Back to Journals » Open Access Emergency Medicine » Volume 12
Lung Ultrasound in Children with Respiratory Tract Infections: Viral, Bacterial or COVID-19? A Narrative Review
Authors Kharasch S, Duggan NM , Cohen AR, Shokoohi H
Received 6 August 2020
Accepted for publication 18 September 2020
Published 14 October 2020 Volume 2020:12 Pages 275—285
DOI https://doi.org/10.2147/OAEM.S238702
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Hans-Christoph Pape
Sigmund Kharasch,1 Nicole M Duggan,2 Ari R Cohen,3 Hamid Shokoohi4
1Division of Pediatric Emergency Medicine, Division of Emergency Ultrasound, Department of Pediatrics, Harvard Medical School, Massachusetts General Hospital, Boston, MA, USA; 2Harvard Affiliated Emergency Medicine Residency Program, Harvard Medical School, Boston, MA, USA; 3Division of Pediatric Emergency Medicine, Department of Emergency Medicine, Harvard Medical School, Massachusetts General Hospital, Boston, MA, USA; 4Division of Emergency Ultrasound, Department of Emergency Medicine, Harvard Medical School, Massachusetts General Hospital, Boston, MA, USA
Correspondence: Hamid Shokoohi Email [email protected]
Abstract: Respiratory tract infections (RTIs) are common complaints among patients presenting to the pediatric emergency department. In the diagnostic assessment of children with RTIs, many patients ultimately undergo imaging studies for further evaluation. Point-of-care lung ultrasound (LUS) can be used safely and with a high degree of accuracy in differentiating etiologies of RTIs in pediatric patients. Ultrasonographical features such as an irregular pleural line, subpleural consolidations, focal and lobar consolidation and signs of interstitial involvement can be used to distinguish between several pathologies. This work offers a comprehensive overview of pediatric LUS in cases of the most common pediatric RTIs including bacterial and viral pneumonia, bronchiolitis, and COVID-19.
Keywords: lung, ultrasound, pediatric, pneumonia, COVID-19, bronchiolitis
Introduction
Respiratory tract infections (RTIs) are among the most common pathology presenting to the pediatric emergency department (ED).1,2 Given the high prevalence and potential severity of these disease processes, imaging modalities facilitating safe and rapid assessment of children with respiratory complaints are indispensable. Point-of-care lung ultrasound (LUS) is widely used to evaluate adult ED patients presenting with respiratory distress.3–5 In fact, standardized protocols using cardiopulmonary ultrasound to guide clinical workups, management, and resuscitation are commonplace in adults.6 Similar to work in adult patients, comparative studies in pediatric populations demonstrate LUS is equivalent, and in many cases, superior to physical examination and chest radiography (CXR) for the diagnosis of several respiratory tract diseases including pneumonia, bronchiolitis, pneumothorax, pulmonary edema, acute chest syndrome, pleural effusion, and pulmonary contusion.7–17 Compared to alternative imaging studies, LUS is a relatively rapid, inexpensive, and easy-to-learn technique, and its ability to eliminate ionizing radiation is a clear safety benefit for pediatric patients. Additionally, as LUS can be performed bedside by the primary healthcare provider, use of LUS over other imaging modalities can minimize exposure of hospital equipment and additional staff to the contagious agents such as COVID-19.18,19
Despite the advantages of LUS over alternative imaging modalities such as CXR or chest computed tomography (CT), in the pediatric ED LUS is often underutilized. Here, we provide a systematic overview of point-of-care LUS use for the most common pediatric respiratory complaints. We include discussion about bacterial and viral pneumonia with special emphasis on coronavirus disease 2019 (COVID-19), and also discuss bronchiolitis. Typical sonographic features of these disorders are discussed and compared. We highlight both the diagnostic potential and pitfalls of using LUS in these commonly encountered pediatric pathologies.
Technique and Equipment
Compared to adults, infants and children have thinner chest walls, a relatively unossified thorax, and smaller lung volumes. These features make them ideal candidates for imaging by LUS. Clearly, the types of machines available are quite variable between settings, although pediatric lung ultrasound can easily be performed with any ultrasound machine available. More recently handheld devices are successfully utilized in cases with suspected COVID-19.
Transducer selection depends on the age and size of the child, as well as the suspected pathology. In LUS, when the subcutaneous tissue, pleura, or subpleural space are being assessed (all structures which are typically very close to the skin surface and thus the ultrasound probe), a high-frequency linear transducer (15–5 MHz) is preferred. This transducer offers higher resolution of shallow structures given its limited depth of penetration. For larger children or when assessing deeper pathologies such as pleural effusion or central consolidations, either a phased array transducer (7.5–2 MHz) which has a relatively small footprint, or a curvilinear transducer (5–2.5 MHz) can be often helpful as they offer deeper imaging penetration, though at the cost of lower resolution images. In neonates, a small-footprint microlinear or “hockey stick” transducer (15–7 MHz) can also be used.13
Children can be scanned in the upright, supine or decubitus position, and often for optimal results, the patient’s position may need to be changed during the examination. Scanning the uncooperative child can be challenging but frequently feasible when performed with the child seated in the caretaker’s lap, during breastfeeding, or through the use of age appropriate distraction techniques in order to minimize anxiety. It may be helpful to give anxious young patients breaks in scanning, for example between the right and left lung. Warm gel can also be used to improve the comfort of the exam. If tolerated by the patient, frequent reassessments with repeated scanning can help clinicians understand either disease progression or improvement after therapies.
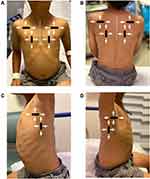
For LUS-mediated diagnosis of most pulmonary pathologies in children, scanning via a standardized survey using a 6-zone scanning protocol on each side originally described by Copetti and Cattarossi is recommended.20 The 6-zone scanning technique assesses anterior, lateral, and posterior lung fields bilaterally, scanning from apex to base in both longitudinal and transverse orientations (Figure 1). Patience and a methodical, thorough approach to scanning will yield highest results when scanning patients of any age.
Pneumonia
Community-acquired pneumonia (CAP) is the leading cause of mortality in children under 5 years of age worldwide.21 Typically, diagnosing CAP in children is based on clinical criteria alone and CXR is not routinely recommended in national and international guidelines.22,23 However, in clinical practice, CXR is commonly performed and in some reports, it is used in up to 90% of children with suspected CAP.24 Improved understanding about the recommended technique, reliability, and common findings of pneumonia on LUS may decrease CXR use among emergency physicians, thus reducing medical costs, radiation exposure, and potentially ED length of stay.
LUS as a Substitute for CXR in Pediatric Pneumonia
A robust body of literature supports the use of LUS as the first-line imaging approach in evaluating children for CAP.19,25–28 A 2015 meta-analysis of 8 studies comprising 795 children with pneumonia demonstrated that LUS has a pooled sensitivity of 96% (95% confidence interval (CI): 94–97%) and specificity of 93% (95% CI: 90–95.7%), with positive and negative likelihood ratios of 15.3 (95% CI: 6.6–35.3) and 0.06 (95% CI: 0.03–0.11), respectively.29 It was concluded that “current evidence supports LUS as an imaging alternative for the diagnosis of childhood pneumonia.” In 2018, a second meta-analysis using 18 studies and 2031 pediatric patients with confirmed pneumonia also confirmed positive test characteristics and similarly concluded that LUS “should be considered as a first-line imaging modality for children with suspected pneumonia.”30 Thus, the literature is fairly unequivocal about the utility of LUS in this context.
Several subsequent studies have directly assessed the diagnostic performance of LUS compared to CXR for the diagnosis of CAP in pediatric populations.19,25–28 Shah et al investigated the use of LUS in 200 pediatric patients, average age 3 years, who were being screened for pneumonia, LUS was found to have a sensitivity of 86% and specificity of 89% for detecting CAP.25 In a subgroup analysis of patients having lung consolidations exceeding 1 cm (as consolidations smaller than 1 cm are not detected on CXR), the specificity increased to 97%.19,25–28 In a study by Jones et al, 191 children were randomized to LUS (investigational arm) only or CXR followed by LUS (control arm).19 Overall, there was a 38% reduction in CXR among investigational subjects with no missed pneumonias. Novice and experienced sonologists achieved a 30% and 61% reduction in CXR use, respectively. At 1–2-week patient follow-up, there were no differences in adverse events or unscheduled health-care visits between study arms.19 Studies such as this not only demonstrate LUS is sensitive for detecting CAP but also LUS use results in similar clinical outcomes when compared to alternative imaging modalities.
Scanning Approach and Strategies
In addition to completing a 6-zone scanning protocol as described above, diligent scanning of the posterior lung zones is particularly important in cases of suspected pneumonia. In a recent study of optimal LUS scanning zones for the diagnosis of CAP, 46.5% of consolidations were identified in the posterior zone, 31% in the anterior zone, and 22.5% in the lateral zone (Figure 2).31 Furthermore, the majority of consolidations were found in lower lobes and 96% of sub-centimeter consolidations were identified on the transverse orientation compared to 76% which were identified in the longitudinal orientation.31 Lung consolidations reach the pleural surface in approximately 98% of cases thus facilitating their detection by LUS.32 In some instances, centrally located (perihilar or paracardiac) consolidations lack contact with the pleural surface and thus visualization by LUS may be difficult given the air surrounding the lesion even if a low-frequency curvilinear probe is used.33,34 In these cases, it may be necessary to pursue additional imaging studies such as CXR or computed tomography.
 |
Figure 2 Identification of pneumonia by affected lung zones demonstrates the majority of consolidations occur in the posterior zones, and in the lower lung (54.3%), compared to the upper (8.6%), or middle (44.3%) lung. (A) 22.5% of visible consolidations occurred in the anterior lung zones, (B) 22.6% of visible consolidations occurred in the lateral lung zones, and (C) 45.6% of visible consolidations occurred in the posterior lung zones. Reproduced from Milner BHA, Tsung JW. Lung consolidation locations for optimal lung ultrasound scanning in diagnosing pediatric pneumonia. J Ultrasound Med. 2017;36(11):2325–2328. © 2017 by the American Institute of Ultrasound in Medicine.31 |
LUS Findings in Bacterial Pneumonia
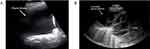
Lung consolidations at various stages ranging from subpleural consolidations, focal consolidations with air bronchograms, to lobar consolidations with hepatization are all findings along the spectrum of pneumonia and air space disease (Figure 3). Several features of discrete LUS consolidations associated with bacterial pneumonia include structures with poorly defined edges, loss of pleural line echogenicity, sonographic air bronchograms (hyperechoic linear elements representing air in bronchioles), and artifacts that appear similar to B-lines (echogenic vertical lines) but instead arise from the deep edge of the consolidation rather than the pleural line (Figure 3).6 These findings can appear near the pleural line, or deeper into lung tissue in cases of central consolidations. Discrete consolidations often demonstrate unilateral findings, where findings in cases of multifocal pneumonia or ARDS are most often bilateral.
LUS Findings in Viral Pneumonia
Lung ultrasound findings in viral pneumonia are well characterized in both adults and pediatrics.35,36 Historically, these findings have been described during times of influenza A epidemics; however, more recently the COVID-19 pandemic has sparked significant discussion surrounding the role of LUS in detecting viral pneumonia.
Typically, viral pneumonia on LUS is characterized by alveolar interstitial syndrome which demonstrates discrete or confluent B-lines (echogenic vertical lines originating from the pleura as opposed to a discrete consolidation, extending to the bottom of the screen and moving in concert with lung sliding) in a specific lung zone (Figure 4). Additionally, sub-centimeter consolidations without air bronchograms, which are often undetectable in CXR, are commonly seen on LUS. These consolidations are topics of debate and are discussed in more detail below. In a study of adult patients during the H1NI influenza A pandemic, Testa et al demonstrated the usefulness of LUS in detecting viral pneumonia. In this work, LUS depicted a pattern of interstitial syndrome in 15 of 16 patients with clinically early viral disease, all of whom had negative CXR but received a final diagnosis of H1N1 pneumonia.35
In a prospective observational cohort study of pediatric patients during the 2009 H1N1 influenza A pandemic, among 20 patients deemed to need CXR, LUS identified viral pneumonia in 15 patients (75%), isolated bacterial pneumonia in one case (5%) and normal findings in 4 cases (20%) cases.36 In this work, viral pneumonia was defined as identifying either sub-centimeter consolidations without air bronchograms, and/or individual or confluent B-lines. Viral pneumonia with superimposed bacterial infiltration was seen in 7 patients (35%). Ultrasound findings of lung consolidation with sonographic air bronchograms correlated 100% with CXR findings of bacterial pneumonia, whereas 8/15 cases (53%) of viral pneumonia had non-specific findings on CXR (primarily peri-bronchial thickening or cuffing). Interobserver agreement distinguishing viral from bacterial pneumonia using LUS was excellent (k=0.82).36
Sub-Centimeter Consolidations – Importance in Pediatric Pneumonia
Sub-centimeter consolidations and their clinical significance have been a subject of controversy among sonographers as in most cases they are seen exclusively on LUS and cannot be visualized on CXR. While sub-centimeter consolidations without air bronchograms appear consistent with viral disease on LUS, the clinical relevance and management of sub-centimeter consolidations with air bronchograms is less well understood. In studies by Shah et al and Jones et al, 6.5% and 14.6% of patients demonstrated sub-centimeter consolidations with air bronchograms, respectively. However, in both studies, only a minority of these patients required hospitalization.19,25 Whether or not sub-centimeter consolidations with air bronchograms represent early or definitive bacterial pneumonia is unknown. It has been suggested these findings may actually represent atelectasis with static air bronchograms, and thus represent atelectasis mistakenly interpreted as signs of bacterial pneumonia.33,37,38 POCUS is intended to define clinical pathology as opposed to simply describing morphology. However, further research to precisely determine the importance and clinical relevance of these findings, and to define whether their presence warrant antibiotic therapy or “watchful waiting approach” is needed.19,31,38
Pleural Effusion – Prevalence and Significance in Pediatric Pneumonia
Simple or complex parapneumonic effusions develop in 2–12% of children with pneumonia, and up to 28% of children requiring hospitalization.30,39,40 LUS is a superior imaging modality to detect and characterize the nature and volume of pleural effusion.41 The LUS appearance of pleural effusions can range from anechoic or hypoechoic fluid in the pleural space to hyperechoic fluid with septations, loculations or fibrin stranding (Figure 5).42 LUS is highly sensitive in detection of small pleural effusions (<10–30 mL) compared to CXR which is able to detect effusions larger than 150–200 mL.43,44 For detecting pleural effusions, CXR has a sensitivity of 65% and specificity of 81% while LUS has a demonstrated sensitivity and specificity as high as 100%.45,46 Comparing LUS with thoracic CT scan in assessing children with suspected complex pneumonia, three different studies revealed no additional clinical benefits in children undergoing CT. Specifically, CT did not provide any additional information compared to LUS for the detection of pleural effusion, lung necrosis or abscess as complications of pneumonia.16,47,48
COVID-19
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has emphasized the need for rapid, accurate, and non-invasive evaluation of lung pathology in populations of all ages. While children of any age are susceptible to COVID-19, several epidemiologic features differentiate disease progression in children from that seen in adults. Studies from China, Italy and the United States (US) have thus far indicated fewer confirmed cases of COVID-19 in pediatrics (2% of diagnosed cases in China, 1.2% of cases in Italy, and 5% of the United States (US) cases), a milder clinical course, and overall better prognosis compared to in adult patients.49–53 Pediatric cases of COVID-19 account for less than 1% of all US hospitalizations for COVID-19.50 Importantly, in children with COVID-19, viral coinfection must be carefully considered.54,55
Benefits of LUS Over Alternative Imaging Modalities in Pediatric COVID-19
LUS offers several advantages over other imaging modalities commonly used in the COVID-19 pandemic. Performed bedside by the primary team, LUS can reduce patient movement within the hospital thereby reducing potential viral exposure of additional health-care workers and difficult-to-sterilize devices such as X-ray or CT scanners. For adult patients admitted to an intensive care unit with COVID-19-associated respiratory failure, daily LUS exam reduced the absolute number of CXR and thoracic CT scans for each patient when compared to a panel of non-COVID-19 respiratory patients from a year earlier.56 As mentioned above, while it seems a smaller proportion of pediatric patients experience respiratory failure requiring intensive-level care, it is feasible that these results may be extrapolated to floor-stable pediatric patients with COVID-19, thus reducing patient exposure to ionizing radiation overall. Further, LUS may play an important role in triage, monitoring disease progression at home, or for out-of-hospital diagnoses for patients with COVD-19. Data in this area are still evolving.57,58
Several studies suggest CXR may be normal in early or mild cases of COVID-19 confirmed by RT-PCR.59–61 In a multi-center study investigating the frequency and distribution of radiographic findings in adult patients with COVID-19, baseline CXR demonstrated only 61% sensitivity for disease compared to a sensitivity of 91% for RT-PCR (P = 0.009).59 Radiographic findings seem to be most prominent at 10–12 days post-symptom onset. With a reported sensitivity as high as 97%, in adult patients, thoracic CT appears more promising than CXR in detecting early COVID-19.62
When compared to CXR, LUS demonstrates higher sensitivity in detecting pathological findings commonly seen in viral pneumonias and COVID-19.14,22,35,63,64 Taken together, the data suggest that both CXR and thoracic CT may have inferior sensitivity in detecting COVID-19 in pediatric populations. Interestingly, in one multi-center retrospective study in 30 pediatric patients with laboratory-confirmed COVID-19, 77% of patients had normal thoracic CT scans. In these patients, initial CT scans were performed within an average of 3.8 to 6 days of symptom onset.65 Eleven patients (33%) underwent follow-up thoracic CT an average of 5.9 days after initial scans, and of these, 10 patients (91%) had no change in findings from their initial thoracic CT scan.
LUS Findings in Pediatric COVID-19
Compared to adult patients, there are few existing studies describing LUS findings in children with COVID-19. Characteristic LUS findings are seen in Figure 6. In adult patients with COVID-19, the most common LUS findings include 1) thickening or irregularity of the pleural line; 2) B-lines (multifocal, discrete or confluent); 3) subpleural consolidations; 4) a multilobar distribution; and 5) rare pleural effusions.66,67 Findings are most often seen in the lung bases and posterior regions. In special pediatric populations, one small study of three neonates with COVID-19 demonstrated B-lines, focal consolidations, and areas of spared lung on LUS during their first week of life.66, 68 In one report of 10 children admitted with RT-PCR-confirmed SARS-CoV-2 infection, in order of frequency findings on LUS included B-lines (70%), pleural irregularities (60%), areas of white lung (10%) and subpleural consolidations (10%).67 Another study of 8 patients aged 0–17 years admitted with laboratory-confirmed SARS-CoV-2 infection revealed a B-line pattern consistent with interstitial syndrome, and subpleural consolidations.58,69 One of the eight patients demonstrated an interstitial B-line pattern on LUS but had a normal CXR. Taken together, all three groups concluded that in the hands of experienced sonographers, consistent findings of COVID-19 on LUS exist, and thus LUS may represent a reasonable approach for diagnosing and monitoring pediatric cases of COVID-19.
Bronchiolitis
Viral bronchiolitis caused by respiratory syncytial virus, human rhinovirus and parainfluenza virus, is the most common lower respiratory tract infection in infants and children 2 years of age and younger.70 The diagnosis of bronchiolitis is primarily a clinical, and current guidelines recommend against the use of routine radiographic testing.71 Often in those who receive a CXR, nonspecific radiographical findings increase the rate of unnecessary antibiotic use in pediatric populations.72,73
Currently, LUS is not routinely used in the assessment of children with suspected bronchiolitis.9–11,37,38,74 Though often relatively small, the available studies on LUS in cases of bronchiolitis demonstrate common findings include focal or confluent B-lines, subpleural consolidations of variable sizes (with or without air bronchograms), and an irregular pleural line.10,11 The presence of LUS findings typically increase with a higher disease severity, and LUS has been shown to predict the need for respiratory support.9–11,75 In bronchiolitis these findings occur more frequently in the posterior zones, thus, similar to scanning for pneumonia, careful attention to scans of the posterior lung zones is key.
A prospective study in 76 infants between 1 and 12 months of age with bronchiolitis presenting to an ED evaluated for the need for respiratory support including high-flow nasal cannula (HFNC) or helmet continuous positive airway pressure (HCPAP) using clinical and LUS scores. Overall, sixty patients (79%) were hospitalized and 32 infants (42%) required either HFNC or HCPAP. LUS score correlated with increased need for respiratory support (p 0.003).11 Taken together, the literature strongly suggests that bronchiolitis is not only amenable to assessment by LUS but also in these cases, LUS may predict clinical outcomes such as need for supplemental oxygen support and trajectory of clinical improvement.
Discussion
Point-of-care LUS is emerging as an indispensable diagnostic tool for the assessment of pediatric patients with respiratory disease. Current evidence suggests that when LUS is performed in a structured manner, it is an accurate and safe alternative to CXR in the diagnosis of several respiratory pathologies in the pediatric emergency department. As seen in Table 1, there are several overlapping features seen on LUS between bacterial pneumonia, viral pneumonia including COVID-19, and bronchiolitis in pediatrics. Varshney et al conducted a prospective study in 94 pediatric patients 2 years of age or less with signs of respiratory tract infection and wheeze to assess the ability of LUS to differentiate between bronchiolitis, asthma, and pneumonia. Of the 72 patients with a clinical diagnosis of bronchiolitis, 46% had a positive LUS that included a constellation of findings such as B-lines, subpleural consolidations, and pleural line abnormalities. LUS accurately identified all cases of pneumonia in this study and was able to rule out asthma as none had identified LUS findings.76
 |
Table 1 Summary of Lung Ultrasound Findings in Pediatric Infectious Pulmonary Pathology |
In pediatrics, it is also important to consider the possibility of concurrent disease processes. Fortunately, LUS not only aids clinicians in differentiating pulmonary pathology but can also be used to identify concomitant disease. Bacterial co-infection is estimated to occur in 9.7% of standard cases, to as high as in 42% in severe cases of bronchiolitis among patients admitted to an ICU.77 Biagi et al assessed the diagnostic accuracy of LUS in children with bronchiolitis and suspected bacterial co-infection.38 In this study, 87 patients with bronchiolitis and suspected bacterial co-infection who underwent CXR as part of their diagnostic workup to assess for co-infection were also assessed with LUS by a pediatrician. Of the twenty-five (29%) patients definitively diagnosed with a concomitant bacterial pneumonia, LUS demonstrated a sensitivity and specificity of 100% and 83.9%, compared to CXR which showed a sensitivity of 96% and specificity of 87.1%.38 Thus, despite the presence of findings consistent with bronchiolitis in these patients, LUS was also able to identify concomitant pneumonia with a similar degree of accuracy as CXR.
Conclusion
While LUS is not routinely incorporated into the diagnostic workup in infants and children with respiratory disease, emerging literature suggest that LUS may likely be an important tool in the clinical assessment of pediatric patients with suspected pneumonia (bacterial or viral), and bronchiolitis. Importantly, as the COVID-19 pandemic continues to evolve, it is likely that LUS will play an even more crucial role in pediatric diagnostic workups given this method’s ability to rapidly and accurately identify pulmonary pathology while minimizing healthcare worker exposure to possible disease. The body of academic work assessing the use of LUS in pediatrics can still be expanded, and further research is warranted. As respiratory disease season for 2020 nears, we anticipate LUS will be an indispensable tool and adjunct to physical exam for differentiating RTIs.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest.
References
1. Alpern ER, Stanley RM, Gorelick MH, et al. Epidemiology of a pediatric emergency medicine research network: the PECARN core data project. Pediatr Emerg Care. 2006;22(10):689–699. doi:10.1097/01.pec.0000236830.39194.c0
2. Nelson K, Neuman MI, Nagler J. Pulmonary emergencies. In: Bachur RG, Shaw KN, editors. Fleisher & Ludwig’s Textbook of Pediatric Emergency Medicine. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins Health; 2015:1905–1952.
3. Jonck Staub L, Biscaro RRM, Kaszubowski E, et al. Lung ultrasound for the emergency diagnosis of pneumonia, acute heart failure, and exacerbations of chronic obstructive pulmonary disease/asthma in adults: a systematic review and meta-analysis. J Emerg Med. 2019;56(1):53–69. doi:10.1016/j.jemermed.2018.09.009
4. Bekgoz B, Kilicaslan I, Bildik F, et al. BLUE protocol ultrasonography in emergency department patients presenting with acute dyspnea. Am J Emerg Med. 2019;37(11):2020–2027. doi:10.1016/j.ajem.2019.02.028
5. Buhumaid RE, Bourque JS-C, Shokoohi H, et al. Integrating point-of-care ultrasound in the ED evaluation of patients presenting with chest pain and shortness of breath. Am J Emerg Med. 2019;37(2):298–303. doi:10.1016/j.ajem.2018.10.059
6. Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest. 2015;147(6):1659–1670. doi:10.1378/chest.14-1313
7. Shah S, Bachur R, Kim D, et al. Lack of predictive value of tachypnea in the diagnosis of pneumonia in children. Pediatr Infect Dis J. 2010;29(5):406–409. doi:10.1097/INF.0b013e3181cb45a7
8. Chavez MA, Naithani N, Gilman RH, et al. Agreement between the World Health Organization algorithm and lung consolidation identified using point-of-care ultrasound for the diagnosis of childhood pneumonia by general practitioners. Lung. 2015;193(4):531–538. doi:10.1007/s00408-015-9730-x
9. Caiulo VA, Gargani L, Caiulo S, et al. Lung ultrasound in bronchiolitis-comparison with chest-x-ray. Eur J Pediatr. 2011;11(11):1427–1433. doi:10.1007/s00431-011-1461-2
10. Basile V, Mauro AD, Scalini E, et al. Lung ultrasound: a useful tool in the diagnosis and management of bronchiolitis. BMC Pediatr. 2015;15(1):63. doi:10.1186/s12887-015-0380-1
11. Supino MC, Buonenso D, Scateni S, et al. Point-of-care lung ultrasound in infants with bronchiolitis in the pediatric emergency department: a prospective study. Eur J Pediatr. 2019;178(5):623–632. doi:10.1007/s00431-019-03335-6
12. Catarossi L, Copetti R, Brusa G. Lung ultrasound diagnostic accuracy in neonatal pneumothorax. Can Rispir J. 2016;6515069.
13. Raimondi F, Rodriguez Fanjul J, Aversa S, et al. Lung ultrasound for diagnosing pneumothorax in the critically ill neonate. J Pediatr. 2016;175:74–78.
14. Martelius L, Heldt H, Lauerma K. B-lines on pediatric lung sonography. Comparison with computed tomography. J Ultrasound Med. 2016;35(1):153–157. doi:10.7863/ultra.15.01092
15. Daswani DD, Shah VP, Avner JR, et al. Accuracy of point-of-care lung ultrasonography for diagnosis of acute chest syndrome in pediatric patients with sickle cell disease and fever. Acad Emerg Med. 2016;23(8):932–940. doi:10.1111/acem.13002
16. Kurian J, Levin TL, Han BK, et al. Comparison of ultrasound and CT in the evaluation of pneumonia complicated by parapneumonic effusion in children. AJR. 2009;193(6):1648–1654. doi:10.2214/AJR.09.2791
17. Stone MB, Secko MA. Bedside ultrasound diagnosis of pulmonary contusion. Pediatr Emerg Care. 2009;25(12):854–855. doi:10.1097/PEC.0b013e3181c39a0c
18. Shu C, Jin F, Linet MS, et al. Diagnostic x-ray and ultrasound exposure and risk of childhood cancer. Br J Cancer. 1994;70(3):531–536. doi:10.1038/bjc.1994.340
19. Pardue Jones B, Tay ET, Klikashvilli I, et al. Feasibility and safety of substituting lung ultrasonography for chest radiography when diagnosing pneumonia in children. A randomized controlled trial. Chest. 2016;150(1):131–150. doi:10.1016/j.chest.2016.02.643
20. Copetti R, Cattarossi L. Ultrasound diagnosis of pneumonia in children. Radiol Med. 2008;113(2):190–198. doi:10.1007/s11547-008-0247-8
21. Liu L, Hogan D, Perin J, et al. Global, regional and national causes of child mortality in 2000–2013, with projections to inform post-2015 priorities: an updated systematic analysis. Lancet. 2015;385(9966):430–440. doi:10.1016/S0140-6736(14)61698-6
22. Chavez MA, Shams N, Ellington LE, et al. Lung ultrasound for the diagnosis of pneumonia in adults: a systematic review and meta-analysis. Respir Res. 2014;15(1):9. doi:10.1186/1465-9921-15-50
23. Andronikou S, Lambert E, Halton J, et al. Guidelines for the use of chest radiographs in community acquired pneumonia in children and adolescents. Pediatr Radiol. 2017;47(11):1405–1411. doi:10.1007/s00247-017-3944-4
24. Bowen SJ, Thomson AH. British thoracic society paediatric pneumonia audit: a review of 3 years of data. Thorax. 2013;68(7):682–683. doi:10.1136/thoraxjnl-2012-203026
25. Shah VP, Tunik MG, Tsung JW. Prospective evaluation of point-of care ultrasonography for the diagnosis of pneumonia in children and young adults. JAMA Pediatr. 2013;167(2):119–125. doi:10.1001/2013.jamapediatrics.107
26. Iorio G, Capasso M, Prisco S, et al. Lung ultrasound findings undetectable by chest radiography in children with community-acquired pneumonia. Ultrasound Med Biol. 2018;44(8):1687–1693. doi:10.1016/j.ultrasmedbio.2018.04.007
27. Iorio G, Capasso M, De Luca G, et al. Lung ultrasound in the diagnosis of pneumonia in children: proposal for a new diagnostic algorithm. Peer J. 2015;3:e1374. doi:10.7717/peerj.1374
28. Espositio S, Simone SP, Borzani I, et al. Performance of lung ultrasonography in children with community-acquired pneumonia. Ital J Pediatr. 2014;40(1):37. doi:10.1186/1824-7288-40-37
29. Pereda MA, Chavez MA, Hooper-Miele CC, et al. Lung ultrasound for the diagnosis of pneumonia in children: a meta-analysis. Pediatrics. 2015;135(4):714–722. doi:10.1542/peds.2014-2833
30. Heuvelings CC, Belard S, Familusi M, et al. Chest ultrasound for the diagnosis of pediatric pulmonary diseases: a systematic review and meta-analysis of diagnostic test accuracy. Br Med Bull. 2018;129:35–51.
31. Milner BHA, Tsung JW. Lung consolidation locations for optimal lung ultrasound scanning in diagnosing pediatric pneumonia. J Ultrasound Med. 2017;36(11):2325–2328. doi:10.1002/jum.14272
32. Lichtenstein DA, Lascols N, Meziere B, et al. Ultrasound diagnosis of alveolar consolidation in the critically ill. Intensive Care Med. 2004;30(2):276–281. doi:10.1007/s00134-003-2075-6
33. Claes AS, Clapuyt P, Menten R, et al. Performance of chest ultrasound in pediatric pneumonia. Eur J Radiol. 2017;88:82–87. doi:10.1016/j.ejrad.2016.12.032
34. Urbankowska E, Krenke K, Drobczynski L, et al. Lung ultrasound in the diagnosis and monitoring of community acquired pneumonia in children. Respir Med. 2015;109(9):1207–1212. doi:10.1016/j.rmed.2015.06.011
35. Testa A, Soldati G, Copetti R, et al. Early recognition of the 2009 pandemic influenza A (H1N1) pneumonia by chest ultrasound. Crit Care. 2012;16(1):R30. doi:10.1186/cc11201
36. Tsung JW, Kessler DO, Shah VP. Prospective application of clinician-performed lung ultrasonography during the 2009 H1N1 influenza A pandemic: distinguishing viral from bacterial pneumonia. Crit Ultrasound J. 2012;4:1–9.
37. Jaszczolt S, Polewczyk T, Dolega-Kozierowska M, et al. Comparison of lung ultrasound and chest x-ray findings in children with bronchiolitis. J Ultrason. 2018;18(74):193–197. doi:10.15557/JoU.2018.0029
38. Biagi C, Pierantoni L, Baldazzi M, et al. Lung ultrasound for the diagnosis of pneumonia in children with acute bronchiolitis. BMC Pulm Med. 2018;18(1):191. doi:10.1186/s12890-018-0750-1
39. Byington CL, Spencer LY, Johnson TA, et al. An epidemiological investigation of a sustained high rate of pediatric parapneumonic empyema: risk factors and microbiological associations. Clin Infect Dis. 2002;34(4):434. doi:10.1086/338460
40. Bradley JS, Byington CL, Shah SS, et al. The management of community-acquired pneumonia in infants and children older than 3 month of age: clinical practice guidelines by the pediatric infectious disease society and the infectious diseases society of America. Clin Infect Dis. 2011;53:e25.
41. Brogi E, Gargani L, Bignami E, et al. Thoracic ultrasound for pleural effusion in the intensive care unit: a narrative review from diagnosis to treatment. Crit Care. 2017;21(1):1–11. doi:10.1186/s13054-017-1897-5
42. Calder A, Owens CM. Imaging of parapneumonic pleural effusions and empyema in children. Pediatr Radiol. 2009;39(6):527–537. doi:10.1007/s00247-008-1133-1
43. Mong A, Epelman M, Kassa D. Ultrasound of the pediatric chest. Pediatr Radiol. 2012;42(11):1287–1297. doi:10.1007/s00247-012-2401-7
44. Coley BD. Chest sonography in children: current indications, techniques, and imaging findings. Radiol Clin North Am. 2011;49(5):825–846. doi:10.1016/j.rcl.2011.06.008
45. Xirouchaki N, Magkanas E, Vapordi K, et al. Lung ultrasound in critically ill patients: comparison with bedside chest radiography. Intensive Care Med. 2011;37(9):1488–1493. doi:10.1007/s00134-011-2317-y
46. Iuri D, De Candia A, Bazzocchi M. Evaluation of the lung in children with suspected pneumonia: usefulness of ultrasonography. Radiol Med. 2009;114(2):321–330. doi:10.1007/s11547-008-0336-8
47. Hajalioghli P, Nemati M, Saleh LD, et al. Can chest computed tomography be replaced by lung ultrasonography with or without plain chest radiography in pediatric pneumonia? J Thorac Imaging. 2016;31(4):247–252. doi:10.1097/RTI.0000000000000209
48. Yan C, Hui R, Lijuan Z, et al. Lung ultrasound vs. chest x-ray in children with suspected pneumonia confirmed by chest computed tomography: a retrospective cohort study. Exp Ther Med. 2020;19:1363–1369.
49. Zhang Y. The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) –China 2020. Chin J Epidemiol. 2020.
50. Livinston E, Bucher K. Coronavirus disease 2019 (COVID-19) in Italy. JAMA. 2020;323(14):1335. doi:10.1001/jama.2020.4344
51. Bialek S, Boundy E, Bowen V, et al. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)-United States. MMRW. 2020;69:343–346.
52. Chang T-H, Wu J-L, Chang L-Y. Clinical characteristics and diagnostic challenges of pediatric COVID-19: a systematic review and meta-analysis. J Formos Med Assoc. 2020;119(5):982–989. doi:10.1016/j.jfma.2020.04.007
53. Lu X, Zhang L, Du H, et al. SARS-CoV-2 infection in children. NEJM. 2020;382(17):17. doi:10.1056/NEJMc2005073
54. Heimdal I, Moe N, Krokstat S, et al. Human coronavirus in hospitalized children with respiratory tract infections: a 9-year population-based study from Norway. J Infect Dis. 2019;219(8):1198–1206. doi:10.1093/infdis/jiy646
55. Xia W, Shao J, Guo Y, et al. Clinical and CT features in pediatric patients with COVID-19 infection: different points from adults. Pediatr Pulmonol. 2020;55(5):1169–1174. doi:10.1002/ppul.24718
56. Mongodi S, Orlando A, Arisi E, et al. Lung ultrasound in patients with acute respiratory failure reduces conventional imaging and health care provider exposure to COVID-19. Ultrasound Med Biol. 2020;46(8):2090–2093. doi:10.1016/j.ultrasmedbio.2020.04.033
57. Shokoohi H, Duggan NM, Gonzalo G-D-CS, et al. Lung ultrasound monitoring in patients with COVID-19 on home isolation. Am J Emerg Med. 2020;
58. Antunez-Montes OY, Buonsenso D, Paz-Ortega SO. Feasibility of lung point-of-care ultrasound for patients with COVID-19 in air medical transport: triage of 2 initially suspected cases on Mexico’s front line. J Ultrasound Med. 2020;
59. Wong HYF, Lam HYS, Fong AH, et al. Frequency and distribution of chest radiographic findings in COVID-19 positive patients. Radiology. 2019;201160.
60. Denina M, Scolfaro C. Lung ultrasound in children with COVID-19. Pediatrics. 2020;1157.
61. Vancheri SG, Savietto G, Ballati F, et al. Radiographic findings in 240 patients with COVID-19 pneumonia: time-dependence after the onset of symptoms. Eur Radiol. 2020;30:1–9.
62. Ai T, Yang Z, Hou H, et al. Correlation of chest CT and RT-PCR testing for coronavirus disease 2019 (COVID-19) in China: a report of 1014 cases. Radiology. 2020;296(2):E32–E40. doi:10.1148/radiol.2020200642
63. Dubinsky TJ, Shah H, Sonneborn R, et al. Correlation of B-lines on ultrasonography with interstitial lung disease on chest radiography and CT imaging. Chest. 2017;152(5):990–998. doi:10.1016/j.chest.2017.05.003
64. Reali F, Papa GFS, Carlucci P, et al. Can lung ultrasound replace chest radiography for the diagnosis of pneumonia in hospitalized children? Respiration. 2014;14(2):112–115. doi:10.1159/000362692
65. Steinberger S, Lin B, Berheim A, et al. CT features of coronavirus disease (COVID-19) in 30 pediatric patients. AJR Am J Roentgenol. 2020;22:1–9. doi:10.2214/AJR.20.23145
66. Peng QI, Wang XT, Zhang LN, et al. Findings on lung ultrasonography of novel corona virus pneumonia during the 2019–2020 epidemic. Intensive Care Med. 2020;12:1–2.
67. Soldati G, Smargiassi S, Inchingolo R, et al. Proposal for international standardization of the use of lung ultrasound for patients with COVID-19. J Ultrasound Med. 2020;10:1002.
68. Gregorio-Hernandez R, Escobar-Izquierdo AB, Cobas-Pazos J, et al. Point-of-care lung ultrasound in three neonates with COVID-19. Eur J Pediatr. 2020;179(8):1279–1285. doi:10.1007/s00431-020-03706-4
69. Musolino AM, Supino AM, Supino MC, et al. Lung ultrasound in children with COVID-19: preliminary findings. Ultrasound Med Biol. 2020;l04:026.
70. Meissner HC. Viral bronchiolitis in children. NEJM. 2016;374(1):62–72. doi:10.1056/NEJMra1413456
71. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474–502. doi:10.1542/peds.2014-2742
72. Dawson KP, Long A, Kennedy J, et al. The chest radiograph in acute bronchiolitis. J Paediatr Child Health. 1990;26(4):209–211. doi:10.1111/j.1440-1754.1990.tb02431.x
73. Brodley WC, Viswanathan M, King VJ, et al. Diagnosis and testing in bronchiolitis: a systematic review. Arch Pediatr Adolesc Med. 2004;158(2):119–126. doi:10.1001/archpedi.158.2.119
74. Di Mauro A, Ammirabile A, Quercia M, et al. Acute bronchiolitis: is there a role for lung ultrasound? Diagnostics. 2019;9(4):172. doi:10.3390/diagnostics9040172
75. Abdel Kader M, Abou Samra MF, Abdel Aal SMS, et al. The utility of lung ultrasound in evaluation of infants with suspected bronchiolitis. EJRNM. 2016;47:1057–1064.
76. Varshney T, Mok E, Shapiro AJ, et al. Point-of-care lung ultrasound in young children with respiratory tract infections and wheeze. Emerg Med J. 2016;33(9):603–610. doi:10.1136/emermed-2015-205302
77. Thorburn K, Harigopal S, Reddy V, et al. High incidence of pulmonary bacterial co-infection in children with severe respiratory syncytial virus (RSV) bronchiolitis. Thorax. 2006;61(7):611–615. doi:10.1136/thx.2005.048397
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.