Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Mechanisms by Which the MBD2/miR-301a-5p/CXCL12/CXCR4 Pathway Regulates Acute Exacerbations of Chronic Obstructive Pulmonary Disease
Authors Shen W , Weng Z, Fan M, Wang S, Wang R, Zhang Y, Tian H, Wang X, Wu X, Yang X, Wei W, Yuan K
Received 19 May 2020
Accepted for publication 23 September 2020
Published 19 October 2020 Volume 2020:15 Pages 2561—2572
DOI https://doi.org/10.2147/COPD.S261522
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Wen Shen,1,* Zhiyin Weng,2,* Minjuan Fan,1 Shukun Wang,1 Ruili Wang,1 Yang Zhang,1 Hong Tian,1 Xi Wang,1 Xin Wu,1 Xiaolei Yang,1 Wei Wei,1 Kaifen Yuan1
1Respiratory Department, The Second Affiliated Hospital of Kunming Medical University, Kunming, People’s Republic of China; 2School of Pharmaceutical Science, Kunming Medical University, Kunming, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Kaifen Yuan
Respiratory Department, The Second Affiliated Hospital of Kunming Medical University, No. 374 Dianmian Road, Kunming 650101, Yunnan Province, People’s Republic of China
Tel +86-13888320577
Email [email protected]
Background: Chronic obstructive pulmonary disease (COPD) is characterized by irreversible expiratory airflow obstruction, and its chronic course is worsened by recurrent acute exacerbations. Our previous microarray assay identified microRNA (miR)-301a-5p as being associated with progression of acute exacerbation of COPD (AE-COPD); however, the mechanism underlying COPD pathogenesis remains unknown.
Methods: Samples of serum and peripheral blood mononuclear cells (PBMCs) were isolated from healthy control subjects and patients with stable COPD (R-COPD) or with an acute exacerbation of COPD (AE-COPD). Human HULEC-5a and human bronchial epithelial (HBE) cells were transfected with methyl-CpG-binding domain protein 2 (MBD2), sh-MBD2, miR-301a-5p mimics or an inhibitor, and then stimulated with cigarette smoke extract (CSE). Conditioned medium co-culture assays were performed by adding the supernatant of medium derived from HULEC-5a cells transfected with miR-301a-5p mimics or inhibitor into wells containing si-c-x-c motif chemokine receptor 4 (CXCR4)-transfected-lung fibroblasts or human leukemic THP-1 cell line macrophages. Transwell assays were performed to analyze cell migration.
Results: Our analysis of clinical samples showed that decreased miR-301a-5p levels in patients with AE-COPD were positively correlated with levels of MBD2 expression, but negatively correlated with levels of chemokine ligand C-X-C motif chemokine ligand 12 (CXCL12) expression. MBD2 overexpression significantly promoted miR-301a-5p production, but suppressed CXCL12 production in HULEC-5a and HBE cells. CXCL12 was confirmed to be a direct target of miR-301a-5p. CXCR4 knockdown significantly enhanced the suppressive effect of miR-301a-5p mimics and attenuated the promotional effects of the miR-301a-5p inhibitor on the migration of circulating fibroblasts and macrophages, as well as the expression levels of phospho-mitogen-activated protein kinase (p-MEK) and phospho-protein kinase B (p-AKT).
Conclusion: In summary, the MBD2/miR-301a-5p/CXCL12/CXCR4 pathway was shown to affect the migration of lung fibroblasts and monocyte-derived macrophages, which may play an important role during COPD exacerbations.
Keywords: chronic obstructive pulmonary disease, microRNA-301a-5p, inflammatory chemokine, cell recruitment
Introduction
Chronic obstructive pulmonary disease (COPD), characterized by chronic bronchial inflammation and irreversible expiratory airflow obstruction, is a common type of airway disease that is predicted to become the third leading cause of death by 2020.1,2 Tobacco smoking is considered to be the main risk factor for COPD progression, as it triggers aberrant inflammation, apoptosis, and oxidative stress.3,4 Clinically, an acute exacerbation of COPD (AE-COPD) is an independent prognostic factor for higher mortality5 and is characterized by an aggravation of respiratory symptoms, usually accompanied by hypoxemia and worsened hypercapnia.6 A previous study showed that AE-COPD frequently worsens the chronic course of COPD, and affects ~ 80% of COPD patients for > 3 years.7 In addition to the hospitalization burden, AE-COPD accounts for > 50% of the total direct costs for COPD treatment in the healthcare system.8 Bronchial relaxation and administration of glucocorticoids and antibiotics have been the current medical therapies used for treating AE-COPD; however, the clinical outcomes of AE-COPD patients remain unsatisfactory due to uncontrolled side effects.9 Therefore, an exploration of the mechanisms that underlie the pathogenesis of AE-COPD is urgently needed to develop new and powerful therapies for the disease.
MicroRNAs (miRNAs) such as miR-181a-2-3p,10 miR-29b,11 and miR-212-5p12 comprise a class of endogenous non-coding small RNAs that are ~ 22 nucleotides in length and play a central roles in regulating several aspects of inflammation, which is as a central feature of COPD. A previous study by Ezzie et al13 showed that 70 miRNAs were differentially expressed in lung tissues from subjects with COPD when compared to lung tissues from smokers without COPD. Nevertheless, there have been no reports on altered miRNA profiles associated with the development of AE-COPD. Thus, our preliminary work focused on identifying the key miRNAs that might be responsible for an acute exacerbation in COPD patients. Among the differentially expressed miRNAs, miR-301a-5p was significantly down-regulated in AE-COPD patients when compared to its expression in COPD patients in a remission phase or in healthy control subjects. A search of relevant literature revealed that altered expression of miR-301a-5p was significantly correlated with serum C-reactive protein levels in patients with Crohn’s disease.14 In hepatocellular carcinoma, miR-301a-5p functions as a direct target of EPB41L4A-AS2, and participates in inhibiting tumor growth and metastasis.15 It is worth noting that methyl-cytosine-phosphate-guanine (CpG)-binding domain protein 2 (MBD2) can promote miR-301a-5p expression by binding to its CpG island to inhibit methylation of the promoter sequence, suggesting that miR-301a-5p is regulated by MBD2.16 As a member of the histone deacetylation complex protein family, MBD2 participates in the pathological processes of several human diseases by inhibiting methylation to promote the expression of multiple genes.17 In a model of severe asthma disease, MBD2 was shown to regulate the differentiation of Th17 cells by promoting the expression of relevant important genes to ultimately affect the inflammatory response.18 Moreover, reduced MBD2 expression has recently been found to enhance airway inflammation in bronchial epithelium in COPD.19
Our previous work explored the downstream target genes of miR-301a-5p, and found there was a targeted regulatory relationship between miR-301a-5p and stromal cell-derived factor-1 (SDF-1/CXCL12), which is a member of the chemokine protein family. CXCL12 is a ligand of the g-protein-coupled receptor or chemokine receptor 4 (c-x-c motif chemokine receptor 4, CXCR4), which is involved in a variety of cellular functions, including embryonic development, immune surveillance, inflammatory responses, and tissue cell homeostasis.20 Isles et al21 showed that CXCR4/CXCL12 signaling might play an important role in neutrophil retention at inflammation sites, thereby suggesting that signaling pathway as a potential new target for the therapeutic removal of neutrophils from the lung in chronic inflammatory disease. Importantly, CXCR4/CXCL12 signaling also participates in the “recruitment” of asthma cells, circulating monocytes, and lung fibroblasts in AE-COPD,22 and also mediates the recruitment of monocytes to oral cancer spheroids.23 Based on these existing reports and our previous studies, we hypothesized that the MBD2/miR-301a-5p/CXCL1/CXCR4 pathway might play a crucial role in the pathogenesis of AE-COPD.
To validate our hypothesis, we analyzed samples of peripheral blood collected from patients with stable COPD and acutely exacerbated COPD, and then explored the correlations among MBD2, miR-301a-5p, and CXCL12 expression levels. We also used an in vitro model of circulating fibroblasts and monocytes to verify the targeted regulation of CXCL12 by miR-301a-5p and its involvement in the migration of circulating fibroblasts and monocytes through CXCR4.
Materials and Methods
Blood Sample Collection
We recruited COPD patients at the Second Affiliated Hospital of Kunming Medical University who were in a COPD remission phase (R-COPD, n = 20) or an acute-exacerbated phase (AE-COPD, n = 20), as well as 20 matched control subjects. All the recruited subjects had a history of smoking with a pack-year index > 20 between January 2017 and September 2018. All the COPD patients were diagnosed according to criteria proposed by Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017 guidelines.24 AE-COPD was defined as an increase in at least two lower respiratory tract symptoms, which included shortness of breath, sputum purulence, cough, wheezing, and chest tightness, or a new onset of two or more such symptoms, with at least one symptom lasting ≥ 3 days. A 4.0 mL sample of fasting peripheral blood was obtained from each participant for use in further analyses. All participants provided their written informed consent for study participation, and the study protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Kunming Medical University (Med-Eth-Re [20180302]).
Sample Pretreatment
Peripheral blood mononuclear cells (PBMCs) were isolated from 2 mL of peripheral blood by gradient centrifugation with Ficoll-Hypaque (Ficoll-Paque PLUS; GE Healthcare Bio-Sciences AB, Uppsala, Sweden), and immediately preserved in liquid nitrogen. Serum was obtained from the remaining 2 mL of peripheral blood and stored at −80°C.
Cell Culture and Treatment
Human pulmonary microvascular endothelial cells (HULEC-5a)25 and human lung bronchial epithelial (HBE) cells26,27 were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The HULEC-5a cells were cultured in MCDB131 medium containing 10% heat-inactivated fetal bovine serum (FBS), EGF (10 μg/mL), and hydrocortisone (1 μg/mL) at 37°C in a 5% CO2 atmosphere. The HBE cells were grown in M199 medium supplemented with EGF (10 μg/mL), hydrocortisone (1 μg/mL), and 10% FBS at 37°C with 5% CO2. Both cell lines were stimulated with cigarette smoke extract (CSE) for 24 h. The smoke extract was prepared according to a previous protocol with minor modifications.28 Human lung fibroblast cells were purchased from ATCC and grown in DMEM medium supplemented with 10% FBS and fibroblast growth factors. Human leukemic THP-1cells were purchased from ATCC and cultured in RMPI-1640 medium containing 10% FBS; after which, they were transferred into serum-free medium containing phorbol myristate acetate (PMA, 100 ng/mL) and 0.3% BSA, and then cultured for 72 h to induce the formation of adherent macrophages.
Transfection
MBD2 expression vector pcDNA3.0-MBD2 and lentivirus-containing short hairpin-MBD2 (sh-MBD2) were synthesized by RiboBio Co., Ltd. (Guangzhou, China). MiR-301a-5p mimics and inhibitor were purchased from VipotionBio Co., Ltd. (Guangzhou, China). For the in vitro experiments, HULEC-5a or HBE cells were divided into the following groups: 1) Control group, consisting of only medium without cells; 2) MBD2 group, consisting of cells transfected with pcDNA3.0-MBD2; 3) sh-MBD2 group, consisting of cells transfected with sh-MBD2; 4) Scramble group, consisting of cells transfected with the negative control; 5) miR-301a-5p mimics, consisting of cells transfected with miR-301a-5p mimics; 6) miR-301a-5p inhibitor group, consisting of cells transfected with the inhibitor. After 48 h of transfection, the cells were stimulated with CSE for use in subsequent experiments.
Conditioned Media System
Small interfering RNA targeting CXCR4 (si-CXCR4) and an si-NC were provided by RiboBio Co., Ltd. (Guangzhou, China). Conditioned media system assays were performed by adding the supernatant of blank medium or medium derived from HULEC-5a cells transfected with the miR-301a-5p mimics, inhibitor or without any treatment into wells containing 2 × 105 lung fibroblasts or THP-1 macrophages transfected with the si-NC or si-CXCR4. After 48 h, the lung fibroblasts or THP-1 macrophages were collected for further analysis.
Luciferase Reporter Assay
The putative binding sites of miR-301a-5p and 3′-UTR sequences of CXCL12 were predicted by TargetScan (http://www.targetscan.org/vert_71/). The 3′-UTR wild type or mutant type sequences of CXCL12 were amplified from HULEC-5a or HBE cells and cloned into pGL3 vectors (Promega, Madison, WI, USA) to generate the corresponding WT and MUT luciferase reporter vectors, respectively. For luciferase reporter assays, HULEC-5a or HBE cells were transfected with luciferase reporter vectors and miR-301a-5p or the NC using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). After 48 h of transfection, the levels of luciferase and renilla activity were measured using a Dual Luciferase Reporter Assay Kit (Promega). The luciferase activity was normalized to Renilla luciferase activity.
Quantitative Reverse Transcription PCR (qRT-PCR)
Total RNA was extracted using TRIzol Reagent (Invitrogen) and complementary DNA (cDNA) was synthesized with a TaqMan microRNA Reverse Transcription Kit or M-MLV Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s instructions. The PCR procedure was performed by using SYBR Green Ready Mix (Applied Biosystems) and the following thermal cycling conditions: 95°C for 5 min, 40 cycles of 95°C for 15 s, and 60°C for 1 min. The levels of MBD2, miR-301a-5p, CXCL12, and CXCR4 expression were quantified using the 2−ΔΔCt method. GAPDH or U6 served as an endogenous control. The primer sequences used in this study are listed in Table 1. Each sample was prepared in triplicate and each experiment was repeated three times.
 |
Table 1 Primers Used for qRT-PCR Analysis |
Enzyme-Linked Immunosorbent Assay (ELISA)
The concentrations of CXCL12 in serum and cell culture supernatant were determined using a commercially available ELISA kit (human CXCL12; R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions.
Immunofluorescence
Immunofluorescence studies were conducted as previously described.29 In brief, cells from the control, MBD2 or sh-MBD2 group were fixed with ice-cold methanol, blocked with 3% bovine serum albumin, and then incubated with primary antibodies against MBD2 and CXCL12 for 2 h. After counterstaining with DAPI, the stained cells were observed under a fluorescence microscope.
Transwell Migration Assay
Transwell assays were performed to evaluate the migration ability of human lung fibroblasts and THP-1 macrophages after exposure to conditioned media. Briefly, cells prepared in serum-free medium were plated into the upper chambers of a 24-well Transwell plate (BD Biosciences, Franklin Lakes, NJ, USA), while 600 μL of medium containing 10% FBS as a chemo-attractant was added to the lower chambers. After 24 h of culture, the cells that migrated into the lower chambers were fixed with 4% paraformaldehyde and stained with 0.1% crystal violet (Beyotime, China). Finally, the numbers of stained cells in six randomly selected fields were counted under a microscope at x200 magnification.
Western Blotting
Total protein was extracted with RIPA lysis buffer and quantified using a BCA assay kit (both from Beyotime, China). Next, an equal amount of protein from each extract was separated by SDS-PAGE, and the protein bands were transferred onto PVDF membranes, which were subsequently blocked with 5% non-fat milk in Tris-buffered saline containing 0.1% Tween 20 (TBST) for 1 h at room temperature. The membranes were then incubated with primary antibodies against MBD2, CXCL12, CXCR4, P-MEK, MEK, AKT, P-AKT, and GAPDH overnight at 4°C, followed by incubation with an HRP-conjugated secondary antibody for 1.5 h, The immunostained protein bands were visualized using enhanced chemiluminescence (ECL) detection reagent.
Statistical Analysis
All experiments were repeated at least three times, and results are expressed as a mean value ± standard deviation (SD). All differential comparisons between two groups were performed using Student’s t-test, and comparisons between multiple groups were performed using one-way analysis of variance (ANOVA) followed by the Bonferroni post hoc test. Analyses of correlations between various factors were performed using Spearman correlation coefficient rank test. P-values < 0.05 were considered to be statistically significant.
Results
The Levels of MBD2, miR-301a-5p, CXCL12, and CXCR4 Expression in Blood Samples from COPD Patients
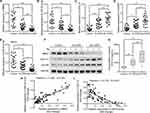
Based on our preliminary results and database predictive analysis, we speculated that the MBD2/miR-301a-5p/CXCL12/CXCR4 pathway might play an important role in COPD pathogenesis. Therefore, we performed a qRT-PCR analysis that examined the expression levels of various molecules in that pathway in PBMCs that were isolated from the control, R-COPD, and AE-COPD groups. Baseline characteristics of the COPD patients and healthy subjects, including their gender, age, weight, height, body mass index (BMI), forced vital capacity (FVC), and forced expiratory volume in the first second (FEV1) are shown in Table 2. There were significant differences in FVC, FEV1, and FEV1/FVC between the healthy subjects and the COPD patients (Table 2). The levels of MBD2 expression in the R-COPD and AE-COPD groups were significantly lower than those in the control group; however, there was no significant difference in MBD2 expression between the R-COPD and AE-COPD groups (Figure 1A). Similarly, miR-301a-5p expression levels were also significantly lower in the R-COPD and AE-COPD groups than in the control group, and miR-301a-5p levels in the AE-COPD group were significantly lower than those in the R-COPD group (Figure 1B). In contrast, both CXCL12 (Figure 1C) and CXCR4 (Figure 1D) expression levels were obviously higher in the R-COPD and AE-COPD groups than in the control group, and AE-COPD was expressed at significantly higher levels than R-COPD. In addition, we analyzed the levels of MBD2, miR-301a-5p, CXCL12, and CXCR4 in serum samples. As shown in Figure 1E, the expression levels of miR-301a-5p in serum were consistent with those in PBMCs. Western blotting further confirmed that MBD2 was down-regulated, while CXCL12 and CXCR4 were up-regulated in the R-COPD and AE-COPD groups when compared with the control group, and AE-COPD showed the more obvious trend (Figure 1F). Given that CXCL12 is both an intracellular and extracellular protein, ELISA assays were performed to analyze its expression levels in serum samples. As shown in Figure 1G, the concentrations of serum CXCL12 in the R-COPD and AE-COPD groups were significantly higher than those in the control group, and CXCL12 concentrations in the AE-COPD group were significantly higher than those in the R-COPD group. Furthermore, Spearman correlation coefficient indicated that miR-301a-5p was significantly positively correlated with the level of MBD2 expression (Figure 1H), but negatively correlated with CXCL12 expression (Figure 1I).
 |
Table 2 Baseline Characteristics by COPD Patients and Healthy Donors |
Effects of CSE on MBD2, miR-301a-5p, CXCL12, and CXCR4 Expression in vitro
As smoking is the leading cause of COPD, we next stimulated HULEC-5a or HBE cells with CSE and then determined the levels of MBD2, miR-301a-5p, CXCL12, and CXCR4 expression in the cells. As expected, CSE treatment significantly suppressed MBD2 and miR-301a-5p expression and promoted CXCL12 expression; however, it did not obviously affect the expression of CXCR4, as determined by qRT-PCR analysis and ELISA (Figure 2A and B).
The Regulatory Effects of MBD2 on miR-301a-5p and CXCL12 Expression in vitro
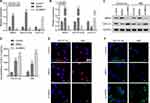
To investigate the role played by MBD2 in CSE-induced expression of miR-301a-5p and CXCL12, HULEC-5a or HBE cells were transfected with MBD2 or sh-MBD2, followed by CSE treatment. Results from qRT-PCR (Figure 3A and B) and Western blot (Figure 3C) analyses indicated that MBD2 transfection significantly up-regulated the levels of MBD2 and miR-301a-5p expression, but down-regulated CXCL12 expression. In contrast, sh-MBD2 transfection produced the opposite results. ELISA results showed extracellular CXCL12 concentrations that were consistent with the concentrations indicated by qRT-PCR and Western blotting results, which further proved that MBD2 could affect CXCL12 expression (Figure 3D). Moreover, immunofluorescence staining of MBD2 and CXCL12 revealed increased MBD2 levels and decreased CXCL12 levels after MBD2 overexpression, but decreased MBD2 levels and increased CXCL12 after sh-MBD2 transfection in both HULEC-5a and HBE cells (Figure 3E and F). These results demonstrated that there was a positive correlation between MBD2 and miR-301a-5p and a negative correlation between miR-301a-5p and CXCL12 in vitro.
CXCL12 Was a Direct Target of miR-301a-5p
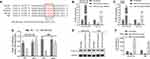
Based on the above results, we predicted the target genes of miR-301a-5p by using the online bioinformatics tool TargetScan. Interestingly, CXCL2 was identified as the putative target of miR-301a-5p. The binding sites for miR-301a-5p in the CXCL12 3ʹUTR (UCAGAG) of CXCL12 mRNA are shown in Figure 4A. Next, a luciferase reporter assay was performed to verify whether miR-301a-5p directly targeted CXCL12. As shown in Figure 4B, the relative luciferase activities were significantly decreased by miR-301a-5p transfection in the WT group, but remained unchanged after co-transfection with the MUT and miR-301a-5p as compared with the NC in both HULEC-5a and HBE cells. In addition, both CXCL12 mRNA (Figure 4C and D) and protein (Figure 4E) expression were negatively regulated by miR-301a-5p in HULEC-5a and HBE cells. Furthermore, the concentrations of CXCL12 were significantly reduced in supernatants derived from miR-301a-5p mimics-transfected HULEC-5a and HBE cells and elevated in supernatants derived from miR-301a-5p inhibitor-transfected HULEC-5a and HBE cells (Figure 4F).
Effects of miR-301a-5p/CXCL12/CXCR4 on the Migration of Circulating Fibroblasts and Macrophages
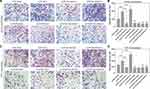
We next evaluated the effects of miR-301a-5p/CXCL12/CXCR4 on the migration of circulating fibroblasts and monocytes, because they are the main recruited circulating cells in lung tissues. In these studies, human lung fibroblasts were used as circulating fibroblasts and monocytes and THP-1 was added to adherent macrophages in vitro. After performing co-culture assays, we found that the numbers of migrated human lung fibroblasts were significantly decreased after miR-301a-5p overexpression, but increased after miR-301a-5p knockdown when compared with a control group. A comparison of the si-NC and si-CXCR4 groups revealed that CXCR4 knockdown strongly suppressed the ability of cells to migrate in cell culture supernatant derived from human lung fibroblasts transfected with the miR-301a-5p mimics or inhibitor (Figure 5A and B) and similar results were obtained for THP-1 macrophages (Figure 5C and D). Furthermore, we also explored whether miR-301a-5p-mediated regulation of circulating fibroblast and macrophage migration was associated with the CXCR4-activated MEK/AKT pathway. Western blot studies indicated that miR-301a-5p mimics obviously suppressed, while the miR-301a-5p inhibitor promoted the expression of CXCR4, P-MEK, and P-AKT. We also found that CXCR4 knockdown enhanced the suppressive effect of miR-301a-5p mimics and attenuated the promotional effects of the miR-301a-5p inhibitor on P-MEK and P-AKT expression in both human lung fibroblasts (Figure 6A) and THP-1 macrophages (Figure 6B). These findings suggest that the activated-MEK/AKT pathway might be the downstream mechanism through which CXCL12 induces the migration of circulating fibroblasts and macrophages.
Discussion
In the present study, we found that miR-301a-5p expression was down-regulated in the peripheral blood of R-COPD and AE-COPD patients when compared with its expression in a control group. Moreover, the reduced levels of miR-301a-5p expression were positively correlated with MBD2. In recent years, the possible role of miRNAs as therapeutic targets for COPD has attracted the attention of researchers.30 Compared to miRNAs found in lung tissues, expression changes in circulating miRNAs found in serum are much easier to detect and could be used for screening purposes. Jia et al12 reported the ectopic expression of several miRNAs, including miR-34a, miR-223, and miR-212-5p in serum samples from COPD patients. Because tobacco smoking can initiate and aggravate the inflammation process in COPD, we stimulated HULEC-5a and HBE cells with CSE. As expected, we found that CSE stimulation significantly down-regulated the levels of MBD2 and miR-301a-5p expression in both HULEC-5a and HBE cells. Zeng et al19 demonstrated that MBD2 expression was decreased in patients with COPD, and also in HBE cells after CSE stimulation in vitro. These results indicated that the decreased expression of MBD2 and miR-301a-5p induced by CSE was involved in COPD, and especially AE-COPD.
By performing gain-of-function and loss-of-function assays, we analyzed the role played by MBD2 in regulating miR-301a-5p and CXCL12 expression in HULEC-5a and HBE cells. Our data showed that MBD2 positively regulated miR-301a-5p expression but negatively regulated CXCL12 expression. In fact, MBD2, as a methylation-dependent reader, actively participates in DNA methylation-mediated gene transcription repression and/or heterochromatin formation.31 Accumulating evidence suggests that MBD2 is associated with several immunological disorders, such as systemic lupus erythematosus32 and autoimmune encephalomyelitis.33 MBD2 was confirmed to bind to the methylated CpG elements of the miR-301a-5p promoter, and knockdown of MBD2 expression was shown to be associated with the suppression of miR-301a-5p in a model of vancomycin-induced acute kidney injury16 Multiple studies have demonstrated a pivotal role for chemokines in COPD. For example, CCL2 levels were found to be increased in various biological specimens (ie, whole blood, induced sputum, and in situ lung tissues) obtained from COPD patients when compared specimens obtained from healthy control subjects.14 Furthermore, serum CXCL8 levels in stable and exacerbation stage COPD patients were found to be elevated relative to their levels in control subjects.34 Another study showed that CXCL10 levels in the sputum of COPD patients were elevated during an exacerbation stage when compared to their levels after recovery.35 In line with these findings, the levels of CXCL12 mRNA and concentrations of CXCL12 protein were also significantly increased in serum samples from AE-COPD patients and in CSE-stimulated HULEC-5a and HBE cells. In contrast, the numbers of CXCR4-positive circulating fibrocytes and the blood levels of CXCL12 were unchanged during the stable state in COPD patients,22 suggesting that their chemotactic properties differ in cells from different sources. Our bioinformatics prediction suggested CXCL12 as a target gene of miR-301a-5p, and that prediction was further validated by a luciferase reporter assay. We found that CXCL12 expression was down-regulated after MBD2 overexpression, which indicated that MBD2 negatively regulated CXCL12 expression by positively regulating miR-301a-5p in CSE-stimulated HULEC-5a and HBE cells (Figure 6C).
Another interesting finding of our study was that miR-301a-5p could effectively suppress the migration of circulating fibroblasts and macrophages by down-regulating CXCL12/CXCR4. It has been reported that increased numbers of circulating fibrocytes can be observed in the peripheral blood of patients with chronic obstructive asthma and idiopathic pulmonary fibrosis, and such increases are observed during an acute exacerbation of disease in those patients.36–38 In addition, the recruitment of circulating monocytes into the airway and lungs is another important process the promotes pulmonary damage in COPD, in which CXCL12/CXCR4 is an important regulator.23,39 Moreover, the CXCL12-CXCR4 axis might participate in recruiting fibrocytes to the lungs during COPD exacerbations.40 During acute exacerbations of COPD, blood fibrocytes are recruited through a CXCR4-dependent pathway.22 Our further analysis showed that miR-301a-5p-regulated circulating fibroblast and macrophage migration was associated with a CXCR4-activated MEK/AKT pathway. Tian et al41 demonstrated that knockdown of CXCR4, a receptor of CXCL12, could reverse the CXCL12-induced migration of oligodendrocyte precursor cells and block the MEK/ERK and PI3K/AKT pathways. Based on that data, we speculated that miR-301a-5p regulated by MBD2 might affect CXCL12/CXCR4 expression in the peripheral blood of COPD patients, and eventually serve to recruit circulating fibroblasts and circulating mononuclear cells into surrounding tissues, ultimately promoting the course of COPD. However, additional studies, and particularly in vivo studies, must be conducted to verify whether the MBD2/miR-301a-5p/CXCL12/CXCR4 pathway is involved in the recruitment of circulating fibroblasts and macrophages.
Conclusion
In conclusion, this study first demonstrated that miR-301a-5p was significantly down-regulated during COPD exacerbations. Our in vitro experiments indicated that the MBD2/miR-301a-5p/CXCL12/CXCR4 pathway might be involved in the recruitment of fibroblasts and macrophages. MiR-301a-5p might help to protect patients against COPD progression by limiting the recruitment of circulating fibroblasts and macrophages.
Ethics Approval and Consent to Participate
All study participants provided their written informed consent for study participation, and study protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Kunming Medical University (Med-Eth-Re [20180302]). All procedures involving human subjects were performed in accordance with ethical standards of the institutional and/or national research committee, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Acknowledgments
This work was supported by grants from the Foundation for Applied Basic Research of Yunnan Province (NO. 2019FB090, 2017FE468-058), the Medical Reserve Talents Training Plan of Yunnan Health Committee (NO. H-2018096), and the National Natural Science Foundation of China (NO. 81860012). Wen Shen and Zhiyin Weng are co-first authors for this study.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
All authors declare that they have no competing interests.
References
1. Pauwels RA, Rabe KF. Burden and clinical features of chronic obstructive pulmonary disease (COPD). Lancet. 2004;364(9434):613–620. doi:10.1016/S0140-6736(04)16855-4
2. Murray CJ, Lopez AD. Alternative projections of mortality and disability by cause 1990-2020: global Burden of Disease Study. Lancet. 1997;349(9064):1498–1504. doi:10.1016/S0140-6736(96)07492-2
3. Fischer BM, Pavlisko E, Voynow JA. Pathogenic triad in COPD: oxidative stress, protease-antiprotease imbalance, and inflammation. Int J Chron Obstruct Pulmon Dis. 2011;6:413–421. doi:10.2147/COPD.S10770
4. Plataki M, Tzortzaki E, Rytila P, Demosthenes M, Koutsopoulos A, Siafakas NM. Apoptotic mechanisms in the pathogenesis of COPD. Int J Chron Obstruct Pulmon Dis. 2006;1(2):161–171.
5. Piquet J, Chavaillon JM, David P, et al. High-risk patients following hospitalisation for an acute exacerbation of COPD. Eur Respir J. 2013;42(4):946–955. doi:10.1183/09031936.00180312
6. Duan Y, Zhou M, Xiao J, et al. Prediction of key genes and miRNAs responsible for loss of muscle force in patients during an acute exacerbation of chronic obstructive pulmonary disease. Int J Mol Med. 2016;38(5):1450–1462. doi:10.3892/ijmm.2016.2761
7. Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363(12):1128–1138. doi:10.1056/NEJMoa0909883
8. Halpin DM. Systemic effects of chronic obstructive pulmonary disease. Expert Rev Respir Med. 2007;1(1):75–84. doi:10.1586/17476348.1.1.75
9. Woodhead M, Blasi F, Ewig S, et al. Guidelines for the management of adult lower respiratory tract infections–full version. Clin Microbiol Infect. 2011;17(Suppl 6):E1–59. doi:10.1111/j.1469-0691.2011.03672.x
10. Kim J, Kim DY, Heo HR, Choi SS, Hong SH, Kim WJ. Role of miRNA-181a-2-3p in cadmium-induced inflammatory responses of human bronchial epithelial cells. J Thorac Dis. 2019;11(7):3055–3069. doi:10.21037/jtd.2019.07.55
11. Kun T, Zhao J, Xie J, Wang J. Jianping, et al. Decreased miR-29b expression is associated with airway inflammation in chronic obstructive pulmonary disease. Am J Physiol Lung Cell Mol Physiol. 2019;316(4):L621–L629. doi:10.1152/ajplung.00436.2018
12. Jia Q, Chang J, Hong Q, Zhang JJ, Zhou H, Chen FH. MiR-212-5p exerts a protective effect in chronic obstructive pulmonary disease. Discov Med. 2018;26(144):173–183.
13. Ezzie ME, Crawford M, Cho JH, et al. Gene expression networks in COPD: microRNA and mRNA regulation. Thorax. 2012;67(2):122–131. doi:10.1136/thoraxjnl-2011-200089
14. Zhen G, Jianfeng G, Yi L, et al. Mucosal MicroRNAs Expression Profiles before and after Exclusive Enteral Nutrition Therapy in Adult Patients with Crohn’s Disease. Nutrients. 2016;8(8):E519. doi:10.3390/nu8080519
15. Wang YG, Wang T, Shi M, Zhai B. Long noncoding RNA EPB41L4A-AS2 inhibits hepatocellular carcinoma development by sponging miR-301a-5p and targeting FOXL1. J Exp Clin Cancer Res. 2019;38(1):153. doi:10.1186/s13046-019-1128-9
16. Wang J, Li H, Qiu S, Dong Z, Xiang X, Zhang D. MBD2 upregulates miR-301a-5p to induce kidney cell apoptosis during vancomycin-induced AKI. Cell Death Dis. 2017;8(10):e3120.
17. Wood KH, Zhou Z. Emerging Molecular and Biological Functions of MBD2, a Reader of DNA Methylation. Front Genet. 2016;7:93. doi:10.3389/fgene.2016.00093
18. Jia A, Wang Y, Sun W, et al. MBD2 Regulates Th17 Cell Differentiation and Experimental Severe Asthma by Affecting IRF4 Expression. Mediators Inflamm. 2017;2017:6249685. doi:10.1155/2017/6249685
19. Zeng Z, Li M, Chen J, et al. Reduced MBD2 expression enhances airway inflammation in bronchial epithelium in COPD. Int J Chron Obstruct Pulmon Dis. 2018;13:703–715. doi:10.2147/COPD.S148595
20. Puchert M, Engele J. The peculiarities of the SDF-1/CXCL12 system: in some cells, CXCR4 and CXCR7 sing solos, in others, they sing duets. Cell Tissue Res. 2014;355(2):239–253. doi:10.1007/s00441-013-1747-y
21. Isles HM, Herman KD, Robertson AL, et al. The CXCL12/CXCR4 Signaling Axis Retains Neutrophils at Inflammatory Sites in Zebrafish. Front Immunol. 2019;10:1784. doi:10.3389/fimmu.2019.01784
22. Dupin I, Allard B, Ozier A, et al. Blood fibrocytes are recruited during acute exacerbations of chronic obstructive pulmonary disease through a CXCR4-dependent pathway. J Allergy Clin Immunol. 2016;137(4):1036–1042. doi:10.1016/j.jaci.2015.08.043
23. Murdoch C, Thornhill MH, Fang HY, Hearnden V, Colley HE. CXCL12/CXCR4 axis mediates recruitment of monocytes to oral cancer spheroids. Oral Dis. 2012;18(6):8–18.
24. Kokturk N, Gurgun A, Sen E, et al. The View of the Turkish Thoracic Society on the Report of the GOLD 2017 Global Strategy for the Diagnosis, Management, and Prevention of COPD. Turkish Thoracic Journal. 2017;18(2):57–64. doi:10.5152/TurkThoracJ.2017.060417
25. Kim YE, Park WS, Ahn SY, et al. WKYMVm hexapeptide, a strong formyl peptide receptor 2 agonist, attenuates hyperoxia-induced lung injuries in newborn mice. Sci Rep. 2019;9(1):6815. doi:10.1038/s41598-019-43321-4
26. Shen W, Liu J, Fan M, et al. MiR-3202 protects smokers from chronic obstructive pulmonary disease through inhibiting FAIM2: an in vivo and in vitro study. Exp Cell Res. 2018;362(2):370–377. doi:10.1016/j.yexcr.2017.11.038
27. Poon J, Campos M, Foronjy RF, et al. Cigarette smoke exposure reduces leukemia inhibitory factor levels during respiratory syncytial viral infection. Int J Chron Obstruct Pulmon Dis. 2019;14:1305–1315. doi:10.2147/COPD.S196658
28. Krimmer DI, Burgess JK, Wooi TK, Black JL, Oliver BG. Matrix proteins from smoke-exposed fibroblasts are pro-proliferative. Am J Respir Cell Mol Biol. 2012;46(1):34–39. doi:10.1165/rcmb.2010-0426OC
29. Zhou Y, He X, Chen Y, Huang Y, Wu L, He J. Exendin-4 attenuates cardiac hypertrophy via AMPK/mTOR signaling pathway activation. Biochem Biophys Res Commun. 2015;468(1–2):394–399. doi:10.1016/j.bbrc.2015.09.179
30. Bracke KR, Mestdagh P. MicroRNAs as future therapeutic targets?in COPD? Eur Respir J. 2017;49(5):1700431. doi:10.1183/13993003.00431-2017
31. Rao X, Zhong J, Zhang S, et al. Loss of methyl-CpG-binding domain protein 2 enhances endothelial angiogenesis and protects mice against hind-limb ischemic injury. Circulation. 2011;123(25):2964–2974. doi:10.1161/CIRCULATIONAHA.110.966408
32. Balada E, Ordi-Ros J, Serrano-Acedo S, Martinez-Lostao L, Vilardell-Tarrés M. Transcript overexpression of the MBD2 and MBD4 genes in CD4+ T cells from systemic lupus erythematosus patients. J Leukoc Biol. 2007;81(6):1609–1616. doi:10.1189/jlb.0107064
33. Zhong J, Yu Q, Yang P, et al. MBD2 regulates TH17 differentiation and experimental autoimmune encephalomyelitis by controlling the homeostasis of T-bet/Hlx axis. J Autoimmun. 2014;53:95–104. doi:10.1016/j.jaut.2014.05.006
34. Zhang J, Bai C. The Significance of Serum Interleukin-8 in Acute Exacerbations of Chronic Obstructive Pulmonary Disease. Tanaffos. 2018;17(1):13–21.
35. Warwick G, Thomas PS, Yates DH. Non-invasive biomarkers in exacerbations of obstructive lung disease. Respirology. 2013;18(5):874–884. doi:10.1111/resp.12089
36. Wang CH, Huang CD, Lin HC, et al. Increased circulating fibrocytes in asthma with chronic airflow obstruction. Am J Respir Crit Care Med. 2008;178(6):583–591. doi:10.1164/rccm.200710-1557OC
37. Moeller A, Gilpin SE, Ask K, et al. Circulating fibrocytes are an indicator of poor prognosis in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2009;179(7):588–594. doi:10.1164/rccm.200810-1534OC
38. Wang CH, Punde TH, Huang CD, et al. Fibrocyte trafficking in patients with chronic obstructive asthma and during an acute asthma exacerbation. J Allergy Clin Immunol. 2015;135(5):
39. Tourkina E, Oates J, Hofbauer A. Caveolin-1 regulates CXCR4/CXCL12-dependent monocyte recruitment in scleroderma patients and in a murine model of interstitial lung disease. Clin Exp Rheumatol. 2010;28(5):S79–S80.
40. Dupin I, Thumerel M, Maurat E, et al. Fibrocyte accumulation in the airway walls of COPD patients. Eur Respir J. 2019;54(3):3. doi:10.1183/13993003.02173-2018
41. Tian Y, Yin H, Deng X, Tang B, Ren X, Jiang T. CXCL12 induces migration of oligodendrocyte precursor cells through the CXCR4activated MEK/ERK and PI3K/AKT pathways. Mol Med Rep. 2018;18(5):4374–4380.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.