Back to Journals » Journal of Pain Research » Volume 14
Low-Dose Interleukin-2 and Regulatory T Cell Treatments Attenuate Punctate and Dynamic Mechanical Allodynia in a Mouse Model of Sciatic Nerve Injury
Authors Hu R, Zhang J, Liu X, Huang D, Cao YQ
Received 10 January 2021
Accepted for publication 13 March 2021
Published 6 April 2021 Volume 2021:14 Pages 893—906
DOI https://doi.org/10.2147/JPR.S301343
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Professor E Alfonso Romero-Sandoval
Rong Hu,1– 3 Jintao Zhang,1,2,4 Xuemei Liu,1,2 Dong Huang,3 Yu-Qing Cao1,2
1Department of Anesthesiology, Washington University School of Medicine, St. Louis, MO, USA; 2Washington University Pain Center, Washington University School of Medicine, St. Louis, MO, USA; 3Department of Pain Management, The Third Xiangya Hospital, Central South University, Changsha, Hunan, People’s Republic of China; 4Department of Anesthesiology, Nanfang Hospital, Southern Medical University, Guangzhou, People’s Republic of China
Correspondence: Yu-Qing Cao
Department of Anesthesiology, Washington University School of Medicine, 660 South Euclid, Box 8054, St. Louis, MO, 63110, USA
Tel +1 314 362 8726
Fax +1 314 362 8334
Email [email protected]
Purpose: Nerve injury-induced mechanical hyper-sensitivity, in particular stroking-induced dynamic allodynia, is highly debilitating and difficult to treat. Previous studies indicate that the immunosuppressive regulatory T (Treg) cells modulate the magnitude of punctate mechanical allodynia resulting from sciatic nerve injury. However, whether enhancing Treg-mediated suppression attenuates dynamic allodynia is not known. In the present study, we addressed this knowledge gap by treating mice with low-dose interleukin-2 (ld-IL2) injections or adoptive transfer of Treg cells.
Methods: Female Swiss Webster mice received daily injections of ld-IL2 (1 μg/mouse, intraperitoneally) either before or after unilateral spared nerve injury (SNI). Male C57BL/6J mice received adoptive transfer of 1 x 106 Treg cells 3 weeks post-SNI. The responses to punctate and dynamic mechanical stimuli on the hindpaw were monitored before and up to 4– 6 weeks post-SNI. We also compared the distribution of Treg cells and CD3+ total T cells after SNI and/or ld-IL2 treatment.
Results: Ld-IL2 pretreatment in female Swiss Webster mice completely blocked the development of SNI-induced dynamic mechanical allodynia and reduced the magnitude of punctate allodynia. Delayed ld-IL2 treatment in female mice significantly attenuated the morphine-resistant punctate and dynamic allodynia at 3– 5 weeks post-SNI. Adoptive transfer of Treg cells to male C57BL/6J mice 3 weeks post-SNI effectively reversed the persistent punctate and dynamic allodynia, supporting that the effect of ld-IL2 is mediated through endogenous Treg cells, and is likely independent of mouse strain and sex. Neither ld-IL2 treatment nor Treg transfer affected the basal responses to punctate or brush stimuli. Ld-IL2 significantly increased the frequency of Treg cells among total CD3+ T cells in the injured sciatic nerves but not in the uninjured nerves or the dorsal root ganglia, suggesting the injured nerve as ld-IL2’s site of action.
Conclusion: Collectively, results from the present study supports Treg as a cellular target and ld-IL2 as a potential therapeutic option for nerve injury-induced persistent punctate and dynamic mechanical allodynia.
Keywords: spared nerve injury, chronic pain, punctate allodynia, dynamic allodynia, low-dose interleukin-2, regulatory T cell
Introduction
About 7–10% of the general population suffer from neuropathic pain that results from damage to the somatosensory nervous system.1 Mechanical hyper-sensitivity, especially the dynamic allodynia evoked by stroking stimuli (brush or gentle touch), is one of the most debilitating and prevalent symptoms of neuropathic pain, and is often resistant to current treatments.2,3 In animal models of neuropathic pain, morphine attenuates punctate allodynia evoked by von Frey filaments at early (< 2 weeks post-injury) but not late stage;4–6 whereas the dynamic allodynia is resistant to morphine treatment at all stages.4,6 Likewise, current pharmacotherapy only achieve less than 50% reduction of pain severity in about 30% neuropathic pain patients.7
Regulatory T (Treg) cells are a specialized subpopulation of CD4+ T cells that express high-level of transcription factor Foxp3 and the high-affinity interleukin-2 (IL2) receptor CD25.8 They employ multiple mechanisms to suppress the activation and proliferation of all other immune cells to maintain immune homeostasis.8 In mouse models of sciatic nerve injury, the number of Treg cells is reduced in the spleen9 but is increased in the injured nerves, the ipsilateral draining lymph nodes, dorsal root ganglia (DRG), and the dorsal horn of the spinal cord.10,11 Nerve injury-induced cutaneous punctate mechanical allodynia is exacerbated by the depletion of endogenous Treg cells, and is attenuated by the increase in Treg cell number.9–14 However, whether enhancing Treg number and/or function ameliorates the morphine-resistant dynamic allodynia has not been tested.
Results from many preclinical and clinical studies have supported low-dose interleukin-2 (ld-IL2) treatment as a safe and effective treatment for multiple autoimmune and neurodegenerative diseases through enhancing Treg-mediated immunosuppression.15–17 Ld-IL2 effectively reverses behavioral sensitization in several mouse models of chronic headache disorders without altering basal nociceptive responses or inducing tolerance.18 A recent study reports that ld-IL2 given soon after nerve injury mildly attenuates early punctate allodynia (< 2 weeks post-injury) through inhibition of the Th1 response at the injured nerve.10 In this study, we investigated whether ld-IL2 treatment and Treg cell transfer are effective for the prolonged punctate and dynamic allodynia resulting from peripheral nerve injury. Since nerve injury-induced mechanical allodynia affects both males and female, we tested the effects of ld-IL2 in outbred female Swiss Webster mice and the effects of Treg adoptive transfer in inbred male C57BL/6J mice, respectively. In the mouse model of spared nerve injury (SNI), ld-IL2 treatment attenuated the punctate allodynia as well as the dynamic allodynia more than 3 weeks after SNI without affecting basal responses to punctate or brush stimuli. The effects of ld-IL2 were recapitulated by the adoptive transfer of Treg cells, independent of mouse sex and/or strain. Ld-IL2 significantly increased the frequency of Treg cells among total CD3+ T cells in the injured sciatic nerves but not in the uninjured nerves or the DRG. Collectively, the present study supports Treg cell as a promising target for treating nerve injury-induced punctate and dynamic allodynia and ld-IL2 as a potential treatment option.
Materials and Methods
Mice
All procedures were carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and were approved by the Institutional Animal Care and Use Committee at Washington University in St. Louis (#19-0895). All efforts were made to minimize the number of mice used and their suffering. To avoid social isolation stress, all mice were group housed (2–5 per cage, same sex) in the animal facility of Washington University in St. Louis on a 12-hour light–dark cycle with constant temperature (23–24°C), humidity (45–50%), and food and water ad libitum. All experiments were performed during the light phase (9 am to 4 pm). Female Swiss Webster mice (Charles River, O’Fallon, MO, USA) and male C57BL/6J mice (Jackson Laboratory, Ellsworth, ME, USA, 8–14 weeks old) were used in the behavioral experiments.
The DEREG (depletion of regulatory T cell) mice contain a transgenic allele that expresses the diphtheria toxin receptor-enhanced green fluorescent protein (DTR-EGFP) fusion protein under the control of the genomic sequences that regulate the expression of endogenous Foxp3.19 DEREG breeders (32050, Jackson Laboratory) were crossed with C57BL/6J mice to generate heterozygotes as a reporter line for Foxp3+ Treg cells.
Mouse Model of SNI
We used the modified SNI model because it is highly reproducible, easy to perform and relatively noninvasive in mice.20 Briefly, mice were anesthetized with 3–4% isoflurane in an induction chamber until losing the righting reflex. Anesthesia was maintained by 1.5–2% isoflurane through a nose cone. After skin and muscle incision to expose the right sciatic nerve in the thigh region, we tightly ligated two branches (sural and common peroneal nerves) with 7-0 silk suture (Braintree Scientific, Braintree, MA, USA) and transected ~2 mm of the nerve distal to the ligature.20 The third branch of the sciatic nerve (tibial nerve) was left intact, and caution was taken not to stretch or contact the spared nerve. In sham mice the right sciatic nerve and its branches were exposed but were not touched. After the surgery, the overlying muscle and skin layers were closed separately with 6-0 vicryl suture and interrupted nylon suture for the skin incision, respectively.
Behavioral Tests
Mice were extensively handled by the experimenters for 2 weeks and were well-habituated to the test room and the test apparatus before each experiment. The experimenters were blinded to the treatments mice received during data collection and analysis.
Response to Punctate Mechanical Stimuli on the Hindpaw
Mice were habituated in individual clear plexiglass boxes (11 x 11 x 15 cm) for 1–2 hours. A series of calibrated von Frey filaments were used to apply mechanical stimuli to the medial plantar surface of the hindpaw (the tibial nerve territory). We used the up-down paradigm to determine the 50% withdrawal threshold.21
Response to Dynamic Mechanical Stimuli on the Hindpaw
Mice were habituated in individual clear plexiglass boxes (11 x 11 x 15 cm) for 1–2 hours. The medial plantar surface of the hindpaw (tibial nerve territory) was stimulated by light stroking with a blunt paintbrush (5/0, Princeton Art & Brush Co., Princeton, NJ, USA) from heel to toe at ~2 cm/second. The behavior was scored as described previously.4 Score 0 indicates no evoked movement or lifting the stimulated paw for less than 1 second. Score 1 indicates a sustained lifting (more than 2 seconds) of the stimulated paw toward the body or a single gentle flinching of the stimulated paw. Score 2 indicates a strong lateral lift of the stimulated paw above the level of the body or a startle-like jump. Score 3 indicates multiple flinching or licking of the stimulated paw. Each paw was tested 3 times, with minimal interval of 3 minutes. The scores of 3 tests were averaged to indicate the response to dynamic mechanical stimuli.
Ld-IL2 Treatment
Recombinant mouse IL2 (carrier-free, Biolegend, San Diego, CA, USA) was freshly diluted from the stock (0.5–1 mg/mL aliquots at −80oC) every day. Each mouse received daily intraperitoneal (ip) injections of 1 µg IL2 in 100 µL saline at various durations. The control mice received daily ip injections of 100 µL saline. Note that on the days that the mouse behaviors were tested, IL2 was always injected after completing the behavioral tests. In the first experiment, female Swiss Webster mice were treated with daily ld-IL2 or saline from 5 days before SNI to 21 days post-surgery. In the second experiment, female Swiss Webster mice were treated with daily ld-IL2 or saline between day 7 and 28 post-surgery.
Isolation and Adoptive Transfer of Treg Cells
First, adult C57BL/6J mice (8–12 weeks old) received daily injections of ld-IL2 for 12 days, as Treg cells from IL2-treated mice have been shown to exhibit stronger suppressive activity compared with those from saline-treated mice.17,22 Next, CD25+CD4+ Treg cells and CD25−CD4+ cells were isolated from splenocytes through CD4+ T cell negative selection followed by a CD25+ T cell positive selection using EasySep mouse CD4+ T cell pre-enrichment and CD25 positive selection kits (18783, Stem Cell Technologies, Cambridge, MA, USA). Male C57BL/6J mice (8–14 weeks old) received 1 x 106 Treg or CD25−CD4+ cells via the tail vein 23 days post-SNI surgery. In a previous study, we have verified that the transferred cells contain 84% CD25+CD4+ cells and 76% Foxp3+CD25+CD4+ cells.18
Tissue Preparation, Immunohistochemistry, and Image Analysis
DEREG mice were euthanized with ip injection of barbiturate (200 mg/kg) and were transcardially perfused with warm 0.1 M phosphate-buffered saline (pH 7.2) followed by cold 4% formaldehyde in 0.1 M phosphate buffer (PB, pH 7.2) for fixation. Lumbar spinal cord, L4 DRGs, the proximal ends of the ligated sciatic nerves (approximately 2 mm length) as well as the contralateral uninjured nerves at the same level were dissected out, post-fixed for 4 hours, protected overnight at 4°C in 0.1 M PB with 30% sucrose and embedded in Optimal Cutting Temperature compound. DRG and nerves were sectioned at 15 µm in the transverse plane using a cryostat, collected on Superfrost Plus glass slides in sequence and stored at −20°C.
One in every 3 serial sections were used for individual immunohistochemistry experiments as described previously.18,23 T cells were identified by the rat anti-CD3 antibody (clone 17A2, 1:200, eBioscience, San Diego, CA, USA) and Treg cells were identified with the chicken anti-EGFP antibody (1:1000, AVES Lab, Tigard, OR, USA) in tissues from DEREG mice. AlexaFluor 568- or 488-conjugated secondary antibodies (Invitrogen, Carlsbad, CA, USA) were used at 1:1000 dilution. CD3+ T cells and EGFP+ Treg cells were quantified under a 40x objective on a Nikon TE2000S inverted epifluorescence microscope. All cells on individual sections were counted. Images of individual sections were captured by an Olympus NanoZoomer Whole-Slide Imaging System (Hamamatsu, Japan). The areas of individual sections were measured with the NDP.view2 software (Hamamatsu). The cell density in individual mice was calculated as total cell number divided by total sectional area. Representative images were adjusted for contrast and brightness using the same parameter within individual experiments. No other manipulations were made to the images. Image analysis was done with experimenters blinded to the experimental groups.
Statistical Analysis
For behavioral experiments, power analysis was conducted to estimate sample size with > 80% power to show an effect size of 0.8, alpha (two-sided) of 0.05, and a simple covariance structure for repeated measures. For immunohistochemistry, sample size was estimated based on our previous experience.
All data were reported as mean ± standard error of the mean. The Shapiro–Wilk test was used to check data normality. Statistical significance between or within experimental groups was assessed by two-tailed t-test, ANOVA (analysis of variance, one-way or two-way, with or without repeated-measures [RM]) with multiple comparisons (Student-Newman-Keuls method, post hoc Dunnett’s test or t-test with Bonferroni correction) where appropriate, using Origin (OriginLab, Northampton, MA, USA) and Statistica (StatSoft, Tulsa, OK, USA) software. Differences with p < 0.05 were considered statistically significant. The statistical analysis for individual experiments was described in figure legends.
Results
Ld-IL2 Pretreatment Attenuates SNI-Induced Punctate Allodynia and Prevents the Development of Dynamic Allodynia
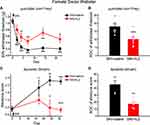
To model neuropathic pain, we ligated and transected two branches of the right sciatic nerve (sural and common peroneal nerves) in female Swiss Webster mice and measured the responses to punctate (von Frey filaments) and dynamic (paintbrush) mechanical stimuli on the medial plantar surface of the right hindpaw, the territory of the spared tibial nerve. SNI resulted in a significant decrease in mechanical threshold to punctate stimuli between 2 and 22 days post-SNI (Figure 1A, SNI+saline group), consistent with the previous study.20 The right paws also exhibited dynamic allodynia to brush stimuli that lasted at least till 31 days post-SNI (Figure 1B, SNI+saline group), in line with the previous report.4
Daily administration of ld-IL2 for up to 8 weeks has been shown to selectively boost Treg cells in patients with many self-reactive T cells.24 Here, we treated female Swiss Webster mice with daily ld-IL2 (1 µg/mouse/day, ip) or saline, starting 5 days before SNI and continuing till 21 days post-surgery.18 Basal punctate and dynamic mechanical sensitivity was not altered by 5 days of ld-IL2 injections (Figure 1, SNI+saline versus SNI+IL2, Day 0). In saline-treated mice, the withdrawal threshold to von Frey filaments decreased to about 20% of the baseline value between 4–22 days post-SNI. In IL2-treated mice, SNI caused an average of 50% reduction of the withdrawal threshold (Figure 1A). The integrated area over the time-effect curves (AOCs) calculated from day 0 baseline to day 22 post-SNI showed that ld-IL2 pretreatment significantly attenuated the magnitude of nerve injury-induced punctate allodynia (Figure 1B). In addition to punctate allodynia, SNI induced dynamic allodynia to brush stimuli in saline-treated mice but not in mice that received ld-IL2 pretreatment (Figure 1C). Both the time course and AOC of dynamic allodynia indicated that ld-IL2 pretreatment prevents the development of nerve injury-induced hyper-sensitivity to brush stimuli (Figure 1C and D).
SNI-Induced Punctate and Dynamic Mechanical Allodynia are Reversed by Ld-IL2 Treatment
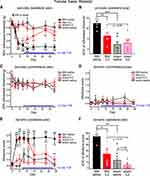
We went on to test whether delayed ld-IL2 treatment reverses SNI-induced punctate or dynamic allodynia. We started the 3-week ld-IL2 treatment 7 days post-SNI, at which time the punctate allodynia was well established on the ipsilateral (right) paws (Figure 2A, SNI+saline and SNI+IL2 groups). SNI-induced punctate allodynia persisted for at least 28 days without treatment (Figure 2A, SNI+saline group). The mechanical threshold started to reverse after 3 days of ld-IL2 treatment (Figure 2A, SNI+IL2 group, day 10). After 7 days of IL2 treatment, the mechanical threshold of mice in the SNI+IL2 group was not different from that of mice in the sham+IL2 group (Figure 2A, day 14–28). The integrated AOC of withdrawal threshold between day 10 and 28 post-SNI in the SNI+IL2 group was significantly lower than that of the SNI+saline group but was comparable to that of the sham+IL2 group (Figure 2B). Notably, ld-IL2 treatment did not change the mechanical threshold of the contralateral (left) paw in mice underwent SNI surgery (Figure 2C, SNI+IL2 group), nor did it affect the paw withdrawal threshold in mice that received sham surgery (Figure 2A–C, sham+saline versus sham+IL2 group).
Similar to punctate allodynia, the dynamic allodynia was fully established on the ipsilateral but not contralateral paws 3 days post-SNI and persisted for at least 29 days without treatment (Figure 2D and E, SNI+saline group). After 7 days of IL2 treatment, the dynamic allodynia was completely reversed and stayed at basal level with continuous IL2 treatment (Figure 2E, day 15–29). The integrated AOC of dynamic allodynia score between day 10 and 28 post-SNI in the SNI+IL2 group was significantly lower than that of the SNI+saline group but was comparable to that of the sham+IL2 group (Figure 2F). Ld-IL2 treatment did not change the responses to brush stimuli of the contralateral paw in mice underwent SNI surgery (Figure 2D, SNI+IL2 group), or of the paws in mice that received sham surgery (Figure 2D–F, sham+saline versus sham+IL2 group). Taken together, we conclude that delayed ld-IL2 treatment substantially attenuates SNI-induced punctate and dynamic allodynia without compromising the baseline responses to punctate or stroking stimuli.
Adoptive Transfer of Treg Cells Reverses the Morphine-Resistant Punctate and Dynamic Mechanical Allodynia
Previous studies indicate that morphine attenuates nerve injury-induced punctate allodynia at early (< 2 weeks post-injury) but not late stage;4–6 whereas neither early- nor late-stage nerve injury-induced dynamic allodynia responds to morphine treatment.4,6 Our data showed that starting ld-IL2 treatment 7 days post-SNI reverses both punctate and dynamic allodynia (Figure 2). Here, we investigated whether enhancing Treg cell number/function is still beneficial in mice after both punctate and dynamic allodynia become morphine-resistant. We enriched CD25+CD4+ Treg cells from IL2-treated donor mice and adoptively transferred the cells (1 x 106 cells/mouse) to male C57BL/6J mice 23 days post-SNI, at which time both punctate and dynamic allodynia become morphine-resistant.4,5 The control group received adoptive transfer of CD25−CD4+ cells (1 x 106 cells/mouse) enriched from IL2-treated donor mice.
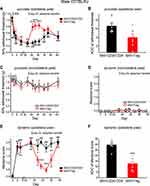
Before the adoptive transfer, all mice exhibited profound punctate allodynia on the paws ipsilateral to the SNI surgery (Figure 3A). In control mice that received CD25−CD4+ cells, the punctate allodynia persisted throughout the experiment (Figure 3A, SNI+CD25−CD4+ group, 26–46 days post-SNI). In mice that received Treg cells, the mechanical threshold was completely reversed 3 days after the transfer (Figure 3A, SNI+Treg group, day 26). The effect of transferred Treg cells lasted for 12 days (Figure 3A, SNI+Treg group, day 26–38), and the punctate allodynia recurred 23 days after Treg transfer (Figure 3A, SNI+Treg group, day 46). The integrated AOC of withdrawal threshold between day 26 and 45 post-SNI showed that transfer of Treg cells significantly attenuated the magnitude of nerve injury-induced punctate allodynia (Figure 3B). Similar to the ld-IL2 treatment, Treg cell transfer did not change the responses to punctate or dynamic mechanical stimuli thresholds of the contralateral paws in mice underwent SNI surgery (Figure 3C and D).
SNI also resulted in significant dynamic allodynia on the ipsilateral paws of all mice (Figure 3E). In control mice that received CD25−CD4+ cells, the dynamic allodynia persisted throughout the experiment (Figure 3E, SNI+CD25−CD4+ group, 27–39 days post-SNI). In mice that received Treg cells, the dynamic allodynia was partially reversed 4 days after the transfer (Figure 3E, SNI+Treg group, day 27), and was completely reversed 2 days later (Figure 3E, SNI+Treg group, day 29). The effect of Treg transfer lasted for 9 days (Figure 3E, SNI+Treg group, day 27–35), and the dynamic allodynia recurred 16 days after Treg transfer (Figure 3E, SNI+Treg group, day 39). The integrated AOC of dynamic allodynia score between day 27 and 39 post-SNI in the SNI+Treg group was significantly lower than that of the control group (Figure 3F). Collectively, these results suggest that enhancing Treg cell number/function can reverse the morphine-resistant punctate and dynamic allodynia long after the nerve injury.
The effects of ld-IL2 and Treg cell transfer on SNI-induced punctate and dynamic allodynia was summarized in Table 1.
 |
Table 1 The Effects of Ld-IL2 and Treg Cell Treatments on SNI-Induced Punctate and Dynamic Allodynia |
Ld-IL2 Treatment Preferentially Increases the Ratio of Treg Cells to Total CD3+ T Cells in the Injured Sciatic Nerve
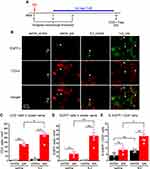
To elucidate the site of action of ld-IL2, we examined the distribution of T cells, especially the Treg cells, in the sciatic nerves and the lumbar L4 DRG of female DEREG transgenic mice that underwent unilateral SNI and received ld-IL2 or saline treatment between day 7 and 29 post-SNI (Figure 4A). DEREG mice selectively express the DTR-EGFP fusion protein in Foxp3+ cells,19 allowing us to identify Treg cells with the EGFP signal. Similar to what we found in wild-type mice, SNI significantly reduced the hindpaw mechanical threshold in DEREG mice (from 1.02 ± 0.09 g at baseline to 0.28 ± 0.03 g at day 7 post-SNI, n = 6; p < 0.001, one-way ANOVA with post hoc Dunnett’s test). Ld-IL2 treatment starting on day 7 post-SNI reversed the threshold to 0.69 ± 0.10 g by day 14 (p < 0.01, day 7 versus day 14 post-SNI, one-way ANOVA with post hoc Dunnett’s test).
Thirty days after the unilateral SNI surgery, we stained the tissues with the CD3 and EGFP antibodies to identify total CD3+ T cells and EGFP+ Treg cells, respectively. Consistent with previous reports,25–27 we could identify very few CD3+ cells (0 ~ 0.7 ± 0.2 cells/section) or EGFP+ cells (0 ~ 0.05 ± 0.05 cells/section) in the dorsal horn of the spinal cord either ipsilateral or contralateral to SNI, suggesting no SNI-induced lymphocyte infiltration to the dorsal horn at this time point. Similarly, uninjured sciatic nerves contained very few CD3+ T cells or EGFP+ Treg cells (Figure 4B–D, saline_contra groups). The densities of total T cells and Treg cells were dramatically increased in the injured sciatic nerves (Figure 4B–D, saline_ipsi groups), but the fraction of Treg cells among total T cells was not altered by SNI (Figure 4E, saline_contra versus saline _ipsi, p = 0.32). Ld-IL2 treatment resulted in an additional 3.6-fold increase in Treg cell density in the injured nerves (Figure 4D, IL2_ipsi versus saline_ipsi) but the density of total CD3+ T cells only increased 1.5-fold (Figure 4C, IL2_ipsi versus saline_ipsi). Consequently, the fraction of Treg cells among total T cells increased more than 2-fold (Figure 4E, IL2_ipsi versus saline_ipsi). Notably, ld-IL2 did not increase the numbers of Treg cells or CD3+ T cells in uninjured nerves (Figure 4B–D, IL2_contra versus saline_contra), neither did it alter the fraction of Treg cells among total CD3+ T cells (Figure 4E, IL2_contra versus saline_contra, p = 0.15).
In contrast to what we observed in the sciatic nerves, nerve injury did not affect the densities of CD3+ T cells and EGFP+ Treg cells in L4 DRG 30 days after SNI (Figure 5A–C, saline_ipsi versus saline_contra). The numbers of Treg cells and total T cells were increased after ld-IL2 in L4 DRG both ipsilateral and contralateral to the SNI (Figure 5A–C, saline versus IL2 groups), but the fraction of Treg cells among total T cells was not altered by either SNI or ld-IL2 (Figure 5D). Collectively, these results suggest that ld-IL2 treatment preferentially enhances Treg-mediated suppression in the injured nerves by increasing the ratio of Treg cells to total CD3+ T cells.
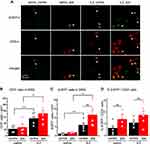 |
Figure 5 Neither SNI nor low-dose interleukin-2 alters the ratio of Treg cells to total CD3+ T cells in DRG. (A) Representative images of EGFP+ and CD3+and cells in L4 DRG ipsilateral (ipsi) and contralateral (contra) to the SNI surgery in female DEREG mice at day 30 post-SNI. (B and C) The densities of CD3+ T cells (B) and EGFP+ Treg cells (C) in the ipsilateral and contralateral L4 DRG of female DEREG mice at day 30 post-SNI (same mice as in Figure 4C–E). *p < 0.05, **p < 0.01, two-way ANOVA and post hoc t-test with Bonferroni correction. (D) The fraction of EGFP+ Treg cells among CD3+ T cells in L4 DRG ipsilateral and contralateral to the SNI surgery (same mice as in Figure 4C-E), respectively. p = 0.76, two-way ANOVA. Abbreviations: SNI, spared nerve injury; Treg, regulatory T cell; DRG, dorsal root ganglion; EGFP, enhanced green fluorescent protein; ANOVA, analysis of variance. |
Discussion
Dynamic mechanical allodynia, the pain evoked by light brushing or stroking of the skin, is one of the most disabling symptoms of neuropathic pain.3,28 Despite the recent progress in elucidating the underlying mechanisms,4,29,30 effective treatment of dynamic allodynia remains elusive.2,7 Many preclinical and clinical studies have identified ld-IL2 treatment as a safe and effective treatment for multiple autoimmune diseases, neurodegenerative diseases as well as chronic headache disorders through enhancing Treg-mediated immunosuppression.15–18 In the present study, we report for the first time that the development of SNI-induced dynamic mechanical allodynia was completely blocked by the ld-IL2 treatment starting prior to the sciatic nerve injury, suggesting that the level of endogenous Treg cells may be one of the factors that determines which individual is at the risk of developing dynamic allodynia after peripheral nerve injury. On the contrary, mice that received ld-IL2 pretreatment still developed punctate mechanical allodynia after SNI, but the magnitude was significantly reduced. This, along with the results from previous work,9–14 implicates the role of endogenous Treg cells in determining the severity of peripheral nerve injury-induced punctate allodynia.
A recent study shows that treating mice with IL2 one day after nerve injury reduces punctate allodynia up to 10 days post-injury,10 at which point it is still responsive to morphine treatment.4–6 Here, we tested the therapeutic use of ld-IL2 in female Swiss Webster mice one week post-SNI, after the punctate and dynamic allodynia were fully established. Both the morphine-sensitive, early punctate allodynia (Figure 2A, day 10 and 14) and the morphine-resistant, late punctate allodynia (Figure 2A, day 22 and 28) were significantly attenuated by the delayed ld-IL2 treatment. Unlike punctate allodynia, nerve injury-induced dynamic allodynia is resistant to morphine treatment at all stages,4,6 but is substantially reduced by the delayed ld-IL2 treatment. Similarly, adoptive transfer of Treg cells to male C57BL/6J mice 3 weeks post-SNI effectively reversed the punctate and dynamic allodynia that are resistant to morphine treatment, supporting that the effect of ld-IL2 is mediated through endogenous Treg cells, and is likely independent of mouse strain and sex. Together, these results expanded the therapeutic values of ld-IL2 and Treg cells to the morphine-resistant punctate and dynamic mechanical allodynia long after the initial nerve injury.
To identify the site of action of ld-IL2 and Treg cells, we quantified the densities of Treg cells and total CD3+ T cells in the sciatic nerves and the lumbar L4 DRG of female DEREG mice. In agreement with previous work,25–27 we found few, if any, T cells in the dorsal horn of the spinal cord after either SNI and/or ld-IL2 treatment, indicating that ld-IL2 and Treg cells act at peripheral sites to attenuate mechanical allodynia. SNI preferentially increased the density of T cells, including Treg cells, in the injured sciatic nerves without altering the fraction of Treg among total T cells, suggesting that SNI-induced Treg increase is not sufficient to counteract SNI-induced increase in effector T cells. Although ld-IL2 led to the infiltration of both Treg cells and total CD3+ T cells in the injured nerves and DRG, it preferentially increased the ratio of Treg cells to total CD3+ T cells in the injured nerves. These results implicate that ld-IL2 and Treg cells attenuate punctate and dynamic allodynia through enhancing immunosuppression at the site of nerve injury. A recent study indicates that Treg cells attenuate punctate mechanical allodynia through interleukin-10-mediated inhibition of the Th1 response at the site of peripheral nerve injury.10 Future in-depth studies is warranted to determine whether ld-IL2 and Treg cells reverse nerve injury-induced dynamic mechanical allodynia through similar or different molecular mechanisms.
In mouse model of chronic migraine, ld-IL2 preferentially expands and activates Treg cells without altering the frequencies of effector T cells.18 In the SNI model, the same dose of IL2 increased the infiltration of effector T cells in addition to Treg cells in the injured nerves and DRG. This likely results from SNI-induced activation of peripheral effector T cells, which start to express the high-affinity IL2 receptor CD25 and become responsive to ld-IL2.8,13 More studies are needed to develop methodology that selectively enhances Treg cell functions while sparing activated effector T cells and to test its therapeutic effects on neuropathic pain. In addition, the sample size in behavioral tests was rather small, and only one peripheral nerve injury model was used in this work. Since we did not observe consistent thermal allodynia in this model, whether ld-IL2 and Treg cell treatment attenuates nerve injury-induced thermal/cold hyper-sensitivity was not investigated. Another limitation is that male and female mice were not tested in parallel in individual assays. Future studies using multiple neuropathic pain models and testing male and female mice in parallel with large sample sizes would be useful to validate the therapeutic values of ld-IL2 and Treg cell treatments. The dose of IL2 used in this study has been shown to result in a 2-fold increase in Treg cell frequency in the peripheral blood,18 similar to the magnitude of ld-IL2-induced Treg expansion reported in human clinical trials.16,31 Both ld-IL2 and Treg cell transfer are well tolerated in patients and show indications of clinical efficacy against multiple autoimmune diseases.16,31,32 Importantly, neither daily ld-IL2 nor Treg transfer altered the basal nociceptive responses to punctate or stroking stimuli on the hindpaw innervated by the uninjured sciatic nerve. This supports further translational and clinical studies to assess whether ld-IL2 can be used to treat both punctate and dynamic mechanical allodynia with minimal adverse effects.
Conclusion
Collectively, the present study identifies Treg cell as a promising target and ld-IL2 as a potential therapy for nerve injury-induced persistent punctate and dynamic mechanical allodynia that are highly debilitating and are largely unresponsive to available treatments.
Abbreviations
DEREG, depletion of regulatory T cell mice; DRG, dorsal root ganglia; DTR-EGFP, diphtheria toxin receptor-enhanced green fluorescent protein; ld-IL2, low-dose interleukin-2; ip, intraperitoneal; PB, phosphate buffer; RM ANOVA, repeated measures analysis of variance; SNI, spared nerve injury; Treg, regulatory T cells.
Data Sharing Statement
All data are available upon reasonable request.
Acknowledgments
The authors thank members of Cao lab for valuable comments on the manuscript. This work was supported by the National Institute of Health (grant number NS103350-02S1, to YQC) and the Xiangya Scholarship Fund (to RH). The sponsors were not involved in any of the stages from study design to submission of the paper for publication.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
Dr Yu-Qing Cao reports a patent Compositions and methods for the treatment of headache disorders and neuropathic pain pending. The authors report no other conflicts of interest in this work.
References
1. van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain. 2014;155(4):654–662. doi:10.1016/j.pain.2013.11.013
2. McNicol ED, Midbari A, Eisenberg E. Opioids for neuropathic pain. Cochrane Database Syst Rev. 2013;8:CD006146.
3. Truini A, Garcia-Larrea L, Cruccu G. Reappraising neuropathic pain in humans–how symptoms help disclose mechanisms. Nat Rev Neurol. 2013;9(10):572–582. doi:10.1038/nrneurol.2013.180
4. Cheng L, Duan B, Huang T, et al. Identification of spinal circuits involved in touch-evoked dynamic mechanical pain. Nat Neurosci. 2017;20(6):804–814. doi:10.1038/nn.4549
5. Due MR, Yang XF, Allette YM, et al. Carbamazepine potentiates the effectiveness of morphine in a rodent model of neuropathic pain. PLoS One. 2014;9(9):e107399. doi:10.1371/journal.pone.0107399
6. Field MJ, Bramwell S, Hughes J, Singh L. Detection of static and dynamic components of mechanical allodynia in rat models of neuropathic pain: are they signalled by distinct primary sensory neurones? Pain. 1999;83(2):303–311. doi:10.1016/S0304-3959(99)00111-6
7. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162–173. doi:10.1016/S1474-4422(14)70251-0
8. Schmidt A, Oberle N, Krammer PH. Molecular mechanisms of treg-mediated T cell suppression. Front Immunol. 2012;3:51. doi:10.3389/fimmu.2012.00051
9. Fischer R, Sendetski M, Del Rivero T, et al. TNFR2 promotes Treg-mediated recovery from neuropathic pain across sexes. Proc Natl Acad Sci U S A. 2019;116(34):17045–17050. doi:10.1073/pnas.1902091116
10. Davoli-Ferreira M, de Lima KA, Fonseca MM, et al. Regulatory T cells counteract neuropathic pain through inhibition of the Th1 response at the site of peripheral nerve injury. Pain. 2020;161(8):1730–1743. doi:10.1097/j.pain.0000000000001879
11. Lees JG, Duffy SS, Perera CJ, Moalem-Taylor G. Depletion of Foxp3+ regulatory T cells increases severity of mechanical allodynia and significantly alters systemic cytokine levels following peripheral nerve injury. Cytokine. 2015;71(2):207–214. doi:10.1016/j.cyto.2014.10.028
12. Austin PJ, Kim CF, Perera CJ, Moalem-Taylor G. Regulatory T cells attenuate neuropathic pain following peripheral nerve injury and experimental autoimmune neuritis. Pain. 2012;153(9):1916–1931. doi:10.1016/j.pain.2012.06.005
13. Laumet G, Ma J, Robison AJ, Kumari S, Heijnen CJ, Kavelaars A. T cells as an emerging target for chronic pain therapy. Front Mol Neurosci. 2019;12:216. doi:10.3389/fnmol.2019.00216
14. Liu XJ, Zhang Y, Liu T, et al. Nociceptive neurons regulate innate and adaptive immunity and neuropathic pain through MyD88 adapter. Cell Res. 2014;24(11):1374–1377. doi:10.1038/cr.2014.106
15. Dansokho C, Ait Ahmed D, Aid S, et al. Regulatory T cells delay disease progression in Alzheimer-like pathology. Brain. 2016;139(Pt 4):1237–1251. doi:10.1093/brain/awv408
16. Klatzmann D, Abbas AK. The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat Rev Immunol. 2015;15(5):283–294. doi:10.1038/nri3823
17. Zhang H, Xia Y, Ye Q, et al. In vivo expansion of regulatory T cells with IL-2/IL-2 antibody complex protects against transient ischemic stroke. J Neurosci. 2018;38(47):10168–10179. doi:10.1523/JNEUROSCI.3411-17.2018
18. Zhang J, Czerpaniak K, Huang L, et al. Low-dose interleukin-2 reverses behavioral sensitization in multiple mouse models of headache disorders. Pain. 2020;161(6):1381–1398. doi:10.1097/j.pain.0000000000001818
19. Lahl K, Loddenkemper C, Drouin C, et al. Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J Exp Med. 2007;204(1):57–63. doi:10.1084/jem.20061852
20. Shields SD, Eckert WA
21. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994;53(1):55–63. doi:10.1016/0165-0270(94)90144-9
22. Tahvildari M, Omoto M, Chen Y, et al. In vivo expansion of regulatory T cells by low-dose interleukin-2 treatment increases allograft survival in corneal transplantation. Transplantation. 2016;100(3):525–532. doi:10.1097/TP.0000000000001044
23. Huang D, Ren L, Qiu CS, et al. Characterization of a mouse model of headache. Pain. 2016;157(8):1744–1760. doi:10.1097/j.pain.0000000000000578
24. Koreth J, Matsuoka K, Kim HT, et al. Interleukin-2 and regulatory T cells in graft-versus-host disease. N Engl J Med. 2011;365(22):2055–2066. doi:10.1056/NEJMoa1108188
25. Gattlen C, Clarke CB, Piller N, et al. Spinal cord T-cell infiltration in the rat spared nerve injury model: a Time Course Study. Int J Mol Sci. 2016;17(3):352. doi:10.3390/ijms17030352
26. Kim CF, Moalem-Taylor G. Detailed characterization of neuro-immune responses following neuropathic injury in mice. Brain Res. 2011;1405:95–108. doi:10.1016/j.brainres.2011.06.022
27. Lopes DM, Malek N, Edye M, et al. Sex differences in peripheral not central immune responses to pain-inducing injury. Sci Rep. 2017;7(1):16460. doi:10.1038/s41598-017-16664-z
28. Jensen TS, Finnerup NB. Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurol. 2014;13(9):924–935. doi:10.1016/S1474-4422(14)70102-4
29. Duan B, Cheng L, Bourane S, et al. Identification of spinal circuits transmitting and gating mechanical pain. Cell. 2014;159(6):1417–1432. doi:10.1016/j.cell.2014.11.003
30. La JH, Chung JM. Peripheral afferents and spinal inhibitory system in dynamic and static mechanical allodynia. Pain. 2017;158(12):2285–2289. doi:10.1097/j.pain.0000000000001055
31. Rosenzwajg M, Lorenzon R, Cacoub P, et al. Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann Rheum Dis. 2019;78(2):209–217. doi:10.1136/annrheumdis-2018-214229
32. Sharabi A, Tsokos MG, Ding Y, Malek TR, Klatzmann D, Tsokos GC. Regulatory T cells in the treatment of disease. Nat Rev Drug Discov. 2018.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.