Back to Journals » International Journal of Nanomedicine » Volume 12
Multifunctional and biomimetic fish collagen/bioactive glass nanofibers: fabrication, antibacterial activity and inducing skin regeneration in vitro and in vivo
Authors Zhou T, Sui B, Mo X ![]() , Sun J
, Sun J
Received 15 January 2017
Accepted for publication 23 March 2017
Published 2 May 2017 Volume 2017:12 Pages 3495—3507
DOI https://doi.org/10.2147/IJN.S132459
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Linlin Sun
Tian Zhou,1 Baiyan Sui,1 Xiumei Mo,2,* Jiao Sun1,*
1Shanghai Biomaterials Research and Testing Center, Shanghai Key Laboratory of Stomatology, Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, 2College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai, People’s Republic of China
*These authors contributed equally to this work
Abstract: The development of skin wound dressings with excellent properties has always been an important challenge in the field of biomedicine. In this study, biomimetic electrospun fish collagen/bioactive glass (Col/BG) nanofibers were prepared. Their structure, tensile strength, antibacterial activity and biological effects on human keratinocytes, human dermal fibroblasts and human vascular endothelial cells were investigated. Furthermore, the Sprague Dawley rat skin defect model was used to validate their effect on wound healing. The results showed that compared with pure fish collagen nanofibers, the tensile strength of the Col/BG nanofibers increased to 21.87±0.21 Mpa, with a certain degree of antibacterial activity against Staphylococcus aureus. It was also found that the Col/BG nanofibers promoted the adhesion, proliferation and migration of human keratinocytes. Col/BG nanofibers induced the secretion of type one collagen and vascular endothelial growth factor by human dermal fibroblasts, which further stimulated the proliferation of human vascular endothelial cells. Animal experimentation indicated that the Col/BG nanofibers could accelerate rat skin wound healing. This study developed a type of multifunctional and biomimetic fish Col/BG nanofibers, which had the ability to induce skin regeneration with adequate tensile strength and antibacterial activity. The Col/BG nanofibers are also easily available and inexpensive, providing the possibility for using as a functional skin wound dressing.
Keywords: electrospun, collagen/bioactive glass nanofibers, antibacterial activity, skin regeneration, angiogenesis
Introduction
Skin wound dressings derived from natural sources have always been a research hotspot in the field of regenerative medicine and traumatology. Among these naturally derived dressings, mammal collagen is already recognized as a good choice in the clinic because of its good wound healing effects. However, there might be some issues regarding the possibility of transmitting animal diseases such as bovine spongiform encephalopathy.1 Recently, it was found that nanofibers made of tilapia skin type I collagen showed good bioactivity with human keratinocytes (HaCaTs),2 which provided the possibility of fabricating wound dressings from fish collagen. However, both pure fish collagen and mammal collagen have deficiencies of low mechanical strength and lack of antibacterial activity. Furthermore, the biological functions of fish collagen on different types of skin cells and the ability to promote full-thickness skin tissue regeneration are still unclear. These key problems are very important in evaluating the value of fish collagen biomaterials and should be solved before clinically using fish collagen as wound dressings.
In order to make up for the deficiencies of fish collagen, which are low mechanical strength and lack of antibacterial activity, some bioactive components can be added to make the fish collagen more suitable for clinical use, such as preventing infection from the exposed wound and also effectively inducing skin regeneration. So far, there has been a single study reporting that Lates calcarifer scale collagen scaffolds impregnated with sago starch-capped silver nanoparticles showed high tensile strength values to be used as wound dressing materials and had antibacterial activity against Staphylococcus aureus.3 However, silver nanoparticles may be associated with the risk of causing toxic effects.4,5 Therefore, it is critical to find an effective and safe additive ingredient to develop novel fish collagen wound healing materials. It was reported that bioactive glass (BG) could be incorporated into the polymer matrix, and this was found to enhance the mechanical and biological properties of the composites.6–9 In particular, BG has been considered to have angiogenic potential.10 BG also has been found to exert an antibacterial effect when challenged with bacteria in some studies.11–13 Therefore, the development of a skin wound dressing with fish collagen and BG seemed to be extremely interesting. It was necessary to study the amount of BG added to fish collagen and the appropriate technology to make the composites achieve the anticipated multifunctional effects, including a certain tensile strength, antibacterial activity and the capability to effectively induce skin regeneration.
It is known that with electrospinning technology, it is easy to incorporate compounds into one blended layer nanofibers and produce an all-in-one wound dressing material with special properties.14,15 The electrospinning nanofibers with very fine diameter and high porous structure have attracted increasing attention for their use in biomedical applications such as medical dressings,16,17 tissue engineering scaffold and controlled drug delivery.18
In this study, fish collagen was obtained from tilapia skin. BG precursor solution was added in a proper mixture ratio to fabricate biomimetic collagen/BG (Col/BG) composite nanofibers by electrospinning. The tensile strength and the antibacterial activity of the Col/BG nanofibers on S. aureus (one of the main bacteria during skin infection) were studied. To explore the possibility of the Col/BG nanofibers being used on full-thickness skin defects, three key cells in the wound healing process, including HaCaTs, human dermal fibroblasts (HDFs) and endothelial cells, were chosen. The viability of cells seeded on the Col/BG nanofibers was analyzed. The relationship between HaCaTs migration and the expression of transforming growth factor-β1 (TGF-β1) and matrix metalloproteinase-9 (MMP-9) induced by Col/BG nanofibers was studied. The ability of HDFs to secrete type one collagen (COL-I) and vascular endothelial growth factor (VEGF) was also investigated. Finally, Sprague Dawley (SD) rat skin defect model was used to verify the effect of the Col/BG nanofibers on inducing skin regeneration. The study provided a scientific basis for the potential use of these novel fish Col/BG nanofibers as a multifunctional skin wound dressing.
Materials and methods
Preparation of tilapia collagen
Tilapia collagen was provided by Shanghai Fisheries Research Institute; the preparation method was the same as reported previously.2 Briefly, tilapia skin was stirred in 0.1 M NaOH solution for 1–2 days and then soaked in 0.5–1 M acetic acid for 4–8 h. The supernatant was collected and 0.1%–0.5% pepsin was added with stirring for 24–48 h. The precipitate was collected and then it was dissolved in 0.5–1 M acetic acid, dialyzed and lyophilized to obtain collagen sponges.
Preparation of BG precursor solution
Briefly, 4.0 g P123 (triblock copolymer of polyethylene oxide–polypropylene oxide–polyethylene oxide, EO20PO70EO20), 6.7 g tetraethyl orthosilicate, 1.4 g Ca(NO3)2·4H2O, 0.73 g triethyl phosphate and 1.0 g 0.5 M HCl were dissolved in 60 g ethanol (Si/Ca/P =80:15:5 mole ratio) and stirred at room temperature for 1 day to form BG precursor solution.
Fabrication of electrospun Col/BG nanofibers
Hexafluoroisopropanol (purity >99.5%) was purchased from Fluorochem Ltd (Hadfield, Derbyshire, UK). Tilapia collagen was dissolved in hexafluoroisopropanol solution to form 8% collagen solution, and then collagen and BG composite solution was prepared by mixing the collagen solution and the BG precursor solution at a volume ratio of 10:1. Then, the polymer solution was taken in a syringe and the electrospun Col/BG nanofibers were developed under high voltage (16–18 kV). The flow rate during electrospinning was 1.0 mL/h, and the acceptable distance between the needle and the aluminum foil collector was 10–15 cm. The nanofibers were crosslinked by glutaraldehyde vapor for 24 h and then stored in a vacuum-drying oven.
Characterization of the Col/BG nanofibers
The morphology of the Col/BG nanofibers was observed by scanning electron microscopy (SEM; JSM-5600; JEOL, Tokyo, Japan). Image analysis software (ImageJ, National Institutes of Health) was used to determine the mean fiber diameter by selecting 100 fibers randomly. The weight loss temperature was determined using a thermogravimetric analyzer (209F1; Netzsch, Selb, Germany). The contact angle was measured by a contact angle measuring instrument (OCA40; Dataphysics, Filderstadt, Germany). The elementary composition was measured with an energy dispersive spectrometer (IE 300X; Oxfordshire, UK). The chemical structure and the phase composition were determined with Fourier transform infrared spectroscopy (Avatar 380, Nicolet, Waltham, MA, USA) and X-ray diffraction (D/Max-2550 PC; Rigaku, Tokyo, Japan), respectively. The tensile strength was analyzed with a universal materials testing machine (Hounsfield, H5K-S UTM, Redhill, UK).
Antibacterial activity assay
S. aureus (ATCC 25923) was seeded on the Col/BG nanofibers and incubated in sterilized brain heart infusion (broth BHI) at 1×106 colony-forming units/mL for 24 h. S. aureus seeded on fish collagen nanofibers was used for comparison, and S. aureus seeded on cover slips was used as the control group. The morphologies of S. aureus were observed by SEM. Furthermore, the absorbance values of the bacterium suspension were tested using a spectrophotometer (UV-160; Shimadzu, Kyoto, Japan) with a wavelength of 600 nm. Then, the bacterium suspension was diluted at a ratio of 1:10 and placed on the agar plate. Colony-forming units were counted after culturing for 24 h at 37°C.
Ionic concentrations analysis
The leaching solutions were prepared by placing the Col/BG nanofibers in BHI at a ratio of 6 cm2/mL and incubating at 37°C for 24 h. The concentrations of calcium (Ca), phosphorus (P) and silicon (Si) released by the Col/BG nanofibers were determined using an inductively coupled plasma emission spectrometer (VISTA-PRO; Agilent Technologies, Santa Clara, CA, USA) to illustrate the relationship between the released ionic concentration and the antibacterial activity of the Col/BG nanofibers. The concentrations of Ca, P and Si in pure BHI were used as a control.
Viability assays of HaCaTs
HaCaTs (purchased from Animal Institute, Kunming, People’s Republic of China) were seeded on the Col/BG nanofibers at a density of 2.5×104 cells/well in 24-well plates in 500 μL of Dulbecco’s Modified Eagle’s Medium (DMEM) high-glucose medium supplemented with 10% fetal bovine serum (Hyclone), 100 U/mL penicillin and 100 mg/mL streptomycin (Hyclone). The cells seeded on the cover slips were used as a control. After the cells were cultured on the Col/BG nanofibers for 24 h, they were fixed with 4% paraformaldehyde for 30 min at 4°C. Then, the cells were washed with phosphate-buffered saline (PBS) three times and the cell morphology was observed by SEM. The cell nuclei were stained with 10 μg/mL 4,6-diamidino-2-phenylindole (Sigma, St Louis, Mo, USA) for 5 min and observed by fluorescence microscopy (Leica Microsystems, Wetzlar, Germany). The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was also performed, wherein 5 mg/mL MTT (Sigma-Aldrich Co., St Louis, MO, USA) was added, with continuous incubation for 4 h. Then, dimethyl sulfoxide was added to dissolve the crystals. The optical density values at 570 and 630 nm were measured using a spectrophotometer (Wellscan MK3; Labsystems, Dragon, Finland). Cell viability was evaluated by MTT according to the optical density values.
Real-time polymerase chain reaction
HaCaTs were seeded on the Col/BG nanofibers in six-well plates at a density of 3×105 cells/well. The cells seeded on the cover slips were used as a control. After 3 days, total RNA was isolated by an RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s guidelines and then reverse-transcribed into cDNA using a PrimeScript first strand cDNA synthesis kit (TaKaRa, Kusatsu, Japan). Real-time polymerase chain reaction kit (SYBR Premix EX Taq; TaKaRa) was used to test the gene expression level by a Bio-Rad sequence detection system (MyiQ2; Bio-Rad Laboratories Inc., Hercules, CA, USA). The expression levels of HaCaT migration-associated genes, such as TGF-β1 and MMP-9, were detected. Glyceraldehyde 3-phosphate dehydrogenase was used as normalization. The primers used in the study are listed in Table 1.
Biological effects of HDFs induced by the Col/BG nanofibers
HDFs (purchased from Kunming Animal Institute) were seeded on the Col/BG nanofibers in 24-well plates at a density of 2×104 cells/well in 500 μL of DMEM supplemented with 10% fetal bovine serum, 100 U/mL penicillin and 100 mg/mL streptomycin. After culturing for 24 h, the cell morphology was observed by SEM and fluorescence microscopy. MTT assay was then performed to evaluate the cell viability. Enzyme-linked immunosorbent assay kits (Cusabio; Huamei Biotech, Wuhan, People’s Republic of China) were used to detect the secretion of COL-I and VEGF by HDFs cultured on the Col/BG nanofibers for 24 h. The cells seeded on the cover slips were used as a control.
Biological effects of human umbilical vein endothelial cells (HUVECs) induced by the Col/BG nanofibers
Ethical approval was given by the independent ethics committee of Shanghai Ninth People’s Hospital affiliated to Shanghai Jiao Tong University, School of Medicine. Informed consent was given by all patients involved. Briefly, human umbilical vein was rinsed with PBS solution containing 100 U/mL penicillin and 100 mg/mL streptomycin three times. The umbilical vein was digested with 0.1% collagenase I (Sigma) for 15 min at 37°C. After that, the cell suspension was collected, centrifuged and then cultured in extracellular matrix (ECM) solution. The cells from three to six generations were used in the experiment. HUVECs were seeded on the Col/BG nanofibers at a density of 2×104 cells/well for 24 h, and then the cell morphology was observed by SEM and fluorescence microscopy. MTT method was used to evaluate the cell viability. In addition to this, after the HDFs were cultured on the Col/BG nanofibers for 24 h, the conditioned medium from HDFs grown on Col/BG nanofibers was collected, HUVECs were further cultured in this conditioned medium and MTT method was used for evaluating the cell viability. The HUVECs cultured by the conditioned media from HDFs not grown on Col/BG nanofibers and the HUVECs seeded on the cover slips were used as controls.
Skin wound healing experiment in SD rats
The experimental protocol was approved by the Medical Animal Care and Welfare Committee of the Ninth People’s Hospital affiliated to the School of Medicine, Shanghai Jiao Tong University. All experiments were conducted in accordance with the Division of Laboratory Animal Medicine guidelines. Twenty-four healthy 6–8-week-old adult male SD rats (n=6 represents the sample number of each group at every time point) with a body weight of 200–250 g (each) were injected with 3% sodium pentobarbital (Sigma-Aldrich Co.) at a dose of 30 mg/kg. Three full-thickness skin defects with a diameter of 1.8 cm were incised on the dorsum of each rat. These wounds were covered with the Col/BG nanofibers or Kaltostat (ConvaTec, Deeside, UK), which was commonly used as a wound dressing in clinic settings. The control group was not covered with any materials. Adhesive Tegaderm (3M) polyurethane films were used to attach the dressings to the wounds. At every time point of 4, 7, 10 and 14 days after surgery, the morphologies of the wounds were examined. Subsequently, the animals were sacrificed to collect the skin tissues.
Histologic examination
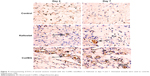
The harvested samples were collected and fixed in 10% formaldehyde for 1 week, paraffin-embedded and sectioned. The sections on days 7 and 14 were stained by hematoxylin–eosin and Masson’s trichrome stain and then dehydrated, washed, mounted and observed under a microscope. Immunohistochemical staining with CD31 antibodies (Abcam, Cambridge, UK) was performed to evaluate the angiogenesis ability of Col/BG nanofibers on days 4 and 7.
Data analysis
All experiments were performed in triplicate and data are expressed as mean ± standard deviation. Statistically significant differences (P<0.05) among different groups were evaluated by Student’s t-test and one-way analysis of variance with Tukey’s post hoc multiple comparison test. All the statistical analyses were performed using SPSS 11.0 software.
Results and discussion
Fish discards which are returned to the sea involve an underutilization of marine resources and represent a serious obstacle to the sustainability of fisheries.19,20 It is possible to use fish discards to isolate collagen, which is further used in various biomedical fields, such as in wound dressing.21 However, effective wound dressings should have suitable tensile strength, a certain degree of antibacterial activity, as well as the ability to promote skin regeneration early. Therefore, in this study, fish collagen and BG were used to fabricate multifunctional and inexpensive electrospun fish Col/BG composite nanofibers. The tensile strength and antibacterial activity of the Col/BG nanofibers and their effects on HaCaTs, HDFs and HUVECs were determined. The healing effects of the Col/BG nanofibers on rat full-thickness skin defect model were investigated to explore their feasibility to be used as a new generation of wound dressing.
Characterization of the Col/BG nanofibers
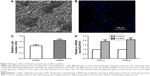
As an additive ingredient, the BG should be mixed well with fish collagen solution to obtain a uniform solution without precipitation for the development of electrospun composite nanofibers. After several preliminary experiments, the proper ratio of fish collagen and BG was chose to fabricate biomimetic electrospun fish Col/BG nanofibers with a diameter of 494±153 nm (Figure 1A). The composite nanofibers had good thermal stability and hydrophilicity (Figure 1B and C), and the contact angle was 32.68°±2.54° (Figure 1C). Energy dispersive spectrometry results showed that the Col/BG nanofibers contained silicon on the surface (Figure 1D). Furthermore, characterization of the chemical structure showed that the Fourier transform infrared spectroscopy and the X-ray diffraction spectra of the Col/BG nanofibers were not significantly changed after crosslinking (Figure 1E and F). The absorption peaks of Si-OH (975 cm−1) and Si-O (1,083.9 cm−1) could be seen clearly (Figure 1E). Also, it was found that the addition of a small amount of BG could significantly increase the tensile strength (21.87±0.21 MPa in dry condition or 4.39±0.23 MPa in wet condition) of the Col/BG composite nanofibers (Figure 1G and H). The reason was that the Col/BG nanofibers may combine the complementary mechanical properties of collagen fibers and BG. Long et al also reported that the tensile strength of collagen/bioglass scaffold was better than that of collagen fiber scaffold.22 Also, BG precursor solution could be distributed evenly in the collagen matrix and the composite nanofibers had a uniform shape, which was conducive to their mechanical property.
Antibacterial activity of the Col/BG nanofibers
As the skin wound is usually not sterile, it may be beneficial for the fish Col/BG nanofibers to have a certain antibacterial activity or preventive effect, besides the ability to increase tensile strength. It was found that the Col/BG nanofibers could inhibit adhesion (Figure 2A and B) and proliferation (Figure 2C, P<0.05) of S. aureus, while the pure fish collagen nanofibers (Col) did not have this effect. This antibacterial effect may be related to the addition of BG, which may exert this effect by its chemical composition and the dissolution conditions in its surroundings.11 The released ions (such as Ca, P and Si) can increase the osmotic pressure, creating an environment where the bacteria cannot grow.23,24 In order to further verify the ions released by the Col/BG nanofibers, inductively coupled plasma emission spectrometry was used to detect the concentration of ions in the BHI; the results showed that the Col/BG nanofibers released a certain amount of Ca, P and Si ions (Table 2). Among them, the concentrations of Ca and Si were higher than that in pure BHI; these increased ions may play a role in the antibacterial activity to S. aureus.
  | Table 2 The ionic concentrations of extracts (mg/L) |
Adhesion and migration of HaCaTs induced by the Col/BG nanofibers
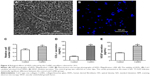
As a biomaterial expected to be used as wound dressing, in addition to their antibacterial activity, it was most important to investigate the ability of the fish Col/BG nanofibers to induce skin regeneration. We know that healing of skin wounds is a dynamic and interactive process mediated by various cell types and the ECM. The proliferation and migration of HaCaTs play a critical role in the re-epithelialization of the wound area. Multiple genes participate in wound healing, such as MMP-9 and TGF-β1. It was reported that TGF-β1 and MMP-9 were involved in keratinocyte migration.25–27 The results indicated that the Col/BG nanofibers not only promoted the adhesion and proliferation (P<0.05) of HaCaTs (Figure 3A–C), but also could upregulate the expression of MMP-9 and TGF-β1 genes (Figure 3D, P<0.05), which may be beneficial for HaCaTs migration.
Biological effects of HDFs induced by the Col/BG nanofibers
For the wound healing process of full-thickness skin defect, the proliferation and migration of HaCaTs were not sufficient and the key cells in the dermal tissues, that is, HDFs, were also required to proliferate and form collagen fibers to reconstruct the dermis. To explore the ability of the Col/BG nanofibers to promote dermal regeneration, their effects on the proliferation of HDFs and the secretion of COL-I were studied. It was found that the Col/BG nanofibers promoted the adhesion and proliferation (P<0.05) of HDFs (Figure 4A–C), as well as induced the secretion of COL-I protein in HDFs (Figure 4D, P<0.05). COL-I protein is a major component of the ECM and can form most of the connective tissue in wound healing.28 Therefore, the capacity of HDFs to secrete COL-I is of vital importance to evaluate the ability of the Col/BG nanofibers to repair the dermis tissue. The results indicated that the Col/BG nanofibers could promote HDFs to secrete COL-I, which facilitated dermis construction. However, the process of dermis regeneration also requires rapid angiogenesis. It is known that blood vessels are required to deliver oxygen and nutrients to the cells of the wound site.29 VEGF plays a prominent role in regulation of physiologic angiogenesis and it is a potent mitogen for vascular endothelial cells.30,31 Therefore, we focused on whether the Col/BG nanofibers can induce the secretion of VEGF in HDFs. Figure 4E shows that the composite nanofibers could significantly stimulate HDFs to secrete VEGF (P<0.05), which may be related to the function of BG. Day reported that 45S5 Bioglass could stimulate the release of VEGF and promote angiogenesis.31 This result was very relevant to the rapid angiogenesis in the wound area.
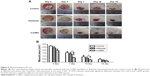
Angiogenesis induced by the Col/BG nanofibers in HUVECs
In order to validate whether the Col/BG nanofibers have an early effect on angiogenesis, HUVECs were selected. On the one hand, the direct effect of the Col/BG nanofibers on HUVECs was observed; on the other hand, it was investigated whether VEGF secreted by HDFs could act on HUVECs and increase their proliferation. As we expected, the results showed that the Col/BG nanofibers directly promoted the adhesion and proliferation (P<0.05) of HUVECs (Figure 5A–C). What is more, the HUVECs proliferated when cultured in the conditioned medium from HDFs grown on the Col/BG nanofibers (Figure 5D, P<0.05). This suggests that the HDF-secreted VEGF in the conditioned medium increased the proliferation of HUVECs. The reason may be related to the Si ions of BG. It was reported that the Si ions played an important role in the induction of angiogenesis by calcium silicate extracts.32 We found that Si existed on the nanofibers (Figure 1D), which may have some effect on inducing early angiogenesis.
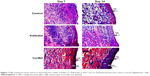
Wound healing effect of the Col/BG nanofibers in SD rat
Finally, SD rat dorsum skin defect model was used to verify the healing effect of Col/BG nanofibers in vivo. Figure 6 shows that the Col/BG nanofibers could significantly promote wound healing. At day 14, the skin wounds in the Col/BG nanofibers group were the smallest. The histologic results confirmed that the Col/BG nanofibers stimulated rapid re-epithelialization (Figure 7). The Col/BG nanofibers also promoted the formation of collagen fibers and early angiogenesis (Figures 8 and 9). This was consistent with the results demonstrated in vitro, that the Col/BG nanofibers could promote HaCaTs migration and the secretion of COL-I and VEGF in HDFs, which also further promoted the proliferation of HUVECs. The structure of nanofibers may contribute to their biological functions. Electrospun nanofibers had unique biomimetic structure, which could provide a suitable microenvironment for cell growth and make human keratinocytes, HDFs and HUVECs attach more easily and proliferate or exert further biological effects. Meanwhile, the biomimetic nanofibrous structure has good cell conductivity and is able to improve blood and other tissue fluid compatibility, which will accelerate wound healing and skin regeneration.15 Compared to other wound dressings produced by conventional methods, on the one hand, electrospun nanofibers have high effective surface area, good water absorbability and can absorb wound exudates efficiently, and on the other hand, the porous structure and small pore size of electrospun nanofibers can meet the requirement of high gas permeation and also protect the wound from bacterial infection.15 These structural features promoted the biological activities of fibrous scaffolds. In this study, multifunctional and biomimetic electrospun fish Col/BG nanofibers were fabricated. By adding BG, the composite Col/BG nanofibers not only exhibited good tensile strength, but also had the ability to release bioactive ions, which improved the antibacterial activity and the ability to induce skin regeneration. Therefore, the Col/BG nanofibers have the potential for use as a functional wound dressing.
Conclusion
In this study, biomimetic electrospun fish Col/BG nanofibers were manufabricated by composting biosafe and inexpensive collagen with BG. The tensile strength of the composite nanofibers was improved and they contained a certain degree of antibacterial activity against S. aureus. The Col/BG nanofibers could not only induce HaCaTs proliferation and migration, but also promote the secretion of COL-I and VEGF in HDFs, which further stimulated the proliferation of HUVECs. In vivo results showed that the Col/BG nanofibers could effectively induce skin regeneration in the wound area. This study suggests that the multifunctional fish Col/BG nanofibers have a great potential for use as a wound dressing.
Acknowledgments
This work was supported by grants from Natural Science Foundation of China (no 31470917) and the Shanghai Sci-Tech Committee Foundation (15DZ2290100). We thank Professor Nanping Wang from Shanghai Fisheries Research Institute for assistance with the supply of tilapia collagen.
Disclosure
The authors report no conflicts of interest in this work.
References
Moon SH, Lee YJ, Rhie JW, et al. Comparative study of the effectiveness and safety of porcine and bovine atelocollagen in Asian nasolabial fold correction. J Plast Surg Hand Surg. 2015;49(3):147–152. | ||
Zhou T, Wang N, Xue Y, et al. Electrospun tilapia collagen nanofibers accelerating wound healing via inducing keratinocytes proliferation and differentiation. Colloids Surf B Biointerfaces. 2016;143:415–422. | ||
Mandal A, Sekar S, Seeni Meera KM, Mukherjee A, Sastry TP, Mandal AB. Fabrication of collagen scaffolds impregnated with sago starch capped silver nanoparticles suitable for biomedical applications and their physicochemical studies. Phys Chem Chem Phys. 2014;16(37):20175–20183. | ||
Cox A, Venkatachalam P, Sahi S, Sharma N. Silver and titanium dioxide nanoparticle toxicity in plants: a review of current research. Plant Physiol Biochem. 2016;107:147–163. | ||
Yu X, Hong F, Zhang YQ. Bio-effect of nanoparticles in the cardiovascular system. J Biomed Mater Res A. 2016;104(11):2881–2897. | ||
Jo JH, Lee EJ, Shin DS, et al. In vitro/in vivo biocompatibility and mechanical properties of bioactive glass nanofiber and poly (epsilon-caprolactone) composite materials. J Biomed Mater Res B Appl Biomater. 2009;91(1):213–220. | ||
Meretoja VV, Helminen AO, Korventausta JJ, Haapa-aho V, Seppala JV, Narhi TO. Crosslinked poly (epsilon-caprolactone/D,L-lactide)/bioactive glass composite scaffolds for bone tissue engineering. J Biomed Mater Res A. 2006;77(2):261–268. | ||
Lu HH, El-Amin SF, Scott KD, Laurencin CT. Three-dimensional, bioactive, biodegradable, polymer-bioactive glass composite scaffolds with improved mechanical properties support collagen synthesis and mineralization of human osteoblast-like cells in vitro. J Biomed Mater Res A. 2003;64(3):465–474. | ||
Ji L, Wang W, Jin D, Zhou S, Song X. In vitro bioactivity and mechanical properties of bioactive glass nanoparticles/polycaprolactone composites. Mater Sci Eng C Mater Biol Appl. 2015;46:1–9. | ||
Gorustovich AA, Roether JA, Boccaccini AR. Effect of bioactive glasses on angiogenesis: a review of in vitro and in vivo evidences. Tissue Eng Part B Rev. 2009;16(2):199–207. | ||
Mortazavi V, Nahrkhalaji MM, Fathi MH, Mousavi SB, Esfahani BN. Antibacterial effects of sol-gel-derived bioactive glass nanoparticle on aerobic bacteria. J Biomed Mater Res A. 2010;94(1):160–168. | ||
Zhang D, Lepparanta O, Munukka E, et al. Antibacterial effects and dissolution behavior of six bioactive glasses. J Biomed Mater Res A. 2010;93(2):475–483. | ||
Allan I, Newman H, Wilson M. Antibacterial activity of particulate Bioglass® against supra-and subgingival bacteria. Biomaterials. 2001;22(12):1683–1687. | ||
Zarandi MA, Zahedi P, Rezaeian I, Salehpour A, Gholami M, Motealleh B. Drug release, cell adhesion and wound healing evaluations of electrospun carboxymethyl chitosan/polyethylene oxide nanofibers containing phenytoin sodium and vitamin C. IET Nanobiotechnol. 2015;9:191–200. | ||
Zahedi P, Rezaeian I, Ranaei-Siadat SO, Jafari SH, Supaphol P. A review on wound dressings with an emphasis on electrospun nanofibrous polymeric bandages. Polymer Adv Tech. 2010;21(2):77–95. | ||
Jeong L, Kim MH, Jung JY, Min BM, Park WH. Effect of silk fibroin nanofibers containing silver sulfadiazine on wound healing. Int J Nanomedicine. 2014;9:5277–5287. | ||
Wang Y, Zhang CL, Zhang Q, Li P. Composite electrospun nanomembranes of fish scale collagen peptides/chito-oligosaccharides: antibacterial properties and potential for wound dressing. Int J Nanomedicine. 2011;6:667–676. | ||
Jannesari M, Varshosaz J, Morshed M, Zamani M. Composite poly (vinyl alcohol)/poly (vinyl acetate) electrospun nanofibrous mats as a novel wound dressing matrix for controlled release of drugs. Int J Nanomedicine. 2011;6:993–1003. | ||
Perez-Galvez R, Garcia-Moreno PJ, Morales-Medina R, Guadix A, Guadix EM. Bile acid binding capacity of fish protein hydrolysates from discard species of the West Mediterranean Sea. Food Funct. 2015;6(4):1261–1267. | ||
Blanco M, Sotelo CG, Pérez-Martín RI. Hydrolysis as a valorization strategy for unused marine food biomass: boarfish and small-spotted catshark discards and by-products. J Food Biochem. 2015;39(4):368–376. | ||
Silva TH, Moreira-Silva J, Marques AL, Domingues A, Bayon Y, Reis RL. Marine origin collagens and its potential applications. Mar Drugs. 2014;12(12):5881–5901. | ||
Long T, Yang J, Shi SS, Guo YP, Ke QF, Zhu ZA. Fabrication of three-dimensional porous scaffold based on collagen fiber and bioglass for bone tissue engineering. J Biomed Mater Res B Appl Biomater. 2015;103(7):1455–1464. | ||
Drago L, Vassena C, Fenu S, et al. In vitro antibiofilm activity of bioactive glass S53P4. Future Microbiol. 2014;9(5):593–601. | ||
Coraca-Huber DC, Fille M, Hausdorfer J, Putzer D, Nogler M. Efficacy of antibacterial bioactive glass S53P4 against S. aureus biofilms grown on titanium discs in vitro. J Orthop Res. 2014;32(1):175–177. | ||
Ponugoti B, Xu F, Zhang C, Tian C, Pacios S, Graves DT. FOXO1 promotes wound healing through the up-regulation of TGF-beta1 and prevention of oxidative stress. J Cell Biol. 2013;203(2):327–343. | ||
Sakai H, Matsuura K, Tanaka Y, Honda T, Nishida T, Inui M. Signaling mechanism underlying the promotion of keratinocyte migration by angiotensin II. Mol Pharmacol. 2015;87(2):277–285. | ||
Salo T, Mäkelä M, Kylmäniemi M, Autio-Harmainen H, Larjava H. Expression of matrix metalloproteinase-2 and -9 during early human wound healing. Lab Invest. 1994;70(2):176–182. | ||
Ayuk SM, Houreld NN, Abrahamse H. Collagen production in diabetic wounded fibroblasts in response to low-intensity laser irradiation at 660 nm. Diabetes Technol Ther. 2012;14(12):1110–1117. | ||
Zhao S, Li L, Wang H, et al. Wound dressings composed of copper-doped borate bioactive glass microfibers stimulate angiogenesis and heal full-thickness skin defects in a rodent model. Biomaterials. 2015;53:379–391. | ||
Ferrara N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol. 2001;280(6):C1358–C1366. | ||
Day RM. Bioactive glass stimulates the secretion of angiogenic growth factors and angiogenesis in vitro. Tissue Eng. 2005;11(5–6):768–777. | ||
Li H, Chang J. Bioactive silicate materials stimulate angiogenesis in fibroblast and endothelial cell co-culture system through paracrine effect. Acta Biomater. 2013;9(6):6981–6991. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.