Back to Journals » OncoTargets and Therapy » Volume 12
Long Non-Coding RNA BCAR4 Promotes Growth, Invasion and Tumorigenicity by Targeting miR-2276 to Upregulate MMP7 Expression in Glioma
Authors Wang Z, Wang L, Liang Z, Xi Y
Received 5 August 2019
Accepted for publication 13 November 2019
Published 12 December 2019 Volume 2019:12 Pages 10963—10973
DOI https://doi.org/10.2147/OTT.S226026
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
Zhifeng Wang, Longlong Wang, Zan Liang, Yanguo Xi
Department of Neurosurgery, Cangzhou Central Hospital, Cangzhou, Hebei Province, People’s Republic of China
Correspondence: Zhifeng Wang; Yanguo Xi
Department of Neurosurgery, Cangzhou Central Hospital, No. 16 Xinhuaxi Road, Cangzhou City, Hebei 061000, People’s Republic of China
Tel +86 317 207 5627
Email [email protected]
Objective: Long non-coding RNA breast cancer anti-estrogen resistance 4 (BCAR4) has been recognized as a proto-oncogene in various malignancies. It has been reported to be highly expressed and promote cell proliferation in glioma. However, its additional roles in gliomagenesis remain largely unclear. This research intends to investigate the impact and internal molecular mechanism of BCAR4 on glioma cell growth, invasion and tumorigenesis.
Methods: BCAR4 expression was examined by qPCR in 30 cases of graded glioma specimens and 7 glioblastoma (GBM) cell lines compared with respective controls. Its potential prognostic value was evaluated by Kaplan–Meier survival analysis. The biological roles of BCAR4 in gliomagenesis were verified by CCK-8, transwell and intracranial xenograft assays successively. qPCR and RNA pull-down assays were applied to study the relationship between BCAR4 and miR-2276. Then, qPCR, Western blot and luciferase reporter assays were used to validate the targeting of matrix metallopeptidase 7 (MMP7) by miR-2276 and the regulation of MMP7 by BCAR4. Finally, MMP7 was restored in BCAR4-silenced GBM cells and the rescue effects were determined by CCK-8 and transwell assays.
Results: BCAR4 expression was increased in glioma tissues and GBM cell lines, and its high expression positively correlated with advanced grades and worse prognosis. Functional assays verified that knockdown of BCAR4-inhibited cell growth and invasion in vitro, and impaired tumor formation in vivo. Mechanistically, we found that BCAR4 could act as a competing endogenous RNA (ceRNA) by targeting miR-2276 to upregulate MMP7 expression. Importantly, MMP7 restoration effectively rescued the inhibitory modulations on GBM cell growth and invasion caused by BCAR4 knockdown.
Conclusion: Our findings identified the essential roles of the BCAR4/miR-2276/MMP7 axis in gliomagenesis and provided novel insights on glioma therapy.
Keywords: glioma, BCAR4, ceRNA, miR-2276, MMP7
Introduction
Deriving from neuroepithelial, glioma is the most common primary tumor in the central nervous system, and malignant glioma is highly lethal.1 Among all the subtypes of gliomas, glioblastoma (GBM) belongs to the World Health Organization (WHO) grade IV gliomas, and is easy to relapse due to its infiltration nature which leads to complete resection almost impossible.2,3 The highly invasive nature also largely contributes to the poor prognosis of GBM patients.4 In spite of the progress achieved in surgical, chemotherapeutic and radio-therapeutic treatment, the overall clinical outcome of GBM patients remains far from satisfactory.4 Therefore, exploring more effective prognostic biomarkers and therapeutic targets especially to overcome the invasion function are urgently needed to improve the current situation.
Long non-coding RNAs (lncRNAs) are a group of RNAs with very limited protein-coding potential containing more than 200 nucleotides.5 As the research goes on, lncRNAs are recognized as important regulators in cancer development by impacting on various cellular processes, such as cell differentiation, proliferation, apoptosis and metastasis.6–8 They function as either tumor suppressors or oncogenes in specific cancers.8 Due to their versatile biological activities and tumor-restricted expression patterns, lncRNAs are deemed as promising biomarkers and therapeutic target in cancers, including in glioma.9–11 For instance, scholars demonstrated that expressions of lncRNAs like HOTAIR, HOXA11-AS and XIST are positively correlated with the malignancy of glioma, while lncRNAs like HOTTIP and GAS5 exhibit the negative correlation. In addition, the tumor-suppressing GAS5 indicates favorable prognosis, whereas oncogenic HOTAIR, HOXA11-AS, H19 and XIST indicate poor prognosis of glioma patients.11 Mechanistically, emerging evidences have supported that lncRNAs are implicated in gene expression regulation by acting as competing endogenous RNAs (ceRNAs) to competitively binding to microRNAs (miRNAs).12–14 MiRNAs are another group of non-coding RNAs with approximately 21 nucleotides in length and suppress target gene expression via base-pairing to the 3ʹ-untranslated region (3ʹ-UTR) of the target mRNAs.15 The interactions among lncRNAs, miRNAs, and the target mRNAs of miRNAs, constitute the ceRNA network, and play critical roles in the development and progression of human cancers.16,17
LncRNA breast cancer anti-estrogen resistance 4 (BCAR4) has been reported to be abnormally overexpressed in several carcinomas, including in glioma.18–21 The known modulation of BCAR4 in gliomagenesis is that it promotes cell proliferation.21 However, BCAR4 also possesses other oncogenic activities such as facilitating cancer cell migration, invasion and epithelial–mesenchymal transition (EMT) in other types of cancers.22,23 Therefore, its roles in gliomas deserve more attention.
In this study, we checked the expression and prognostic significance of BCAR4 in gliomas, and verified its biological functions in cell growth, invasion and tumorigenicity by employing both in vitro and in vivo assays. Bioinformatics screening predicted that miR-2276 might interact with BCAR4, and MMP7 was one of the targets of miR-2276. Substantial experiments were performed to validate the regulatory ceRNA network of BCAR4/miR-2276/MMP7 in GBM cells. Finally, MMP7 was restored in BCAR4-silenced GBM cells and the rescue effects on cell growth and invasion were examined. Our study therefore demonstrated a novel BCAR4/miR-2276/MMP7 signaling axis contributing to gliomagenesis and provided valuable insights in discovering potential biomarkers and therapeutic targets for this disease.
Materials and Methods
Patients and Tissues
Ten non-tumoral brain tissues (from cerebral trauma patients without neoplasm) and 30 glioma tissues (from glioma patients) were collected from Cangzhou Central Hospital from Jan 2017 to Dec 2018. After surgical excision, all samples were histopathologically diagnosed by pathologists and classified according to WHO classification. All patients did not receive chemotherapy or radiotherapy prior to sample collection. Tissue samples were frozen immediately in liquid nitrogen and stored at −80°C for RNA extraction. The basal clinical features of the 30 glioma patients enrolled in this study are listed in Table 1. Written informed consent had been obtained from all the included patients and the experiment was conducted in accordance with the Declaration of Helsinki. This study was also approved by the Ethics Committee of Cangzhou Central Hospital and the written informed consents were collected from all patients.
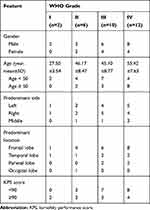 |
Table 1 The Clinical Features of the 30 Glioma Specimens Enrolled in This Study |
Cell Culture and Transfection
GBM cell lines (U87, U251, U343, LN229, LN308, A172 and SNB19) and normal human astrocytes (NHA) were from the Cell Bank of Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, China). DMEM medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Thermo Fisher Scientific, Waltham, MA, USA), 100 U/mL penicillin, and 100 mg/mL streptomycin (Thermo Fisher Scientific, Waltham, MA, USA) was used to culture cells at 37°C with 5% CO2. For transfections, cells were cultured using complete medium without antibiotics at least 24 hrs prior to transfection. Cells were washed twice by PBS and transiently transfected with siRNAs, miRNAs or plasmids by using Lipofectamine™ 2000 Transfection Reagent (Thermo Fisher Scientific, Waltham, MA, USA) as per the manufacturer’s instructions.
SiRNAs, miRNAs, Lentivirus and Plasmids
BCAR4 siRNAs, non-labeled or biotin-labeled miR-2276-3p mimics and negative control oligos were all purchased from GenePharma (Shanghai, China). Lentivirus containing BCAR4 shRNA or negative control lentivirus were purchased from GenePharma (Shanghai, China). U87 cells were infected with lentivirus at an MOI of 10 using 10 μg/mL polybrene (Sigma, St. Louis, MO, USA). The wild type or mutated-type sequence of 3ʹ-UTR region of MMP7 mRNA was cloned into pmirGLO vector (Promega, Madison, WI, USA). The wild-type sequence of MMP7 was cloned into pcDNA3.1 vector (Thermo Fisher Scientific, Waltham, MA, USA).
Quantitative Real-Time PCR (qPCR)
Total RNA was extracted from tissue samples or cells by using TRIzol reagent (Thermo Fisher Scientific, Waltham, MA, USA) as per the manufacturer’s instructions. 1 μg of RNA were reverse transcribed to detect miR-2276-3p and U6B expression level using specific primer kit (RiboBio, Guangzhou, China). 2 μg of RNA were reverse transcribed to detect BCAR4, MMP7 and β-actin expression level using general reverse transcription kit (Promega, Madison, WI, USA). qPCR was performed using SYBR Green PCR Master Mix (Takara, Otsu, Japan) on the Applied Biosystems 7500 Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA). The level of miR-2276-3p was normalized to that of U6B. The expressions of BCAR4 and MMP7 were standardized to the control values of β-actin. The relative expression of target genes was calculated with the 2−ΔΔCt methods.
CCK-8 Assay
Transfected U87 and LN229 cells were seeded at a density of 5×103 cells per well in a 96-well plate containing 100 μL DMEM complete medium. Cell Counting Kit-8 (Dojindo, Tokyo, Japan) reagent was added at Days 1–5 after seeding (Day 0) and incubated at 37°C for 2 hrs. The data of optical density of OD450 were measured by a microplate reader (Molecular Device, LLC, California, USA). Each experiment was performed three times with six replicates.
Transwell Assay
Transwell assay was performed to detect the cell invasion ability. Invasion assay was carried out by using transwell chambers (Corning, New York, USA). 3×103 transfected U87 or LN229 cells seeded into the 1 mg/mL matrigel-coated upper chamber were cultured at 70% confluence in 500 μL FBS free medium. Medium with 10% FBS (200 μL) was added to the lower chamber. After 24 hrs culturing, the invaded cells were fixed with 4% paraformaldehyde, followed by staining with 0.25% crystal violet. Evaluation of cell invasion ability was performed by counting invaded cells under an inverted microscope (Olympus, 100×), and five different microscopic views were selected randomly for analysis.
Western Blot
Total protein was extracted using RIPA lysis buffer (Beyotime Biotechnology, Nanjing, China) as per the manufacturer’s instructions. Protein samples were separated by 9% SDS-PAGE and then transferred onto nitrocellulose membranes (EMD Millipore, Billerica, USA). After incubated and blocked in the 5% skim milk in PBST buffer for 1 hr at room temperature, the membranes were incubated with anti-MMP7 antibody (Cell Signaling Technology, Danvers, MA, USA; 1:1000) and anti-β-actin antibody (Sigma, St. Louis, MO, USA; 1:8000) at 4°C overnight, respectively. On the following day, the membranes were incubated with HPR-conjugated secondary antibodies at room temperature for 1 hrs. The blots were measured using an enhanced chemiluminescence detection system (Pierce, Rockford, IL, USA).
Dual-Luciferase Reporter Assays
U87 and LN229 cells were seeded in 24-well plates at a density of 1.5×105 cells per well 24 hrs before transfection. Cells were transfected with MMP7-3ʹ-UTR-WT or MMP7-3ʹ-UTR-MT, followed by transfection with miR-2276 mimics or negative control oligos. 48 hrs later, cells were harvested, and firefly and renilla luciferase activities were measured using a dual-luciferase reporter assay system (Promega, Madison, WI, USA) as per the manufacturer’s instructions.
RNA Pull-Down Assay with Biotinylated miR-2276
U87 and LN229 cells were transfected with biotin-labeled wild-type miR-2276-3p (Bio-miR-2276-WT), mutated-type miR-2276-3p (Bio-miR-2276-MT) or antagonistic miR-2276-3p probe (Bio-miR-NC). 48 hrs after transfection, cells were harvested and incubated with a specific lysate buffer (Ambion, Austin, TX, USA) for 10 mins. After that, the cell lysates were mixed with M-280 streptavidin magnetic beads (Sigma, St. Louis, MO, USA) for 3 hrs at 4°C. The pull-down products were subjected to qPCR for BCAR4 expression.
Intracranial Xenograft Assay in Nude Mice
The constructed U87 sub-cell lines expressing luciferase and indicated shRNAs (NC-sh and BCAR4-sh2) were transplanted intracranially (5×104 cells per mouse) into the brains of 4-week old female BALB/C athymic nude mice using cranial guide screws. The intracranial tumor volume was detected by bioluminescence imaging every week after transplantation (at days 7, 14, 21). All animal experimental procedures were approved by the Institutional Animal Care and Use Committee of Cangzhou Central Hospital. All experiments were performed following guidelines and regulations of Committee of Cangzhou Central Hospital.
Statistical Analysis
Data were presented as mean±SD (standard deviation). Significant differences were determined by Student’s t-test or one-way ANOVA by using GraphPad Prism v6 software (GraphPad Software, Inc., San Diego, CA). Survival analysis was performed by Kaplan–Meier method and log-rank test. Pearson correlation test was performed to confirm the correlation between BCAR4 and miR-2276-3p expressions. A P value of <0.05 was considered statistically significant.
Results
Overexpression of BCAR4 in Gliomas and Its Prognostic Significance
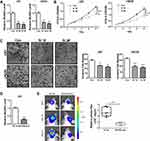
To validate the expression of BCAR4 in gliomas, we firstly examined its expression levels in 30 cases of graded glioma specimens and 10 non-tumoral brain tissues by qPCR. We found that BCAR4 was overexpressed in the 30 gliomas compared with normal brains (Figure 1A). Importantly, among these 30 cases of gliomas, BCAR4 expression was higher in advanced grades (WHO grades III–IV) than in lower grades (WHO grades I–II; Figure 1B). Besides, grade IV gliomas expressed higher BCAR4 than grade III gliomas (Figure 1B). In other words, its expression positively correlated with glioma grades. We also checked its expression in GBM cell lines and the normal human astrocytes (NHA). The result came out the same. BCAR4 expression was increased in the tested GBM cell lines (Figure 1C). Therefore, the expression detection confirmed the overexpression of BCAR4 in gliomas, which was in consistence with the previous finding by Wei et al.21 We then analyzed its prognostic significance by employing Kaplan–Meier survival analysis. As shown in Figure 1D, patients with higher BCAR4 levels showed a significant poorer progression-free survival (PFS) and overall survival (OS) rate. These initial results implied that BCAR4 might be crucial for gliomagenesis.
BCAR4 Knockdown Impairs GBM Cell Growth, Invasion and Tumorigenicity
Then, we systematically explored the biological functions of BCAR4 in gliomagenesis. Two different small interfering RNAs (siRNAs) targeting BCAR4 were used to transiently knockdown BCAR4 in GBM cells. U87 and LN229 cell lines were selected since they contained relatively higher levels of BCAR4 (Figure 1C). The inhibition efficiency was checked by qPCR (Figure 2A). CCK-8 assay verified that BCAR4 knockdown led to growth inhibition in both U87 and LN229 cell lines (Figure 2B). Transwell assay demonstrated that BCAR4 knockdown prominently suppressed GBM cell invasion (Figure 2C). To further make sure whether BCAR4 was critical for gliomagenesis in vivo, we constructed the stable BCAR4 knockdown U87 sub-cell line by utilizing its shRNA expressing lentivirus (Figure 2D). The following intracranial xenograft experiment showed that stable BCAR4 knockdown significantly inhibited tumor formation (Figure 2E). Therefore, our functional assays indicated the oncogenic roles of BCAR4 in gliomagenesis.
BCAR4 Modulates miR-2276/MMP7 Pathway in GBM Cells
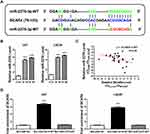
To investigate the molecular mechanism of BCAR4 in promoting gliomagenesis, we screened possible miRNA regulators of BCAR4 by using DIANA-LncBase v224 and found that miR-2276 was a potential target of BCAR4 (Figure 3A). qPCR showed that BCAR4 knockdown led to increased expression of miR-2276 in both U87 and LN229 cell lines (Figure 3B). In addition, in the abovementioned 30 gliomas, BCAR4 expression was inversely correlated with miR-2276 (Figure 3C), further demonstrating the negative regulation between BCAR4 and miR-2276. RNA pull-down confirmed the interaction between BCAR4 and miR-2276 (Figure 3A and D).
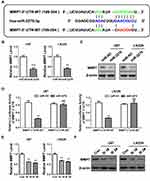
Then, we predicted the mRNA targets of miR-2276 by using TargetScan Release 7.2 and among all the targets of miR-2276, we mainly focused on matrix metallopeptidase 7 (MMP7; Figure 4A). As is known, MMP7 is implicated in glioma cell proliferation and invasion and closely associated with gliomagenesis.25 qPCR and Western blot assays revealed that miR-2276 mimics transfection led to reduced expression of MMP7 mRNA and protein in both U87 and LN229 cell lines (Figure 4B and C). Luciferase reporter assay also indicated that MMP7 was a direct target of miR-2276 in GBM cells (Figure 4A and D). Intriguingly, BCAR4 knockdown obviously inhibited the expression of MMP7 in both U87 and LN229 cell lines (Figure 4E and F). Therefore, our results identified that BCAR4 might act as a competing endogenous RNA (ceRNA) by interacting with miR-2276 to upregulate MMP7 expression in glioma cells.
MMP7 Restoration Rescues the Tumor Suppressing Effects of BCAR4 Knockdown
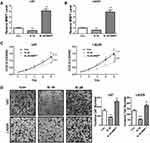
As we have found that BCAR4 was crucial for GBM cell growth, invasion and tumorigenicity, and it positively regulated MMP7 expression via targeting miR-2276, we finally intend to verify whether MMP7 could be the downstream effector of BCAR4 in promoting gliomagenesis. MMP7 was restored in BCAR4-silenced U87 and LN229 cells (Figure 5A and B). CCK-8 and transwell assays indicated that MMP7 overexpression resulted in enhanced GBM cell growth and invasion, and its restoration in BCAR4-silenced cells effectively abolished the inhibition of cell growth and invasion caused by BCAR4 knockdown in both U87 and LN229 cells (Figure 5C and D). Altogether, our study uncovered that MMP7 was a critical effector in mediating the oncogenic roles of BCAR4 in glioma.
Discussion
The present study described the essential roles of the lncRNA BCAR4 in gliomagenesis. Firstly, BCAR4 was confirmed to be overexpressed in glioma tissues and GBM cell lines, and its higher expression was positively associated with the advanced glioma grades and worse prognosis of the patients. In vitro and in vivo loss-of-function assays revealed that BCAR4 knockdown inhibited GBM cell growth, invasion and tumor formation significantly. Importantly, we identified a novel molecular mechanism that BCAR4 acted as a ceRNA by targeting miR-2276 to upregulate MMP7 expression in glioma cells (Figure 6). Finally, functional assays demonstrated that MMP7 played an essential role in the oncogenic roles of BCAR4 in glioma. Taken together, our findings illustrated that the newly disclosed BCAR4/miR-2276/MMP7 axis-facilitated gliomagenesis and could be developed as a novel therapeutic target for glioma treatment.
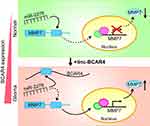 |
Figure 6 Schematic illustration of the molecular pathway in which BCAR4/miR-2276/MMP7 forms the ceRNA relationship in glioma cells. |
Along with the research goes on, lncRNAs are widely recognized as promising diagnostic and prognostic biomarkers in glioma.26 For instance, lncRNA HOTAIR was found to be increased in GBM patients’ serum and it might be applied as an effective diagnostic and prognostic biomarker for glioma patients.27 Herein, we validated that BCAR4 was overexpressed in glioma, keeping in accordance with the previous study by Wei et al.21 More importantly, we found that its higher expression positively correlated with advanced glioma grades and worse clinical outcome of the patients. These findings verified the potential clinical significance of BCAR4 in predicting the prognosis of glioma patients.
There are also amounting evidences that lncRNA are vital contributors for glioma pathogenesis and progression.26 For BCAR4, Wei et al have proved that BCAR4 could promote glioma cell proliferation.21 Its biological functions have also been investigated in other types of cancers, including in chondrosarcoma and lung cancer, in which BCAR4 facilitates cancer cell migration, invasion and EMT.22,23 In this study, we presented valid evidences to support that BCAR4 facilitated GBM cell growth, invasion and tumorigenicity, fully expanding its oncogenic roles in glioma.
Among the known molecular mechanisms of lncRNAs, ceRNA attracts intensive attention recently. Through targeting miRNAs, lncRNAs could positively regulate the expression of the downstream targets and exert their functions.12–14 For BCAR4, it has been demonstrated that it could promote the stemness of colorectal cancer cells through targeting to miR-665/STAT3 signaling.20 Herein, we identified BCAR4 as the sponge for miR-2276 in glioma. Up to date, there are no reports regarding the roles of miR-2276 in human cancers. It has merely been mentioned that miR-2276 was decreased in breast cancer samples, especially in those at higher grades and with lymph node metastasis.28 Our results showed that miR-2276 could be induced by BCAR4 knockdown, and it was downregulated in gliomas and exhibiting inverse correlation with BCAR4 expression. Subsequently, we further found that MMP7 was a direct target of miR-2276 in GBM cells. Since MMP7 is known to be highly involved in gliomagenesis,25 we focused on the regulation of MMP7 by miR-2276 and BCAR4. The results confirmed that MMP7 was downregulated by miR-2276 whereas upregulated by BCAR4 in GBM cells, demonstrating the existence of the BCAR4/miR-2276/MMP7 axis in glioma. Importantly, MMP7 restoration effectively rescued the impaired GBM cell growth and invasion caused by BCAR4 knockdown. Till now, we proved that MMP7 was an essential downstream effector of BCAR4 in promoting gliomagenesis.
In summary, our results provide solid evidences into the oncogenic roles of BCAR4 via targeting miR-2276 to upregulate MMP7 in gliomagenesis. These findings may improve the understanding of the glioma pathogenesis and improve the clinical prognosis and therapy for the patients.
Ethical Approval and Written Informed Consent
All procedures performed in this study involving human participants were in accordance with the 1964 Helsinki declaration. Written informed consent was obtained from all patients enrolled in this study.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.v68.6
2. Wen PY, Kesari S. Malignant gliomas in adults. N Engl J Med. 2008;359(5):492–507. doi:10.1056/NEJMra0708126
3. Clarke J, Butowski N, Chang S. Recent advances in therapy for glioblastoma. Arch Neurol. 2010;67(3):279–283. doi:10.1001/archneurol.2010.5
4. Tykocki T, Eltayeb M. Ten-year survival in glioblastoma. A systematic review. J Clin Neurosci. 2018;54:7–13. doi:10.1016/j.jocn.2018.05.002
5. Chan JJ, Tay Y. Noncoding RNA:RNA regulatory networks in cancer. Int J Mol Sci. 2018;19(5):E1310. doi:10.3390/ijms19051310
6. Peng S, Cao L, He S, et al. An overview of long noncoding RNAs involved in bone regeneration from mesenchymal stem cells. Stem Cells Int. 2018;2018:8273648. doi:10.1155/2018/8273648
7. Tang Q, Hann SS. HOTAIR: an oncogenic long non-coding RNA in human cancer. Cell Physiol Biochem. 2018;47(3):893–913. doi:10.1159/000490131
8. Wu Y, Shao A, Wang L, et al. The role of lncRNAs in the distant metastasis of breast cancer. Front Oncol. 2019;9:407. doi:10.3389/fonc.2019.00407
9. Lu T, Wang Y, Chen D, Liu J, Jiao W. Potential clinical application of lncRNAs in non-small cell lung cancer. Onco Targets Ther. 2018;13(11):8045–8052. doi:10.2147/OTT.S178431
10. Li ZX, Zhu QN, Zhang HB, Hu Y, Wang G, Zhu YS. MALAT1: a potential biomarker in cancer. Cancer Manag Res. 2018;10:6757–6768. doi:10.2147/CMAR.S169406
11. Li J, Zhu Y, Wang H, Ji X. Targeting long noncoding RNA in glioma: a pathway perspective. Mol Ther Nucleic Acids. 2018;13:431–441. doi:10.1016/j.omtn.2018.09.023
12. Sen R, Ghosal S, Das S, Balti S, Chakrabarti J. Competing endogenous RNA: the key to posttranscriptional regulation. ScientificWorldJournal. 2014;2014:896206. doi:10.1155/2014/896206
13. Ergun S, Oztuzcu S. Oncocers: ceRNA-mediated cross-talk by sponging miRNAs in oncogenic pathways. Tumour Biol. 2015;36(5):3129–3136. doi:10.1007/s13277-015-3346-x
14. Yang C, Wu D, Gao L, et al. Competing endogenous RNA networks in human cancer: hypothesis, validation, and perspectives. Oncotarget. 2016;7(12):13479–13490. doi:10.18632/oncotarget.7266
15. Huang J, Lyu H, Wang J, Liu B. MicroRNA regulation and therapeutic targeting of survivin in cancer. Am J Cancer Res. 2014;5(1):20–31.
16. Qu J, Li M, Zhong W, Hu C. Competing endogenous RNA in cancer: a new pattern of gene expression regulation. Int J Clin Exp Med. 2015;8(10):17110–17116.
17. Shuwen H, Qing Z, Yan Z, Xi Y. Competitive endogenous RNA in colorectal cancer: a systematic review. Gene. 2018;645:157–162. doi:10.1016/j.gene.2017.12.036
18. Ju L, Zhou YM, Yang GS. Up-regulation of long non-coding RNA BCAR4 predicts a poor prognosis in patients with osteosarcoma, and promotes cell invasion and metastasis. Eur Rev Med Pharmacol Sci. 2016;20(21):4445–4451.
19. Zou R, Chen X, Jin X, et al. Up-regulated BCAR4 contributes to proliferation and migration of cervical cancer cells. Surg Oncol. 2018;27(2):306–313. doi:10.1016/j.suronc.2018.05.013
20. Ouyang S, Zhou X, Chen Z, Wang M, Zheng X, Xie M. LncRNA BCAR4, targeting to miR-665/STAT3 signaling, maintains cancer stem cells stemness and promotes tumorigenicity in colorectal cancer. Cancer Cell Int. 2019;19:72. doi:10.1186/s12935-019-0784-3
21. Wei L, Yi Z, Guo K, Long X. Long noncoding RNA BCAR4 promotes glioma cell proliferation via EGFR/PI3K/AKT signaling pathway. J Cell Physiol. 2019;234:23608–23617. doi:10.1002/jcp.28929
22. Shui X, Zhou C, Lin W, Yu Y, Feng Y, Kong J. Long non-coding RNA BCAR4 promotes chondrosarcoma cell proliferation and migration through activation of mTOR signaling pathway. Exp Biol Med (Maywood). 2017;242(10):1044–1050. doi:10.1177/1535370217700735
23. Yang H, Yan L, Sun K, et al. lncRNA BCAR4 increases viability, invasion, and migration of non-small cell lung cancer cells by targeting glioma-associated oncogene 2 (GLI2). Oncol Res. 2019;27(3):359–369. doi:10.3727/096504018X15220594629967
24. Paraskevopoulou MD, Vlachos IS, Karagkouni D, et al. DIANA-LncBase v2: indexing microRNA targets on non-coding transcripts. Nucleic Acids Res. 2016;44(D1):D231–D238. doi:10.1093/nar/gkv1270
25. Mu N, Gu J, Liu N, et al. PRL-3 is a potential glioblastoma prognostic marker and promotes glioblastoma progression by enhancing MMP7 through the ERK and JNK pathways. Theranostics. 2018;8(6):1527–1539. doi:10.7150/thno.22699
26. Peng Z, Liu C, Wu M. New insights into long noncoding RNAs and their roles in glioma. Mol Cancer. 2018;17(1):61. doi:10.1186/s12943-018-0812-2
27. Tan SK, Pastori C, Penas C, et al. Serum long noncoding RNA HOTAIR as a novel diagnostic and prognostic biomarker in glioblastoma multiforme. Mol Cancer. 2018;17(1):74. doi:10.1186/s12943-018-0822-0
28. Torkashvand S, Damavandi Z, Mirzaei B, Tavallaei M, Vasei M, Mowla SJ. Decreased expression of bioinformatically predicted piwil2-targeting microRNAs, miR-1267 and miR-2276 in breast cancer. Arch Iran Med. 2016;19(6):420–425.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.