Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Nanoshell-mediated photothermal therapy can enhance chemotherapy in inflammatory breast cancer cells
Authors Fay B, Melamed J, Day E
Received 24 July 2015
Accepted for publication 9 October 2015
Published 6 November 2015 Volume 2015:10(1) Pages 6931—6941
DOI https://doi.org/10.2147/IJN.S93031
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Brittany L Fay, Jilian R Melamed, Emily S Day
Biomedical Engineering, University of Delaware, Newark, DE, USA
Abstract: Nanoshell-mediated photothermal therapy (PTT) is currently being investigated as a standalone therapy for the treatment of cancer. The cellular effects of PTT include loss of membrane integrity, so we hypothesized that nanoshell-mediated PTT could potentiate the cytotoxicity of chemotherapy by improving drug accumulation in cancer cells. In this work, we validated our hypothesis using doxorubicin as a model drug and SUM149 inflammatory breast cancer cells as a model cancer subtype. In initial studies, SUM149 cells were exposed to nanoshells and near-infrared light and then stained with ethidium homodimer-1, which is excluded from cells with an intact plasma membrane. The results confirmed that nanoshell-mediated PTT could increase membrane permeability in SUM149 cells. In complementary experiments, SUM149 cells treated with nanoshells, near-infrared light, or a combination of the two to yield low-dose PTT were exposed to fluorescent rhodamine 123. Analyzing rhodamine 123 fluorescence in cells via flow cytometry confirmed that increased membrane permeability caused by PTT could enhance drug accumulation in cells. This was validated using fluorescence microscopy to assess intracellular distribution of doxorubicin. In succeeding experiments, SUM149 cells were exposed to subtherapeutic levels of doxorubicin, low-dose PTT, or a combination of the two treatments to determine whether the additional drug uptake induced by PTT is sufficient to enhance cell death. Analysis revealed minimal loss of viability relative to controls in cells exposed to subtherapeutic levels of doxorubicin, 15% loss of viability in cells exposed to low-dose PTT, and 35% loss of viability in cells exposed to combination therapy. These data indicate that nanoshell-mediated PTT is a viable strategy to potentiate the effects of chemotherapy and warrant further investigation of this approach using other drugs and cancer subtypes.
Keywords: nanoshells, photothermal therapy, hyperthermia, chemotherapy, sensitization, breast cancer
Introduction
Although chemotherapy is a frontline component of current cancer treatment, its effectiveness is often limited by the development of cellular resistance and production of off-target toxicity. The side effects of chemotherapy range from minor reactions, such as nausea and hair loss, to extreme complications including fatigue and cognitive dysfunction. Many of these toxicities occur because chemotherapy is systemically delivered and lacks specificity for tumor cells. A technology that could potentiate chemotherapy specifically at tumor sites so that systemically nontoxic doses of drugs could be administered to patients would greatly improve both treatment outcome and patient quality of life by overcoming resistance and minimizing side effects. Here, we report the application of nanoshell-mediated photothermal therapy (PTT) to address this unmet clinical need.
Previous research has demonstrated that applying heat in combination with chemotherapy can result in synergistic effects on tumor cells.1 The controlled delivery of heat specifically to tumor sites, however, was difficult to achieve until the recent development of nanoparticle-mediated PTT.2,3 Nanoparticle-mediated PTT is a technique in which plasmonic nanoparticles are used to damage cancer cells via tumor-localized hyperthermia. In this therapy, nanoparticles are delivered to tumors either intratumorally or intravenously by exploiting the enhanced permeability and retention effect.4 Subsequent exposure of the tumor to light at the nanoparticles’ resonant wavelength causes synchronized oscillation of the nanoparticles’ conduction-band electrons that results in the production of heat sufficient to damage the cancer cells.2,5 To maximize the success of PTT, nanoparticles are designed to absorb near-infrared (NIR) wavelengths of light, which penetrate more deeply into tissue than other wavelengths.6 Since heat is produced only where activating light and nanoparticles are both present, damage to healthy cells outside the tumor is minimal. The mode of cell death induced by PTT can be tuned between apoptosis or necrosis by altering parameters such as the light exposure conditions with low-dose PTT/mild hyperthermia favoring apoptosis.7,8 Thus, PTT is advantageous for cancer treatment because it is highly effective, minimally invasive, and offers limited side effects. Accordingly, there are several gold-based, NIR-absorbing nanoparticles being investigated for PTT, including silica core-gold shell nanoshells,5,9,10 gold nanorods,11 gold–gold sulfide nanoparticles,12 hollow gold nanospheres,13 and gold nanocages.14 We selected nanoshells for use in our study, which demonstrates that low-dose PTT can sensitize cancer cells to chemotherapy by promoting drug accumulation in cells, because they are the furthest along in clinical development and are currently being evaluated in multiple clinical trials.15,16
One of the cellular effects of PTT is loss of membrane integrity, so we hypothesized that nanoshell-mediated PTT could potentiate the success of chemotherapy by promoting drug accumulation in cancer cells. This physical method of promoting drug uptake should be less susceptible to the resistance mechanisms that plague small molecule-based therapeutics, rendering PTT an excellent strategy for chemosensitization. One recent publication demonstrated that PTT mediated by solid gold nanoparticles excited with broad spectrum (575–1,200 nm) pulsed light could reduce the half maximal inhibitory concentration (IC50) of cyclophosphamide in vitro, providing proof-of-principle that PTT can sensitize cancer cells to chemotherapy when the two treatments are applied individually.17 A second study demonstrated that mild hyperthermia mediated by gold nanorods could promote apoptotic signaling in cancer cells to provide synergy with cisplatin both in vitro and in vivo.18 This finding was in agreement with a previous study that revealed synergy between nanorod-mediated hyperthermia and cisplatin.19 Here, we aimed to expand upon this exciting field of study by demonstrating that nanoshell-mediated PTT could sensitize cancer cells to subtherapeutic doses of doxorubicin (dox). We selected nanoshells for this work because they are more efficient photothermal transducers than solid gold spheres, can be activated with continuous wave 800 nm light, and PTT mediated by nanoshells is currently being investigated as a standalone cancer therapy in two clinical trials.15,16 Dox was selected as the model drug because despite being the frontline chemotherapy for patients with a diverse range of cancer types, it is often rendered ineffective with prolonged treatment. In addition, its use is often limited by the development of cardiotoxicity, so there exists an urgent need for a novel therapeutic platform that can render cancer cells susceptible to lower doses of dox. Finally, we also aimed to determine whether the synergistic effects observed between low-dose PTT/mild hyperthermia and cisplatin or cyclophosphamide could be extended to other classes of chemotherapeutic agents, as well as to other cancer subtypes.
Notably, we delivered PTT and chemotherapy to cancer cells individually in our studies in order to elucidate the fundamental additive or synergistic effects between these two treatment strategies. This information is important because new therapeutic approaches like PTT are often tested as an adjuvant to chemotherapy in early clinical trials. Excitingly, several researchers have begun to investigate the use of combination therapeutics in which drugs are physically bound to or entrapped inside nanoparticles so that the conjugates can act as simultaneous photothermal transducers and drug delivery vehicles. Examples of these combination therapeutics include drug-loaded or drug-conjugated gold nanospheres,20 gold half-shell nanoparticles,21 chitosan/gold nanorod hybrid nanospheres,22 dendrimer-modified nanorods,23 gold nanocages,24 graphene oxide nanoparticles,25 and FeCo/graphitic shell nanocrystals.26 These systems demonstrate that simultaneous drug delivery and PTT is feasible and the positive results obtained thus far support continued development of such strategies. However, it will likely take many years of development before complex nanostructures can be evaluated clinically and extensive modifications made to nanoparticles and/or drugs in order to facilitate their combination may negatively impact the function of the individual components. To avoid these complications and reveal the fundamental benefits of combining chemotherapy with PTT, we chose to deliver unmodified nanoparticles and drugs as individual systems in our studies. If localized PTT can enhance drug accumulation specifically in tumor cells, it may be a useful strategy to maintain therapeutic efficacy while allowing systemically nontoxic doses of drugs to be utilized. The ability to eliminate tumors without inducing toxicity to normal tissue would be highly desirable in a clinical setting.
In summary, in this work we explore the application of nanoshell-mediated PTT as a chemosensitization strategy. We selected to use an in vitro model of inflammatory breast cancer (IBC), a rare and lethal subtype of breast cancer, for our studies to evaluate synergy between nanoshell-mediated PTT and dox. Due to its aggressive nature, the current standard of care for IBC consists of a multimodality approach including surgery, radiation, and chemotherapy with anthracyclines like dox. Despite this intense treatment regimen, most women with IBC eventually experience recurrence and die from their disease. Consequently, the 5-year survival rate for IBC patients is less than 50% and the 10-year survival rate is less than 35%.27,28 By comparison, the 10-year survival rate for all subtypes of breast cancer combined is 83%.29 In addition to poor treatment outcomes, IBC patients often face detrimental side effects, such as cardiotoxicity, because high drug doses may be required for effective tumor elimination. This toxicity can limit the long-term use of chemotherapy and reduce the overall success of treatment. In this work, we aimed to demonstrate that nanoshell-mediated PTT could potentiate the success of dox in IBC. By lowering the dosage of chemotherapy required for tumor elimination, the use of PTT could mitigate off-target effects while simultaneously improving patient outcomes. Here, we describe the results of our in vitro studies, which confirm that nanoshell-mediated PTT is an effective strategy for chemosensitization of IBC.
Materials and methods
Characterization of nanoshells
Nanoshells consisting of ~120 nm silica cores and ~15 nm thick gold shells were purchased from Nanospectra Biosciences, Inc. (Houston, TX, USA). The nanoshells’ extinction spectrum was characterized using a Cary 60 UV-Vis spectrophotometer (Agilent Technologies, Santa Clara, CA, USA) to confirm that peak extinction was near 808 nm, the wavelength of the laser utilized for nanoshell excitation in these studies. The nanoshells were visualized with scanning electron microscopy using a Hitachi S4700 Field-Emission Scanning Electron Microscope (Tokyo, Japan) and the diameter of 40 nanoparticles was measured using ImageJ software (NIH, Bethesda, MD, USA).
Cell culture
SUM149 IBC cells were generously gifted from the laboratory of Kenneth van Golen (Department of Biological Sciences, University of Delaware, Newark, DE, USA). We selected SUM149 cells for our studies because they are inherently resistant to dox.30 Accordingly, they provide a relevant model to evaluate whether mild PTT can sensitize IBC cells to dox. In addition, SUM149 cells are the most commonly used of the eight existing preclinical models of IBC. This is because they can be maintained in two- and three-dimensional culture, form tumors and metastasize in mice, and contain a high fraction of cancer stem cells, which are the subpopulation of cells implicated in treatment resistance and metastasis.31,32 Cells were maintained in Ham’s F12 growth medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 5% fetal bovine serum (Life Technologies), 1% l-glutamine (Life Technologies), 1 μg/mL hydrocortisone (Sigma-Aldrich Co., St Louis, MO, USA), 1% insulin–transferrin–selenium (Life Technologies), and 1% penicillin/streptomycin (Life Technologies). Cells were incubated at 37°C in a 5% CO2 environment. No ethical approval was required from the institutional review board of the University of Delaware for the use of this cell line.
Visualization of nanoshells in cell cultures
Cells were plated in a 24-well plate at a density of 4×105 cells per well and cultured for 24 hours. Cells were incubated with media or media supplemented with 1×109 nanoshells for 4 hours and then each well was rinsed three times with phosphate-buffered saline (PBS, Sigma-Aldrich Co.) to remove nanoshells that had not bound to or entered cells. Cells were fixed with a 4% paraformaldehyde solution for 15 minutes and then rinsed thoroughly with deionized (DI) water. A silver staining kit (Ted Pella, Inc., Redding, CA, USA) was used to stain the samples; silver stain nucleates on the gold surface of nanoshells to enable their visualization by light microscopy. After 15 minutes, the samples were rinsed thoroughly with DI water to stop the staining process. Samples were imaged using an AxioObserver Z1 microscope (Carl Zeiss Microscopy, LLC, Thornwood, NY, USA).
Assessment of IBC cell membrane permeability following PTT
This experiment aimed to demonstrate that IBC cells are sensitive to PTT. Cells were plated in a 96-well plate at a density of 3×104 cells per well and cultured for 48 hours. Cells were incubated with media or media supplemented with 7.5×108 nanoshells for 4 hours. Each well was then rinsed once with Dulbecco’s PBS (D-PBS) and replenished with fresh media. Cells were exposed to an 808 nm laser (B&W Tek, Newark, DE, USA) with a 2 mm diameter spot size at 80 W/cm2 for 3 minutes and then incubated for 30 minutes at 37°C. A fluorescent viability kit (Life Technologies) that contained calcein AM and ethidium homodimer-1 (EthD-1) was used to assess cell membrane permeability. In live cells, the nonfluorescent calcein AM molecule is converted to green fluorescent calcein after hydrolysis by intracellular esterases. Thus, live cells exhibit green fluorescence after metabolizing calcein AM. In comparison, EthD-1 is weakly fluorescent until it binds to DNA, at which point it emits strong red fluorescence. Because EthD-1 is impermeable to cells with an intact plasma membrane, it can be used to indicate loss of membrane integrity. In this work, following a 30-minute incubation with calcein AM and EthD-1, samples were rinsed, submerged with D-PBS, and then imaged using an AxioObserver Z1 microscope (Zeiss).
Assessment of rhodamine uptake by IBC cells following PTT
To demonstrate that low-dose PTT can increase IBC cell membrane permeability and consequently enhance drug uptake, we used flow cytometry to analyze the accumulation of rhodamine 123, a fluorescent dye, in cells treated with PTT or control therapies. Rhodamine 123 was substituted for dox in these studies because it is brighter than dox and allows analysis of uptake independent of the effects dox may have on cell function. Cells were plated in a 96-well plate at a density of 5×104 cells per well, cultured for 24 hours, and incubated with media or media supplemented with 1×109 nanoshells for 4 hours. Each well was washed once with D-PBS and then replenished with fresh media. Specified samples were irradiated with an 808 nm laser with a 7 mm spot size (to ensure exposure of the entire well to light) at 5.5 W/cm2 for 3 minutes. Thus, the four treatment groups included: media only, light only, nanoshells only, and PTT (ie, nanoshells and light). Following treatment, cells were incubated with media supplemented with 1 μM rhodamine 123 for 20 minutes, washed once with D-PBS, and then trypsinized and suspended in D-PBS. Cells were pelleted by centrifugation, suspended in 4% paraformaldehyde, and incubated for 15 minutes to ensure fixation. Cells were centrifuged again to remove the paraformaldehyde, suspended in D-PBS, and stored at 4°C overnight. The fluorescence of each sample was then measured using an Accuri C6 flow cytometer (BD Biosciences, San Jose, CA, USA). The mean fluorescence of cells that were never exposed to rhodamine 123 was also measured and subtracted from all other samples to account for background signal. The background-subtracted fluorescence intensity in each treatment group was then normalized to that of cells treated with only media and rhodamine 123. This experiment was repeated twice, with each biological replicate consisting of three technical replicates (three wells of each treatment type).
Visualization of dox in IBC cells following PTT
To ensure that PTT-induced membrane permeabilization enhances cellular drug uptake in addition to dye uptake, dox was visualized in PTT-treated IBC cells using fluorescence microscopy. Fluorescence microscopy was used instead of flow cytometry for these experiments because it enables quantification of dox distribution in different cellular compartments. This information is important because dox must enter the nucleus to elicit its effects. Cells were plated on a glass chamber slide at a density of 5×104 cells per well and cultured overnight. Cells were incubated in media supplemented with 5×109 nanoshells for 4 hours, then washed with D-PBS, and replenished with fresh media. PTT-treated cells were irradiated with an 808 nm laser with a 7 mm spot size at 5.5 W/cm2 for 3 minutes and compared to control cells that received nanoshells with no laser irradiation. Immediately following PTT, cells were incubated with 5 μM dox for 1 hour, washed with D-PBS, fixed with 4% formaldehyde, and mounted with ProLong Gold Antifade with 4’,6-diamidino-2-phenylindole (DAPI). Dox was visualized by fluorescence microscopy using a Zeiss AxioObserver Z1 with a Chroma filter set (495/20 nm bandpass excitation, 515 nm dichromatic beamsplitter, 540/30 nm bandpass emission). DAPI and differential interference contrast (DIC) images were also acquired so that intracellular dox fluorescence intensity could be quantified in specific regions-of-interest (ROIs) using ImageJ. ROIs included either whole cell outlines obtained from DIC images or nuclear outlines obtained from DAPI images. These ROIs were overlaid onto dox images and the mean fluorescence in each ROI was determined using ImageJ. Whole cell and nuclear analysis was performed on 332 and 339 cells, respectively, using images acquired with a 40× objective.
Evaluation of PTT combined with chemotherapy
The following experimental design was utilized to demonstrate that PTT could potentiate the cytotoxicity of dox in IBC cells. Cells were plated in a 96-well plate at a density of 2×104 cells per well and cultured for 24 hours. Cells were incubated with media or media supplemented with 1×109 nanoshells for 4 hours. Each well was washed once with D-PBS and then replenished with either media or media supplemented with 0.75 μM dox. All samples were irradiated with an 808 nm laser with a 7 mm spot size (to ensure exposure of the entire well to light) at 5.5 W/cm2 for 3 minutes. Thus, the four treatment groups consisted of: control (light only), light and 0.75 μM dox, PTT (ie, both nanoshells and NIR light), and PTT and 0.75 μM dox (ie, nanoshells, NIR light, and dox). Samples were incubated overnight at 37°C and then cell viability was analyzed using an AlamarBlue kit (Life Technologies), following the manufacturer’s instructions with a 3-hour incubation. Resazurin, the active ingredient in AlamarBlue, is nonfluorescent until it enters cells and is reduced to red-fluorescent resorufin. Thus, the fluorescence intensity provides a measure of cell viability. Fluorescence of treated samples was quantified using a BioTek Synergy H1 microplate reader (Winooski, VT, USA). This experiment was repeated four times, with each biological replicate consisting of three technical replicates (three wells of each treatment type).
Statistical analysis
Cell fluorescence data obtained from flow cytometry and cell viability data obtained from the AlamarBlue assay were analyzed using JMP software. An analysis of variance (ANOVA) with post hoc Tukey was performed for both experiments in order to determine which treatment groups were significantly different from each other at the 95% confidence level. A Student’s t-test was used to analyze differences in intracellular dox determined by fluorescence microscopy.
Results
Nanoshell characterization
Visualization of nanoshells with scanning electron microscopy (Figure 1A) and subsequent analysis with ImageJ software indicated a homogenous size distribution. The nanoshells had a diameter of 154±5 nm (mean ± standard deviation). Extinction characteristics were determined with a spectrophotometer and revealed the nanoshells had a peak plasmon resonance at ~775 nm (Figure 1B).
  | Figure 1 Characterization of the nanoshells used in this work. |
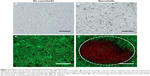
Nanoshell-mediated PTT increases membrane permeability of IBC cells
SUM149 IBC cells were incubated with nanoshells and silver stained in order to demonstrate effective binding of the nanoparticles to the cells. Figure 2A displays cells that were not exposed to nanoshells, while Figure 2B shows nanoshell-treated cells. The positive stain (dark regions) in Figure 2B confirms that nanoshells were able to adhere nonspecifically to SUM149 cells. The level of nanoshells present was sufficient to enable cellular membrane damage via PTT (Figure 2C and D). Upon NIR irradiation at 80 W/cm2 for 3 minutes, SUM149 cells not exposed to nanoshells maintained viability and membrane integrity, indicated by positive green calcein fluorescence and lack of red EthD-1 fluorescence (Figure 2C). In contrast, cells incubated with nanoshells prior to light exposure experienced loss of membrane integrity within the laser spot (outlined by the white dotted line), indicated by red EthD-1 fluorescence (Figure 2D). Cells outside the laser spot maintained their viability and membrane integrity, confirming that nanoshells alone are not cytotoxic.
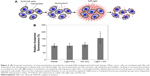
PTT increases rhodamine and dox accumulation in IBC cells
We hypothesized that low-dose PTT could increase cell membrane permeability to enhance drug accumulation in IBC cells (Figure 3A). To investigate the effect of low-dose PTT on drug uptake, we compared rhodamine 123 fluorescence in cells treated with: 1) media only, 2) NIR light only, 3) nanoshells only, or 4) PTT (ie, nanoshells and NIR light). Flow cytometry was used to determine the mean fluorescence of each group as described in the “Materials and methods” section. As shown in Figure 3B, cells exposed to only media, NIR light, or nanoshells displayed similar levels of rhodamine fluorescence, and the differences between groups were not significant according to ANOVA. Thus, neither light nor nanoshells alone are sufficient to enhance rhodamine uptake in cells. In contrast, cells treated with PTT displayed 56% higher rhodamine fluorescence than the media only group, confirming that PTT can enhance dye accumulation in cells. An ANOVA with post hoc Tukey validated that the fluorescence signal in the PTT-treated cells was significantly different from that in all other groups. The P-value for each comparison was: P=0.0025 for PTT versus media, P=0.0029 for PTT versus light only, and P=0.0112 for PTT versus nanoshells only.
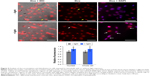
To demonstrate that these results apply to a clinically relevant drug, we used fluorescence microscopy to compare the accumulation and distribution of dox in cells treated with PTT or exposed to nanoshells without light irradiation. Cells exposed to nanoshells but not NIR light displayed some nuclear dox fluorescence, and minimal dox was observed within cytoplasmic regions (examples indicated by yellow arrows in Figure 4). Notably, both nuclear and cytoplasmic dox fluorescence increased in cells exposed to nanoshells and NIR light (eg, cytoplasmic regions with enhanced dox fluorescence are indicated by white arrows in Figure 4). Quantification of fluorescence intensity in whole cells or nuclei with ImageJ revealed that dox fluorescence increased by 20% in cells treated with PTT relative to those not exposed to NIR light. A Student’s t-test revealed that this difference in accumulation was significant, with a P-value <0.0001 for both nuclear and whole cell analysis.
PTT sensitizes IBC cells to dox
To investigate the chemosensitization effect of PTT, four experimental groups were compared: cells incubated with: 1) media only, 2) 0.75 μM dox (an intentionally subtherapeutic dose), 3) 1×109 nanoshells, and 4) both 1×109 nanoshells and 0.75 μM dox. Each group was exposed to NIR irradiation at 5.5 W/cm2 in the entire well for 3 minutes. After incubating overnight, cell viability was assessed with an AlamarBlue assay and results were normalized to the media control group (Figure 5). Cells incubated with the intentionally subtherapeutic dose of dox experienced minimal cytotoxicity, as indicated by viability of 97.7%±3.7%. Cells treated with only low-dose PTT demonstrated viability of 84.8%±2.9%. Finally, cells that received dox treatment and PTT demonstrated 64.5%±8.1% viability, which is ~20% less than the viability observed after PTT alone. An ANOVA with post hoc Tukey revealed that the viabilities of cells treated with either media or dox were significantly different from the viabilities of cells treated with standalone PTT (P<0.01) or combination PTT plus dox (P<0.001). In addition, the PTT and PTT plus dox groups were significantly different from each other, with a P-value of 0.0003.
To evaluate whether the impact of PTT and dox on cells was synergistic or additive, we utilized the method introduced by Hahn et al1 in their seminal paper on thermochemotherapy. Per this method, expected additive effects can be calculated by multiplying the surviving fraction from two individual treatments. Accordingly, in our studies, if the effect of PTT and dox was additive, we would expect to observe viability in the combined treatment group of 82.9%±5.5% (mean ± standard deviation). The observed viability, however, was 64.5%±8.1%, ~20% lower than predicted for an additive effect. The use of a Student’s t-test to compare the projected additive effect versus the observed effect across all four biological replicates of the experiment revealed that the difference in survival was significant, with a P-value of 0.009. Therefore, we can conclude that PTT and dox are synergistic, rather than additive, against IBC cells.
Discussion
Our studies demonstrate that IBC cells experience loss of plasma membrane integrity upon exposure to nanoshell-mediated PTT (Figures 2–4), resulting in increased drug accumulation in cells (Figures 3 and 4) and decreased cell viability (Figure 5) following combination therapy. The increased cell membrane permeability, evaluated qualitatively by ethidium homodimer-1 staining under high-dose irradiation conditions (Figure 2) and quantitatively by rhodamine 123 uptake (Figure 3) and dox uptake (Figure 4) under low-dose irradiation conditions, was observed only where nanoshells and light were combined; this result is in agreement with our previous studies exploring nanoshell-mediated PTT and confirms that laser irradiation alone and nanoshells alone are insufficient to induce damage of IBC cellular membranes.9,33 To evaluate the combined effects of PTT and dox, subtherapeutic doses of each treatment were administered either alone or in combination. By using a dose of dox that causes minimal to no cell death, enhancement of chemotherapy via PTT could be assessed.34,35 Because the combination treatment group (PTT and dox) produced a 20% decrease in viability compared to the PTT only group (Figure 5), it can be concluded that nanoshell-mediated PTT enhanced the effectiveness of the chemotherapy treatment. Furthermore, using the method of Hahn et al1 to calculate the predicted additive effect of PTT and dox, we would expect viability of ~83% under the conditions used in our studies. The combination of PTT and dox experimentally resulted in ~65% cell viability, demonstrating a synergistic effect. Our proposed mechanism for this chemosensitization is that the heat-induced increase in membrane permeability caused by PTT enables more dox to be taken up and retained by the cells (Figure 3A), resulting in increased cytotoxic effects at doses that would ordinarily be subtherapeutic.36–38 This mechanism is supported by the 56% increase in rhodamine fluorescence observed inside cells following PTT (Figure 3B) and the 20% increase in dox fluorescence observed inside cells following PTT (Figure 4). Further, we have demonstrated that the 20% increase in intracellular dox localizes to the nucleus to effectively induce cytotoxicity. Therefore, we conclude that nanoshell-mediated PTT can enhance chemotherapy in IBC cells.
Our findings have clinical relevance in that the use of PTT to potentiate chemotherapy can reduce the dosage of drug necessary to achieve a therapeutic response. This is important because cardiotoxicity remains an unresolved limitation of dox chemotherapy and reduces the amount of drug that can be delivered systemically over time. Our data suggest that using PTT as a chemosensitization strategy in IBC may overcome this limitation by increasing the efficacy of lower doses of dox. Importantly, nanoshell application should not introduce additional cardiotoxicity. Nanoshells delivered intravenously have shown no toxicity to the heart in studies performed in mice, rats, and Beagle dogs for time durations up to 404 days.9,39 Further, a recently completed evaluation of the safety of nanoshells in humans demonstrates that nanoshells have an excellent clinical safety profile.40 Since nanoshells display no toxicity to the heart or other vital organs, it may be concluded that combining PTT with low-dose dox should result in an overall improvement in therapeutic ratio compared to conventional treatment protocols for IBC. Accordingly, using PTT to sensitize IBC tumors to dox may improve both the treatment outcome and patient quality of life by enhancing therapeutic impact while minimizing side effects.
Conclusion
This work confirms that nanoshell-mediated PTT is a viable strategy to potentiate the effect of chemotherapy in IBC. While SUM149 cells were utilized as proof-of-principle here, we anticipate that PTT will prove useful to enhance chemotherapy in other IBC and non-IBC cancers, as the physical mechanism of chemosensitization it provides should be consistent across cell types. In the future, this strategy could be employed to improve patient outcomes by enhancing tumor regression while enabling treatment systemically with nontoxic doses of chemotherapeutic agents. Previous work has demonstrated that PTT can be used to increase vascular density and promote drug delivery to tumors,41 and our work suggests that the cellular level effects of PTT may provide additional therapeutic benefits once drugs arrive at the tumor site. Together, these findings support the continued development of combined PTT and systemic low-dose chemotherapy using other drugs in addition to dox, and also for other cancer types in addition to IBC. Furthermore, our results suggest that researchers should continue to explore the development of more advanced nanoparticle-drug conjugates for combined PTT and chemotherapy42 since the dual therapeutic approach provides more than an additive effect on cancer cell viability.
Acknowledgments
This project was supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health (NIH) under grant number U54-GM104941. The Delaware INBRE program, with a grant from NIGMS (P20-GM103446) from the NIH, also supported this project. The authors acknowledge Deborah Powell from the Delaware Bio-Imaging Center for assistance with scanning electron microscopy.
Disclosure
BLF received support from the University of Delaware Summer Scholars program. JRM received support from the Department of Defense through a National Defense Science and Engineering Graduate Fellowship. The authors report no other conflicts of interest in this work.
References
Hahn GM, Braun J, Har-Kedar I. Thermochemotherapy: Synergism between hyperthermia (42°–43°) and adriamycin (or bleomycin) in mammalian cell inactivation. Proc Natl Acad Sci U S A. 1975;72:937–940. | ||
Hirsch LR, Stafford RJ, Bankson JA, et al. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc Natl Acad Sci U S A. 2003;100:13549–13554. | ||
Day ES, Morton JG, West JL. Nanoparticles for thermal cancer therapy. J Biomech Eng. 2009;131:074001. | ||
Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46:6387–6392. | ||
Day E, Thompson P, Zhang L, et al. Nanoshell-mediated photothermal therapy improves survival in a murine glioma model. J Neurooncol. 2011;104:55–63. | ||
Weissleder R. A clearer vision for in vivo imaging. Nat Biotechnol. 2001;19:316–317. | ||
Pérez-Hernández M, del Pino P, Mitchell SG, et al. Dissecting the molecular mechanism of apoptosis during photothermal therapy using gold nanoprisms. ACS Nano. 2015;9:52–61. | ||
Melamed JR, Edelstein RS, Day ES. Elucidating the fundamental mechanisms of cell death triggered by photothermal therapy. ACS Nano. 2015;9:6–11. | ||
Day ES, Zhang L, Thompson PA, et al. Vascular-targeted photothermal therapy of an orthotopic murine glioma model. Nanomedicine. 2012;7:1133–1148. | ||
O’Neal DP, Hirsch LR, Halas NJ, Payne JD, West JL. Photo-thermal tumor ablation in mice using near infrared-absorbing nanoparticles. Cancer Lett. 2004;209:171–176. | ||
Dickerson EB, Dreaden EC, Huang XH, et al. Gold nanorod assisted near-infrared plasmonic photothermal therapy (PPTT) of squamous cell carcinoma in mice. Cancer Lett. 2008;269:57–66. | ||
Day ES, Bickford LR, Slater JH, Riggall NS, Drezek RA, West JL. Antibody-conjugated gold–gold sulfide nanoparticles as multifunctional agents for imaging and therapy of breast cancer. Int J Nanomedicine. 2010;5:445–454. | ||
Lu W, Xiong C, Zhang G, et al. Targeted photothermal ablation of murine melanomas with melanocyte-stimulating hormone analog-conjugated hollow gold nanospheres. Clin Cancer Res. 2009;15:876–886. | ||
Chen J, Glaus C, Laforest R, et al. Gold nanocages as photothermal transducers for cancer treatment. Small. 2010;6:811–817. | ||
Nanospectra Biosciences, Inc. Efficacy study of AuroLase therapy in subjects with primary and/or metastatic lung tumors. Available from: https://clinicaltrials.gov/ct2/show/NCT01679470; NLM Identifier: NCT01679470. Accessed December 20, 2014. | ||
Nanospectra Biosciences, Inc. Pilot study of AuroLase therapy in refractory and/or recurrent tumors of the head and neck. Available from: https://clinicaltrials.gov/ct2/show/NCT00848042; NLM Identifier: NCT00848042. Accessed December 20, 2014. | ||
Banu H, Stanley B, Faheem SM, Seenivasan R, Premkumar K, Vasanthakumar G. Thermal chemosensitization of breast cancer cells to cyclophosphamide treatment using folate receptor targeted gold nanoparticles. Plasmonics. 2014;9:1341–1349. | ||
Mehtala JG, Torregrosa-Allen S, Elzey BD, Jeon M, Kim C, Wei A. Synergistic effects of cisplatin chemotherapy and gold nanorod-mediated hyperthermia on ovarian cancer cells and tumors. Nanomedicine. 2014;9:1939–1955. | ||
Hauck TS, Jennings TL, Yatsenko T, Kumaradas JC, Chan WCW. Enhancing the toxicity of cancer chemotherapeutics with gold nanorod hyperthermia. Adv Mater. 2008;20:3832–3838. | ||
You J, Zhang G, Li C. Exceptionally high payload of doxorubicin in hollow gold nanospheres for near-infrared light-triggered drug release. ACS Nano. 2010;4:1033–1041. | ||
Park H, Yang J, Lee J, Haam S, Choi IH, Yoo KH. Multifunctional nanoparticles for combined doxorubicin and photothermal treatments. ACS Nano. 2009;3:2919–2926. | ||
Guo R, Zhang L, Qian H, Li R, Jiang X, Liu B. Multifunctional nanocarriers for cell imaging, drug delivery, and near-IR photothermal therapy. Langmuir. 2010;26:5428–5434. | ||
Li X, Takashima M, Yuba E, Harada A, Kono K. PEGylated PAMAM dendrimer-doxorubicin conjugate-hybridized gold nanorod for combined photothermal-chemotherapy. Biomaterials. 2014;35:6576–6584. | ||
Wang Z, Chen Z, Liu Z, et al. A multi-stimuli responsive gold nanocage-hyaluronic platform for targeted photothermal and chemotherapy. Biomaterials. 2014;35:9678–9688. | ||
Zhang W, Guo Z, Huang D, Liu Z, Guo X, Zhong H. Synergistic effect of chemo-photothermal therapy using PEGylated graphene oxide. Biomaterials. 2011;32:8555–8561. | ||
Sherlock SP, Tabakman SM, Xie L, Dai H. Photothermally enhanced drug delivery by ultrasmall multifunctional FeCo/graphite shell nanocrystals. ACS Nano. 2011;5:1505–1512. | ||
Gonzalez-Angulo AM, Hennessy BT, Broglio K, et al. Trends for inflammatory breast cancer: is survival improving? Oncologist. 2007;12:904–912. | ||
Bristol IJ, Woodward WA, Strom EA, et al. Locoregional treatment outcomes after multimodality management of inflammatory breast cancer. Int J Radiat Oncol Biol Phys. 2008;72:474–484. | ||
American Cancer Society. Cancer Facts & Figures 2014. Atlanta, GA: American Cancer Society; 2014. | ||
Pan Q, Bao LW, Kleer CG, Brewer GJ, Merajver SD. Antiangiogenic tetrathiomolybdate enhances the efficacy of doxorubicin against breast carcinoma. Mol Cancer Ther. 2003;2:617–622. | ||
Robertson FM, Chu K, Fernandez SV, et al. Genomic profiling of pre-clinical models of inflammatory breast cancer identifies a signature of epithelial plasticity and suppression of TGF-β signaling. J Clin Exp Pathol. 2012;2:5. | ||
Lacerda L, Woodward WA. Models of inflammatory breast cancer. In: Ueno NT, Cristofanilli M, editors. Inflammatory Breast Cancer: An Update. New York, NY: Springer Science + Business Media; 2012:139–150. | ||
Lowery AR, Gobin AM, Day ES, Halas NJ, West JL. Immunonanoshells for targeted photothermal ablation of tumor cells. Int J Nanomedicine. 2006;1:149–154. | ||
Chou T. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70:440–446. | ||
Tallarida RJ. Drug synergism: its detection and applications. J Pharmacol Exp Ther. 2001;298:865–872. | ||
Bettaieb A, Wrzal PK, Averill-Bates DA. Hyperthermia: cancer treatment and beyond. In: Rangel L, editor. Cancer Treatment: Conventional and Innovative Approaches. Rijeka: InTech; 2013. | ||
Bischof JC, Padanilam J, Holmes WJ, et al. Dynamics of cell membrane permeability changes at supraphysiological temperatures. Biophys J. 1995;68:2608–2614. | ||
Issels RD. Hyperthermia adds to chemotherapy. Eur J Cancer. 2008;44:2546–2554. | ||
Gad SC, Sharp KL, Montgomery C, Payne JD, Goodrich GP. Evaluation of the toxicity of intravenous delivery of Auroshell particles (gold-silica nanoshells). Int J Toxicol. 2012;31(6):584–594. | ||
Stern JM, Solomonov VVK, Sazykina E, Schwartz JA, Gad SC, Goodrich GP. Initial evaluation of the safety of nanoshell-directed photothermal therapy in the treatment of prostate disease. Int J Toxicol. 2015. doi:10.1177/1091581815600170. | ||
Park JH, von Maltzahn G, Xu MJ, et al. Cooperative nanomaterial system to sensitize, target, and treat tumors. Proc Natl Acad Sci U S A. 2010;107(3):981–986. | ||
Strong LE, West JL. Hydrogel-coated near infrared absorbing nanoshells as light-responsive drug delivery vehicles. ACS Biomaterials Science and Engineering. 2015;1:685–692. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.