Back to Journals » OncoTargets and Therapy » Volume 13
Lung Cancer Cells Derived Circulating miR-21 Promotes Differentiation of Monocytes into Osteoclasts
Authors Zhao Q, Liu C, Xie Y, Tang M, Luo G, Chen X, Tian L, Yu X
Received 29 September 2019
Accepted for publication 6 March 2020
Published 31 March 2020 Volume 2020:13 Pages 2643—2656
DOI https://doi.org/10.2147/OTT.S232876
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jianmin Xu
Qian Zhao,1,2,* Chang Liu,1,* Ying Xie,1 Mengjia Tang,1 Guojing Luo,1 Xiang Chen,1 Li Tian,1 Xijie Yu1
1Laboratory of Endocrinology and Metabolism, Department of Endocrinology, National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University, Chengdu, People’s Republic of China; 2Department of General Practice, West China Hospital, Sichuan University, Chengdu, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xijie Yu
Laboratory of Endocrinology and Metabolism, Department of Endocrinology, National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University, No. 37 Guoxue Xiang, Chengdu 610041, People’s Republic of China
Tel +86-28-8542-2362
Fax +86-28-8542-3459
Email [email protected]
Objective: Osteoclastogenesis is a key process in osteolytic bone metastasis (BM). Previous studies indicated that some miRNAs could regulate cancers progression and osteoclastogenesis. Our purpose was to investigate the roles of lung cancer cells-derived circulating miR-21 on osteoclastogenesis and its clinical significance in BM patients.
Materials and Methods: The difference of miRNA expression in two lung cancer cell lines SBC-5 (with characteristic BM ability) and SBC-3 (without BM ability) were analyzed by microarray and qRT-PCR. Circulating miR-21 levels of lung cancer patients with or without BM were compared by qRT-PCR. The TRAP staining was used to investigate the effects of conditioned media from lung cancer cell lines or patients’ plasma with different miR-21 levels on osteoclastogenesis. ROC curve was used to evaluate the diagnostic performance of circulating miR-21 in BM patients.
Results: We found that miR-21 expression was specifically higher in SBC-5 than that in SBC-3 cells. The supernatants of SBC-5 cells with higher-level miR-21 promoted osteoclastogenesis. Moreover, we demonstrated that the circulating miR-21 level was significantly higher in BM patients than that in non-BM patients. The plasma from BM patients with higher-level miR-21 could also promote osteoclastogenesis. Mechanistically, lung cancer cells-derived circulating miR-21 could be transferred into osteoclast precursor cells and promote osteoclastogenesis probably by inhibiting PTEN. Finally, clinical data showed that circulating miR-21 had a potential for the diagnosis of BM.
Conclusion: Overall, our findings suggested that circulating miR-21 played important roles in osteoclastogenesis of lung cancer patients and may serve as a biomarker to diagnose BM of lung cancer.
Keywords: microRNA, bone metastasis, lung cancer, osteoclast, biomarker
Introduction
Lung cancer was the main cause of malignancy-related deaths worldwide. Approximately 1.5 million people were diagnosed with lung cancer each year and 86.7% of them could not survive.1 In the United States, 27% of cancer deaths were due to lung cancer. Metastasis was the major reason for its high mortality. Bone was the most frequent site for metastasis, and bone metastasis (BM) was presented in 30–40% lung cancer patients.2 Although the survival rate was rising with the improvement of neoadjuvant and surgical treatment, metastasis, especially BM indicated a poor prognosis. It was found that the mean survival time of BM patients was no more than 12 months from BM diagnosis to death due to various complications.3 The detection of BM mainly depended on bone scintigraphy screening, MRI or positron emission tomography-computed tomography (PET-CT). Bone scintigraphy had high sensitivity, but lacking specificity. MRI or PET-CT was limited due to their cost-effectiveness or inconvenience.4 Bone turnover markers had also been used to evaluate bone lesion progression. However, currently, none of these markers could detect BM occurrence independently of bone imageological examination.4 So the diagnostic approach for the accurate, rapid and sensitive identification of BM was needed. BM was not usually diagnosed until complications occurred due to its non-symptom stage. When lung cancer had spread to bone, treatment options were limited and mainly palliative.
MiRNAs were small, non-coding RNAs that paired with the target gene mRNAs, resulting in mRNAs degradation, or inducing gene silence by inhibiting translation. Interacting with a negative regulatory sequence in the 3ʹ-non-coding region of target mRNAs, miRNAs were involved in a wide range regulation of normal and abnormal cell functions. Moreover, previous studies had indicated that dysregulation of miRNA was related to initiation, progression, and metastasis of tumor.5 Furthermore, circulating miRNAs were described as potential mediators of BM in some studies.
As an oncogenic miRNA, miR-21 had been proved to play an important role in tumor development through regulating multifold cell behaviors such as proliferation, invasion, and metastasis.6 Furthermore, high level of circulating miR-21 had been observed in lung cancer patients in several studies. However, its role in BM of lung cancer had not yet been fully elucidated. The present study aimed to investigate the role of miR-21 in BM and the clinical significance of circulating miRNA-21 as an available non-invasive biomarker for the early diagnosis of BM in lung cancer patients.
Materials and Methods
Ethics Statement
The study was approved by the hospital research ethics committee of West China Hospital of Sichuan University and conducted in accordance with the International Ethical Guidelines for Biomedical Research Involving Human Subjects. All patients included in this study had signed the informed consent form.
Cell Culture and Transfection
Human small cell lung cancer (SCLC) cells: SBC-5, SBC-3, and mouse-derived monocyte cell line RAW264.7 were purchased from the Zishi Biological Technology Co., Ltd., Shanghai, PRC. SBC-5, SBC-3, and RAW264.7 cells were maintained in Dulbecco’s modified Eagle medium (DMEM) (HyClone, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, USA). Human primary monocytes were isolated from peripheral blood of patients included in this study. These monocytes were maintained in DMEM medium with 10% FBS. Cell transfection with miR-21 mimic or miR-21 inhibitor from RiboBio (Guangzhou, China) was performed by riboFECTTM CP Transfection Kit (RiboBio, China), according to the manufacturer’s instructions.
Patients
Forty-nine patients diagnosed with lung cancer at West China Hospital of Sichuan University were enrolled in this study. These patients had not received any chemotherapy, radiotherapy or surgery before enrollment. For all cases of this study, primary lesions of lung cancer were confirmed by pathological approach. Either SPECT or PET-CT was performed to all of the patients. With positive findings of imaging examinations, the clinical diagnosis of lung cancer BM was made. The clinical data were recorded. Peripheral blood samples were collected. The plasma and monocytes were isolated and stored at −80°C. These patients were divided into three groups (14 patients in non-metastasis group, 14 patients in other sites metastasis group, and 21 patients in BM group) according to the metastatic status of lung cancer. Between the 3 groups, the difference of age, sex distribution and pathological type distribution was not significant.
Analysis of miRNA and mRNA Expression
MiRNAs and mRNAs from cells, culture supernatants and patients’ plasma were extracted. Microarrays and qRT-PCR were conducted following the procedure described by our team previously7 or according to the protocol provided by the manufacturer. The 2−ΔΔCt method was used to analyze the data.
Induced Differentiation of Precursor Cells to Osteoclasts with Conditioned Media
The osteoclast precursors – RAW264.7 cells and peripheral blood primary monocytes from patients were conducted to differentiate into osteoclasts with conditioned media (CM) according to the protocol described by our team previously.7 The conditioned media were prepared as 10% cancer cell culture supernatants or patients’ plasma with 10% FBS, 80% DMEM, 10 ng/mL macrophage colony-stimulating factor (M-CSF), and 10 ng/mL RANK ligand (RANKL) for each type of cells. The control unconditioned media were constituted of 90% DMEM with 10% FBS, 10 ng/mL M-CSF, and 10 ng/mL RANKL. In the end, cells were fixed and stained for TRAP (tartrate-resistant acid phosphatase) with acid phosphatase, leukocyte (TRAP) kit (Sigma-Aldrich, Darmstadt, Germany). TRAP+ cells (with 3 or more nuclei) were counted under bright-field microscopy.
Cross-Culture of Peripheral Blood Monocytes and Plasma from Lung Cancer Patients
Step 1: Induce differentiation of peripheral blood monocytes from BM patients under 3 conditions. First group: The monocytes from BM patients were treated with plasma from BM patients. Second group: The monocytes from BM patients were treated with plasma from non-BM patients. The third group: The monocytes from BM patients were treated with plasma from non-BM patients, transfected with miR-21 mimic. Step 2: Induce differentiation of peripheral blood monocytes from non-BM patients under 3 conditions. First group: The monocytes from non-BM patients were treated with plasma from non-BM patients. Second group: The monocytes from non-BM patients were treated with plasma from BM patients. The third group: The monocytes from non-BM patients were treated with plasma from BM patients, transfected with miR-21 inhibitor.
Statistical Analysis
All data were presented as the mean values ± SD. All statistical analyses were performed with the SPSS 21 software (SPSS, Chicago, IL). Unpaired t-test was used for comparisons between two groups. One-way analysis of variance (ANOVA) with a post hoc Tukey’s test was used to compare multiple groups. All tests were two-sided. A P value less than 0.05 was considered to be statistically significant. The correlation analysis was performed by Pearson’s correlation coefficient. The receiver operating characteristic (ROC) curve was used to evaluate the diagnostic performance, such as area under the curve (AUC), cutoff point, sensitivity, and specificity.
Results
Highly Expressed and Secreted miRNAs by Lung Cancer Cells with BM Ability
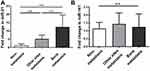
Our prior study demonstrated that SBC-5 cells can colonize into skeletal and non-skeletal tissues, whereas SBC-3 cells can only colonize into non-skeletal tissues after xenografts in NOD-SCID IL2Rγnull immunodeficient mice.7 This fact indicates that SBC-5 cells may produce some factors to communicate with skeleton. We first used miRNA microarray to analyze miRNAs that may regulate such communication. Fourteen miRNAs were found to be highly expressed in SBC-5 cell line (P<0.01) (Figure 1A). Seven miRNAs were increased by more than 5 folds: miR-200c, miR-141, miR-27a, miR-125b, miR-21, miR-221, and miR-23a (P <0.0001). These miRNAs were selected for further study. The changes in four of these miRNAs were further confirmed by qRT-PCR analyses (P <0.01, Figure 1B): miR-21, miR-23a, miR-141, and miR-221. We hypothesized that SBC-5 cells may secrete miRNAs to communicate with bone cells. The miRNAs in cancer cells culture supernatants were extracted and tested by qRT-PCR. The miR-21 level in the supernatants of SBC-3 cells was 1.02 ± 0.17, while in the supernatants of SBC-5 cells it was 14.69 ± 1.87 (P = 0.0184, Figure 1C). MiR-141 was also detected to be higher in the supernatant of SBC-5 but its difference was negative in the plasma of lung cancer patients with or without BM (Figure 2B). MiR-23a and miR-221 were not detected in the supernatants of SBC-5 and SBC-3 (data not shown). Therefore, the subsequent studies mainly focused on miR-21.
MiR-21 Secreted by SBC-5 Cells Could Promote Osteoclast Differentiation
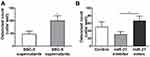
To determine the function of miRNAs secreted by SBC-5 cells, the supernatants from lung cancer cells were used to induce osteoclastogenesis from RAW264.7 cells. It was found that SBC-5 supernatants promoted more osteoclasts formation compared to SBC-3. 48.33 ± 11.06 mature osteoclasts per-well were observed with SBC-3 CM, while with SBC-5 CM, there were 101.33 ± 18.93 mature osteoclasts per-well (P=0.0138, Figure 3A). In the culture with SBC-5 CM, transfection of miR-21 inhibitor reduced osteoclasts compared to the control group, whereas transfection of miR-21 mimic increased osteoclasts. The number of osteoclasts was 50.67 ± 26.03 per-well in the control group, while osteoclasts increased to 58.00 ± 17.78 per-well with miR-21 mimic. With miR-21 inhibitor, osteoclasts decreased to 41.33 ± 11.37 per-well (Figure 3B). The difference between the three groups was significant (P=0.019). These data suggested that miR-21 secreted by SBC-5 could promote osteoclastogenesis.
High Level of miR-21 in the Plasma of Lung Cancer Patients with BM
To verify its clinical significance of higher miR-21 secreted by cancer cells, we recruited lung cancer patients and investigated miR-21 level in the plasma. The miR-21 level in the non-metastasis group was 1.24 ± 0.91, and 5.13 ± 2.40 in other sites metastasis group, whereas it was 12.46 ± 7.25 in the BM group. The level of miR-21 in the BM group was significantly higher than the other two groups (P<0.001, Figure 2A). The miR-141 levels were 1.15 ± 0.39, 1.44 ± 0.69, 1.24 ± 0.83 in non-metastasis, other sites metastasis and BM groups, respectively. There was no significant difference between the three groups (P=0.5487, Figure 2B). Therefore, miR-141 was excluded from the subsequent study. The different levels of circulating miR-21 in the three groups suggested its clinical significance in BM of lung cancer.
MiR-21 in the Plasma of BM Patients Could Promote Osteoclastogenesis
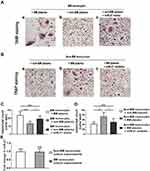
A cross-culture of peripheral blood monocytes and plasma from lung cancer patients with the intervention of exogenous miR-21 mimic or inhibitor was conducted to determine the role of circulating miR-21 in lung cancer patients with BM. It was observed that osteoclasts induced from BM patients’ monocytes with non-BM patients’ plasma CM (Figure 4Ab) were reduced, compared to that with BM patients’ plasma CM (Figure 4Aa), while transfection of miR-21 mimic rescued the decline of osteoclasts (Figure 4Ac). Accordingly, osteoclasts induced from non-BM patients’ monocytes with BM patients’ plasma CM (Figure 4Bb) was more than that with non-BM patients’ plasma CM (Figure 4Ba), while transfection of miR-21 inhibitor could reduce osteoclasts (Figure 4Bc). And the difference analysis is presented in Figure 4C and D. To eliminate the interference of miR-21 secreted from monocytes themselves during the differentiation, peripheral blood monocytes were cultured and induced under the condition without CM. The miR-21 level in the culture supernatants was tested. There was no significant difference between non-BM group (0.993± 0.089) and BM group (0.980± 0.115, P= 0.9316) (Figure 4E). These results suggested it was circulating miR-21 in plasma of BM patients that could promote the differentiation of osteoclasts. The miR-21 secreted by lung cancer cells may play an important role in bone metastasis in lung cancer patients.
Circulating miR-21 Affected PTEN to Promote Osteoclastogenesis
To explore the mechanism by which miR-21 promotes osteoclastogenesis, we searched the potential regulatory targets of miR-21 through bioinformatics analysis. We speculated that phosphatase and tensin homolog (PTEN), a tumor suppressor, may be a possible target of miR-21 in osteoclastogenesis. MiR-21 targets PTEN in lung cancer cells and hepatocellular cancer cells to enhance their proliferation and reduce apoptosis.8,9 Very recently PTEN has been shown to play an important role in osteoclast differentiation.10,11 To further validate our prediction, we performed another cross-culture of peripheral blood monocytes and plasma from lung cancer patients with the intervention of exogenous miR-21 inhibitor. The mRNA expressions of PTEN and osteoclast-related genes were measured by qRT-PCR to explore their correlation with miR-21.
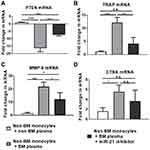
The mRNA expression of PTEN in the non-BM monocytes treated with non-BM plasma group was considered as a baseline, so the relative quantification of PTEN mRNA in other groups was expressed as negative. There was a significant difference between the three groups (P<0.0001) (Figure 5A). This indicated that up-regulation of circulating miR-21 in BM patients resulted in a down-regulation of PTEN expression in monocytes-osteoclasts. High level of plasma miR-21 from BM patients up-regulated mRNA expression of osteoclast-related genes: TRAP, CTSK, and MMP-9. A significant difference was observed in the levels of TRAP and MMP-9 mRNA between three groups (P<0.001, P<0.05, respectively) (Figure 5B and C). This result suggested that miR-21 in the plasma of BM patients may promote osteoclastogenesis by up-regulating MMP-9 and TRAP. There may be an association between down-regulation of PTEN and up-regulation of MMP-9 and TRAP. As for CTSK mRNA, no significant difference was observed between the three groups (P=0.0582). However, there was still a significant difference between non-BM monocytes plus non-BM plasma group and non-BM monocytes plus BM plasma group (P<0.05) (Figure 5D). These results suggested that circulating miR-21 promoted osteoclastogenesis through down-regulation of PTEN probably.
Correlation Between Circulating miR-21 Level and Clinical Characteristics
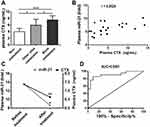
To further clarify the clinical significance of the high level of plasma miR-21 in BM patients, we investigate its association with some clinical characteristics. The plasma levels of C-terminal telopeptide of type I collagen (CTX) were 4.082± 0.9203 ng/mL, 6.046± 1.709 ng/mL and 7.658± 1.164 ng/mL in non-metastasis, other sites metastasis and BM group, respectively. The difference between the three groups was significant (P<0.05, Figure 6A). As for plasma CTX levels in patients with bisphosphonate treatment, it was 8.657 ± 0.8407 ng/mL before treatment and significantly decreased to 6.636 ± 0.5399 ng/mL after treatment (P=0.0021, Figure 6C). Accordingly, plasma level of miR-21 before treatment was 13.57 ± 9.45, and it was significantly decreased to 7.37 ± 5.50 after treatment in BM patients (P=0.023, Figure 6C). Pearson correlation analysis showed that there was a significant positive correlation between plasma miR-21 level and CTX level in these patients (r= 0.5524, 95% CI: 0.1704–0.7901, P= 0.0077, Figure 6B). This meant that circulating miR-21 level had a significant positive correlation with osteoclast activity.
To evaluate the diagnostic value of circulating miR-21 to screen BM in lung cancer patients, ROC curve analysis was performed. It determined that a cut-off value of 8.515 for miR-21 was optimal in the differentiation between lung cancer patients with or without BM, with a high AUC of 0.9091, 95% confidence interval (CI): 0.8170 to 1.001, p< 0.0001, 76.19% sensitivity and 95.45% specificity, as shown in Figure 6D. These results suggested that circulating miR-21 may serve as a biomarker for the diagnosis of BM in lung cancer patients.
Discussion
Lung cancer is one of the most malignant tumors worldwide due to the high incidence of metastases.12 Bone is the most common site for metastasis in lung cancer patients. Up to 60–75% of these patients developed BM at the later stages.13 Early diagnosis is crucial to prevent and treat BM. MiRNAs had been demonstrated to be critical regulators in cancer development and progression.14 Previously, miRNAs in the blood as biomarkers for some types of cancers had been reported. In recent years, several circulating miRNAs had been described to be associated with BM. However, it was still unclear about the clinical significance of circulating miRNAs in BM of lung cancer. In the present study, we found that miR-21 was specifically over-expressed and highly secreted by SBC-5 cells, a lung cancer cell line with characteristic BM ability. Higher-level miR-21 in the supernatant of SBC-5 cells could promote osteoclastogenesis. We also found that the level of circulating miR-21 in the plasma of BM patients was significantly higher than that of non-BM patients. Moreover, we observed that higher-level miR-21 from the plasma of BM patients could promote osteoclastogenesis. Mechanistically, circulating miR-21 from lung cancer promoted osteoclastogenesis through down-regulation of PTEN probably. As osteoclastogenesis was a key step in osteolytic BM, our results implied that the circulating miR-21 played critical roles in BM of lung cancer (Figure 7). Furthermore, we demonstrated that there was a significant positive correlation between plasma miR-21 level and CTX level in lung cancer patients. We presented a significant value of AUC (=0.9091, with 76.19% sensitivity and 95.45% specificity) of plasma circulating miRNA-21 to screen BM in lung cancer patients. This suggested that miR-21 may serve as a biomarker for BM diagnosis in lung cancer patients.
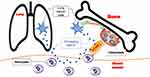 |
Figure 7 Schematic model of lung cancer cells-derived circulating miR-21 promoting bone metastasis by activating differentiation of monocytes into osteoclasts. |
Previous studies had shown the roles of tumor-derived miR-21 in multiple steps of tumor progression, including tumor growth at primary and secondary sites, invasion, migration, and extracellular matrix modification. MiR-21 promoted tumor growth by repressing many tumor suppressor genes, including PTEN,8,9/CDK2AP1,15 and B-cell translocation gene-2 (BTG-2),16 and inhibiting B-cell lymphoma-2 (BCL-2),17 an apoptosis gene. MiR-21 also increased cell motility and invasion by down-regulation of metastasis-related tumor suppressor genes, including maspin and programmed cell death 4 (PDCD4).18,19
MiR-21 level was found to be associated with tumorigenesis and metastasis in lung cancer. Zhang et al reported that miR-21 was higher in non-small cell lung cancer (NSCLC) tumor tissue. Notably, patients with advanced clinical TNM stage or distal metastasis showed higher miR-21 expression. Moreover, they identified an inverse correlation between miR-21 and PTEN levels in tumor tissue and further showed that miR-21 post-transcriptionally down-regulated PTEN to stimulate tumor cell growth in NSCLC.20 Tian et al also observed that miR-21 was up-regulated in NSCLC tissues compared with non-cancerous lung tissues. The patients with high miR-21 expression had a higher tumor grade and were at higher risk of lymph node metastasis.21
Over-expressing miR-21 in lung cancer cells could be released into circulation, leading to increased miR-21 levels in blood.22 Shen et al investigated miRNA profiles in paired lung tumor tissues and plasma specimens from NSCLC patients.23 MiR-21 displayed significant concordance between plasma and the corresponding tumor tissues, which could distinguish the NSCLC patients from the healthy controls. Wang et al also found that the level of miR-21 was higher in NSCLC serum samples than in control serum samples.24 Moreover, they noted that high serum miR-21 was significantly correlated with TNM stage and lymph node metastasis in NSCLC patients.
As expected, BM also presented a high level of miR-21 in lung cancer patients. Guo et al showed that the expression of miR-21 in NSCLC patients with BM was significantly higher than that in patients with bone tuberculosis.25 However, they only tested osseous tissues and did not analyze miR-21 level in blood.
BM was a complex multistep process and osteoclasts played important roles in this process.26 Osteoclasts were originated from osteoclast progenitor cells in the monocyte-macrophage lineage. RANKL-RANK-OPG (osteoprotegerin) signaling was critical in osteoclast formation and function. Once disseminated tumor cell reached bone marrow, they extravasated and colonized in this new microenvironment to form micrometastases. They interacted with bone cells, especially osteoclasts. Cancer cells secreted factors (RANKL, PTHrP, M-CSF) that acted in a paracrine way to promote osteoclastogenesis and activate osteoclast-mediated bone resorption.26 On the other hand, the active osteoclasts and resorbed bone matrix released growth factors and cytokines including transforming growth factor-beta (TGF-β) and insulin-like growth factors (IGFs). This accelerated the survival and growth of cancer cells and supported more bone destruction. Eventually, osteolytic BM was established and a vicious cycle was initiated. Bidirectional interactions between cancer cells and osteoclasts resulted in bone destruction and BM.27 Osteoclastogenesis was a key step in osteolytic metastasis.
MiRNAs played important roles in tumor progression and osteoclastogenesis. Some miRNAs may regulate RANKL–induced osteoclastogenesis. Recent studies suggested that cancer derived-miR-21 may function as a messenger between cancer cells and osteoclasts to promote osteoclastogenesis. Sugatani et al reported that RANKL–induced osteoclastogenesis was mediated by miR-21.28 MiR-21 was identified to down-regulate PDCD4 to remove the repression from c-Fos, a critical transcription factor for osteoclastogenesis and osteoclast-specific downstream genes. Besides, Hu et al indicated that the prevention of bone resorption in miR-21 knockout (miR-21−/−) mice was attributed to an inhibition of osteoclast function through miR-21 targeting PDCD4, despite the existence of RANKL.29
In addition to PDCD4, in our study, we found lung cancer cells-derived circulating miR-21 could promote osteoclastogenesis through PTEN probably. The working mechanism of PTEN on osteoclastogenesis had been described in recent studies. It was demonstrated that down-regulated PTEN promoted osteoclastogenesis through PI3K/Akt/NF-κB pathway,30 PI3K/Akt/GSK3β pathway,10 and PI3K/AKT/FoxO1 pathway.11
Recent studies have elucidated the impact of specific miRNA–mRNA interactions during in osteoclastogenesis. RANKL/RANK signaling plays a dominantrole in the differentiation, function and survival of osteoclasts. Several miRNAs directly and indirectly target the master osteoclast transcription factor NFATc1, downstream of the RANKL/RANK signaling.31 Among the target genes of miRNA-21, PTEN, a tumor suppressor and key modulator of cell survival and cell cycle progression have been well documented.32,33 There is an inverse relation between miR-21 and PTEN expression in cancer or noncancerous cells.34,35 MiR-21 promotes human lung cancer cell proliferation, cell cycle progression, and EMT, through direct regulation of PTEN/Akt/GSK3β signaling cascades.36 In recent studies, PTEN was identified as an important regulatory factor in RANKL-induced osteoclastogenesis.37 Overexpression of PTEN inhibits RANKL-induced osteoclast formation by its negative regulation of RANKL-induced AKT and NF-κB signaling.38 In a previous study, upregulated PTEN inhibited RANKL-induced PI3K/AKT activation. This reduced the mRNA and protein expression level of NFATc1, which correspondingly suppressed the expression of its downstream genes, including TRAP and CTSK.38 In addition to AKT/NF-κB/NFATc1 axis, AKT/GSK3β/NFATc1 signaling cascade also plays a critical role in osteoclastogenesis.39,40
Similar to our findings, a previous study found that miR-214 can function through PI3K/Akt/NF-kB/NFATc1 pathway by PTEN. MiR-214 resulted in down-regulated PTEN level and increased osteoclast activity. Inhibition of miR-214 levels significantly reduced osteoclast activity. Moreover, the expression of TRAP, MMP9 and CTSK was significantly up-regulated by miR-214 mimic and down-regulated by anti-miR-214, respectively.30 In a later study, with the delivery system, anti-miR-214 was delivered to osteoclasts, and bone microarchitecture and bone mass were improved in ovariectomized osteoporosis mice. In addition, osteoclast-related genes were also down-regulated, such as TRAP and CTSK.41 In addition, a recent study reported that miR-142-5p was also involved in the efficient regulation of osteoclast differentiation through targeting PTEN. And miR-142-5p regulated osteoclast differentiation through PTEN/PI3K/AKT/FoxO1 pathway. Furthermore, miR-142-5p increased the expression of NFATc1, CTSK, TRAP and MMP-9 by decreasing the PTEN protein level.11 In brief, PTEN is a well-documented target gene of miR-21. PTEN regulates osteoclastogenesis via various signaling pathways. Therefore, it is reasonable to speculate that miR-21 can promote osteoclastogenesis through down-regulation of PTEN.
TRAP, MMP9 and CTSK are important markers for the differentiation and activity of osteoclast.30 The mature osteoclast is described histologically as a multinucleated, TRAP-positive cell.42 Other characteristics of the osteoclast include the expression of enzymes such as CTSK and matrix MMP-9 that play a role in matrix degradation, and the vacuolar proton pump for the transport of protons to the resorption lacunae.42 CTSK is a lysosomal cysteine protease that is abundantly and selectively expressed in osteoclasts.43 MMP-9 is a matrix metalloproteinase that is also abundantly expressed in osteoclasts.43
TRAP, CTSK and MMP9 are also expressed in some cancer cells arising from different organs. Regardless of their source, TRAP, CTSK and MMP9 are reported to contribute to bone metastasis of cancers. The expression level of TRAP correlates with the severity of the tumor.44 In lung cancer, patients with high TRAP expression had a significantly lower overall survival than the patients with low TRAP expression.45 In addition, TRAP 5b was proposed as a serum marker for bone metastases in various types of primary cancers.46,47 MMPs contribute to ECM degradation, invasive growth and tumor angiogenesis,48 of which MMP-9 promotes tumor growth in the bone microenvironment. This promotion is related to osteoclast activity and this activity is the main responsible for bone resorption, which is the trigger of the denominated “vicious circle”.48 MMP9 facilitates the formation of bone metastasis by promoting osteoclastogenesis and directly degrading the bone matrix.49 Cancers with expression of CTSK have a high propensity to metastasize to bone. The role for CTSK in bone metastasis has been mainly attributed to the expansion of tumour within the bone.50 Moreover, preclinical studies show that CTSK inhibitors reduce breast cancer-induced osteolysis and skeletal tumor burden.51 In addition, the intraosseous mRNA levels of TRAP and CTSK in bone metastasis patients were also higher than those in breast cancer patients without BM and cancer-free individuals, respectively.52 In brief, TRAP, CTSK and MMP9 are osteoclast-specific differentiation markers, and their expressions can indirectly reflect the degree of osteoclasts differentiation and activity. Furthermore, they play important roles in bone metastasis of cancers. Therefore, we selected TRAP, CTSK, and MMP-9 as osteoclast-related genes.
Although our knowledge of BM had been increasing, it was still very difficult to cure BM. If BM patients could be detected at an early stage, it presented a better prognosis and overall survival rate. Therefore, it was essential to discover effective biomarkers for early diagnosis. MiRNA profiles reflected a specific profile for individual tumor status.53 Due to their stability in different biological fluids including blood, they were considered as an appropriate diagnostic biomarker. The changes of miRNAs may be observed at an early stage before clinical symptoms emerged. The easily available of miRNAs in blood made them useful biomarkers to screen metastasis in a non-invasive approach.
Some studies had reported that certain circulating miRNAs could be used as novel biomarkers to diagnose BM. In breast cancer patients, Zhao et al found serum miR-10b concentrations were significantly higher in patients with BM than that in patients without BM or healthy subjects.54 Serum miR-10b showed a significant AUC (=0.769) for BM with 64.8% sensitivity and 69.5% specificity. In our study, ROC analysis presented a high value of AUC (=0.9091, 76.19% sensitivity and 95.45% specificity) of plasma circulating miRNA-21 to screen BM patients, which suggested its potential application as a biomarker for BM diagnosis in lung cancer. Moreover, we found that there was a significant positive correlation between plasma miR-21 level and CTX level in lung cancer patients with BM. This meant circulating miR-21 level had a positive correlation with osteoclast activity. Besides, we found that bisphosphonate treatment significantly decreased plasma miR-21 and CTX levels in lung cancer patients with BM synchronously. This further confirmed the close relationship between miR-21 and cancer metastasis status.
There was some limitation worth to be mentioned in our study. First, this study was single-center and the number of patients was still not large enough, there may exist selection bias. Moreover, monocytes could differentiate into several types of cells including macrophages and osteoclasts. We only focused on the effects of circulating miR-21 on the differentiation of monocytes into osteoclasts. Although we identified miR-21 as a regulator of PTEN, we did not exclude other possible targets in addition to PTEN. In the present study we only used SCLC cell lines, it is necessary to verify some results in NSCLC cell lines. For technological and ethical reasons, we cannot obtain proper or enough clinical samples to test miR-21 level in the tumor tissue.
Conclusion
In conclusion, our study demonstrated that lung cancer cells-derived circulating miR-21 could promote osteoclastogenesis and inhibiting miR-21 would suppress osteoclastogenesis. Mechanistically, circulating miR-21 could be transferred into osteoclast precursor cells and promote osteoclast formation. This process was probably mediated through miR-21 targeting PTEN. As osteoclastogenesis was a key step in osteolytic BM, this novel finding would extend our knowledge on the roles of cancer-derived circulating miR-21 in BM and may provide a therapeutic approach for BM in lung cancer. Furthermore, our analysis demonstrated that circulating miR-21 was up-regulated in lung cancer patients with BM, and we found a high value of AUC (=0.9091, 76.19% sensitivity, and 95.45% specificity) of circulating miR-21 to screen BM patients. This suggested that circulating miR-21 may be a promising non-invasive biomarker for BM diagnosis in lung cancer patients.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81572639, 81770875), the Science and Technology Department of Sichuan Province (2018SZ0142, 20YYJC1063), the Sichuan University (2018SCUH0093), and the National Clinical Research Center for Geriatrics of West China Hospital (No. Z2018B05), and 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (2020HXFH008, ZYGD18022).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Zhang L, Gong Z. Clinical characteristics and prognostic factors in bone metastases from lung cancer. Med Sci Monit. 2017;23:4087–4094. doi:10.12659/MSM.902971
2. Hao C, Cui Y, Hu MU, et al. OPN-a splicing variant expression in non-small cell lung cancer and its effects on the bone metastatic abilities of lung cancer cells in vitro. Anticancer Res. 2017;37(5):2245–2254. doi:10.21873/anticanres
3. Zhou Y, Yu Q-F, Peng A-F, Tong W-L, Liu J-M, Liu Z-L. The risk factors of bone metastases in patients with lung cancer. Sci Rep. 2017;7(1):8970. doi:10.1038/s41598-017-09650-y
4. Huang Q, Ouyang X. The role of miRNAs in bone metastasis and their significance in the detection of bone metastasis: a review of the published data. Future Oncol. 2015;11(1):141–151. doi:10.2217/fon.14.161
5. Harquail J, Benzina S, Robichaud GA, Morin PJ. MicroRNAs and breast cancer malignancy: an overview of miRNA-regulated cancer processes leading to metastasis. Cancer Biomark. 2012;11(6):269–280. doi:10.3233/CBM-120291
6. Javid J, Sughayer MA, Mir R, Alauddin MS. Clinical significance of circulatory miRNA-21 as an efficient non-invasive biomarker for the screening of lung cancer patients. Asian Pac J Cancer Prev. 2018;19(9):2607–2611. doi:10.22034/APJCP.2018.19.9.2607
7. Gong M, Ma J, Guillemette R, et al. miR-335 inhibits small cell lung cancer bone metastases via IGF-IR and RANKL pathways. Mol Cancer Res. 2014;12(1):101–110. doi:10.1158/1541-7786.MCR-13-0136
8. Meng F, Henson R, Wehbe-Janek H, Ghoshal K, Jacob ST, Patel T. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology. 2007;133(2):647–658. doi:10.1053/j.gastro.2007.05.022
9. Li X, Zang A, Jia Y, et al. Triptolide reduces proliferation and enhances apoptosis of human non-small cell lung cancer cells through PTEN by targeting miR-21. Mol Med Rep. 2016;13(3):2763–2768. doi:10.3892/mmr.2016.4844
10. Lu S-H, Hsia Y-J, Shih K-C, Chou T-C. Fucoidan prevents RANKL-stimulated osteoclastogenesis and LPS-induced inflammatory bone loss via regulation of Akt/GSK3β/PTEN/NFATc1 signaling pathway and calcineurin activity. Mar Drugs. 2019;17(6). doi:10.3390/md17060345
11. Lou Z, Peng Z, Wang B, Li X, Li X, Zhang X. miR-142-5p promotes the osteoclast differentiation of bone marrow-derived macrophages via PTEN/PI3K/AKT/FoxO1 pathway. J Bone Miner Metab. 2019;37(5):815–824. doi:10.1007/s00774-019-00997-y
12. Jin X, Yu Y, Zou Q, et al. MicroRNA-105 promotes epithelial-mesenchymal transition of nonsmall lung cancer cells through upregulating Mcl-1. J Cell Biochem. 2018;120(4):5880–5888. doi:10.1002/jcb.27873
13. Weilbaecher KN, Guise TA, McCauley LK. Cancer to bone: a fatal attraction. Nat Rev Cancer. 2011;11(6):411–425. doi:10.1038/nrc3055
14. Liu C-H, Huang Q, Jin Z-Y, et al. Circulating microRNA-21 as a prognostic, biological marker in cholangiocarcinoma. J Cancer Res Ther. 2018;14(1):220–225. doi:10.4103/0973-1482.193125
15. Zheng J, Xue H, Wang T, et al. miR-21 downregulates the tumor suppressor P12 CDK2AP1 and stimulates cell proliferation and invasion. J Cell Biochem. 2011;112(3):872–880. doi:10.1002/jcb.22995
16. Liu M, Wu H, Liu T, et al. Regulation of the cell cycle gene, BTG2, by miR-21 in human laryngeal carcinoma. Cell Res. 2009;19(7):828–837. doi:10.1038/cr.2009.72
17. Shi L, Chen J, Yang J, Pan T, Zhang S, Wang Z. MiR-21 protected human glioblastoma U87MG cells from chemotherapeutic drug temozolomide induced apoptosis by decreasing Bax/Bcl-2 ratio and caspase-3 activity. Brain Res. 2010;1352:255–264. doi:10.1016/j.brainres.2010.07.009
18. Zhu Q, Wang Z, Hu Y, et al. miR-21 promotes migration and invasion by the miR-21-PDCD4-AP-1 feedback loop in human hepatocellular carcinoma. Oncol Rep. 2012;27(5):1660–1668. doi:10.3892/or.2012.1682
19. Yang Y, Meng H, Peng Q, et al. Downregulation of microRNA-21 expression restrains non-small cell lung cancer cell proliferation and migration through upregulation of programmed cell death 4. Cancer Gene Ther. 2015;22(1):23–29. doi:10.1038/cgt.2014.66
20. Zhang J-G, Wang J-J, Zhao F, Liu Q, Jiang K, Yang G-H. MicroRNA-21 (miR-21) represses tumor suppressor PTEN and promotes growth and invasion in non-small cell lung cancer (NSCLC). Clin Chim Acta. 2010;411(11–12):846–852. doi:10.1016/j.cca.2010.02.074
21. Tian L, Shan W, Zhang Y, Lv X, Li X, Wei C. Up-regulation of miR-21 expression predicate advanced clinicopathological features and poor prognosis in patients with non-small cell lung cancer. Pathol Oncol Res. 2016;22(1):161–167. doi:10.1007/s12253-015-9979-7
22. Browne G, Taipaleenmäki H, Stein GS, Stein JL, Lian JB. MicroRNAs in the control of metastatic bone disease. Trends Endocrinol Metab. 2014;25(6):320–327. doi:10.1016/j.tem.2014.03.014
23. Shen J, Todd NW, Zhang H, et al. Plasma microRNAs as potential biomarkers for non-small-cell lung cancer. Lab Invest. 2011;91(4):579–587.
24. Wang Z‐X, Bian H‐B, Wang J‐R, Cheng Z‐X, Wang K‐M, De W. Prognostic significance of serum miRNA‐21 expression in human non‐small cell lung cancer. J Surg Oncol. 2011;104(7):847–851. doi:10.1002/jso.22008
25. Guo Q, Zhang H, Zhang L, et al. MicroRNA-21 regulates non-small cell lung cancer cell proliferation by affecting cell apoptosis via COX-19. Int J Clin Exp Med. 2015;8(6):8835–8841.
26. Patel LR, Camacho DF, Shiozawa Y, Pienta KJ, Taichman RS. Mechanisms of cancer cell metastasis to the bone: a multistep process. Future Oncol. 2011;7(11):1285–1297. doi:10.2217/fon.11.112
27. Kagiya T. MicroRNAs and osteolytic bone metastasis: the roles of MicroRNAs in tumor-induced osteoclast differentiation. J Clin Med. 2015;4(9):1741–1752. doi:10.3390/jcm4091741
28. Sugatani T, Vacher J, Hruska KA. A microRNA expression signature of osteoclastogenesis. Blood. 2011;117(13):3648–3657. doi:10.1182/blood-2010-10-311415
29. Hu C-H, Sui B-D, Du F-Y, et al. miR-21 deficiency inhibits osteoclast function and prevents bone loss in mice. Sci Rep. 2017;120(1):43191. doi:10.1038/srep43191
30. Zhao C, Sun W, Zhang P, et al. miR-214 promotes osteoclastogenesis by targeting Pten/PI3k/Akt pathway. RNA Biol. 2015;12(3):343–353. doi:10.1080/15476286.2015.1017205
31. Hrdlicka HC, Lee S-K, Delany AM. MicroRNAs are critical regulators of osteoclast differentiation. Curr Mol Biol Rep. 2019;5(1):65–74. doi:10.1007/s40610-019-0116-3
32. Shi B, Wang Y, Zhao R, Long X, Deng W, Wang Z. Bone marrow mesenchymal stem cell-derived exosomal miR-21 protects C-kit+ cardiac stem cells from oxidative injury through the PTEN/PI3K/Akt axis. PLoS One. 2018;13(2):e0191616. doi:10.1371/journal.pone.0191616
33. Liu H, Le cheng CD, Zhang H. Suppression of miR-21 expression inhibits cell proliferation and migration of liver cancer cells by targeting Phosphatase and Tensin Homolog (PTEN). Med Sci Monit. 2018;24:3571–3577. doi:10.12659/MSM.907038
34. Lu J-M, Zhang -Z-Z, Ma X, Fang S-F, Qin X-H. Repression of microRNA-21 inhibits retinal vascular endothelial cell growth and angiogenesis via PTEN dependent-PI3K/Akt/VEGF signaling pathway in diabetic retinopathy. Exp Eye Res. 2020;190:107886. doi:10.1016/j.exer.2019.107886
35. Liu H-Y, Zhang -Y-Y, Zhu B-L, et al. miR-21 regulates the proliferation and apoptosis of ovarian cancer cells through PTEN/PI3K/AKT. Eur Rev Med Pharmacol Sci. 2019;23(10):4149–4155. doi:10.26355/eurrev_201905_17917
36. Cretoiu D. miR-21 regulates growth and EMT in lung cancer cells via PTEN Akt GSK3 beta signaling. Front Biosci. 2019;24(8):1426–1439. doi:10.2741/4788
37. Blüml S, Friedrich M, Lohmeyer T, et al. Loss of phosphatase and tensin homolog (PTEN) in myeloid cells controls inflammatory bone destruction by regulating the osteoclastogenic potential of myeloid cells. Ann Rheum Dis. 2015;74(1):227–233. doi:10.1136/annrheumdis-2013-203486
38. Meng J, Zhang W, Wang C, et al. Catalpol suppresses osteoclastogenesis and attenuates osteoclast-derived bone resorption by modulating PTEN activity. Biochem Pharmacol. 2020;171:113715. doi:10.1016/j.bcp.2019.113715
39. Jang HD, Noh JY, Shin JH, Lin JJ, Lee SY. PTEN regulation by the Akt/GSK-3β axis during RANKL signaling. Bone. 2013;55(1):126–131. doi:10.1016/j.bone.2013.02.005
40. Moon JB, Kim JH, Kim K, et al. Akt induces osteoclast differentiation through regulating the GSK3β/NFATc1 signaling cascade. J Immunol. 2012;188(1):163–169. doi:10.4049/jimmunol.1101254
41. Cai M, Yang L, Zhang S, Liu J, Sun Y, Wang X. A bone-resorption surface-targeting nanoparticle to deliver anti-miR214 for osteoporosis therapy. Int J Nanomedicine. 2017;12:7469–7482. doi:10.2147/IJN.S139775
42. Bonewald LF. Chapter 313 - cell–cell and cell–matrix interactions in bone. In: Bradshaw RA, Dennis EA, editors. Handbook of Cell Signaling. Volume 1.
43. Tomita A, Kasaoka T, Inui T, et al. Human breast adenocarcinoma (MDA-231) and human lung squamous cell carcinoma (Hara) do not have the ability to cause bone resorption by themselves during the establishment of bone metastasis. Clin Exp Metastasis. 2008;25(4):437–444. doi:10.1007/s10585-008-9148-4
44. Zenger S, He W, Ek-rylander B, et al. Differential expression of tartrate-resistant acid phosphatase isoforms 5a and 5b by tumor and stromal cells in human metastatic bone disease. Clin Exp Metastasis. 2011;28(1):65–73. doi:10.1007/s10585-010-9358-4
45. Gao Y-L, Liu M-R, Yang S-X, Dong Y-J, Tan X-F. Prognostic significance of ACP5 expression in patients with lung adenocarcinoma. Clin Respir J. 2018;12(3):1100–1105. doi:10.1111/crj.2018.12.issue-3
46. Holen I, Walker M, Nutter F, et al. Oestrogen receptor positive breast cancer metastasis to bone: inhibition by targeting the bone microenvironment in vivo. Clin Exp Metastasis. 2016;33(3):211–224. doi:10.1007/s10585-015-9770-x
47. Haider M-T, Holen I, Dear TN, Hunter K, Brown HK. Modifying the osteoblastic niche with zoledronic acid in vivo—potential implications for breast cancer bone metastasis. Bone. 2014;66(100):240–250. doi:10.1016/j.bone.2014.06.023
48. Pego ER, Fernández I, Núñez MJ. Molecular basis of the effect of MMP-9 on the prostate bone metastasis: a review. Urol Oncol. 2018;36(6):272–282. doi:10.1016/j.urolonc.2018.03.009
49. Wu Z, Wang T, Fang M, et al. MFAP5 promotes tumor progression and bone metastasis by regulating ERK/MMP signaling pathways in breast cancer. Biochem Biophys Res Commun. 2018;498(3):495–501. doi:10.1016/j.bbrc.2018.03.007
50. Podgorski I, Linebaugh BE, Sloane BF. Cathepsin K in the bone microenvironment: link between obesity and prostate cancer? Biochem Soc Trans. 2007;35(Pt 4):701–703. doi:10.1042/BST0350701
51. Le Gall C, Bonnelye E, Clézardin P. Cathepsin K inhibitors as treatment of bone metastasis. Curr Opin Support Palliat Care. 2008;2(3):218–222. doi:10.1097/SPC.0b013e32830baea9
52. Liu J, Li D, Dang L, et al. Osteoclastic miR-214 targets TRAF3 to contribute to osteolytic bone metastasis of breast cancer. Sci Rep. 2017;7:40487. doi:10.1038/srep40487
53. Chan S-H, Wang L-H. Regulation of cancer metastasis by microRNAs. J Biomed Sci. 2015;22(1):9. doi:10.1186/s12929-015-0113-7
54. Zhao F-L, Hu G-D, Wang X-F, Zhang X-H, Zhang Y-K, Yu Z-S. Serum overexpression of microRNA-10b in patients with bone metastatic primary breast cancer. J Int Med Res. 2012;40(3):859–866. doi:10.1177/147323001204000304
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.