Back to Journals » Clinical Ophthalmology » Volume 12
Long-term outcomes of intended undercorrection intraocular lens implantation in pediatric cataract
Authors Lekskul A , Chuephanich P, Charoenkijkajorn C
Received 1 June 2018
Accepted for publication 15 August 2018
Published 2 October 2018 Volume 2018:12 Pages 1905—1911
DOI https://doi.org/10.2147/OPTH.S176057
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Apatsa Lekskul, Pichaya Chuephanich, Chao Charoenkijkajorn
Department of Ophthalmology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Purpose: To evaluate the postoperative refraction of intended undercorrection after intraocular lens (IOL) implantation in pediatric cataract patients.
Design: A cross-sectional study (data collected by retrospective chart review).
Patients and methods: The medical records of children aged under 10 years, who underwent cataract surgery with IOL implantation at the Ramathibodi Hospital between January 2000 and May 2018, were reviewed. IOL power calculations were 30%, 25%, 20%, 15% and 10% undercorrected if children were aged 6–12, 13–24, 25–36, 37–48 and 49–60 months, respectively. Two diopters (D) undercorrection was used in children aged between 5 and 8 years and one diopter undercorrection was used in children aged between 8 and 10 years. The main outcome measure was the postoperative refractive errors at the last follow-up visit.
Results: In total, 50 children (21 females and 29 males, 16 unilateral and 34 bilateral, 84 eyes) met the inclusion criteria for this study. Mean age at the time of surgery was 77.82±31.24 months. Mean follow-up time was 56.56±45.83 months. The main outcome in this study was the postoperative refractive error in children aged 7 years or more. We found 74 eyes of 44 children who were aged 7 years or more at last follow-up visit. In total, 45 eyes were myopic (-0.25 to -8.25 D) with a mean refraction of -2.26±2.16 D. A further 21 eyes were hyperopic (+0.25 to +3.25 D), with a mean refraction of +1.05±0.79 D and eight eyes were emmetropic or having only astigmatism.
Conclusion: The major postoperative refractive error at the last follow-up time was myopia. We have to adjust the IOL calculation formula to specify more undercorrection, with the aim of achieving more optimal refractive outcomes in adulthood.
Keywords: pseudophakic, refraction, formula, myopia, emmetropization
Introduction
Pediatric cataract is one of the leading causes of childhood blindness worldwide, and it can lead to the development of amblyopia, especially in the developing countries. In visually significant cataract, surgery must be performed as soon as possible to remove the lens opacity, and effective correction of aphakia is the keys to successful visual rehabilitation. Primary intraocular lens (IOL) implantation is an increasingly common procedure and is currently the preferred means to correct the optical errors in children undergoing cataract surgery.1 However, there is no consensus regarding the optimal initial postoperative refractive goal for children undergoing IOL implantation. Even with recent advances in surgical technique, instrumentation and implant design, one major challenge in pediatric cataract surgery is to obtain the desired long-term postoperative refractive status.2
Ocular growth is an important factor that complicates IOL power determination. As the eyes grow, the axial length increases rapidly in the first 2 years of life then slowly tapering and stabilizing at 7–10 years of age.3 Additionally, the cornea flattens and lens power decreases. These processes are responsible for the overall change in refractive status as the eye grows, causing the eye to become more myopic. This process is known as emmetropization. In children, implantation of an IOL at emmetropic power reveals significant myopia at ocular maturity, as the average calculated lens power decreases around 20 diopters (D) from birth to adulthood.4 For this reason, many pediatric ophthalmologists recommend initial undercorrection to avoid long-term myopic shift.
Despite many suggested guidelines, there is no consensus on what degree of undercorrection should be used in different age groups. Dahan and Drusedau5 proposed that undercorrection of IOL power which is 20% less than the emmetropic IOL power for children aged less than 2 years and 10% less for children aged more than 2 years. Enyedi et al4 proposed to target a postoperative refraction of +6 D in a 1 year-old, +5 D for a 2 year-old, +4 D for a 3-year old, +3 D for a 4 year-old, +2 D for a 5 year-old, +1 D for a 6 year-old, Plano lens for a 7-year-old and −1 to −2 D for children older than 8 years of age.
Based on our guidelines, the undercorrected IOL power was calculated by anticipating the expected myopic shift. The following IOL corrections were applied to each age group: undercorrection of 30% in children aged 6 months to 1 year, 25% less in children aged 1–2 years, 20% less in children aged 2–3 years, 15% less in children aged 3–4 years, 10% less in children aged 4–5 years, 2 D undercorrection in children aged 5–8 years, and 1 D undercorrection in children aged 8–10 years. This IOL calculation guideline was applied, which was used for over 10 years, from the data obtained from Gordon and Donzis that showed the refractive status, axial length, and keratometric changes in normal eyes in different age groups from birth until adulthood.3 Gordon and Donzis also proposed that suggested IOL powers calculated using the Sanders-Retzlaff-Kraff (SRK) formula that utilizes the axial length and keratometric data from each age group. This formula was applied to compare the suggested IOL powers for children and adults and adjusted the percentage for more comprehensive usage in clinical practice. For example, in children aged 1–2 years, the data suggested a mean IOL power of 25.1 D, whereas the mean IOL power of a normal adult eye in this study was 18.8 D. It was assumed that if the IOL power was undercorrected by 25% (25.1×0.75=18.8), the desired adult IOL power (18.8 D) will be obtained and this will result in emmetropia as the children reach adulthood.
A retrospective analysis was performed to evaluate the postoperative refraction after intended undercorrection for IOL implantation in pediatric cataract patients. The undercorrection formula was evaluated in this study to determine if it can obtain the desired refractive power as the children age.
Patients and methods
Patient recruitment
After obtaining approval from the Institutional Review Board, a retrospective noncomparative study was performed. The medical records of children aged under 10 years, who underwent cataract surgery and IOL implantation in the Ramathibodi Hospital between January 2000 and May 2018, were reviewed. The children who were free of any other ocular abnormality were included in the study. Also, children who underwent lens aspiration with primary IOL implantation, posterior capsulotomy, and anterior vitrectomy were included in the study. The IOL was placed in a capsular bag in all patients. Preoperative axial length was measured by immersion ultrasound A-scan and the SRK/T formula was used to calculate the IOL power in all patients. Patients with other ocular abnormalities, including: retinal diseases that might limit the visual potential of the eye; retinal detachment; persistent fetal vasculature; glaucoma, optic nerve diseases, corneal abnormalities, active uveitis or signs suggestive of a previous episode of uveitis, history of laser treatment or cryotherapy for retinopathy of prematurity, those with surgical complications, IOL placement in sulcus, those who did not come for a follow-up were excluded from the study. The undercorrected IOL powers were calculated by anticipating the expected myopic shift.
Data collection
The data collected included baseline characteristics, surgical procedures, surgical outcomes (visual outcomes, perioperative and postoperative complications), postoperative refraction, and best corrected visual acuity (BCVA).
Outcome measures
The primary outcome measure was the postoperative refractive errors in pseudophakic eyes at the last follow-up visit in children aged over 7 years. It is believed that after 7 years of age the refractive status will become more stable. According to data from the study by Gwiazda et al,6 children reach an emmetropic plateau between the age of 5 and 7 years, upon which there is no significant change in refractive status. The follow-up data in this study were obtained postoperatively at 1 month (±2 weeks), 6 months, 1 year and each year following. The follow-up data included postoperative refraction and BCVA.
Results are presented as mean and variations as SD. The refractive errors were calculated as the spherical power in D. In this study, we excluded cylindrical power as the change in astigmatic power appeared to be unrelated to emmetropization of spherical errors.7
Ethics statement
Approval for this study was obtained from the Institutional Review Board of the Faculty of Medicine at Ramathibodi Hospital, Mahidol University and was conducted according to the tenets of the Declaration of Helsinki. The Institutional Review Board did not require patient consent to review their medical records as this was a retrospective study and because each patient could not be identified from their medical records.
Results
A total of 50 children (21 females and 29 males, 16 unilateral and 34 bilateral; 84 eyes) met the inclusion criteria. Mean age at the time of surgery was 77.82±31.24 months (range 4–131 months). Mean follow-up time was 56.56±45.83 months (range 1–180 months) as shown in Table 1. The age distribution of the children aged 7 years or more at the last follow-up visit is shown in Figure 1. The results of the final postoperative refractive errors were as follows: 45 eyes (60.8%) were myopic (−0.25 to −8.25 D), with a mean refractive error of −2.26±2.16 D; 21 eyes (28.3%) were hyperopic (+0.25 to +3.25 D), with a mean refractive error of +1.05±0.79 D; eight eyes (10.8%) were emmetropic or having only astigmatism. It was also found that 47 (63.5%) eyes achieved a final refractive error of ±1 D and 60 (81.0%) eyes achieved refractive error of ±2 D (Figure 2).
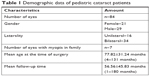  | Table 1 Demographic data of pediatric cataract patients |
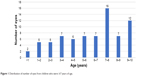  | Figure 1 Distribution of number of eyes from children who were ≥7 years of age. |
  | Figure 2 Distribution of the final refractive status in children >7 years of age who underwent cataract surgery with intended undercorrection of IOL power. |
The majority of the children in this study (85%) achieved a BCVA better than 20/40 postoperatively, whereas only 8% of children had a BCVA between 20/50 and 20/70 (Figure 3). Four (7%) children had a BCVA worse than 20/100, all of which had a unilateral cataract before surgery and the lower BCVA was thought to be due to amblyopia. The mean postoperative final refraction from each age group was as follows: −3.00±1.41 D in children aged less than 1 year, −2.50±2.08 D in children aged 1–2 years, −2.31±2.19 D in children aged 2–3 years, −1.66±1.53 D in children aged 3–4 years, −2.25±2.91 D in children aged 4–5 years, −0.37±2.88 D in children aged 5–6 years, −0.45±0.64 D in children aged 8–9 years, and −0.30±1.11 D in children aged 9–10 years (Figure 4). The comparison of postoperative initial spherical power and final spherical power from 61 eyes is shown in Figure 5 (the data from the 13 remaining eyes was not available) to demonstrate the refractive changes that occur overtime postoperatively.
  | Figure 3 Distribution of postoperative best corrected visual acuity. |
  | Figure 4 Distribution of mean final refraction according to age at the time of surgery. |
  | Figure 5 Postoperative initial spherical power compared with final spherical power in each study participant. |
Discussion
The guideline used in this study were derived from the study of axial length, keratometry, and suggested IOL power proposed by Gordon and Donzis.3 This study aimed for more undercorrection than the guidelines suggested by Enyedi et al4 and Dahan and Drusedau.5 A justification for selecting the guidelines proposed by Gordon and Donzis3 was that Asian children are more likely to develop myopia later in life than other populations.8
As this approach may raise concerns about residual hypermetropia that could exacerbate refractive amblyopia in children, especially in unilateral cases that might not wear postoperative corrective spectacles or contact lens. In our practice, it was found that most children can tolerate glasses well, even with large differences in the refractive power between the eyes. Progression of amblyopia following insertion of undercorrected IOL power was also rare. Furthermore, when the eyes continue to grow and approach emmetropia, binocular vision and stereopsis often improve.9
In our study, we found that 36 of the 51 (70.6%) eyes from children aged 7 years or older had myopia (−0.25 to −8.25 D), with a mean refractive error of −2.47±2.34 D. Thirteen (25.5%) eyes were hyperopic with a mean refractive error of +1.1±0.88 D and two (3.9%) eyes were emmetropic. One possible explanation could be related to the high prevalence of myopia among Asian populations. More studies are warranted to determine the appropriate adjustment of IOL power in Asian children, so that the dependency on spectacles, contact lenses or refractive surgery can be reduced when these pseudophakic children become older.
Postoperatively, 28 (54.9%) eyes achieved refraction within ±1 D and 68.6% within ±2 D (Figure 2). Although about half of the eyes achieved an acceptable final refractive status, the majority of these eyes were myopic. Fortunately, 85% of children managed to achieve a postoperative BCVA better than 20/40, with only 7% having a BCVA worse than 20/100. The major cause of a BCVA worse than 20/100 was deprivation amblyopia. This emphasizes that amblyopia management is an important part in managing pediatric cataract.
It was also observed that the patient’s age at the time of surgery played a role in the determination of the postoperative refractive status. It was found more postoperative myopia in children who underwent cataract surgery at a younger age and children who were operated on at the aged of 5 years or more exhibited lower refractive errors (Figure 4). For example, the mean final refractive error was −2.50±2.08 D in children aged 1–2 years and −0.45±0.64 D in children aged 8–9 years. These findings are consistent with those of Enyedi et al,4 who found an overall postoperative myopic shift was greatest in the youngest patients aged up to 8 years. One possible explanation is that there are difficulties in the measurement of ocular parameters (axial length, keratometry) in young children and this may lead to more errors in the IOL power calculation. However, our findings are in contrast to those of Sachdeva et al,10 who found more hypermetropia in children aged less than 2 years and suggested that less undercorrection was required in this age group. It was concluded that more data are needed to confirm the trends in refractive errors in this age group.
Moreover, in this study, there are seven eyes from four children with a family history of myopia. The mean final refractive state of these eyes was −0.78±2.84 D. The mean follow-up time in this group was 97.25±43.79 months. This result contradicts our hypothesis that children with a family history of myopia, even those with a long follow-up period, will develop more myopic shift than children without family history. Interestingly, we found that six out of seven children, who were aged more than 10 years with unilateral cataract, showed more myopia (from −0.75 to −5.75 D) in pseudophakic eye compared to the fellow normal eye. This may due to the growth of the cataractous eye being different to the normal eye. The increasing elongation of the posterior segment relative to the anterior segment, given the IOL is in the same position, may play an important role in magnifying the myopic shift that occurs with ocular growth. Another possibility is our formula needs to be adjusted to aim for more undercorrection to achieve emmetropia in the long-term.
One study using an animal model showed that the removal of cataractous lens retards the rate of axial length elongation.11 Furthermore, a recent study by Lambert12 demonstrated that children with unilateral congenital cataract generally have a shorter axial length in their cataractous eye and the difference usually remain even after surgery. According to this study, the shorter axial length may lead to greater hyperopic shift in pseudophakic eyes, which is in contrast to the result of our study that showed more myopia in pseudophakic eyes. It is proposed that this may be the result of other factors that contribute to final refractive status such as the ocular growth, which may be influenced races and genetic factors. Further studies are required to clarify how a cataract can alter the normal ocular growth pattern.
In this study, there is also a small group of eyes with large amount of refractive error (five eyes with myopia more than 5 D) and we assume that this may be the result of axial length and keratometric measurement errors. In adults, inaccurate axial length measurements can account for 3–4 D for each millimeter and these errors can magnify to 4–14 D per millimeter in smaller pediatric eyes.2 In addition, the refractive error may result from the IOL calculation formula, which was derived from studies in adults and thus, may not be appropriate for shorter pediatric eyes.
The limitations of this study include its small sample size and the uneven distribution of the age of the children, as 35 of the 44 children are aged 7 years or more at the time of surgery. This may cause bias in the results interpretation and in application of these findings into clinical practice.
Conclusion
Choosing an appropriate IOL power is challenging in children. To date, there is no standardization in IOL power adjustment for intended undercorrection to account for myopic shift. In our practice, the majority of postoperative refractive error in children aged 10 years or more at the last follow-up time was myopic after the intended undercorrection of IOL power. The IOL power calculation formula needs to be adjusted in this study for more undercorrection to achieve an optimal refractive outcome in adulthood. Following our finding, we have decided to increase our undercorrection percentages by 5% increments in each age group. More studies are needed to compare these guidelines for undercorrection in other cohorts.
Acknowledgment
The authors thank David Dimasi, PhD, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Wilson ME, Apple DJ, Bluestein EC. Intraocular lens implantation: has it become the standard of care for children? Ophthalmology. 1996;103(11):1719–1720. | ||
Al Shamrani M, Al Turkmani S. Update of intraocular lens implantation in children. Saudi J Ophthalmol. 2012;26(3):271–275. | ||
Gordon RA, Donzis PB. Refractive development of the human eye. Arch Ophthalmol. 1985;103(6):785–789. | ||
Enyedi LB, Peterseim MW, Freedman SF, Buckley EG. Refractive changes after pediatric intraocular lens implantation. Am J Ophthalmol. 1998;126(6):772–781. | ||
Dahan E, Drusedau MU. Choice of lens and dioptric power in pediatric pseudophakia. J Cataract Refract Surg. 1997;23(Suppl 1):618–623. | ||
Gwiazda J, Thorn F, Bauer J, Held R. Emmetropization and the progression of manifest refraction in children followed from infancy to puberty. Clin Vision Sci. 1993;8:337–344. | ||
Mutti DO, Mitchell GL, Jones LA, et al. Refractive astigmatism and the toricity of ocular components in human infants. Optom Vis Sci. 2004;81(10):753–761. | ||
Twelker JD, Mitchell GL, et al. CLEERE Study Group, et al. Children’s ocular components and age, gender, and ethnicity. Optom Vis Sci. 2009;86(8):918–935. | ||
Brown SM, Archer S, del Monte MA. Stereopsis and binocular vision after surgery for unilateral infantile cataract. J Aapos. 1999;3(2):109–113. | ||
Sachdeva V, Katukuri S, Kekunnaya R, Fernandes M, Ali MH. Validation of guidelines for undercorrection of intraocular lens power in children. Am J Ophthalmol. 2017;174:17–22. | ||
Kugelberg U, Zetterstrom C, Lundgren B. Syren-Nordqvist S: eye growth in the aphakic newborn rabbit. J Cataract Refract Surg. 1996;22:337–341. | ||
Lambert SR. Changes in ocular growth after pediatric cataract surgery. Dev Ophthalmol. 2016;57:29–39. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
