Back to Journals » International Journal of Nanomedicine » Volume 13
Nanoselenium prevents eimeriosis-induced inflammation and regulates mucin gene expression in mice jejunum
Authors Alkhudhayri AA , Dkhil MA , Al-Quraishy S
Received 12 January 2018
Accepted for publication 16 February 2018
Published 3 April 2018 Volume 2018:13 Pages 1993—2003
DOI https://doi.org/10.2147/IJN.S162355
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Thomas Webster
Abdulsalam A Alkhudhayri,1 Mohamed A Dkhil,1,2 Saleh Al-Quraishy1
1Department of Zoology, College of Science, King Saud University, Riyadh, Saudi Arabia; 2Department of Zoology and Entomology, Faculty of Science, Helwan University, Cairo, Egypt
Background: Although elemental selenium has been found to be effective against Eimeria, no study has yet investigated the effects of selenium nanoparticles (SeNPs) on the Eimeria parasite. The aim of this study, therefore, was to evaluate the ameliorative effect of SeNPs compared with elemental selenium on mice jejunum infected with sporulated oocysts of Eimeria papillata.
Methods: The mice were divided into 4 groups, with the first being the non-infected, control group, and the second, third, and fourth groups being orally inoculated with 1,000 sporulated oocysts of E. papillata. The third and fourth groups also received, respectively, an oral dose of 0.1 mg/kg sodium selenite and 0.5 mg/kg SeNPs daily for 5 consecutive days.
Results: The infection induced severe histopathological jejunal damage, reflected in the form of destroyed jejunal mucosa, increased jejunal oxidative damage, a reduction in the number of jejunal goblet cells, and increased production of pro-inflammatory cytokines, quantified by real-time polymerase chain reaction. Treatment of mice with SeNPs significantly decreased the oocyst output in the feces by ~80%. Furthermore, the number of parasitic stages counted in stained jejunal paraffin sections was significantly decreased after the mice were treated with SeNPs. In addition, the number of goblet cells increased from 42.6±7.3 to 95.3±8.5 cells/10 villus-crypt units after treatment. By day 5 post-infection with E. papillata, SeNPs could be seen to have significantly increased the activity of glutathione peroxidase from 263±10 to 402.4±9 mU/mL. Finally, SeNPs were able to regulate the gene expression of mucin 2, interleukin 1β, interleukin 6, interferon-γ, and tumor necrosis factor α in the jejunum of mice infected with E. papillata.
Conclusion: The results collectively showed that SeNPs are more effective than sodium selenite with regard to their anti-coccidial, anti-oxidant, and anti-inflammatory role against eimeriosis induced in the jejunum of mice.
Keywords: selenium nanoparticles, eimeriosis, jejunum, mice, inflammation, mucin 2 gene
Introduction
Coccidiosis is a severe condition affecting a large number of vertebrates. It is caused by the coccidian parasite of the genus Eimeria and affects the digestive tract of its hosts, causing diarrhea and weight loss. Eimerian parasites are both species and site specific, are able to reproduce rapidly, and can easily infect young animals. The parasite spreads from one animal to the next by contact with infected feces,1 and this direct fecal–oral life cycle is ideal for spreading rapidly through susceptible hosts.2
Domestic animals suffering from coccidiosis exhibit low meat and milk production due to malabsorption from the intestinal tract, and coccidiosis is, therefore, a significant issue in the meat and dairy industry, internationally. Attempts to tackle coccidiosis, along with other infectious diseases of animals, have resulted in a tremendous increase in the value of veterinary drugs bought worldwide, rising from $8.65 billion in 1992 to $20.1 billion in 2010. By 2018, this figure is expected to increase further to $42.9 billion.3–5 Despite these large sums, the side effects of synthetic anticoccidial drugs cause serious problems in animals, and therefore natural products that can serve as promising sources of novel anti-eimerial agents are increasingly being sought.1 These products should not only target the parasite, but also play a role in protecting the organs of the host from the consequences of the infection.1
Nanotechnologies are now offering new opportunities for innovative medical treatments to fight parasitic infection. In particular, nanoparticles are considered to be promising agents for the treatment of several parasitic diseases, since particles with nanometer dimension have novel properties that are different from those of both isolated atoms and bulk material. For example, nano-sized selenium exhibits an important biological activity by scavenging free radicals in a size-dependent fashion.6 It has also shown promise as a therapeutic regenerative material against several diseases, including cancer, by preventing cellular damage.7 Rayman reported that selenium nanoparticles (SeNPs) have anti-oxidant and anti-inflammatory effects,8 and, recently, Dkhil et al showed that nanoselenium could act against murine schistosomiasis.9 Furthermore, SeNPs are also known to exhibit anti-leishmanial effects.10
Although Dkhil et al reported the anti-eimerial role of zinc oxide nanoparticles,11 to our knowledge, no study has yet investigated the effects of SeNPs on the Eimeria parasite, although elemental selenium has been found to be effective against Eimeria.12 Since the potential effect of a substance depends upon its size,13 the aim of this study was to evaluate the ameliorative effect of SeNPs compared to elemental selenium on mice jejunum infected with sporulated oocysts of E. papillata.
Materials and methods
SeNPs and characterization
SeNPs with particle sizes of 5–50 nm were purchased from Nanocs Inc. (Boston, MA, USA). The SeNPs were separated using a high-speed centrifuge and re-dispersed in aqueous medium using a sonicator.13 Transmission electron microscopy (TEM) was used to characterize the shape and size of the SeNPs, using a JEOL JEM-2100 (JEOL Ltd., Tokyo, Japan) high-resolution TEM at an accelerating voltage of 200 kV.
Animals
Thirty-two male C57BL/6 mice, weighing 20–25 g and aged 10–12 weeks, were obtained from the animal facilities of the Zoology Department, College of Science, King Saud University, Riyadh, Saudi Arabia. The mice were fed a standard diet and water ad libitum. All animal procedures and experiments in this study were approved by the ethical committee at the Department of Zoology, King Saud University (DGS 1438/6/20) and were conducted according to the guidelines for animal experimentation issued by King Saud University.
Mice infection and experiment design
To prepare the parasite infective stage (sporulated oocysts) of E. papillata, fecal pellets of infected mice were suspended in 2.5% (w/v) potassium dichromate and diluted in saturated sodium chloride for oocyst flotation. Using a McMaster counting chamber, the oocysts were counted and expressed as number of oocysts per gram of wet feces.14
The animals were divided into 4 groups, with 10 mice per group. The first group was the non-infected, control group, which was gavaged with 100 μL of distilled water. The second, third, and fourth groups were orally inoculated with 1,000 sporulated oocysts of E. papillata suspended in 100 μL of distilled water.15 The third and fourth groups received, respectively, an oral dose of 0.1 mg/kg sodium selenite (NaSe)12 and 0.5 mg/kg SeNPs suspended in 100 μL of distilled water daily for 5 consecutive days.7 The dose was chosen according to the results of our previous experiments (Figure S1). For clarification, a scheme presented in Figure S2 shows the steps, duration, and dose administration.
Evaluation of the histopathological changes and goblet cell responses
Pieces of jejuna were freshly cut and fixed in 10% neutral buffered formalin for 24 hours. Specimens were then dehydrated with ethanol and embedded in paraffin. After this, 5 μm paraffin-embedded sections were stained with H&E. The number of E. papillata parasitic stages in 10 well-orientated villus-crypt units (VCU) was counted. Scoring for jejunum inflammatory lesions was carried out according to Dommels et al.16 The histological score was indicated for infiltrations of mononuclear cells, neutrophils, eosinophils, and plasma cells, as well as for tissue destruction and tissue repair. A rating score between 0 (no change from normal tissue) and 3 (for lesions present in most areas and all jejunal layers) was given for each aspect of inflammatory lesions, tissue destruction, and tissue repair. In order to better reflect the histopathological changes associated with eimeriosis, the sum of the inflammatory lesions was doubled to give more weight to this parameter.
Paraffin sections were stained with Alcian blue to help identify goblet cells and the number of goblet cells in 10 VCU was counted for the jejunum of each animal.17
Evaluation of glutathione peroxidase (GPx) activity
Mice jejuna were weighed and homogenized in an ice-cold medium that contained 50 mM Tris–HCl (Sigma, St Louis, MO, USA) at pH 7.4. The jejunum homogenate was centrifuged at 2,000×g for 10 min at 4°C. The supernatants were used for this assay.
GPx was measured according to the method described by Paglia and Valentine.18 This is an indirect measure of the activity of GPx. In brief, GPx stimulates the reduction of glutathione (GSH). Oxidized GSH changes back to the reduced state (GSH) by means of the nicotinamide adenine dinucleotide phosphate (NADPH)-dependent reductase. The oxidation of NADPH to NADP+ is accompanied by a decrease in absorbance at 340 nm, providing a spectrophotometric means for monitoring GPx enzyme activity. To assay GPx, a jejunum tissue homogenate is added to a solution containing GSH, GSH reductase, and NADPH, and the enzymatic reaction is initiated by adding the substrate. This reaction results in a decreased absorbance at 340 nm, which is directly related to GPx activity.
Gene expression analysis
The total RNA was isolated from pieces of mice jejuna using Trizol (Thermo Fisher Scientific, Waltham, MA, USA). Contaminating genomic DNA was digested with DNase, using the DNA-free™ kit (Applied Biosystems, Darmstadt, Germany). Then, cDNA was synthesized using the QuantiTect® Reverse Transcription kit (Qiagen, Hilden, Germany). Real-time polymerase chain reaction (PCR) was performed using the QuantiTect™ SYBR® Green PCR kit (Qiagen) using a TaqMan 7500 (Applied Biosystems) and the gene-specific QuantiTect™ primer assay (Qiagen), according to the manufacturer’s instructions and with the following primers purchased from Qiagen: MUC2 (Mm_Muc2_2_SG, Cat. No. Mm_Muc2_2_SG), IL-1β (Mm_Il1β_2_SG, Cat. No. QT01048355), IL-6 (Mm_Muc4_2_SG, Cat. No. QT00138663), IFN-γ (Mm_Ifng_1_SG, Cat. No. QT01038821), TNF-α (Mm_Tnf_1_SG, Cat. No. QT00104006), iNOS (Mm_Nos2_1_SG, Cat. No. QT00100275), CASP3 (Mm_Casp3_1_SG, QT00260169), BCL2 (Mm_Bcl2_1_SG, Cat. No. QT00156282), BAX (Mm_Bax_1_SG, Cat. No. QT00102536), and GAPDH (Mm_Gapdh_3_SG, Cat. No. QT01658692). Following an initial incubation at 50°C for 2 min, Taq polymerase was activated at 95°C for 10 min, followed by 45 cycles of 95°C for 15 s, 60°C for 35 s, and 72°C for 30 s. The PCR product was measured as SYBR green fluorescence intensity at the end of the extension phase. Relative quantitative evaluation of the amplification data was performed using the TaqMan 7500 system software v.1.2.3f2 (Applied Biosystems) and following the 2−ΔΔc T method.19 Expression of the genes was compared to that of glyceraldehyde 3-phosphate dehydrogenase.20
Statistical analysis
Significance was evaluated by one-way analysis of variance, and statistical comparisons between the groups were performed by Duncan’s test, using a statistical package program (SPSS version 17.0). All values are expressed as the mean and the standard error of the mean (SEM). All p-values are two-tailed and p≤0.05 is considered as significant for all statistical analyses in this study.
Results
Figure S1 shows that while SeNPs exert a dose-dependent inhibitory effect on oocyst output, NaSe reduces the number of oocysts only at a dose of 0.1 mg/kg. Based on this initial assessment, mice infected with E. papillata were administered doses of 0.1 mg/kg of NaSe or 0.5 mg/kg of SeNPs. In the latter case, TEM was first used to confirm the size (10–30 nm) and spherical shape of the SeNPs (Figure 1).
  | Figure 1 Size characterization of SeNPs by TEM. Scale bar=100 nm. |
The infection induced a high number of oocysts to be output in the feces (1,693×103±339 oocysts/g feces) on day 5 post-infection (p.i.). Treatment of the infected mice with 0.5 mg/kg SeNPs significantly (p≤0.01) reduced the oocyst count in the expelled feces by ~80% when compared with that in the infected group (Table 1). The results of treatment with nano-sized selenium were better than those with 0.1 mg/kg NaSe (Table 1).
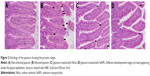
Examination of the H&E-stained jejunal sections revealed that there were ~69.4±7 E. papillata development stages per 10 VCU in the infected mice villi (Figure 2). Treatment of the infected mice with NaSe and SeNPs significantly decreased the number of development stages by 25±5 and 16±4 stages/10 VCU, respectively (Table 1).
Histopathological alterations were evident in the infected jejuna, including vacuolated injured jejunal mucosa, disintegration of submucosa with the presence of lymphatic infiltrations and some damaged muscle fibers. In addition to the apparent destruction and shedding of the epithelia of the jejunum due to the parasitic invasion, the inflammatory status of the jejunum was assessed using a semi-quantitative scoring system devised by Dommels et al.16 Based on this scoring, while the infected group was found to have suffered moderate inflammatory injury (Figure 3), it was evident that both NaSe and SeNPs could significantly improve the inflammatory lesions induced in mice by E. Papillata infection (Figure 3).
  | Figure 3 Jejunum histological injury scores on day 5 post-infection for control, non-infected, and infected mice, and for infected mice treated with either NaSe or SeNPs. |
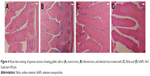
Examination of sections stained with Alcian blue (Figure 4) showed that on day 5 p.i. with E. papillata, the number of goblet cells per 10 VCU was decreased compared with that in the control non-infected mice. Nonetheless, the number of goblet cells was significantly higher in the group treated with SeNPs than in either the untreated infected group or the NaSe-treated group (Figure 5).
Figure 6 shows the downregulation of muc2 mRNA in the jejuna of mice infected with E. papillata. The gene was significantly upregulated (by ~2-folds); however, after the mice were treated with SeNPs, the NaSe-treated group also showed increased expression of muc2 but to a lesser degree than that shown by the SeNP-treated group (Figure 6).
The level of GPx was significantly reduced in the jejunal homogenate taken from the E. papillata infected group, compared with the control. The GPx level was significantly restored, however, in both the NaSe- and SeNPs-treated groups, compared with the untreated infected group (Figure 7).
Infection-induced inflammation was recorded in the mice jejuna on day 5 p.i. through the increased mRNA expression of interleukin 1β, interferon-γ, and tumor necrosis factor α. However, treatment with NaSe or SeNPs significantly ameliorated the expression of these inflammation-linked genes (Figure 8).
Discussion
More attention is needed on the use of nanoselenium as an anti-parasitic agent, due to its low toxicity and high bioavailability.21,22 In the present study, we used SeNPs for the first time as an anti-eimerial agent.
E. papillata infection begins with the oral uptake of sporulated oocysts, resulting in the release of sporozoites that, in turn, invade the site-specific jejunal epithelial cells.23 The intracellular stages of the parasites rapidly multiply in asexual schizogonic cycles before oocysts are formed.24 Finally, the oocysts are released with the feces and can be highly infectious. The treatment for eimeriosis currently depends on anti-coccidial drugs such as toltrazuril.25 Such drugs, however, induce problematic residual storage of these compounds or their metabolites in animal tissues and/or in their products, such as eggs.11 As a result, natural products and nanoparticles are now being considered as promising sources of novel anti-eimerial agents.11,26–28 Compared to many of the extracts from natural products that have been used against eimeriosis, like extracts from neem,26 pomegranate,27 and garlic,28 SeNPs were found to be more effective as they significantly reduce the oocyst output by about 80% (Table 1).
Selenium has already been shown to be able to reduce parasitic infection in the intestine. Smith et al revealed that mice were able to expel the nematode worm Heligmosomoides bakeri more efficiently following selenium treatment.29 Our previous research, meanwhile, has indicated that selenium can significantly lower oocyst output in the feces of mice infected with E. papillata.12 The results of this new study reinforce the earlier work by revealing that a dose of 0.1 mg/kg of NaSe caused a substantial lowering of oocyst output (which may be attributed to the increased mice immune response to the parasite),30,31 but that SeNPs were an even more effective inhibitor of E. papillata infection (Table 1; Figures 2 and 3). As reported above, these doses were chosen on the basis of our previous research,12 but also on the basis of the intention to use a supranutritional dose, since it has been confirmed that supranutritional levels of Se or nano-Se cause no toxicity when given to mice (0.5–1.5 mg Se/kg, daily, for 7 days).32 Moreover, Hassnin et al7 reported that, SeNPs are highly bioactive agents, with low toxicity and good absorptive ability due to the interaction between SeNPs and −NH2, C=O, −COO, and −C−N− groups of proteins. In general, our results are in agreement with other investigations reporting that SeNPs are much more efficient than NaSe.33,34
SeNPs could improve the infection-induced histopathological changes in mice jejunum. This was evidenced by the significant reduction of the jejunum histological injury score (Figure 3) and the improvement in the hypoplasia of goblet cells induced by infection. Such ameliorative effects on the histopathological changes may be due to the previously proved anticoccidial effects of selenium and the associated reduction in the number of intracellular parasitic stages, and hence the parasitic load and consequent damage to the infected jejunal tissue.12 Comparing our results concerning SeNPs with that of Dkhil et al11 using zinc oxide nanoparticles as an anti-coccidial agent, it is evident that SeNPs offer the potential for a more pronounced improvement in the histopathological alterations induced in the jejunum by E. papillata infection.
Speckmann and Steinbrenner have reported on the role of selenium and selenoproteins during inflammation; showing that intestinal epithelial cells use dietary selenium compounds for the synthesis of selenoprotein P, which might support the intestinal immune system by providing selenium to antibody-producing plasma cells.35 Some studies have also provided evidence that the mucus released by goblet cells can function as a defensive barrier,36,37 bearing in mind that, during eimeriosis, the Eimeria parasite invades and destroys goblet cells.26,38 In this context, it is interesting that the present results indicate the upregulation of the goblet cell-specific gene muc2 in mice treated with SeNPs. Several diseases are associated with altered mucin gene expression, including colon cancer.38–40 Jaspers et al reported that selenium controls mucus secretion during respiratory diseases.41 Moreover, Lee et al reported on the protective role of selenium against mucosal injury.42
In the jejunum, toxic effects due to the accumulation of reactive oxygen species (ROS) cause destructive changes in the epithelia.26 In this regard, feed additives are one of the tools used by nutritionists to combat cellular oxidative stress. Nève et al and Yan et al reported that selenium, as a component of selenoproteins, has a metabolic role in preventing oxidative damage to body tissues and Huang et al have reported that SeNPs are able to suppress inflammation by restraining nitric oxide production and enhancing anti-oxidant enzyme activity.43–45 In this context, GPx is one of the family of antioxidant enzymes that uses GSH to reduce peroxides.46 Generally, GPx has an important role in the reduction of ROS levels and in mitigating inflammatory pathways in the intestinal mucosa. The GPx enzymes are able to use selenium at their active sites to detoxify ROS.43 In our study, SeNPs are shown significantly to increase the activity of GPx in mice infected with E. papillata (Figure 7).
Eimeriosis also stimulates the production of pro-inflammatory cytokines that play an important role in the jejunal inflammation that is a feature of the disease.28 The inflammation caused by eimeriosis is associated with migration of blood leukocytes to the jejunal mucosa, and these activated leukocytes then guide epithelial cells to cascades of biochemical and cellular events, leading to the production of superoxide radical anions (O2−).28 It is well known that the increased levels of pro-inflammatory cytokines observed in this study on day 5 p.i. with E. papillata (Figure 8) amplify the inflammatory cascade and result in damage to the intestinal tissues of mice (Figure 3). In this context, Lee et al have reported on the anti-inflammatory role of selenium during intestinal injury,42 and selenium has also been shown to significantly downregulate levels of pro-inflammatory cytokines and affect gene expression, signaling pathways and cellular functions involved in inflammation.47 Comparing our results when using SeNPs against eimeriosis and both our previous work using zinc oxide nanoparticles,11 and some natural extracts,28 it is evident that SeNPs have a more potent anti-inflammatory effect.
Our study has built on this work to reveal the anti-inflammatory effect of both NaSe and SeNPs in the context of inflammation induced by Eimeria infection. In this regard, we showed that treatment with selenium is effective in ameliorating the upregulation of genes associated with inflammation (Figure 8) and this was further reflected in the histological results (Figure 3). Furthermore, we showed that SeNPs had a more marked beneficial effect than NaSe across all the investigated pro-inflammatory genes, and in terms of the histological outcomes. Generally, when comparing the current results to those using natural products as anti-eimerial agents, it is evident that SeNPs can more effectively improve eimeriosis-induced injuries.26–28
Conclusion
Our results collectively show that SeNPs are more effective than NaSe, at least at a parasitological level, at reducing both the number of E. papillata parasitic stages in the jejunum and in decreasing the number of oocysts expelled in mice feces, and therefore could be used as an anti-eimerial, anti-oxidant, and anti-inflammatory agent involved in immunoregulation.43 Further studies are required to understand the mechanism of action of SeNPs on both the host and the parasite.
Acknowledgment
This project was supported by King Saud University, Deanship of Scientific Research, College of Science Research Center.
Disclosure
The authors report no conflicts of interest in this work.
References
Pakandl M. Selection of a precocious line of the rabbit coccidium Eimeria flavescens Marotel and Guilhon (1941) and characterisation of its endogenous cycle. Parasitol Res. 2005;97(2):150–155. | ||
Mehlhorn H. Encyclopedic Reference of Parasitology. 4th ed. 2014. | ||
Animal health market to hit $43 billion in five years. Western FarmPress. 2012. Available from: http://westernfarmpress.com/management/animal-health-market-hit-43-billion-five-years. Accessed December 3, 2017. | ||
Reports Q. China veterinary drug industry 2012 market research report. 2012. Available from: http://www.qyresearchreports.com/report/china-veterinary-drug-industry-2012-market-research-report.htm. Accessed December 3, 2017. | ||
Hao H, Cheng G, Iqbal Z, et al. Benefits and risks of antimicrobial use in food-producing animals. Front Microbiol. 2014;5:288. | ||
Peng D, Zhang J, Liu Q, Taylor E. Size effect of elemental selenium nanoparticles (Nano-Se) at supranutritional levels on selenium accumulation and glutathione S-transferase activity. J Inorg Biochem. 2007;101(10):1457–1463. | ||
Hassnin K, Hashem K, Abdel-Kawi S. The prospective protective effect of selenium nanoparticles against chromium-induced oxidative and cellular damage in rat thyroid. Int J Nanomed. 2013;8:1713–1720. | ||
Rayman M. Selenium and human health. Lancet. 2012;379(9822):1256–1268. | ||
Dkhil M, Al-Quraishy S, Bauomy A, Diab M. Protective role of selenium nanoparticles against Schistosoma mansoni induced hepatic injury in mice. Biomed Res. 2016;27(1):214–219. | ||
Beheshti N, Soflaei S, Shakibaie M, et al. Efficacy of biogenic selenium nanoparticles against Leishmania major: in vitro and in vivo studies. J Trace Elem Med Biol. 2013;27(3):203–207. | ||
Dkhil M, Al-Quraishy S, Wahab R. Anticoccidial and antioxidant activities of zinc oxide nanoparticles on Eimeria papillata-induced infection in the jejunum. Int J Nanomed. 2015;10:1961–1968. | ||
Dkhil M, Abdel-Baki A, Wunderlich F, Sies H, Al-Quraishy S. Dietary selenium affects intestinal development of Eimeria papillata in mice. Parasitol Res. 2013;113(1):267–274. | ||
Jiang J, Oberdörster G, Elder A, Gelein R, Mercer P, Biswas P. Does nanoparticle activity depend upon size and crystal phase? Nanotoxicology. 2008;2(1):33–42. | ||
Schito M, Barta J, Chobotar B. Comparison of four murine Eimeria species in immunocompetent and immunodeficient mice. J Parasitol. 1996;82(2):255–262. | ||
Schito M, Barta J. Nonspecific immune responses and mechanisms of resistance to Eimeria papillata infections in mice. Infect Immun. 1997;65:3165–3170. | ||
Dommels Y, Butts C, Zhu S, et al. Characterization of intestinal inflammation and identification of related gene expression changes in mdr1a(−/−) mice. Genes Nutr. 2007;2(2):209–223. | ||
Allen A, Hutton D, Leonard A, Pearson J, Sellers L. The role of mucus in the protection of the gastroduodenal mucosa. Scand J Gastroenterol Suppl. 1986;125:71–78. | ||
Paglia D, Valentine W. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70(1):158–169. | ||
Livak K, Schmittgen T. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25(4):402–408. | ||
Krücken J, Delić D, Pauen H, et al. Augmented particle trapping and attenuated inflammation in the liver by protective vaccination against Plasmodium chabaudi malaria. Malaria J. 2009;8(1):54. | ||
Hu C, Li Y, Xiong L, Zhang H, Song J, Xia M. Comparative effects of nano elemental selenium and sodium selenite on selenium retention in broiler chickens. Anim Feed Sci Technol. 2012;177(3–4):204–210. | ||
Gangadoo S, Dinev I, Chapman J, et al. Selenium nanoparticles in poultry feed modify gut microbiota and increase abundance of Faecalibacterium prausnitzii. Appl Microbiol Biotechnol. 2018;102(3):1455–1466. | ||
Seemann E, Kurth T, Entzeroth R. Insight into the ultrastructural organisation of sporulated oocysts of Eimeria nieschulzi (Coccidia, Apicomplexa). Parasitol Res. 2012;111(5):2143–2147. | ||
Frölich S, Johnson M, Robinson M, Entzeroth R, Wallach M. The spatial organization and extraction of the wall-forming bodies of Eimeria maxima. Parasitology. 2013;140(7):876–887. | ||
Alnassan A, Shehata A, Kotsch M, et al. Efficacy of early treatment with toltrazuril in prevention of coccidiosis and necrotic enteritis in chickens. Avian Pathol. 2013;42(5):482–490. | ||
Dkhil M, Al-Quraishy S, Abdel Moneim A, Delic D. Protective effect of Azadirachta indica extract against Eimeria papillata-induced coccidiosis. Parasitol Res. 2012;112(1):101–106. | ||
Amer OS, Dkhil MA, Hikal WM, Al-Quraishy S. Anti-oxidant and anti-inflammatory activities of pomegranate (Punica granatum) on Eimeria papillata-induced infection in mice. Biomed Res Int. 2015;2015:7. | ||
Al-Quraishy S, Delic D, Sies H, Wunderlich F, Abdel-Baki A, Dkhil M. Differential miRNA expression in the mouse jejunum during garlic treatment of Eimeria papillata infections. Parasitol Res. 2011;109(2):387–394. | ||
Smith A, Cheung L, Beshah E, Shea-Donohue T, Urban J. Selenium status alters the immune response and expulsion of adult Heligmosomoides bakeri worms in mice. Infect Immun. 2013;81(7):2546–2553. | ||
Hawkes W, Kelley D, Taylor P. The effects of dietary selenium on the immune system in healthy men. Biol Trace Elem Res. 2001;81(3):189–213. | ||
Frölich S, Entzeroth R, Wallach M. Comparison of protective immune responses to apicomplexan parasites. J Parasitol Res. 2012;2012:1–11. | ||
Zhang J, Wang X, Xu T. Elemental selenium at nano size (Nano-Se) as a potential chemopreventive agent with reduced risk of selenium toxicity: comparison with se-methylselenocysteine in mice. Toxicol Sci. 2007;101(1):22–31. | ||
Arnér E, Holmgren A. Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem. 2000;267(20):6102–6109. | ||
Copeland P. Regulation of gene expression by stop codon recoding: selenocysteine. Gene. 2003;312:17–25. | ||
Speckmann B, Steinbrenner H. Selenium and selenoproteins in inflammatory bowel diseases and experimental colitis. Inflamm Bowel Dis. 2014;20(1):110–119. | ||
Deplancke B, Gaskins H. Microbial modulation of innate defense: goblet cells and the intestinal mucus layer. Am J Clin Nutr. 2001;73:1131S–1141S. | ||
Cheng H. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine II. Mucous cells. Am J Anatomy. 1974;141(4):481–501. | ||
Kim Y, Gum J, Brockhausen I. Mucin glycoproteins in neoplasia. Glycoconj J. 1996;13(5):693–707. | ||
Hanski C, Riede E, Gratchev A, et al. MUC2 gene suppression in human colorectal carcinomas and their metastases: in vitro evidence of the modulatory role of DNA methylation. Lab Invest. 1997;77(6):685–695. | ||
Jeffery P, Li D. Airway mucosa: secretory cells, mucus and mucin genes. Europ Resp J. 1997;10(7):1655–1662. | ||
Jaspers I, Zhang W, Brighton L, Carson J, Styblo M, Beck M. Selenium deficiency alters epithelial cell morphology and responses to influenza. Free Rad Biol Med. 2007;42(12):1826–1837. | ||
Lee J, Chun H, Choi H, et al. Selenium administration attenuates 5-flurouracil-induced intestinal mucositis. Nutr Cancer. 2017;69(4):616–622. | ||
Nève J. Selenium as a “nutraceutical”: how to conciliate physiological and supra-nutritional effects for an essential trace element. Curr Opin Clin Nutr Metab Care. 2002;5(6):659–663. | ||
Yan L, Johnson L. Selenium bioavailability from naturally produced high-selenium peas and oats in selenium-deficient rats. J Agricult Food Chem. 2011;59(11):6305–6311. | ||
Huang Z, Rose A, Hoffmann P. The role of selenium in inflammation and immunity: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Sign. 2012;16(7):705–743. | ||
Al-Quraishy S, Othman M, Dkhil M, Abdel Moneim A. Olive (Olea europaea) leaf methanolic extract prevents HCl/ethanol-induced gastritis in rats by attenuating inflammation and augmenting antioxidant enzyme activities. Biomed Pharmacother. 2017;91:338–349. | ||
Zhu C, Zhang S, Song C, et al. Selenium nanoparticles decorated with Ulva lactuca polysaccharide potentially attenuate colitis by inhibiting NF-κB mediated hyper inflammation. J Nanobiotechnol. 2017;15(1):20. |
Supplementary materials
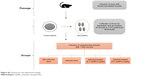  | Figure S2 Scheme for the experiment design. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.