil nanoemulsion: antibacterial activity against Staphylococcus aureus and wound healing activity in Wistar rats. Ultrason Sonochem. 2014;21(3):1044–1049. | ||
Ghayempour S, Montazer M, Mahmoudi Rad M. Encapsulation of Aloe vera extract into natural Tragacanth Gum as a novel green wound healing product. Int J Biol Macromol. 2016;93:344–349. | ||
Arana L, Salado C, Vega S, et al. Solid lipid nanoparticles for delivery of Calendula officinalis extract. Colloids Surf B Biointerfaces. 2015;135:18–26. | ||
Singla R, Soni S, Kulurkar PM, et al. In situ functionalized nanobiocomposites dressings of bamboo cellulose nanocrystals and silver nanoparticles for accelerated wound healing. Carbohydr Polym. 2017;155:152–162. | ||
Singla R, Soni S, Patial V, et al. In vivo diabetic wound healing potenof nanobiocomposites containing bamboo cellulose nanocrystals impregnated with silver nanoparticles. Int J Biol Macromol. 2017;105(Pt 1):45–55. | ||
Cui M-D, Pan Z-H, Pan L-Q. Danggui Buxue extract-loaded liposomes in thermosensitive gel enhance in vivo dermal wound healing via activation of the VEGF/PI3K/Akt and TGF-β/Smads signaling pathway. Evid Based Complement Alternat Med. 2017;2017(10):8407249–13. | ||
Gumus ZP, Guler E, Demir B, et al. Herbal infusions of black seed and wheat germ oil: their chemical profiles, in vitro bio-investigations and effective formulations as phyto-nanoemulsions. Colloids Surf B Biointerfaces. 2015;133:73–80. | ||
Buranasukhon W, Athikomkulchai S, Tadtong S, Chittasupho C. Wound healing activity of Pluchea indica leaf extract in oral mucosal cell line and oral spray formulation containing nanoparticles of the extract. Pharm Biol. 2017;55(1):1767–1774. | ||
Faezizadeh Z, Gharib A, Godarzee M. In-vitro and in-vivo evaluation of silymarin nanoliposomes against isolated methicillin-resistant Staphylococcus aureus. Iran J Pharm Res. 2015;14(2):627. | ||
Moulaoui K, Caddeo C, Manca ML, et al. Identification and nanoentrapment of polyphenolic phytocomplex from Fraxinus angustifolia: in vitro and in vivo wound healing potential. Eur J Med Chem. 2015;89:179–188. | ||
Li M, Li H, Li X, et al. A bioinspired alginate-gum arabic hydrogel with micro-/nanoscale structures for controlled drug release in chronic wound healing. ACS Appl Mater Interfaces. 2017;9(27):22160–22175. | ||
Panichpakdee J, Pavasant P, Supaphol P. Electrospun cellulose acetate fiber mats containing emodin with potential for use as wound dressing. Chiang Mai J Sci. 2016;43(1):1249–1259. | ||
Dai X-Y, Nie W, Wang Y-C, Shen Y, Li Y, Gan S-J. Electrospun emodin polyvinylpyrrolidone blended nanofibrous membrane: a novel medicated biomaterial for drug delivery and accelerated wound healing. J Mater Sci Mater Med. 2012;23(11):2709–2716. | ||
Gharib A, Faezizadeh Z, Godarzee M. Therapeutic efficacy of epigalgallate-loaded nanoliposomes against burn wound infection by methicillin-resistant Staphylococcus aureus. Skin Pharmacol Physiol. 2013;26(2):68–75. | ||
Naumov AA, Shatalin YV, Potselueva MM. Effects of a nanocomplex containing antioxidant, lipid, and amino acid on thermal burn wound surface. Bull Exp Biol Med. 2010;149(1):62–66. | ||
Suwantong O, Ruktanonchai U, Supaphol P. In vitro biological evaluation of electrospun cellulose acetate fiber mats containing asiaticoside or curcumin. J Biomed Mater Res A. 2010;94(4):1216–1225. | ||
Dai X, Liu J, Zheng H, et al. Nano-formulated curcumin accelerates acute wound healing through Dkk-1-mediated fibroblast mobilization and MCP-1-mediated anti-inflammation. NPG Asia Mater. 2017;9(3):e368. | ||
Ranjbar-Mohammadi M, Rabbani S, Bahrami SH, Joghataei MT, Moayer F. Antibacterial performance and in vivo diabetic wound healing of curcumin loaded gum tragacanth/poly(ε-caprolactone) electrospun nanofibers. Mater Sci Eng C Mater Biol Appl. 2016;69:1183–1191. | ||
Mitra T, Manna PJ, Raja STK, Gnanamani A, Kundu PP. Curcumin loaded nano graphene oxide reinforced fish scale collagen – a 3D scaffold biomaterial for wound healing applications. RSC Adv. 2015;5(119):98653–98665. | ||
Li X, Chen S, Zhang B, et al. In situ injectable nano-composite hydrocomposed of curcumin, N,O-carboxymethyl chitosan and oxidized alginate for wound healing application. Int J Pharm. 2012;437(1–2):110–119. | ||
Lin Y-H, Lin J-H, Hong Y-S. Development of chitosan/poly-γ-glutamic acid/pluronic/curcumin nanoparticles in chitosan dressings for wound regeneration. J Biomed Mater Res B Appl Biomater. 2017;105(1):81–90. | ||
Bui HT, Chung OH, dela Cruz J, Park JS. Fabrication and characterization of electrospun curcumin-loaded polycaprolactone-polyethylene glycol nanofibers for enhanced wound healing. Macromol Res. 2014;22(12):1288–1296. | ||
Kianvash N, Bahador A, Pourhajibagher M, et al. Evaluation of propylene glycol nanoliposomes containing curcumin on burn wound model in rat: biocompatibility, wound healing, and anti-bacterial effects. Drug Deliv Transl Res. 2017;7(5):654–663. | ||
Thomas L, Zakir F, Mirza MA, Anwer MK, Ahmad FJ, Iqbal Z. Development of curcumin loaded chitosan polymer based nanoemulsion gel: In vitro, ex vivo evaluation and in vivo wound healing studies. Int J Biol Macromol. 2017;101:569–579. | ||
El-Refaie WM, Elnaggar YSR, El-Massik MA, Abdallah OY. Novel curcumin-loaded gel-core hyaluosomes with promising burn-wound healing potential: development, in-vitro appraisal and in-vivo studies. Int J Pharm. 2015;486(1–2):88–98. | ||
Perumal G, Pappuru S, Chakraborty D, Maya Nandkumar A, Chand DK, Doble M. Synthesis and characterization of curcumin loaded PLA-hyperbranched polyglycerol electrospun blend for wound dressing applications. Mater Sci Eng C Mater Biol Appl. 2017;76:1196–1204. | ||
Gong C, Wu Q, Wang Y, et al. A biodegradable hydrogel system containing curcumin encapsulated in micelles for cutaneous wound healing. Biomaterials. 2013;34(27):6377–6387. | ||
Suwantong O, Ruktanonchai U, Supaphol P. In vitro biological evaluation of electrospun cellulose acetate fiber mats containing asiaticoside or curcumin. J Biomed Mater Res A. 2010;94(4):1216–1225. | ||
Panichpakdee J, Pavasant P, Supaphol P. Electrospinning of asiaticoside/2-hydroxypropyl-β-cyclodextrin inclusion complex-loaded cellulose acetate fiber mats: release characteristics and potential for use as wound dressing. Polym Kor. 2014;38(3):338–350. | ||
Liu X, Lin T, Fang J, et al. In vivo wound healing and antibacterial performances of electrospun nanofibre membranes. J Biomed Mater Res A. 2010;94(2):499–508. | ||
Vatankhah E, Prabhakaran MP, Jin G, Mobarakeh LG, Ramakrishna S. Development of nanofibrous cellulose acetate/gelatin skin substitutes for variety wound treatment applications. J Biomater Appl. 2014;28(6):909–921. | ||
Liao N, Unnithan AR, Joshi MK, et al. Electrospun bioactive poly (ε-caprolactone)–cellulose acetate–dextran antibacterial composite mats for wound dressing applications. Colloids Surf A Physicochem Eng Asp. 2015;469:194–201. | ||
Gomaa SF, Madkour TM, Moghannem S, El-Sherbiny IM. New polylactic acid/cellulose acetate-based antimicrobial interactive single dose nanofibrous wound dressing mats. Int J Biol Macromol. 2017;105:1148–1160. | ||
Unnithan AR, Barakat NA, Pichiah PB, et al. Wound-dressing materiwith antibacterial activity from electrospun polyurethane-dextran nanofiber mats containing ciprofloxacin HCl. Carbohydr Polym. 2012;90(4):1786–1793. | ||
Nosar MN, Salehi M, Ghorbani S, Beiranvand SP, Goodarzi A, Azami M. Characterization of wet-electrospun cellulose acetate based 3-dimensional scaffolds for skin tissue engineering applications: influence of cellulose acetate concentration. Cellulose. 2016;23(5):3239–3248. | ||
Samadian H, Salehi M, Farzamfar S, et al. In vitro and in vivo evaluation of electrospun cellulose acetate/gelatin/hydroxyapatite nanocomposite mats for wound dressing applications. Artif Cells Nanomed Biotechnol. 2018;44:1–11. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.