Back to Journals » OncoTargets and Therapy » Volume 13
Long Noncoding RNA LINC00460 Promotes Hepatocellular Carcinoma Progression via Regulation of miR-342-3p/AGR2 Axis
Authors Yang J, Li K, Chen J, Hu X, Wang H, Zhu X
Received 19 November 2019
Accepted for publication 30 January 2020
Published 6 March 2020 Volume 2020:13 Pages 1979—1991
DOI https://doi.org/10.2147/OTT.S239258
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Leo Jen-Liang Su
Jing Yang,1,2 Kun Li,3 Jian Chen,4 Xiaoxiong Hu,5,6 He Wang,2 Xuan Zhu1
1Department of Gastroenterology, The First Affiliated Hospital of Nanchang University, Nanchang, Jiangxi 330006, People’s Republic of China; 2Department of Gastroenterology and Hepatology, The People’s Hospital of Yichun City, Yichun 336000, People’s Republic of China; 3Department of General Surgery, The People’s Hospital of Yichun City, Yichun 336000, People’s Republic of China; 4Department of Oncology, The People’s Hospital of Yichun City, Yichun 336000, People’s Republic of China; 5Clinic Research Center of People’s Hospital of Yichun City, Yichun 336000, People’s Republic of China; 6Department of Infection Disease, The People’s Hospital of Yichun City, Yichun 336000, People’s Republic of China
Correspondence: Xuan Zhu
Department of Gastroenterology, The First Affiliated Hospital of Nanchang University, No. 17 Yongwaizheng Road, Donghu District, Nanchang 330006, Jiangxi, People’s Republic of China
Tel +86-0791-88692748
Email [email protected]
Background: Hepatocellular carcinoma (HCC) is the leading cause of cancer-related death worldwide. LINC00460, a novel long non-coding RNA (lncRNA), was recently confirmed as an oncogene in various cancers. However, the biological function and underlying mechanism of LINC00460 in HCC is largely obscure.
Methods: Fifty pairs of tumor tissue and adjacent normal tissues from HCC patients, as well as six HCC cell lines and a normal human hepatic epithelial cell line were subjected to qRT-PCR assay to evaluate the expression levels of LINC00460. CCK-8 assays were used to detect the proliferation of HCC cells. Transwell assay was used to measure the migration and invasion abilities of HCC cells. RNA pull-down and luciferase assays were performed to verify the direct interaction between LINC00460 and miR-342-3p. A xenograft model of HCC was established to validate the in vivo function of LINC00460 in HCC progression.
Results: We firstly detected LINC00460 expression was significantly upregulated in both HCC tumor tissues and cell lines. The upregulation of LINC00460 was positively associated with HCC progression. Functionally, LINC00460 facilitated HCC cell proliferation, migration, and invasion capacities, which due to that LINC00460 could physically bind to and repress miR-342-3p to elevate the expression of AGR2.
Conclusion: Our data firstly reveal the clinical relevance, biological function, and regulatory mechanism of LINC00460 in HCC development. LINC00460 promotes HCC progression by elevating AGR2 expression via sponging miR-342-3p, providing a promising therapeutic target for HCC treatment.
Keywords: hepatocellular carcinoma, LncRNA, LINC00460, miR-342-3p, AGR2, migration, invasion
Background
Hepatocellular carcinoma (HCC) is the second leading cause of cancer-related death worldwide, with high morbidity and mortality.1,2 Despite great advances in HCC treatment, the prognosis and survival rate of HCC patients is still unsatisfactory due to metastasis and recurrence.3 Therefore, it is necessary to identify novel biomarkers for predicting the prognosis of HCC patients and developing molecular target therapy.
Long non-coding RNAs (lncRNAs), which defined as non-coding RNAs more than 200 nucleotides, belong to a kind of non-coding RNAs, which can affect various biological processes involving epigenetic modification, transcriptional regulation, protein translation and degradation.4,5 Increasing novel LncRNAs are discovered and identified as tumor suppressors or oncogenes in human cancers,6,7 offering the possibility of lncRNAs as novel biomarkers and therapeutic targets for cancer. Recently, LINC00460, a cancer-related lncRNA, has been highlighted to be involved in carcinogenesis and promotes tumor proliferation, invasion, and migration, and its upregulation is generally associated with tumor grade and poor prognosis.8,9 However, the role and underlying molecular mechanism of LINC00460 in HCC remains unclear. MicroRNAs are short small nonprotein-coding RNAs with around 22 nucleotides, and negatively regulate target gene expression by targeting its 3ʹ untranslated region (UTR) of mRNA. Increasing evidence has demonstrated that miRNAs play crucial roles in various biological processes, involving cell growth, differentiation, and apoptosis.10 But whether LINC00460 could interact with miRNA in HCC still remains to be explored.
Here, we aimed to explore the biological function, mechanism and clinical implication of LINC00460 in HCC progression. We firstly demonstrated that LINC00460 expression was significantly increased in HCC tumor tissues and cell lines, which was closely correlated with advanced clinical features of HCC. Functionally, LINC00460 could bind to and sequester miR-342-3p to elevate the expression of AGR2, thereby promoting HCC cell proliferation, migration, and invasion.
Materials and Methods
Clinical Patient Samples
Fifty pairs of HCC and adjacent para-carcinoma tissue samples were collected in the Department of General Surgery, The People's Hospital of Yichun City and Department of General Surgery, the Third Affiliated Hospital of Nanchang University. These tissues were instantly placed into liquid nitrogen to protect RNA integrity and stored at −80°C for further analysis. All patient sample collections were approved by the ethics committee of the Department of General Surgery, The People's Hospital of Yichun City and the Department of Gastroenterology, the First Affiliated Hospital of Nanchang University. The clinicopathological features of the patients were listed in Table 1. Written informed consent had been obtained from all patients.
 |
Table 1 The Clinicopathological Features of the HCC Patients |
Cell Lines and Transfection
One human normal hepatic epithelial cell line LO2 and six HCC cell lines (HepG2, Huh7, SMMC7721, BEL-7402, HCCLM3, SK-HEP-1) were purchased from Cell Bank of Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (FBS) (Gibco, USA) in a humidified 37 °C incubator with 5% CO2. For LINC00460 overexpression, the full-length sequence of LINC00460 was cloned into pcDNA3.1 (Invitrogen, CA, USA) plasmid to generate pcDNA3.1-LINC00460. All small interfering RNAs (siRNAs) for cell transfection were obtained from Shanghai Biotend Biotechnology Co, Ltd. Cell transfection was conducted with Lipofectamine 2000 Reagent (Invitrogen, CA, USA) as the manufacturer’s protocol. Briefly, 3 x 105 cells were seeded in a six-well tissue culture plate, and incubated the cells until 60–80% confluent. Then we prepared the following solutions: (1) Solution A, we used 2 μg plasmid or diluted 10 μL of siRNA duplex (100 pmol siRNA) into 250 μL optiMEM; (2) Solution B, we diluted 5 μL of Lipofectamine 2000 into 250 μL optiMEM., each solution was incubated at room temperature for 5 min. Then we mixed Solution A directly to Solution B for 20 min at room temperature. Subsequently, 500 μL mixture was added to each well containing cells and medium. Finally, cells were incubated for 6 h at 37°C in a CO2 incubator and replace the medium. Cells were subjected to the following experiments after 48 h of transfection.
The siRNA sequences for transfection were following:
LINC00460-siRNA:
5′- AGACCTAATAGCCAATAAG-3′;
AGR-siRNA:
5ʹ-UGAUGAAUAAUCAUCAAGGGUCCUUGAUGAUUAUUCAUCACU-3ʹ;
scramble-siRNA:
5′-GGACUCUCGGAUUGUAAGAUU-3′.
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT‑PCR)
Total RNAs were extracted from tissues and cell lines using TRIzol reagent (Invitrogen, CA, USA). The first-strand cDNA was reverse transcribed from RNAs using reverse transcriptase kit (Takara, Otsu, Japan). qRT-PCR was performed using SYBR Green SuperMix (Roche, Basel, Switzerland) as the manufacturer’s protocols. GAPDH and U6 were used as the internal control for lncRNA/mRNA and miRNAs, respectively. Besides, U6 and 18s rRNA were set as reference genes of the nucleus and cytoplasm respectively. Each sample was run in triplicate using samples from independent experiments. The relative mRNA expressions were expressed as a function of threshold cycle (Ct) and analyzed by 2−ΔΔCt method. The primer sequences are listed in Table 2.
 |
Table 2 The RT-qPCR Primer Sequences |
CCK-8 Assay
Cell proliferation capacity was evaluated by a Cell Counting Kit-8 (CCK8, Dojindo, Japan) as the manufacturer’s protocol. Cells were seeded at a density of 2×103/well in 96-well plates. Cells were cultured for indicated times (24, 48 or 72 hrs). Then, 10 μL of CCK8 solution was added to each well, and incubated for 1 h at 37 °C before the absorbance was measured at 450 nm wavelength with the microplate reader (Synergy H4 Hybrid Reader, BioTek, Winooski, USA). Each data point presents the mean ± s.d. of three independent experiments.
Colony Formation
A total of 500 cells were plated into 6-well plates. After culture for 14 days in a humidified 37 °C incubator with 5% CO2, the colonies were fixed with 10% formaldehyde for 30 min and then stained with 0.5% crystal violet (Beyotime, China) for 30 min. The colonies were photographed by the microscope (Olympus, Tokyo, Japan).
Transwell Assay
Transwell assay was conducted for the detection of cell migration and invasion. Transwell chambers (Millicell Hanging Cell Culture Insert, polyethylene terephthalate 8.0 µm, Millipore, MA, USA) coated with Matrigel (BD biosciences, USA) were prepared as the manufacturer’s instructions. After starvation, 1×105 cells in 200 μL of serum-free medium were placed in the top chamber. The chamber was inserted into a 12-well filled with 600 μL medium added with 10% FBS. After 24 h incubation, noninvasive cells were removed from the upper surface of the membrane with cotton swabs, and invasive cells on the lower membrane surface were fixed with methanol, stained with 0.5% crystal violet (Beyotime, China), photographed and counted under five random 200× microscopic fields per well using a Nikon Inverted Research Microscope Eclipse Ti microscope. The cell migration assay was simultaneously conducted as above, except for the chambers without Matrigel.
Luciferase Reporter Assays
4 × 104 HCC cells were plated in 24-well plates the day before transfection with the reporter plasmid with either miR-342-3p or NC mimics. Then cells were transfected with Lipofectamine 2000 (Invitrogen, USA) as the manufacturer’s instructions. After 48 h of transfection, luciferase activities were analyzed using the Dual-Luciferase Reporter Assay System (Promega, USA) as the manufacturer’s protocol.
Cellular Nucleo-Cytoplasmic Fractionation
Cells were fractionated using NE-PER Nuclear and Cytoplasmic Extraction Reagents (ThermoFisher, USA) as the manufacturer’s protocol. 4×106 cells were re-suspended in buffer C (20 mM Tris-HCl pH 7.5, 75 mM NaCl, 5 mM MgCl2, 0.5% p/w sodium deoxycholate, 0.2% Triton, 1 mM DTT, 0.5% glycerol) added protease inhibitor cocktail (Sigma, USA) and RNase inhibitor (Thermo Scientific, USA). After centrifugation, supernatants containing cytoplasmic lysates were harvested. Then, pelleted nuclei were washed extensively with PBS. Pelleted nuclei were resuspended in buffer N (10 mM Tris-HCl pH 8, 25 mM NaCl, 5 mM MgCl2, 1% p/w sodium deoxycholate, 1% Triton, 0.2% SDS, 1 mM DTT) added protease and RNase inhibitors and sonicated.
RNA Pull-Down Assay
LINC00460 and Negative Control (NC) were biotin-labeled using Biotin RNA Labeling Mix (Roche, Basel, Switzerland) and T7/SP6 RNA polymerase (Roche) to be bio-LINC00460 and bio-NC by GenePharma (Shanghai, China). Then, HepG2 and Huh7 cells were mixed and incubated with biotinylated RNAs for 48 h. Next, cells were collected and incubated with Streptavidin agarose beads (Invitrogen, USA) for 1 h 37 °C. After beads were washed with buffer, the bound RNAs were quantified and analyzed by qRT-PCR assays.
Fluorescence in situ Hybridization (FISH)
The subcellular localization of LINC00460 was assessed using the FISH kit (BIS-P0001, Guangzhou Bersin Biotechnology Co., Ltd., Guangzhou, China). The cells slide was supplemented with LINC00460 probe hybridization solution labeled by Digoxigenin, while the antagonistic LINC00460 probe used as a negative control (NC). The slide was hybridized at 42 °C for 16 h and immersed in 2 × SSC, followed by immersion in 70% ethanol for 3 min and stained with 4′6-diamidino-2-phenylindole (DAPI) for 10 min. The slide was photographed using the Zeiss LSM880 NLO confocal microscope (Leica, Germany).
Western Blot Analysis
When transfection finished, HepG2 and Huh7 cells were lysed in RIPA buffer (Beyotime, China). Total protein concentration was qualified with a BCA Protein Assay kit (Beyotime, China). Next, protein samples were separated by SDS-PAGE and transferred to PVDF membranes (Millipore, USA). The membranes were blocked with 5% non-fat milk for 1 h at room temperature, and then incubated with primary antibodies (AGR2, 1:5000 dilution, ab76473; GAPDH, 1:2000 dilution, ab8245) at 4 °C overnight. Anti-Rabbit and anti-mouse IgG were used as the secondary antibody, respectively. All these antibodies were purchased from Abcam (Cambridge, UK). The protein bands were quantified with the ImageJ software (USA).
Tumor Xenograft Experiments
Male BALB/c nude mice (6-week old) were used for xenograft experiments. All animal protocols were approved by the Institutional Animal Care and Use Committee at the First Affiliated Hospital of Nanchang University. We used the guideline 3Rs (REPLACEMENT, REDUCTION AND REFINEMENT) for the welfare of the laboratory animals. Briefly, 5 × 106 HepG2 cells were subcutaneously injected into the flank of mice. After around one week, the tumor was visible (100–200 mm3) and injected with the si-NC and si-LINC00460 twice per week. Tumor volumes were measured every 3 days. Tumor volume was calculated using the formula: Tumor volume (mm3) = (width) × (height)2/2. After 15 days, mice were sacrificed for the further analysis and tumor weight was measured.
Statistical Analyses
SPSS 21.0 software was used for all data analyses. Clinicopathological correlations were calculated by the Pearson Chi-square Test (Linear Regression). Significance of difference was calculated with Student’s t-test unless otherwise indicated. P values less than 0.05 were considered significant (*P < 0.05; **P < 0.01; ***P < 0.001). The data show the mean ± s.d. of three independent biological experiments.
Result
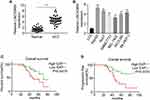
LINC00460 Is Significantly Upregulated in HCC and Associated with the Poor Clinical Outcome
We first examined the LINC00460 expression in 50 pairs of HCC clinical samples by qRT-PCR assay. LINC00460 was remarkably upregulated in HCC tissues compared to that in adjacent normal tissues (Figure 1A), which was consistent with that in the six HCC cell lines (HepG2, Huh7, SMMC7721, BEL-7402, HCCLM3, and SK-HEP-1) relative to that in a normal human hepatic epithelial cell line LO2 (Figure 1B). To further explore the clinical significance of LINC00460 expression in HCC, we served the median value of LINC00460 in HCC tissue samples in Figure 1A as the cut-off value, and divided the 50 cases of HCC patients into two groups: the LINC00460 low expression group (n = 25) and LINC00460 high expression group (n = 25). Kaplan-Meier analyses results of relevant clinicopathological features of HCC patients demonstrated that increased expression of LINC00460 was positively associated with tumor differentiation, lymph node metastasis and tumor-node-metastasis (TNM) stage (P < 0.01, Table 1), but there was no significant association between LINC00460 expression and age or gender of patients. The further statistical analyses revealed that patients with high LINC00460 had shorter overall survival and progress free survival relative to patients with low LINC00460 expression (Figure 1C and D). These results demonstrated that LINC00460 exerts a tumor-promoting effect on HCC progression.
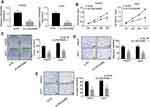
Knockdown of LINC00460 Inhibits HCC Cell Proliferation, Migration and Invasion
To explore the role of LINC00460 in HCC, we performed loss of function experiments in HepG2 and Huh7 cells (Figure 2A). CCK8 assay results demonstrated that knockdown of LINC00460 significantly decreased the cell proliferation in HepG2 and Huh7 cells (Figure 2B). Colony formation assay indicated that downregulated LINC00460 inhibited the colony numbers of HepG2 and Huh7 cells (Figure 2C). We next performed transwell assays to explore how LINC00460 impacts HCC cell migration and invasion. Results showed that the migration and invasion cell numbers of HepG2 and Huh7 cells treated with si-LINC00460 were markedly suppressed compared with that in the si-NC group (Figure 2D and E). Collectively, these data suggested that knockdown of LINC00460 inhibits cell proliferation, migration, and invasion in HCC.
Overexpression of LINC00460 Promotes HCC Cell Proliferation, Migration and Invasion
To further investigate the function of LINC00460 in HCC progression, we overexpressed LINC00460 in HepG2 and Huh7 cells with a relative high LINC00460 expression level (Figure 3A). The results of CCK-8 assays indicated that LINC00460 overexpression significantly enhanced the HCC cell viability in a time-dependent manner (Figure 3B). Moreover, more colonies were formed after LINC00460 upregulation (Figure 3C), suggesting that LINC00460 promotes the proliferation of HCC cells. We next detected the migration and invasion abilities of HCC cells with LINC00460 overexpression. Results showed that LINC00460 overexpression markedly repressed the migration and invasion of HepG2 and Huh7 cells (Figure 3D and E). These results indicated that LINC00460 promotes proliferation, invasion, and migration of HCC cells.
LINC00460 Directly Interacts with miR-342-3p in HCC Cells
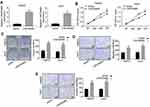
Emerging evidence demonstrated that long non-coding RNAs have been shown to serve as miRNA sponges in regulating the expression and biofunction of miRNAs.11,12 qRT-PCR and FISH assays results showed that LINC00460 was mainly located in the cytoplasm of HepG2 and Huh7 cells (Figure 4A and B), suggesting LINC00460 may be a sponge of miRNAs. Thus, we analyzed the potential binding miRNA partner of LINC00460. Results showed that LINC00460 may bind to miR-342-3p using the miRanda software (http://www.microrna.org/) (Figure 4C). Luciferase reporter assays results confirmed that overexpression of miR-342-3p obviously repressed the luciferase activity of wide-type (WT) LINC00460, but mutation of this binding motif diminished the inhibition (Figure 4C). In addition, RNA pull-down assays showed that the miR-342-3p expression was more enriched on the biotin-labeled LINC00460 probe (Figure 4D). Furthermore, LINC00460 overexpression led to the decreased miR-342-3p expression in HepG2 and Huh7 cells (Figure 4E). In 50 pairs of clinical samples, miR-342-3p was remarkably downregulated in HCC tumor tissues (Figure 4F), and there was a strong negative correlation between LINC00460 and miR-342-3p (Figure 4G, Pearson, P < 0.001). All these results indicated that LINC00460 directly interacts with and inhibits miR-342-3p expression.
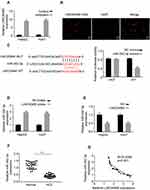 |
Figure 4 LINC00460 directly interacts with miR-342-3p in HCC cells. (A) The mRNA levels of nuclear control (U6), cytoplasmic control (GAPDH) and LINC00460 were analyzed using qRT‐PCR in nuclear and cytoplasmic fractions. (B) FISH assay was performed to determine the cellular location of LINC00460. Blue: DAPI (nuclease); Red: LINC00460-FAM (LINC00460). Scale bar, 20 μm. (C). A predicted binding site of miR-342-3p within LINC00460 by bioinformatic analysis using the miRanda software (http://www.microrna.org/). Luciferase activity was determined in HepG2 and Huh7 cells after transfection with miR-342-3p mimic or miRNA negative control (miR-NC). The binding sequences “ACACAGAG” in LINC00460 were mutated to “GTGTGAGA” for generating MUT‐LINC00460. (D and E) RNA pull-down assays were used to determine the interaction between LINC00460 and miR-342-3p. (F) Relative mRNA expression of miR-342-3p in HCC tissues and adjacent normal tissues was assessed using qRT-PCR assay. (G) Pearson correlation analysis between LINC00460 and miR-342-3p expressions in 50 pairs of HCC tissues. All data are representative of three independent experiments and shown as mean ± s.d. *P < 0.05, **P < 0.01, and ***P < 0.001. |
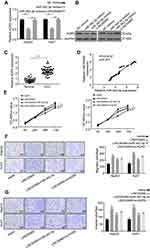
LINC00460 Increases AGR2 Expression via Sponging miR-342-3p in HCC Cells
Previous research demonstrated that AGR2 is a potential target of miR-342-3p.13 Furthermore, overexpression of miR-342-3p significantly inhibited AGR2 expression on both mRNA and protein levels in HepG2 and Huh7 cells (Figure 5A and B). However, after co-transfection with LINC00460, this inhibitory effect on AGR2 expression was partially decreased (Figure 5A and B), implying that LINC00460 regulates AGR2 expression through miR-342-3p. In addition, we examined the expression level of LINC00460 in 50 pairs of HCC clinical samples by qRT-PCR assay. We found that AGR2 was upregulated in HCC tumor tissues (Figure 5C). AGR2 and LINC00460 were positively associated in HCC sample tissues (Pearson, P < 0.001, Figure 5D), suggesting that LINC00460 elevated AGR2 expression by sponging miR-342-3p. To determine whether LINC00460 impacts HCC progression through AGR2/miR-342-3p axis, we co-transfected with miR-342-3p or si-AGR2 in LINC00460-overexpressed HCC cells (Figure 5E). CCK8 assays results showed that miR-342-3p overexpression or AGR2 knockdown rescued the enhanced proliferative effect of LINC00460 overexpression in HepG2 and Huh7 cells (Figure 5E). Furthermore, the elevated migration and invasion abilities were also suppressed after co-transfection with miR-342-3p mimic or si-AGR2 (Figure 5F and G). These data indicated that LINC00460 increases AGR2 expression via sponging miR-342-3p, consequently facilitating the proliferation of HCC cells.
Depletion of LINC00460 Inhibits Tumor Growth in vivo
To validate the in vivo function of LINC00460 in HCC progression, a xenograft model of HCC was established using the LINC00460 knockdown (si-LINC00460) and control (si-NC) HepG2 cells. Tumor sizes were measured every 3 days. The results showed that the LINC00460 knockdown resulted in the reduction of both tumor size and weight (Figure 6A). Consistent with cellular assays results, LINC00460 knockdown decreased AGR2 expression but increased miR-342-3p expression in the HepG2 xenograft tumors (Figure 6B). Besides, as depicted in Figure 6C, the protein expression of AGR2 was downregulated in the si-LINC00460 group compared to that in the si-NC control. All results indicated that LINC00460 promotes HCC tumor growth in vivo by upregulating AGR2.
Discussion
Emerging evidence demonstrated that lncRNAs could function as endogenous miRNA sponges by binding to miRNAs in the cytoplasm and regulating their function.12 As key regulators to regulate some key genes and signal pathways associated with the initiation and development of cancers, the abnormal expression of lncRNAs are closely related to tumor differentiation, proliferation, metastasis, and drug resistance, and other aspects,6,11,12,14,15 thus becoming potential therapeutic targets and prognostic biomarkers for cancers. Recently, a novel lncRNA LINC00460 has been highlighted to be involved in carcinogenesis.8,9 The expression of LINC00460 was reported to be upregulated in NSCLC tissues. LINC00460 can promote the NSCLC tumor metastasis through affecting EMT.8 In addition, LINC00460 has been found significantly upregulated in prostate cancer and closely related to tumor progression and poor outcomes of the patients through the PI3K/AKT signaling pathway.9 These results suggested that LINC00460 serves as an oncogene and could be a novel diagnostic biomarker in cancers. However, an in-depth investigation of LINC00460 clinical significance and underlying biological function in HCC has not been fully undertaken. In our study, we showed that the expression of LINC00460 was markedly upregulated in HCC tissues and cells, which was associated with clinicopathological features and shorter survival of HCC. Knockdown of LINC00460 impaired HCC cell proliferation, migration, and invasion. The nude mice xenografts further revealed that knockdown of LINC00460 inhibited tumor growth in vivo. These results demonstrated that LINC00460 functions as an oncogene that promotes HCC progression.
The involvement of microRNAs in cancers and their significance as clinical biomarkers are becoming increasingly appreciated.10,16 LncRNAs could act as miRNA sponges and regulate their downstream target gene expression, thus participating in the tumor progression.12 LINC00460 could promote HCC progression through sponging miR-485-5p to elevate PAK1.17 Besides, miR-342-3p has been served as a tumor suppressor in HCC tumorigenesis, and its low expression is associated with poor prognosis of HCC.18 Moreover, LINC00460 was found to promote gastric cancer cell proliferation, migration and invasion by sponging miR-342-3p to up-regulate KDM2A expression in GC cells.19 Recently, anterior gradient protein-2 (AGR2), an androgen-regulated gene, has been identified as an oncogene and generally overexpressed in various cancers.20–23 AGR2 promotes the metastasis of breast cancer cells.21 Besides, High AGR2 expression has been reported to show lower overall survival of CRC patients. AGR2 enhances the canonical Wnt/β-catenin dependent cell proliferation and differentiation, as well as regulates the stemness of colon cancer stem cells.22 Moreover, AGR2 expression has been observed relatively high in prostate cancer and lung cancer.20,23 It has been reported that AGR2 is a direct target of miR-342-3p.13 In our study, LINC00460 was proved to mainly distribute in the cytoplasm, thus regulating the post-transcription of miRNAs. Bioinformatic analysis (miRanda software) predicted that miR-342-3p was the only downstream miRNA target of LINC00460. RNA pull-down and luciferase assays were used to verify their interaction in HCC. Functionally, we found that LINC00460 markedly promoted the HCC proliferation, migration, and invasion by increasing AGR2 expression via sponging miR-342-3p, which were abrogated by co-transfection with miR-342-3p or si-AGR2.
Collectively, our findings firstly determined the expression level, clinical relevance, and function mechanism of LINC00460 in HCC, and revealed that LINC00460 facilitates HCC progression by regulating miR-342-3p/AGR2 axis, providing a potential therapeutic strategy for HCC treatment.
Conclusion
Our study demonstrated that LINC00460 is highly expressed in HCC tissues and cells. Its upregulation is closely associated with the TNM stage, metastasis and poor survival of HCC patients. LINC00460 serves as an oncogenic lncRNA that promotes HCC tumorigenesis via sponging miR-342-3p to elevate the AGR2 expression, which provides a promising therapeutic target for HCC treatment.
Abbreviations
AGR2, anterior gradient protein-2; CCK-8, Cell counting kit-8; DMEM, Dulbecco’s Modified Eagle’s Medium; FISH, Fluorescence in situ hybridization; HCC, Hepatocellular carcinoma; miRNA, microRNA; MUT, mutation; NC, Negative Control; qRT-PCR, quantitative reverse transcription polymerase chain reaction; siRNAs, Small interfering RNAs; TNM, tumor-node-metastasis; UTR untranslated region; WT, wide-type.
Ethics Approval and Consent to Participate
The current study was approved by the ethics committee of the Department of General Surgery, the People's Hospital of Yichun City, and the Department of Gastroenterology, The First Affiliated Hospital of Nanchang University. Written informed consent was obtained from all patients and conducted in accordance with the Declaration of Helsinki.
Acknowledgment
This research was sponsored by National Natural Science Foundation of China (grant number: 81660110).
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was supported by the National Natural Science Foundation of China (grant number: 81660110).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. eng. doi:10.3322/caac.21262
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34. doi:10.3322/caac.v69.1
3. Forner A, Bruix J. Biomarkers for early diagnosis of hepatocellular carcinoma. Lancet Oncol. 2012;13(8):750–751. doi:10.1016/S1470-2045(12)70271-1
4. Lorenzi L, Avila Cobos F, Decock A, et al. Long noncoding RNA expression profiling in cancer: challenges and opportunities. Genes Chromosomes Cancer. 2019;58(4):191–199. doi:10.1002/gcc.22709
5. Derrien T, Johnson R, Bussotti G, et al. The GENCODE v7 catalog of human long noncoding RNAs: analysis of their gene structure, evolution, and expression. Genome Res. 2012;22(9):1775–1789. doi:10.1101/gr.132159.111
6. Wang KC, Chang HY. Molecular mechanisms of long noncoding RNAs. Mol Cell. 2011;43(6):904–914. doi:10.1016/j.molcel.2011.08.018
7. Sun W, Yang Y, Xu C, Guo J. Regulatory mechanisms of long noncoding RNAs on gene expression in cancers. Cancer Genet. 2017;216–217:105–110. doi:10.1016/j.cancergen.2017.06.003
8. Yue QY, Zhang Y. Effects of Linc00460 on cell migration and invasion through regulating epithelial-mesenchymal transition (EMT) in non-small cell lung cancer. Eur Rev Med Pharmacol Sci. 2018; (2284-0729 (Electronic)): 22(4):1003–1010. eng.
9. Dong Y, Quan HY. Downregulated LINC00460 inhibits cell proliferation and promotes cell apoptosis in prostate cancer. Eur Rev Med Pharmacol Sci. 2019;23(14):6070–78. eng. [(2284-0729 (Electronic))]. doi:10.26355/eurrev_201907_18420
10. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–297. doi:10.1016/S0092-8674(04)00045-5
11. Geisler S, Coller J. RNA in unexpected places: long non-coding RNA functions in diverse cellular contexts. Nat Rev Mol Cell Biol. 2013;14(11):699–712. doi:10.1038/nrm3679
12. Hansen TB, Jensen TI, Clausen BH, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495(7441):384–388. doi:10.1038/nature11993
13. Xue X, Fei X, Hou W, Zhang Y, Liu L, Hu R. miR-342-3p suppresses cell proliferation and migration by targeting AGR2 in non-small cell lung cancer. Cancer Lett. 2018;412:170–178. doi:10.1016/j.canlet.2017.10.024
14. Kondo Y, Shinjo K, Katsushima K. Long non-coding RNAs as an epigenetic regulator in human cancers. Cancer Sci. 2017;108(10):1927–1933. doi:10.1111/cas.2017.108.issue-10
15. Hu G, Niu F, Humburg BA, et al. Molecular mechanisms of long noncoding RNAs and their role in disease pathogenesis [Review]. Oncotarget. 2018;9(26):18648–18663. doi:10.18632/oncotarget.v9i26
16. Adams BD, Kasinski AL, Slack FJ. Aberrant regulation and function of microRNAs in cancer. Curr Biol. 2014;24(16):R762–76. doi:10.1016/j.cub.2014.06.043
17. Tu J, Zhao Z, Xu M, Chen M, Weng Q, Ji J. LINC00460 promotes hepatocellular carcinoma development through sponging miR-485-5p to up-regulate PAK1. Biomed Pharmacother. 2019;118:109213. doi:10.1016/j.biopha.2019.109213
18. Gao Y, Zhang Sg, Wang ZH, Liao JC, Liao JC. Down-regulation of miR-342-3p in hepatocellular carcinoma tissues and its prognostic significance. Eur Rev Med Pharmacol Sci. 2017;21(9):2098–102. eng. (2284-0729 (Electronic)).
19. Wang F, Liang S, Liu X, Han L, Wang J, Du Q. LINC00460 modulates KDM2A to promote cell proliferation and migration by targeting miR-342-3p in gastric cancer. Onco Targets Ther. 2018;11:6383–6394. doi:10.2147/OTT
20. Zhang JS, Gong A, Cheville JC, Smith DI, Young CY. AGR2, an androgen-inducible secretory protein overexpressed in prostate cancer. Genes Chromosomes Cancer. 2005;43(3):249–259. doi:10.1002/gcc.20188
21. Liu D, Rudland Ps, Sibson DR, Platt-Higgins A, Barraclough R. Human homologue of cement gland protein, a novel metastasis inducer associated with breast carcinomas. Cancer Res. 2005;65(9):3796–805. eng. [(0008-5472 (Print))]. doi:10.1158/0008-5472.CAN-04-3823
22. Dahal Lamichane B, Jung SY, Yun J, et al. AGR2 is a target of canonical Wnt/beta-catenin signaling and is important for stemness maintenance in colorectal cancer stem cells. Biochem Biophys Res Commun. 2019;515(4):600–606. doi:10.1016/j.bbrc.2019.05.154
23. Alavi M, Mah V, Maresh EL, et al. High expression of AGR2 in lung cancer is predictive of poor survival. BMC Cancer. 2015;15:655. doi:10.1186/s12885-015-1658-2
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.