Back to Journals » Journal of Inflammation Research » Volume 13
LncRNA SOX2OT rs9839776 Polymorphism Reduces Sepsis Susceptibility in Southern Chinese Children
Authors Wu Z, Yu Y , Fu L, Mai H , Huang L, Che D, Tao J, Gu X
Received 14 September 2020
Accepted for publication 18 November 2020
Published 10 December 2020 Volume 2020:13 Pages 1095—1101
DOI https://doi.org/10.2147/JIR.S281760
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Zhiyuan Wu,1,* Yongqin Yu,1,* Lanyan Fu,2 Hanran Mai,2 Li Huang,1 Di Che,2 Jianping Tao,1 Xiaoqiong Gu2– 4
1Department of Pediatric Intensive Care Unit, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, People’s Republic of China; 2Department of Clinical Biological Resource Bank, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, People’s Republic of China; 3Department of Blood Transfusion, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, People’s Republic of China; 4Department of Clinical Laboratory, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaoqiong Gu; Jianping Tao Email [email protected]; [email protected]
Background: Sepsis in children is one of the main causes of death in pediatric intensive care units (PICUs); however, the pathogenesis of sepsis is not fully clear. Previous studies revealed that many genetic variations were related to sepsis susceptibility. A long non-coding RNA SOX2 overlapping transcript (SOX2OT) may play a role in mitochondrial homeostasis and antioxidative activity, but the relationship between the lncRNA SOX2OT polymorphism and sepsis susceptibility has not been reported.
Methods: In this study, 474 pediatric sepsis patients and 678 healthy controls were recruited from southern China. After genotyping, the strength of the associations was evaluated through odds ratios (ORs) and 95% confidence intervals (CIs).
Results: The SOX2OT rs9839776 T allele was associated with decreased susceptibility to sepsis in southern Chinese children (TT/CT vs CC adjusted OR = 0.778, 95% CI = 0.610– 0.992; P = 0.0431). Moreover, the difference in susceptibility was greater in children of age > 60 months (adjusted OR = 0.458, 95% CI = 0.234– 0.896; P = 0.0225), survivors (adjusted OR = 0.758, 95% CI = 0.585– 0.972; P = 0.0358), males (adjusted OR = 0.655, 95% CI = 0.479– 0.894; P = 0.0077) and the sepsis subgroup (adjusted OR = 0.548, 95% CI = 0.343– 0.876; P = 0.0120).
Conclusion: The rs9839776 T allele may contribute to decreased sepsis risk in Chinese children. Future studies with a larger sample size are needed to verify these results.
Keywords: sepsis, SOX2OT, rs9839776, susceptibility, polymorphism
Introduction
Sepsis, severe organ dysfunction due to the body’s maladaptive response to infection, is one of the main causes of death in pediatric intensive care units (PICUs). According to disease severity, sepsis patients can be divided into three groups: sepsis, severe sepsis, and septic shock.1 Although sepsis treatment in the PICU has been improved by advanced mechanical ventilation and extracorporeal membrane oxygenation (ECMO), curing pediatric sepsis patients once severe sepsis or septic shock develops remains a difficult task; the mortality rate has ranged from 10 to 20% during the past few decades in the PICU.2,3 The World Health Organization (WHO) estimated that one million people die each year from neonatal sepsis.4 Therefore, early diagnosis and prognosis improvements have become a challenging subject in clinical work. Multiple factors influence sepsis in children, including pathogen infection, immature inflammatory responses and immunosuppression. In recent years, some studies have focused on the relationship between genetic variation and the incidence of sepsis in children. For example, the genetic variant rs16944 in the IL1B gene and the genetic variant rs1205 in the C-reactive protein gene have been confirmed as risk factors in infant patients.5,6 These previous studies indicate that additional genes may influence the mechanism of sepsis development in children, but relevant studies are limited.
Long noncoding RNAs (lncRNAs) are defined as RNA molecules longer than 200 nucleotides with little or no translation capacity. Although lncRNAs do not encode proteins, they can affect a variety of biological processes, including chromatin modification, alternative splicing, and mRNA stability.7 LncRNAs have been shown to be associated with various diseases, such as inflammatory diseases and tumors, by changing the epigenetic regulation and cellular processes.8,9 Several studies have revealed that some lncRNAs, such as those in MALAT1, NEAT1, and HOTAIR, contribute to the development of sepsis in different ethnic groups.10–12 According to these studies, some lncRNAs can play a role in modifying susceptibility to sepsis and even predict prognosis. For instance, SOX2OT was reported to be correlated with the expression of the SRY-box 2 gene, and overexpression of these transcripts may promote cell proliferation.13 In a previous study, Mengfei Chen reported that SOX2OT contributed to the progression of mitochondrial dysfunction by inhibiting SOX2 expression in septic cardiomyopathy.14 The expression of SOX2OT is also reportedly related to retinal degeneration, non-small cell lung cancer and central nervous system diseases.15–17 In recent years, many studies have concentrated on the association between cytokine and protein single nucleotide polymorphisms (SNPs) and sepsis.5,18 However, the relationship between lncRNA SNPs and sepsis has not been reported. Therefore, whether the lncRNA SOX2OT polymorphism can affect sepsis is unclear.
This study aimed to identify the association between the lncRNA SOX2OT polymorphism and sepsis. We chose the rs9839776 polymorphism of the SOX2OT gene in a case-control study including 474 cases and 678 controls from a population of children in southern China.
Materials and Methods
Study Population
A total of 474 sepsis patients and 678 healthy controls were recruited at the Guangzhou Women and Children’s Medical Center between January 2016 and December 2018. Sepsis patients were diagnosed according to the International Sepsis Definitions Conference in 2012, and the sepsis, severe sepsis and septic shock groups were classified according to international guidelines.1 This study was approved by the Ethics Committee of Guangzhou Women and Children’s Medical Center (Permission number:2,015,042,202) and based on the guidelines of the Declaration of Helsinki. Each parent or guardian of the patients and control subjects provided informed consent for study participation. Personal information, clinical data, and demographic information were collected from the hospital inpatient record system.
Genotyping and DNA Extraction
Peripheral blood was collected in an EDTA tube from each sepsis patient and control. Following the guidance of the manufacturer’s instructions, genomic DNA was extracted with a Blood DNA Isolation Kit (Tiangen, Beijing, China), and then we stored these samples in the −80°C equipment before genotyping. A 384-well plate on an ABI Q6 instrument (Thermo Fisher Scientific, United States) was used for amplifying DNA samples according to the protocol of the TaqMan real-time polymerase chain reaction protocol. Specific fluorescent probes labeled with VIC or FAM for rs9839776, which can differentiate wild-type and variant alleles, were purchased from ABI (ThermoFisher Scientific, United States). The PCR reaction system include 2 × Genotyping PreMix, PCR primer pool and template DNA. The total reaction volume of each sample was 5 mL. The qPCR reaction step were as follows: predeformation needs 95°C for 2 min, then 40 cycles of 95°C for 15 s and 60°C for 30 s were set for amplify, and 60°C for 5 min for extension sufficiently.
Statistical Analysis
The data analysis was performed using SAS software (version 9.1; SAS Institute, Cary, NC). We calculated the odds ratios (ORs) and 95% confidence intervals (CIs) to evaluate the association between the SOX2OT gene polymorphism (rs9839776) and sepsis susceptibility. In addition, multivariate unconditional logistic regression analysis was used to calculate the adjusted ORs after adjustment for age and sex. Furthermore, analyses stratified by age, sex, sepsis subtype, prognosis and number of organs with dysfunction were performed. All statistical analyses were performed using a two-sided chi square test, and a P-value less than 0.05 was regarded as statistically significant.
Results
Population Characteristics
As shown in Table 1, age, sex, sepsis subgroup prognosis and the number of organs with dysfunction were assessed in our total sample of 1152 children, including 474 sepsis patients and 678 healthy controls. No difference in age (35.04 ± 34.26 months vs 35.53 ± 29.37 months, P = 0.1811) or sex (P = 0.111) was found between the two groups. Among the sepsis cases, 98 children (20.68%) were diagnosed with sepsis, 291 children (61.39%) were diagnosed with severe sepsis, and 85 children (17.93%) were diagnosed with septic shock. In addition, based on the number of organs with dysfunction, 276 children (58.23%) had 1–2 organs with dysfunction, while 95 children (20.04%) had 3 or more organs with dysfunction.
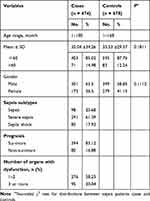 |
Table 1 Frequency Distribution of Selected Characteristics in Sepsis Cases and Healthy Controls |
Association Between SOX2OT Gene Polymorphism and Sepsis Susceptibility
As shown in Table 2, the independent correlation between the SOX2OT genotype frequency distribution of the SNP rs9839776 C>T and sepsis was assessed by the goodness-of-fit chi square test. The rs9839776 C>T polymorphism frequencies followed the Hardy-Weinberg equilibrium (PHWE = 0.5275). The genotype analysis of the lncRNA SOX2OT polymorphism revealed a significant difference in the carriers of the rs9839776 genotypes exhibiting TT/CT allele (adjusted OR = 0.778, 95% CI = 0.610–0.992, and P = 0.0431), suggesting that rs9839776 TT/CT genotypes decrease sepsis susceptibility.
 |
Table 2 Genotype Frequency Distribution of SOX2OT in Sepsis Cases and Healthy Controls |
Stratified Analysis of the Selected Polymorphism and Sepsis
As shown in Table 3, we further explored the association between the lncRNA SOX2OT rs9839776 polymorphism and susceptibility to sepsis stratified by age, sex, sepsis subtype, prognosis and number of organs with dysfunction. After adjusting for age and sex, we observed that compared with the rs9839776 CC allele, the TT/CT alleles were more predominant for children > 60 month for age (adjusted OR = 0.458, 95% CI = 0.234–0.896; P = 0.0225). The TT/CT alleles also are a protective factor in males (adjusted OR = 0.655, 95% CI = 0.479–0.894; P = 0.0077). For the sepsis subgroup, the TT/CT variant alleles were associated with a decreased risk of sepsis (adjusted OR = 0.548, 95% CI = 0.343–0.876; P = 0.0120); however, no significant association was found between severe sepsis, septic shock and the TT/CT variant genotypes. Moreover, our stratified analyses revealed that among the survivors who suffered from sepsis, the TT/CT variant had a protective effect (adjusted OR = 0.758, 95% CI = 0.585–0.972; P = 0.0358). Significant associations were not found for the other stratifications by age, prognosis and number of organs with dysfunction.
 |
Table 3 Stratification Analysis of Susceptibility in Sepsis Patients |
Discussion
Recently, some genetic factors were reported to closely affect the sepsis development; however, the association of genetic variations in SOX2OT with sepsis susceptibility among children is not fully known. A previous study claimed that SOX2OT can mediate mitochondrial dysfunction during inflammation, indicating the potential mechanism underlying the pathophysiology of sepsis.14 Nonetheless, the relationship between sepsis and SOX2OT in children remains unclear. We chose rs9839776 as our study object to design a case-control study and investigated whether the rs9839776 C>T variant genotypes can reduce sepsis risk in southern Chinese children.
In this case study, we compared the blood samples between 474 sepsis patients and 678 healthy controls via qPCR analysis and chi square testing. We verified that the lncRNA SOX2OT rs9839776 T allele is associated with a decreased risk of sepsis in children and that the protective effect is more obvious in males and the age more than 60 months. Furthermore, the stratified analysis revealed that the rs9839776 T allele is a protective factor during early stages of sepsis in the subgroups. To the best of our knowledge, our study is the first to investigate the association between the lncRNA SOX2OT gene polymorphism (rs9839776) and sepsis susceptibility in southern Chinese children.
The SOX2OT gene is located in the 3q26.3–q27 locus of the human chromosome and harbors the SOX2 gene, extending in a highly conserved region of over 700 kb that is one of the major regulators of pluripotency.19,20 Marjan E indicated that SOX2OT plays an important role in the induction and maintenance of SOX2 expression in breast cancer.21 A previous study identified that lncRNA SOX2OT expression in non-small cell lung cancer can act as a biomarker and could be used as an independent prognostic factor.15 The study by Xiaomin Shi demonstrated that SOX2OT upregulation promotes metastasis in hepatocellular carcinoma cells and leads to worse outcomes.22 Moreover, SOX2OT SNP was reported to increase the susceptibility to the development of some diseases. For example, V Boraska clarified that in a genome-wide association study, rs9839776 in SOX2OT is associated with anorexia nervosa susceptibility.23 Xiuwu Tang reported that rs9839776 increases the risk of breast cancer in Chinese women.24 A recent study conducted by our group revealed that an increased risk of recurrent miscarriage in the southern Chinese population can be attributed to the rs9839776 CT genotype, which may act as a prognostic biomarker in recurrent miscarriage patients.25 Therefore, we speculate that the rs9839776 polymorphism has different functions in different diseases. In this study, we found that this variant can decrease the risk of sepsis in southern Chinese children.
Previous studies have analyzed lncRNA SOX2OT expression. For example, a higher expression level of SOX2OT mRNA was identified in somatic cancers such as breast cancer, lung squamous cell carcinoma and osteosarcoma.19,26 In the Mina study, SOX2OT was overexpressed in breast cancer tissues and correlated with breast cancer susceptibility;27 however, research on SOX2OT is mostly limited to the area of oncology, and the role of lncRNA in inflammatory diseases remains largely unknown. Huyu Lin reported that LncRNA SOX2OSOX2OT downregulation can suppress the proliferation, apoptosis, oxidative stress, and inflammatory respond of vascular smooth muscle cell through regulating miR-145.28 Moreover, Chao-Peng Li speculated that SOX2OT knockdown can protect retinal ganglion cells against high glucose-induced injury in mice and has an antioxidative function by regulating NRF2/HO-1 signaling activity.17 Mengfei Chen reported that upregulated SOX2OT expression in LPS-induced septic cardiomyopathy and downregulated expression in cardiomyocytes could restore mitochondrial homeostasis.14 High glucose levels are a risk factor in sepsis and mitochondrial dysfunction is involved in organ dysfunction of sepsis.29 These researches indicated that SOX2OT expression level is related to sepsis development. Based on a previous study, we hypothesized that SOX2OT is a detrimental factor and that the rs9839776 polymorphism alters the expression of SOX2OT in sepsis. We posited that among children with sepsis, those with the rs9839776 CT/TT allele may have a different survival rate than those with the CC allele. The observed improved survival rate among CT/TT allele patients confirmed our hypothesis. Prognosis prediction and reducing the fatality rate remains an important problem in the context of sepsis. Our findings can provide a new viewpoint to solve this problem, and further research in this regard is warranted.
Although this study is the first to report the relationship between lncRNA SOX2OT and sepsis risk in southern Chinese children, a number of limitations should be explained. First, a case-control study was performed to evaluate the association between lncRNA SOX2OT and sepsis risk in southern Chinese children. We did not detect SOX2OT expression in sepsis patients, and a definite mechanism underlying this polymorphism was not explored. Second, our study referred only to the SOX2OT rs9839776 polymorphism. The SOX2OT gene is largely polymorphic, is harbored in the SOX2 gene, and extends in a highly conserved region of over 700 kb; thus, other SNPs potentially influence the expression of SOX2OT. Potential chain SNPs based on the linkage disequilibrium theory should be considered in future studies. Third, this retrospective study included only southern Chinese children. Gene polymorphisms are diverse, and therefore our results cannot be generalized to other ethnicities simply. Finally, our study sample was not sufficiently large, and a larger sample size is needed in future studies.
In conclusion, this study demonstrated that the SOX2OT rs9839776 T variant is a protective factor associated with sepsis in Chinese children. The protective effect was reflected more substantially in children of age > 60 months, survivors and male patients. Moreover, the protective effect was pronounced in the sepsis subgroup compared to the severe sepsis and septic shock subgroups. The association between the SOX2OT gene variant and sepsis should be validated in a larger sample size and in different ethnicities in future studies.
Acknowledgments
The authors acknowledge the Clinical Biological Resource Bank of Guangzhou Women and Children’s Medical Center for providing all the clinical samples.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
This study was funded by the Guangdong Provincial Science and Technology Plan (China, 2017A030223003), Guangzhou Medical and Health Technology Projects (China, 20191A011033 and 20191A011021), Guangzhou Science and Technology Program Key Projects (China, 201904010486 and 201804010035), Guangdong Natural Science Foundation (China, 2019A1515012061), and Guangzhou Institute of Pediatrics/Guangzhou Women and Children’s Medical Center Fund (China, grant number YIP-2018-019, GCP-2019-003, YIP-2019-050, GCP-2019-006).
Disclosure
The authors have no conflicts of interest to declare.
References
1. Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med. 2013;39:165–228. doi:10.1007/s00134-012-2769-8.
2. Emr BM, Alcamo AM, Carcillo JA, Aneja RK, Mollen KP. Pediatric sepsis update: how are children different? Surg Infect. 2018;19:176–183. doi:10.1089/sur.2017.316.
3. Ruth A, McCracken CE, Fortenberry JD, Hall M, Simon HK, Hebbar KB. Pediatric severe sepsis. Pediatr Crit Care Me. 2014;15:828–838. doi:10.1097/PCC.0000000000000254.
4. Edmond K, Zaidi A. New approaches to preventing, diagnosing, and treating neonatal sepsis. PLoS Med. 2010;7:e1000213. doi:10.1371/journal.pmed.1000213.
5. Varljen T, Sekulovic G, Rakic O, et al. Genetic variant rs16944 in IL1B gene is a risk factor for early-onset sepsis susceptibility and outcome in preterm infants. Inflamm Res. 2020;69:155–157. doi:10.1007/s00011-019-01301-4.
6. Liu C, Jin P, Luo Y, et al. Association of single-nucleotide polymorphisms of C-reactive protein gene with susceptibility to infantile sepsis in Southern China. Med Sci Monit. 2018;24:590–595. doi:10.12659/MSM.908602.
7. Guttman M, Rinn JL. Modular regulatory principles of large non-coding RNAs. Nature. 2012;482:339–346. doi:10.1038/nature10887.
8. Boeckel J, Jaé N, Heumüller AW, et al. Identification and characterization of hypoxia-regulated endothelial circular RNA. Circ Res. 2015;117:884–890. doi:10.1161/CIRCRESAHA.115.306319.
9. Zacharopoulou E, Gazouli M, Tzouvala M, Vezakis A, Karamanolis G. The contribution of long non-coding RNAs in inflammatory bowel diseases. Digest Liver Dis. 2017;49:1067–1072. doi:10.1016/j.dld.2017.08.003.
10. Chen J, Gu X, Zhou L, et al. Long non-coding RNA-HOTAIR promotes the progression of sepsis by acting as a sponge of miR-211 to induce IL-6R expression. Exp Ther Med. 2019;18:3959–3967. doi:10.3892/etm.2019.8063.
11. Chen H, Wang X, Yan X, Cheng X, He X, Zheng W. LncRNA MALAT1 regulates sepsis-induced cardiac inflammation and dysfunction via interaction with miR-125b and p38 MAPK/NFκB. Int Immunopharmacol. 2018;55:69–76. doi:10.1016/j.intimp.2017.11.038.
12. Zhang C, Niu F. LncRNA NEAT1 promotes inflammatory response in sepsis-induced liver injury via the Let-7a/TLR4 axis. Int Immunopharmacol. 2019;75:105731. doi:10.1016/j.intimp.2019.105731.
13. Han L, Zhang W, Zhang B, Zhan L. Long non-coding RNA SOX2OT promotes cell proliferation and motility in human ovarian cancer. Exp Ther Med. 2018;15:2182–2188. doi:10.3892/etm.2017.5618.
14. Chen M, Guan Y, Li A, et al. LncRNA SOX2OT mediates mitochondrial dysfunction in septic cardiomyopathy. DNA Cell Biol. 2019;38:1197–1206. doi:10.1089/dna.2019.4839.
15. Xie Y, Zhang Y, Du L, et al. Circulating long noncoding RNA act as potential novel biomarkers for diagnosis and prognosis of non-small cell lung cancer. Mol Oncol. 2018;12:648–658. doi:10.1002/1878-0261.12188.
16. Qureshi IA, Mattick JS, Mehler MF. Long non-coding RNAs in nervous system function and disease. Brain Res. 2010;1338:20–35. doi:10.1016/j.brainres.2010.03.110.
17. Li C, Wang S, Wang W, Song S, Liu X. Long noncoding RNA-Sox2OT knockdown alleviates diabetes mellitus-induced Retinal Ganglion Cell (RGC) injury. Cell Mol Neurobiol. 2017;37:361–369. doi:10.1007/s10571-016-0380-1.
18. Shao Y, Zhao T, Zhang W, et al. Presence of the apolipoprotein E-epsilon4 allele is associated with an increased risk of sepsis progression. Sci Rep. 2020;10(1):15735. doi:10.1038/s41598-020-72616-0.
19. Shahryari A, Jazi MS, Samaei NM, Mowla SJ. Long non-coding RNA SOX2OT: expression signature, splicing patterns, and emerging roles in pluripotency and tumorigenesis. Front Genet. 2015;6. doi:10.3389/fgene.2015.00196.
20. Fantes J, Ragge NK, Lynch S, et al. Mutations in SOX2 cause anophthalmia. Nat Genet. 2003;33:462–463. doi:10.1038/ng1120.
21. Askarian-Amiri ME, Seyfoddin V, Smart CE, et al. Emerging role of long non-coding RNA SOX2OT in SOX2 regulation in breast cancer. PLoS One. 2014;9:e102140. doi:10.1371/journal.pone.0102140.
22. Shi XM, Teng F. Up-regulation of long non-coding RNA Sox2ot promotes hepatocellular carcinoma cell metastasis and correlates with poor prognosis. Int J Clin Exp Pathol. 2015;8:4008–4014.
23. Boraska V, Franklin CS, Floyd JAB, et al. A genome-wide association study of anorexia nervosa. Mol Psychiatr. 2014;19:1085–1094. doi:10.1038/mp.2013.187.
24. Tang X, Gao Y, Yu L, et al. Correlations between lncRNA-SOX2OT polymorphism and susceptibility to breast cancer in a Chinese population. Biomark Med. 2017;11:277–284. doi:10.2217/bmm-2016-0238.
25. Fang Z, Che D, Qing S, et al. The lncRNA SOX2OT rs9839776 C>T polymorphism indicates recurrent miscarriage susceptibility in a Southern Chinese population. Media Inflamm. 2019;2019:1–6. doi:10.1155/2019/9684703.
26. Wang Z, Tan M, Chen G, Li Z, Lu X. LncRNA SOX2-OT is a novel prognostic biomarker for osteosarcoma patients and regulates osteosarcoma cells proliferation and motility through modulating SOX2. Iubmb Life. 2017;69:867–876. doi:10.1002/iub.1681.
27. Mina D, Naiemeh H, Hamidreza K, et al. Correlations between overexpression of SOX2OT long non-coding RNA and susceptibility to breast cancer. Accel Technol Biotechnol Bioassays Med Chem Nat Prod Res. 2020;23:1–7. doi:10.2174/1386207323666200514075042.
28. Lin H, You B, Lin X, et al. Silencing of long non-coding RNA Sox2ot inhibits oxidative stress and inflammation of vascular smooth muscle cells in abdominal aortic aneurysm via microRNA-145-mediated Egr1 inhibition. Aging (Albany NY). 2020;12(13):12684–12702. doi:10.18632/aging.103077.
29. Ameziane-El-Hassani R, Dupuy C. Detection of reactive oxygen species in cells undergoing oncogene-induced senescence. Methods Mol Biol. 2017;1534:139–145. doi:10.1007/978-1-4939-6670-7_13.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
