Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Mu-Opioid Receptor Polymorphism Moderates Sensitivity to Parental Behaviors During Characterization of Personality Traits
Authors Noto K, Suzuki A, Shirata T , Matsumoto Y, Takahashi N, Goto K , Otani K
Received 2 June 2020
Accepted for publication 28 August 2020
Published 22 September 2020 Volume 2020:16 Pages 2161—2167
DOI https://doi.org/10.2147/NDT.S265774
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Taro Kishi
Keisuke Noto,1 Akihito Suzuki,1 Toshinori Shirata,1 Yoshihiko Matsumoto,1 Nana Takahashi,1 Kaoru Goto,2 Koichi Otani1
1Department of Psychiatry, Yamagata University School of Medicine, Yamagata 990-9585, Japan; 2Department of Anatomy and Cell Biology, Yamagata University School of Medicine, Yamagata 990-9585, Japan
Correspondence: Toshinori Shirata Email [email protected]
Purpose: Attachment research shows that attachment experiences with parents in childhood influence the characterization of personality traits. Meanwhile, it is known that mu-opioid receptor function is involved in human attachment. Furthermore, a few studies suggest that the A118G polymorphism of the mu-opioid receptor gene (OPRM1) is associated with altered mu-opioid receptor function. Thus, we examined if the OPRM1 polymorphism moderates the sensitivity to parental behaviors and thereby contributes to the characterization of personality traits.
Materials and Methods: Participants were 725 healthy Japanese. Parenting practices of their parents were evaluated by the Parental Bonding Instrument (PBI) with the care and protection subscales. Personality was evaluated using the Temperament and Character Inventory (TCI). The OPRM1 A118G polymorphism was detected by a PCR method.
Results: Multiple regression analyses revealed significant effects of the interaction between the OPRM1 genotype and maternal protection on scores of the self-directedness and cooperativeness dimensions, while significant main effects of the OPRM1 genotype on scores of the TCI were not found. Further analyses showed that there were significant negative correlations between maternal protection scores and the two dimensional scores in the A/A and A/G genotypes with higher correlation coefficients in the former, but not in the G/G genotype.
Conclusion: The present study suggests that the OPRM1 polymorphism contributes to the characterization of personality traits by moderating the sensitivity to parental behaviors, especially maternal protection.
Keywords: OPRM1, attachment, personality, PBI, TCI, gene–environment interaction
Introduction
The attachment theory by Bowlby1 suggests that to obtain felt security a child instinctively displays attachment behaviors such as crying, clinging, and following, which result in attaining proximity to a parent figure. When the parent figure reacts and supplies care, the child starts to explore the surrounding while being watched affectionately by the parent figure. Thus, to provide a secure base and to encourage a child to explore from that base are important roles of parents.1 This attachment relationship may be observed in altered intensity and forms with significant others throughout life. A child who experienced the relationship of favorable attachment mentioned above tends to grow up to be self-reliant, secure, and co-operative. Conversely, a child who experienced lack of care and overprotection by parents tends to grow up to be over-dependent, insecure, and anxious, and is prone to develop depression or anxiety disorders in stressful situations. Within this framework of attachment theory, Parker et al2 suggested the Parental Bonding Instrument (PBI) to evaluate parenting practices individuals received in childhood. Subsequent studies show that parental rearing evaluated by the PBI is related to psychiatric disorders such as major depression, obsessive-compulsive disorder, and eating disorders.3–7 With regard to other aspects, our studies suggest that anomalous parenting as assessed by the PBI is associated with some personality dimensions of the Temperament and Character Inventory (TCI) and the NEO Personality Inventory.8,9 These empirical studies suggest a close connection between human attachment and the characterization of personality, supporting Bowlby’s1 attachment theory with practical data.
The brain opioid theory of social attachment posits that brain opioids are deeply implicated in the elaboration of social bonding processes including infant–mother attachment in mammals.10 This theory is supported by many animal experiments, eg, exogenous opiates reduce distress vocalizations of socially isolated puppies, and mouse pups lacking the mu-opioid receptor gene (OPRM1) display deficits in attachment behaviors toward mothers.11,12 Furthermore, a human study using positron emission tomography with [11C] carfentanil, a selective ligand to mu-opioid receptor, shows that low mu-opioid receptor availability is correlated with attachment avoidance characterized by reduced attachment emotions and behaviors.13,14 These studies suggest that mu-opioid receptor function is involved in human attachment, eg, occurrence of attachment behaviors and emotional responses to attachment experiences.
The A118G polymorphism in the exon 1 of the OPRM1 (rs1799971) results in an Asn40Asp aminoacid exchange at the putative glycosylation site in the extracellular terminus.15 It is reported that the cell culture systems expressing the G allele of this polymorphism have lower levels of forskolin-induced cAMP activation, agonist-induced receptor activation, and surface receptor expression.16 In human autopsy brain tissues, the G allele is associated with 1.5-fold lower levels of mRNA and 10-fold lower levels of protein of mu-opioid receptor compared to the A allele.17 In studies using positron emission tomography, participants with the G allele show a reduction of baseline mu-opioid receptor availability compared to those homozygous for the A allele.18,19 Furthermore, Cimino et al20 recently showed that the G allele is associated with an insecure attachment style in children with or without disruptive mood regulation disorder.
The discussions so far may suggest that the A118G polymorphism of the OPRM1 is involved in human attachment and plays some roles in the characterization of personality. To date, association studies between the OPRM1 A118G polymorphism and personality yielded negative results.21,22 However, these studies did not take gene–environment interaction(s) into account, and their negative results may be ascribable to this deficit. In fact, we previously reported that the interaction between the BDNF Val66Met genotype and parental rearing influenced formation of some personality traits, though this genotype per se did not show such effects.23,24
Therefore, the purpose of this study was to test the hypothesis that the OPRM1 A118G polymorphism moderates the sensitivity to parental behaviors during childhood and thereby contributes to the characterization of personality traits as assessed by the TCI.
Materials and Methods
Participants
Originally, 818 participants were enrolled in the study. They were recruited from medical students or staff in Yamagata University Hospital and its affiliated hospitals by KN, AS, TS, NT, and KO. Criteria of exclusion were 1) serious physical illnesses as clarified by a self-report checklist, 2) current or past history of psychiatric disorders of the Diagnostic and Statistical Manual of Mental Disorders (DSM)-IV, as clarified by the psychiatric screening previously described,4,25 and 3) parental divorce or death before the age of 16. The DSM-IV was used in this study, since the DSM-5 was not yet available in 2009 when the enrollment started. Eight participants had serious physical illnesses, 39 had psychiatric disorders, 14 had parents divorced or deceased, and 24 had missing data. In eight participants, DNA extraction or PCR amplification was failed. The remaining 725 cases were used for statistical analyses. Four hundred and twenty-three were males and 302 were females. The mean age±SD was 26.7±7.9 years. This study was conducted in accordance with the Declaration of Helsinki, and received the approval of the Ethics Committee of Yamagata University School of Medicine (approval no. 31/26.12.2002). Written informed consent was obtained from all participants.
Assessment of Parental Behaviors
Parenting practices of the participants’ parents during the first 16 years were evaluated by the Japanese version of the PBI, which has high reliability and validity.26 The PBI has the care subscale in which a high score shows involvement and care and a low score shows rejection and indifference, and the protection subscale in which a high score means intrusion, control, and overprotection and a low score means encouragement of autonomy and independence. Cronbach’s alphas in the present study were 0.925 for the paternal care, 0.851 for the paternal protection, 0.892 for the maternal care, and 0.874 for the maternal protection.
Assessment of Personality Traits
Personality traits were evaluated using the Japanese version of the Temperament and Character Inventory (TCI),27 which has high internal consistency and construct validity. The TCI is a self-report scale based on psychobiological theory of personality.8 This theory suggests that personality is composed of two factors, ie, temperament and character. Temperament represents automatic responses to emotional stimuli, and is moderately heritable. Character represents self-concepts about goals and values, and is moderately influenced by sociocultural learning. Temperament has four dimensions, ie, novelty seeking, harm avoidance, reward dependence, and persistence. Novelty seeking is the exploratory activity and intense excitement in response to novel stimuli, harm avoidance is the inhibition of behavior in response to aversive stimuli or no reward, reward dependence is the maintenance of behavior in response to reward and succorance, and persistence is the perseveration with behavior despite frustration and fatigue. Character has three dimensions, ie, self-directedness, cooperativeness, and self-transcendence. Self-directedness is the concept of the self as an autonomous individual, cooperativeness is the concept of the self as an integral part of humanity or society, and self-transcendence is the concept of the self as an integral part of a unified whole. In the present participants, Cronbach’s alphas for the novelty seeking, harm avoidance, reward dependence, persistence, self-directedness, cooperativeness, and self-transcendence were 0.719, 0.838, 0.694, 0.524, 0.798, 0.797, and 0.782, respectively.
Analysis of Genotyping
DNA was extracted from peripheral blood using a QIAamp DNA Blood Kit (Qiagen, Tokyo, Japan). The OPRM1 A118G polymorphism was determined by a real time PCR and cycling probe technology for SNP typing (Takara Bio Inc, Otsu, Japan). The primers and probes were 5ʹ-gTC AgT ACC ATg gAC AgC Ag −3ʹ (forward primer), 5ʹ- TgA CAA TCA CAT ACA TgA CCA g −3ʹ (reverse primer), 5ʹ -Tgg CAA CCT -(FAM) 3ʹ, and 5ʹ -Tgg CgA CC -(ROX) 3ʹ, respectively, where underlined letters in the probes indicate the RNA. The amplification and detection were performed along the standard protocol of the manufacturer’s instruction (Takara Bio Inc, Otsu, Japan). After the completion of the assessments of the PBI and TCI, KN, TS, and YM, who were blind to these data, performed genotyping analyses at the Department of Psychiatry of Yamagata University School of Medicine.
Statistical Analyses
The effects of interactions between the OPRM1 polymorphism and the PBI factors on the TCI dimensions were tested by forced multiple linear regression analysis,28 in which dependent variables were seven TCI dimensional scores, and independent variables were the OPRM1 genotype, four subscale scores of the PBI, products of the OPRM1 genotype and the PBI factors, age and sex. The OPRM1 genotypes were coded according to the number of the G allele (A/A=0, A/G=1, G/G=2), and dummy variables were used for sex (female=0, male=1). The PBI subscale scores were centered prior to analyses. In this model, all variables had variance inflation factor values of less than 2.0 and, thus, the effects of multicollinearity did not influence results of regression. When a significant interaction effect between the OPRM1 genotype and a PBI factor was found in some TCI dimension, the correlation between scores of the PBI factor and those of the TCI dimension in each OPRM1 genotype was tested using linear regression analysis.
All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) version 22 (IBM Japan, Tokyo, Japan). A P-value less than 0.05 was considered significant. Bonferroni correction was used for multiple testing.
Results
Demographic characteristics, the PBI, and TCI scores and the OPRM1 genotypes of the participants are shown in Table 1. The distribution of the OPRM1 genotype was in the Hardy-Weinberg equilibrium (χ2=1.581, P=0.209).
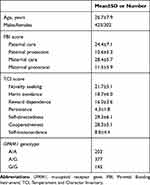 |
Table 1 Demographic Characteristics, the PBI and TCI Scores, and the OPRM1 Genotypes of the Subjects |
In multiple regression analyses, the main effects of the OPRM1 genotype on the TCI scores were not significant, but effects of the interaction between the OPRM1 genotype and maternal protection on scores of the self-directedness (P<0.05) and cooperativeness (P<0.01) dimensions were significant (Table 2). In subsequent analyses, the effects of maternal protection on the self-directedness and cooperativeness dimensions were genotype-specific, ie, there were significant (P<0.005) negative correlations between maternal protection scores and these dimensional scores in the A/A and A/G genotypes with higher correlation coefficients in the former, but not in the G/G genotype (Figure 1).
 |
Table 2 Multiple Regression Analyses of the TCI Scores |
 |
Figure 1 Effects of maternal protection on self-directedness (A) and cooperativeness (B) in the three genotypes of the mu-opioid receptor gene. |
Discussion
As expected, the present study revealed significant effects of the interaction between the OPRM1 polymorphism and maternal protection on self-directedness and cooperativeness, despite the lack of main effects of this polymorphism on any personality dimensions. The pattern of interaction was exactly the same in the two dimensions, ie, maternal protection had significant negative effects in the A/A and A/G genotypes with stronger degrees in the former, but not in the G/G genotype. These results suggest that the OPRM1 polymorphism does not directly contribute to the characterization of personality traits, but indirectly contributes to the characterization of self-directedness and cooperativeness by moderating the sensitivity to maternal protection. As far as we know, the present study is the first to show the presence of a gene–environment interaction regarding this genetic polymorphism.
The results of this study summarized above suggest that the G allele of the OPRM1 polymorphism is associated with lower sensitivity to maternal protection. The present result is in line with the result by Troisi et al29 that the G allele is less sensitive to poor parental rearing. In relation to this, Moles et al12 suggest that OPRM1 knocked-out mice have lower sensitivity to maternal separation, resulting in fewer distress calls. Meanwhile, the OPRM1 G allele is also associated with lower function of mu-opioid receptor in humans.17–19 Taken together, it is considered that the G allele of the OPRM1 polymorphism induces lower mu-opioid receptor function, which in turn induces lower sensitivity to maternal protection.
In the present study, the participants with the OPRM1 A allele showed not only lower scores of self-directedness and cooperativeness in response to higher maternal protection but also higher scores of these dimensions in response to lower maternal protection. This result suggests that OPRM1 is one of the plasticity genes, as suggested by Belsky et al,30 and this feature may explain the lack of simple correlations between the OPRM1 genotype and personality traits.21,22 On the other hand, several studies show that low scores of self-directedness and cooperativeness are predictive of the presence of personality disorders,31,32 which are often comorbid with various psychiatric disorders such as major depression, anxiety disorders, eating disorder, and substance abuse.33 Thus, if individuals with the OPRM1 A allele receive high maternal protection during childhood, they may become more susceptible to various psychiatric disorders. Conversely, if they receive low maternal protection during childhood, they may be protected against development of these disorders.
Last and least, it should be stressed that self-directedness and cooperativeness, which were specifically influenced by the interaction effect between the OPRM1 genotype and maternal protection, are closely connected with human attachment. Namely, these dimensions represent the self-concept as an autonomous individual and that as an integral part of society,8 which are the developmental tasks accomplished through favorable attachment experiences, especially with parents.1 Also, these dimensions are closely related to working models of the self and other, which underlie and determine adult attachment styles.34 Therefore, the present study indirectly supports the view that the mu-opioid receptor plays an important role in human attachment.10 In this regard, our study is in line with that by Cimino et al20 suggesting the importance of the OPRM1 polymorphism in human attachment.
There are two possible limitations in the present study. Firstly, the assessment of parental behaviors was performed by the PBI, which relies on respondent’s memory. Therefore, the parental characteristics reported by our participants might be colored by their personality to some extent, though PBI ratings correlate with the actual parental rearing assessed by significant others with acceptable validity.35 Secondly, the present results obtained in Japanese may be difficult to extend to other ethnic groups, because a considerable ethnic diversity is reported in the allele frequencies of the OPRM1 polymorphism.15
Conclusion
The OPRM1 genetic polymorphism contributes to the characterization of self-directedness and cooperativeness by moderating the sensitivity to parental behaviors, especially maternal protection.
Data Sharing Statement
The datasets generated and/or analyzed during the current study are not publicly available because the authors are not allowed to publish the raw data of subject’s genetic information by the ethics committee of the Yamagata University School of Medicine. Data are, however, available from the corresponding author upon reasonable request.
Acknowledgments
This study was supported by funding from the Ministry of Education, Culture, Sports, Science, and Technology of Japan. The funding had no effect on the present study.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bowlby J. The making and breaking of affectional bonds. I. Aetiology and psychopathology in the light of attachment theory. An expanded version of the Fiftieth Maudsley Lecture, delivered before the Royal College of Psychiatrists, 19 November 1976. Br J Psychiatry. 1977;130(3):201–210. doi:10.1192/bjp.130.3.201
2. Parker G, Tupling H, Brown LB. A parental bonding instrument. Br J Med Psychol. 1979;52(1):1–10. doi:10.1111/j.2044-8341.1979.tb02487.x
3. Oshino S, Suzuki A, Ishii G, Otani K. Influences of parental rearing on the personality traits of healthy Japanese. Compr Psychiatry. 2007;48(5):465–469. doi:10.1016/j.comppsych.2007.04.008
4. Takahashi N, Suzuki A, Matsumoto Y, Shirata T, Otani K. Perceived parental affectionless control is associated with high neuroticism. Neuropsychiatr Dis Treat. 2017;13:1111–1114. doi:10.2147/NDT.S132511
5. Grotmol KS, Ekeberg Ø, Finset A, et al. Parental bonding and self-esteem as predictors of severe depressive symptoms: a 10-year follow-up study of Norwegian physicians. J Nerv Ment Dis. 2010;198(1):22–27. doi:10.1097/NMD.0b013e3181c8189c
6. Chen D, Bienvenu OJ, Krasnow J, et al. Parental bonding and hoarding in obsessive-compulsive disorder. Compr Psychiatry. 2017;73:43–52. doi:10.1016/j.comppsych.2016.11.004
7. Tetley A, Moghaddam NG, Dawson DL, Rennoldson M. Parental bonding and eating disorders: a systematic review. Eat Behav. 2014;15(1):49–59. doi:10.1016/j.eatbeh.2013.10.008
8. Cloninger CR, Przybeck TR, Svrakic DM, Wetzel RD. The Temperament and Character Inventory (TCI): A Guide to Its Development and Use. St. Louis, Missouri: Center for Psychobiology of Personality, Washington University; 1994.
9. McCrae RR, Costa PT. NEO Inventories for the NEO Personality Inventory-3 (NEO-PI-3), NEO Five-Factor-3 (NEO-FFI-3), NEO Personality Inventory-Revised (NEO PI-R), Professional Manual. Lutz, Florida: Psychological Assessment Resources; 2010.
10. Nelson EE, Panksepp J. Brain substrates of infant-mother attachment: contributions of opioids, oxytocin, and norepinephrine. Neurosci Biobehav Rev. 1998;22(3):437–452. doi:10.1016/S0149-7634(97)00052-3
11. Panksepp J, Herman B, Conner R, Bishop P, Scott JP. The biology of social attachments: opiates alleviate separation distress. Biol Psychiatry. 1978;13(5):607–618.
12. Moles A, Kieffer BL, D’Amato FR. Deficit in attachment behavior in mice lacking the mu-opioid receptor gene. Science. 2004;304(5679):1983–1986. doi:10.1126/science.1095943
13. Nummenmaa L, Manninen S, Tuominen L, et al. Adult attachment style is associated with cerebral μ-opioid receptor availability in humans. Hum Brain Mapp. 2015;36(9):3621–3628. doi:10.1002/hbm.22866
14. Mikulincer M, Shaver PR. Attachment in Adulthood: Structure, Dynamics, and Change. New York: Guilford Press; 2007.
15. Gelernter J, Kranzler H, Cubells J. Genetics of two mu opioid receptor gene (OPRM1) exon I polymorphisms: population studies, and allele frequencies in alcohol- and drug-dependent subjects. Mol Psychiatry. 1999;4(5):476–483. doi:10.1038/sj.mp.4000556
16. Kroslak T, Laforge KS, Gianotti RJ, Ho A, Nielsen DA, Kreek MJ. The single nucleotide polymorphism A118G alters functional properties of the human mu opioid receptor. J Neurochem. 2007;103(1):77–87. doi:10.1111/j.1471-4159.2007.04738.x
17. Zhang Y, Wang D, Johnson AD, Papp AC, Sadée W. Allelic expression imbalance of human mu opioid receptor (OPRM1) caused by variant A118G [published correction appears in J Biol Chem. 2005280(46):38888]. J Biol Chem. 2005;280(38):32618–32624. doi:10.1074/jbc.M504942200
18. Peciña M, Love T, Stohler CS, Goldman D, Zubieta JK. Effects of the Mu opioid receptor polymorphism (OPRM1 A118G) on pain regulation, placebo effects and associated personality trait measures. Neuropsychopharmacology. 2015;40(4):957–965. doi:10.1038/npp.2014.272
19. Weerts EM, McCaul ME, Kuwabara H, et al. Influence of OPRM1 Asn40Asp variant (A118G) on [11C]carfentanil binding potential: preliminary findings in human subjects. Int J Neuropsychopharmacol. 2013;16(1):47–53. doi:10.1017/S146114571200017X
20. Cimino S, Carola V, Cerniglia L, Bussone S, Bevilacqua A, Tambelli R. The μ-opioid receptor gene A118G polymorphism is associated with insecure attachment in children with disruptive mood regulation disorder and their mothers. Brain Behav. 2020;10(7):e01659. doi:10.1002/brb3.1659
21. Hernandez-Avila CA, Covault J, Gelernter J, Kranzler HR. Association study of personality factors and the Asn40Asp polymorphism at the mu-opioid receptor gene (OPRM1). Psychiatr Genet. 2004;14(2):89–92. doi:10.1097/01.ypg.0000107931.32051.c7
22. Jorm AF, Prior M, Sanson A, et al. Lack of association of a single-nucleotide polymorphism of the mu-opioid receptor gene with anxiety-related traits: results from a cross-sectional study of adults and a longitudinal study of children. Am J Med Genet. 2002;114(6):659–664. doi:10.1002/ajmg.10643
23. Suzuki A, Matsumoto Y, Shibuya N, et al. The brain-derived neurotrophic factor Val66Met polymorphism modulates the effects of parental rearing on personality traits in healthy subjects. Genes Brain Behav. 2011;10(4):385–391. doi:10.1111/j.1601-183X.2010.00673.x
24. Suzuki A, Matsumoto Y, Shibuya N, et al. Interaction effect between the BDNF Val66Met polymorphism and parental rearing for interpersonal sensitivity in healthy subjects. Psychiatry Res. 2012;200(2–3):945–948. doi:10.1016/j.psychres.2012.03.014
25. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders.
26. Ogawa M. A study on reliability and validity of Japanese version of PBI (Parental Bonding Instrument). Seishinkachiryogaku. 1981;6:1193–1201.
27. Kijima N, Saito R, Takeuchi M, et al. Cloninger’s seven factor model of temperament and character and Japanese version of Temperament and Character Inventory (TCI). Jpn J Psychiatr Diagn. 1996;7:379–399.
28. Aiken LS, West SG. Multiple Regression: Testing and Interpreting Interactions. Thousand Oaks, California: SAGE Publishing; 1991.
29. Troisi A, Frazzetto G, Carola V, et al. Variation in the μ-opioid receptor gene (OPRM1) moderates the influence of early maternal care on fearful attachment. Soc Cogn Affect Neurosci. 2012;7(5):542–547. doi:10.1093/scan/nsr037
30. Belsky J, Jonassaint C, Pluess M, Stanton M, Brummett B, Williams R. Vulnerability genes or plasticity genes? Mol Psychiatry. 2009;14(8):746–754. doi:10.1038/mp.2009.44
31. Gutiérrez F, Navinés R, Navarro P, et al. What do all personality disorders have in common? Ineffectiveness and uncooperativeness. Compr Psychiatry. 2008;49(6):570–578. doi:10.1016/j.comppsych.2008.04.007
32. Svrakic DM, Draganic S, Hill K, Bayon C, Przybeck TR, Cloninger CR. Temperament, character, and personality disorders: etiologic, diagnostic, treatment issues. Acta Psychiatr Scand. 2002;106(3):189–195. doi:10.1034/j.1600-0447.2002.02196.x
33. Sadock BJ, Va S, Ruiz P. Kaplan and Sadock’s Comprehensive Textbook of Psychiatry.
34. Otani K, Suzuki A, Matsumoto Y, Enokido M, Kuwahata F, Takahashi N. Associations of working models of the self and other with Cloninger’s personality dimensions. Compr Psychiatry. 2015;56:175–178. doi:10.1016/j.comppsych.2014.09.022
35. Parker G. The measurement of pathogenic parental style and its relevance to psychiatric disorder. Social Psychiatry. 1984;19(2):75–81. doi:10.1007/BF00583818
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
