Back to Journals » OncoTargets and Therapy » Volume 10
Low-grade central osteosarcoma in proximal humerus: a rare entity
Authors Tang F, Min L, Zhou Y, Luo Y, Tu CQ ![]()
Received 29 May 2017
Accepted for publication 26 September 2017
Published 24 October 2017 Volume 2017:10 Pages 5165—5172
DOI https://doi.org/10.2147/OTT.S142818
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Ingrid Espinoza
Fan Tang,1,2 Li Min,1,2 Yong Zhou,1 Yi Luo,1 Chongqi Tu1
1Department of Orthopedics, West China Hospital, Sichuan University, Chengdu, People’s Republic of China; 2Sarcoma Biology Laboratory, Center for Sarcoma and Connective Tissue Oncology, Massachusetts General Hospital, Boston, MA, USA
Abstract: Low-grade central osteosarcoma is a rare subtype of tumor with low-grade malignancy. Currently, wide resection with negative resection margin is the standard treatment for this disease. The role of neoadjuvant chemotherapy in low-grade central osteosarcoma was controversial and was mostly considered for tumors containing high-grade focal areas. Local tumor recurrences often exhibited a tumor with higher histologic grade or differentiation with the potential for metastases. In low-grade central osteosarcoma, timely wide resection after definite diagnosis can result in 5-year survival for almost 90%. However, the relatively nonspecific radiological and pathological findings make diagnosis very difficult. MDM2 and CDK4 are specific and provide sensitive markers for the diagnosis of low-grade central osteosarcoma, helping to differentiate low-grade central osteosarcoma from some benign lesions, including fibrous dysplasia, bone giant cell tumor, and chondrosarcoma. Here, we report the case of a 19-year-old woman with low-grade central osteosarcoma located at the proximal humerus. The affected site was rare, but the sensitive biomarkers CDK4 and MDM2 were positive. The patient recovered well after wide tumor resection following a proximal humerus endoprosthesis replacement. Our case highlighted the management strategies in low-grade central osteosarcoma. Being familiar with radiographic features, understanding the biological characteristics, and mastering diagnostic biomarkers can help oncologists avoid embarrassing situations in treatment when this rare tumor is highly suspected, even when located at an uncommon site. The discussion in this report focuses on radiographic and pathological features, advances of biomarkers that help in differential diagnosis, and current treatment options in low-grade central osteosarcoma.
Keywords: low-grade central osteosarcoma, proximal humerus, differential diagnosis, wide resection, MDM2, CDK4
Introduction
Low-grade central osteosarcoma (LGCOS) was firstly described as an “intraosseous well-differentiated osteosarcoma” by Unni et al.1 The tumor is rare and accounts for less than 2% of all osteosarcomas.2 There is a strong predilection for the long bones, particularly the distal femur and proximal tibia which are involved in more than half of the cases.3 Other affected sites include the rib, skull, ankle, and mandible, with a few individual cases documented in the literature.4–7 Unlike the majority of osteosarcomas, LGCOS is less aggressive, has limited metastatic potential, and a relatively good prognosis.1 However, local tumor recurrences are very often found to exhibit a higher histologic grade tumor or differentiation, with the potential for metastases.8,9 Wide resection with a negative margin is recognized as the standard treatment for LGCOS currently. Although relatively rare, the high differentiation and relatively low malignancy of this tumor contribute to an extremely high rate of initial misdiagnoses, especially presenting similarly as fibrous dysplasia, and sometimes as giant cell tumor and chondrosarcoma in its clinical manifestation.10 If the diagnosis of an LGCOS is highly suspected but cannot be established according to its radiologic findings, even when a biopsy suggests a benign lesion, a richer biopsy sample or immunohistochemical staining is helpful for diagnosis.11 Here, we describe a rare case of primary LGCOS affecting the proximal humerus in a young female who finally underwent tumor en bloc resection followed by prosthetic reconstruction. The location of the tumor in our patient was uncommon, but the patient recovered well after our definite diagnosis and timely treatment. We reviewed the literature and discussed the differential radiographic diagnosis, diagnostic markers, and appropriate treatment options for this rare entity.
Case presentation
A 19-year-old woman was admitted to our department with a chief complaint of left shoulder pain for 11 months. She had suffered a fall when skating 20 days earlier, leading to exacerbation of pain following activity. She was transferred from a local hospital, and the X-ray revealed a lytic destruction in her left proximal humerus with a suspected pathological fracture at the humeral neck. Chondrosarcoma was suspected on pathological diagnosis after fine-needle aspiration. However, she refused treatment at her local hospital and was transferred to our department for further treatment. There was a slight swelling over her left upper arm without any erythema. A slight activity restriction was detected on physical examination. Then, a shoulder–elbow belt was used to temporarily fix her affected arm.
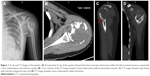
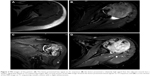
Plain radiographs in our hospital showed a lytic, expansible lesion with a sclerotic margin in the proximal humerus (Figure 1). The following radiographic examinations including computed tomography (CT), magnetic resonance imaging (MRI), and bone scan were performed. CT images revealed there was predominantly lytic mixed with coarse trabeculation and densely sclerotic slight cortical destruction in the tumor (Figure 1). MRI showed that the lesion presented low signals on T1-sequence, and mixed signals on T2-sequence, images with a slight enhancement (Figure 2). An increased nuclear uptake at the destruction area was found on bone scan (Figure 3). Cancer markers including CEA, AFP, CA-125, and CA199 were all normal. CT of chest indicated there was no lung metastasis.
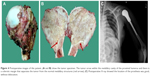
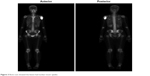  | Figure 3 Bone scan showed the lesion had nuclear tracer uptake. |
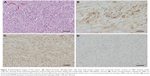
Open biopsy was undertaken and pathological examination showed a spindle cell tumor of low-grade malignancy. Then, proximal humerus tumor en bloc resection followed by modular prosthetic replacement was carried out. The operation was performed with the patient under general anesthesia in a lateral position. The length of the tumor segment was 5.4 cm, and the resection length was 7.5 cm so that there was a 2 cm-tumor-negative margin (Figure 4). The attachments of the latissimus dorsi and teres major on the humerus were severed to remove the tumor. The rotator cuff and axillary nerve were not affected by tumor. The bone defect present after tumor resection was reconstructed using a modular proximal humerus endoprosthesis (cement type; Chunglizhengda Medical Instruments Co., Beijing, People’s Republic of China). Then, the rotator cuff was sutured to the head of the prosthesis with 2–0 non-absorbable sutures (Ethibond* Excel, Polyester Suture, Green Braided, Johnson & Johnson, USA) to improve the stability of the shoulder joint. Muscles and soft tissues were reconstructed in situ. No mesh was used during the operation. The final pathological diagnosis was LGCOS with strong CDK4 and SMA positivity and focal mild MDM2 positivity on immumohistochemical staining (Figure 5). The distal operated edge of the humerus was pathologically confirmed to be tumor negative. Olympus cellSens Entry (Version 510, OLYMPUS CORPORATION) was used to analysis the positive integrated optical density (IOD); the values of CDK4 was 0.32, SMA was 0.24, and MDM2 was 0.01, respectively.
The patient recovered well and was discharged 5 days after the surgery. She did not receive further chemotherapy and was closely followed-up postoperatively. Imaging studies were focused on tumor recurrence and the stability of the prosthetic implant. Up to the time of writing, 12 months after the surgery, the patient was capable of all activities of daily living needed for self-care and the MSTS (Musculoskeletal Tumor Society) score was 25.0. There were no signs of tumor recurrence and the positioning of the prosthetic remains good, with no dislocation, loosening, or fracture. Moreover, the follow-up is being continued.
Written informed consent with regard to publication of this case report and accompanying images was obtained from the patient.
Discussion
Thus far, only 11 cases of LGCOS located at the proximal humerus have been documented in the English literature, including two cases arising secondarily from other lesions (Table 1).3,10,12,13 The patient in our study was a 19-year-old woman with an 11-month history of pain in her shoulder. There is no gender predilection for LGCOS, and patients are usually affected in the third or early fourth decade of life. They frequently have a long history of nonspecific symptoms, the most common being pain and a slow-growing mass.14
  | Table 1 Previous cases of LGCOS located at the proximal humerus |
Diagnosing LGCOS remains challenging due to the relatively nonspecific radiological findings. The rate of misdiagnosis of LGCOS surprisingly reached 50%.10 Andresen et al identified four radiographic patterns of LGCOS: First, lytic with varying amounts of thick and coarse trabeculation; Second, predominantly lytic with few thin, incomplete trabecula; Third, densely sclerotic; and, fourth, mixed lytic and sclerotic.15 The case in our study presented as mainly lytic with trabeculation and sclerotic in CT images, revealing that our case includes all four radiographic patterns (Figure 1). Aggressive features in the present case, including cortical destruction, soft-tissue extension, and periosteal reaction, may be lacking in a patient with LGCOS. Overall, the case in our study presented benign-appearing, but focally aggressive, tumor features including cortical destruction and slight soft-tissue extension. In most cases, LGCOS mimics fibrous dysplasia.16,17 However, the radiographic differential diagnoses of our case during the initial diagnosis period were aneurysmal bone cyst, chondrosarcoma, and giant cell tumor of the bone. X-rays of giant cell tumors in long bones usually show an expansile, often lobulated, area of osteolysis with a narrow zone of transition. There is little or no evidence of matrix calcification within the tumor and relatively little trabeculation in lytic lesions.18 Aneurysmal bone cyst arises in the under-30 age group and presents with pain and swelling. Aneurysmal bone cyst is often eccentrically located in the metaphysis of a long bone, including in the proximal humerus, adjacent to the non-fused physeal growth plate. Moreover, there is an expansile lytic lesion with a thin sclerotic margin within the tumor.19 However, the lack of nuclear uptake on bone scan and obvious long T1 and long T2 signals on MRI can distinguish the aneurysmal bone cyst from LGCOS in our case. In chondrosarcoma, plain radiograph demonstrates a predominantly lytic lesion that involves the metaphysis and proximal diaphysis, but contains multiple areas of chondroid matrix mineralization, which was lacking in our case.20
Another problem with LGCOS is the difficulty in establishing a definite pathological diagnosis. Any difficult or non-diagnostic biopsies of solitary bone lesions should be referred to specialist tumor units for a second opinion. Several biological markers have been investigated to distinguish LGCOS from some benign diseases, such as osteonectin, c-fos, and ezrin, but none of these markers have been shown to be useful. AgNOR and MIB-1 have been reported to be helpful in distinguishing fibrous dysplasia from low-grade osteosarcomas.21 The pathological images showed strong CDK4-positive and weak focal MDM2-positive staining on immunohistochemical analysis. Positive immunohistochemical staining for CDK4 or MDM2 may be used as a diagnostic marker, although negative immunostaining cannot rule out this tumor. Dujardin et al figured out that immunohistochemical expression of MDM2 and CDK4 is specific and provides sensitive markers for the diagnosis of low-grade osteosarcomas, helping to differentiate LGCOS from benign fibrous lesions, particularly in cases with atypical radioclinical manifestation.22 MDM2 alterations might be involved in the tumorigenesis of LGCOS.23 CDK4 expression may be associated with the clinical features of LGCOS. Fifty percent of the tumors showed diffuse immunohistochemical staining for CDK4. Among them, 75% of CDK4-positive tumors presented as lytic lesions on a plain radiograph. Moreover, 33% of tumors showing a sclerotic pattern on a plain radiograph were positive for CDK4.24 Yoshida et al reported that MDM2 and CDK4 were positive, based on immunohistochemical labeling, in 70% and 87% of low-grade osteosarcomas, respectively. The combination of the two markers thus shows 100% sensitivity and 97.5% specificity for the diagnosis of low-grade osteosarcoma.13 Mutational analysis may be another additional helpful parameter in individual cases for the differential diagnosis of fibrous dysplasia and LGCOS, as GNAS mutations that are highly specific for fibrous dysplasia and rarely occur in low-grade osteosarcomas.25 In addition, there was a low prevalence of Gsα gene mutations in LGCOS in contrast to monostotic fibrous dysplasia.26
The patient in our study underwent wide tumor resection followed by modular prosthetic reconstruction without postoperative chemotherapy. The tumor may recur to be a higher-grade disease with a tendency for distant metastasis. In the Mayo Clinic series, 15% of recurrences appeared as a conventional osteosarcoma with poor prognosis.27 There is agreement that wide resection is the only accepted treatment of LGCOS. Under this circumstance, the prognosis of this rare but low-grade malignant tumor was excellent, with 90% overall survival at 5 years.2 Intralesional excision alone leads to a poorer prognosis. Therefore, wide tumor resection with a negative margin was necessary, even after intralesional excision.
The role of chemotherapy in LGCOS is controversial. In fact, high-grade foci (grade 3 according to Broder’s grading system) are sometimes detected in LGCOS (grade 1 and 2). According to the clinical series outcome reported by Righi et al, in 20 cases of low-grade osteosarcoma with high-grade foci (grade 3) in less than 50% of the tumor, nine cases did not receive adjuvant chemotherapy, with only one death attributed to unrelated causes. In the other 13 cases of low-grade osteosarcoma with high-grade foci (grade 3) in more than 50% of the tumor, 12 patients received adjuvant chemotherapy: two had recurrence, four developed multiple lung metastases, and three succumbed to the disease. Therefore, patients with a diagnosis of LGCOS where the high-grade (grade 3) component is less than 50% of the resected specimen may not require chemotherapy, achieving good prognosis by means of en bloc resection only.3
Conclusion
LGCOS is, indeed, a rare tumor with radiological and pathological features indicating benign lesion. Wide tumor resection with negative margin was the standard treatment for this disease. Timely treatment is undertaken on the premise of accurate and definite diagnosis, but the relatively nonspecific radiological and pathological findings make diagnosis very difficult. CDK4 or MDM2 may be used as diagnostic immunohistochemical markers. This study highlights the importance of mastering radiographic features, understanding the biological characteristics, and being familiar with diagnostic biomarkers of LGCOS when this rare tumor is highly suspected, even when located at an uncommon site in the body.
Acknowledgments
The authors wish to acknowledge funding in support of Fan Tang by a scholarship from the China Scholarship Council (201606240118). Li Min is supported by the New Teacher Fund of the Ministry of Education in China (number 20120181120028).
Disclosure
The authors report no conflicts of interest in this work.
References
Unni KK, Dahlin DC, McLeod RA, Pritchard DJ. Intraosseous well-differentiated osteosarcoma. Cancer. 1977;40(3):1337–1347. | ||
Choong PF, Pritchard DJ, Rock MG, Sim FH, McLeod RA, Unni KK. Low grade central osteogenic sarcoma. A long-term followup of 20 patients. Clin Orthop Relat Res. 1996;(322):198–206. | ||
Righi A, Paioli A, Dei Tos AP, et al. High-grade focal areas in low-grade central osteosarcoma: high-grade or still low-grade osteosarcoma? Clin Sarcoma Res. 2015;5:23. | ||
Bugnone AN, Temple HT, Pitcher JD. Low-grade central osteosarcoma of the foot and ankle: radiographic and pathologic features in two patients: case report and literature review. Foot Ankle Int. 2005;26(6):494–500. | ||
Diniz AF, Filho JA, Alencar Rde C, et al. Low-grade central osteosarcoma of the mandible: a case study report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103(2):246–252. | ||
Moghadamfalahi M, Alatassi H. Low-grade central osteosarcoma of the rib: a case report and brief review of the literature. Case Rep Pathol. 2013;2013:798435. | ||
Fukunaga M. Low-grade central osteosarcoma of the skull. Pathol Res Pract. 2005;201(2):131–135. | ||
Iemoto Y, Ushigome S, Fukunaga M, Nikaido T, Asanuma K. Case report 679. Central low-grade osteosarcoma with foci of dedifferentiation. Skeletal Radiol. 1991;20(5):379–382. | ||
Ogose A, Hotta T, Emura I, Imaizumi S, Takeda M, Yamamura S. Repeated dedifferentiation of low-grade intraosseous osteosarcoma. Hum Pathol. 2000;31(5):615–618. | ||
Malhas AM, Sumathi VP, James SL, et al. Low-grade central osteosarcoma: a difficult condition to diagnose. Sarcoma. 2012;2012:764796. | ||
Hayashi K, Tsuchiya H, Yamamoto N, et al. Diagnosis and treatment of low-grade osteosarcoma: experience with nine cases. Int J Clin Oncol. 2014;19(4):731–738. | ||
Endo M, Yoshida T, Yamamoto H, et al. Low-grade central osteosarcoma arising from bone infarct. Hum Pathol. 2013;44(6):1184–1189. | ||
Yoshida A, Ushiku T, Motoi T, et al. Immunohistochemical analysis of MDM2 and CDK4 distinguishes low-grade osteosarcoma from benign mimics. Mod Pathol. 2010;23(9):1279–1288. | ||
Brooks S, Starkie CM, Clarke NM. Osteosarcoma after the fourth decade. A clinico-pathological review. Arch Orthop Trauma Surg. 1985;104(2):100–105. | ||
Andresen KJ, Sundaram M, Unni KK, Sim FH. Imaging features of low-grade central osteosarcoma of the long bones and pelvis. Skeletal Radiol. 2004;33(7):373–379. | ||
Muramatsu K, Hashimoto T, Seto S, Gondo T, Ihara K, Taguchi T. Low-grade central osteosarcoma mimicking fibrous dysplasia: a report of two cases. Arch Orthop Trauma Surg. 2008;128(1):11–15. | ||
Franceschina MJ, Hankin RC, Irwin RB. Low-grade central osteosarcoma resembling fibrous dysplasia. A report of two cases. Am J Orthop (Belle Mead NJ). 1997;26(6):432–440. | ||
Purohit S, Pardiwala DN. Imaging of giant cell tumor of bone. Indian J Orthop. 2007;41(2):91–96. | ||
Mendenhall WM, Zlotecki RA, Gibbs CP, Reith JD, Scarborough MT, Mendenhall NP. Aneurysmal bone cyst. Am J Clin Oncol. 2006;29(3):311–315. | ||
Soldatos T, McCarthy EF, Attar S, Carrino JA, Fayad LM. Imaging features of chondrosarcoma. J Comput Assist Tomogr. 2011;35(4):504–511. | ||
Okada K, Nishida J, Morita T, Kakizaki H, Ishikawa A, Hotta T. Low-grade intraosseous osteosarcoma in northern Japan: advantage of AgNOR and MIB-1 staining in differential diagnosis. Hum Pathol. 2000;31(6):633–639. | ||
Dujardin F, Binh MB, Bouvier C, et al. MDM2 and CDK4 immunohistochemistry is a valuable tool in the differential diagnosis of low-grade osteosarcomas and other primary fibro-osseous lesions of the bone. Mod Pathol. 2011;24(5):624–637. | ||
Park HR, Jung WW, Bertoni F, et al. Molecular analysis of p53, MDM2 and H-ras genes in low-grade central osteosarcoma. Pathol Res Pract. 2004;200(6):439–445. | ||
Jeon DG, Koh JS, Cho WH, et al. Clinical outcome of low-grade central osteosarcoma and role of CDK4 and MDM2 immunohistochemistry as a diagnostic adjunct. J Orthop Sci. 2015;20(3):529–537. | ||
Salinas-Souza C, De Andrea C, Bihl M, et al. GNAS mutations are not detected in parosteal and low-grade central osteosarcomas. Mod Pathol. 2015;28(10):1336–1342. | ||
Pollandt K, Engels C, Kaiser E, Werner M, Delling G. Gsalpha gene mutations in monostotic fibrous dysplasia of bone and fibrous dysplasia-like low-grade central osteosarcoma. Virchows Arch. 2001;439(2):170–175. | ||
Kurt AM, Unni KK, McLeod RA, Pritchard DJ. Low-grade intraosseous osteosarcoma. Cancer. 1990;65(6):1418–1428. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.