Back to Journals » Cancer Management and Research » Volume 11
LncRNA LOC105372579 promotes proliferation and epithelial-mesenchymal transition in hepatocellular carcinoma via activating miR-4316/FOXP4 signaling
Authors E C, Yang J, Li H, Li C
Received 12 December 2018
Accepted for publication 25 February 2019
Published 11 April 2019 Volume 2019:11 Pages 2871—2879
DOI https://doi.org/10.2147/CMAR.S197979
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Ahmet Emre Eşkazan
Changyong E,1 Jinghui Yang,1 Hang Li,2 Chunsheng Li3
1Department of Hepatobiliary and Pancreatic Surgery, China-Japan Union Hospital of Jilin University, Changchun 130033, People’s Republic of China; 2Department of Cerebral Surgery, Jilin Cancer Hospital, Changchun 130000, People’s Republic of China; 3Department of Gastrointestinal Colorectal and Anal surgery, China-Japan Union Hospital of Jilin University, Changchun 130033, People’s Republic of China
Background: Recently, a growing number of long noncoding RNAs (lncRNAs) have been identified to be important for human cancer development. However, how lncRNA regulates hepatocellular carcinoma (HCC) progression still remains largely unclear. We aimed to investigate the function of LOC105372579 in HCC progression.
Materials and methods: The expression levels of lncRNA LOC105372579 in HCC tissues and cell lines were analyzed by qRT-PCR. The effects of LOC105372579 silencing on proliferation, migration and invasion were determined by using cell counting kit-8, colony formation assay and Transwell assay. Moreover, the xenograft mouse model was used to detect how LOC105372579 regulates HCC growth in vivo.
Results: LOC105372579 was highly expressed in HCC tissues and cell lines. Moreover, upregulated levels of LOC105372579 predicted poor prognosis. LOC105372579 silencing suppressed the proliferation of HCC cells in vitro. We also validated that LOC105372579 knockdown inhibited the migration, invasion, and epithelial–mesenchymal transition of HCC cells. Xenograft assay demonstrated that LOC105372579 promotes tumor growth in vivo. Mechanistically, we identified that LOC105372579 is a sponge for miR-4316 and that FOXP4 is a direct target of miR-4316.
Conclusion: Thus, our findings supported that LOC105372579 contributes to HCC cell proliferation, migration, invasion, and EMT by activating miR-4316/FOXP4 signaling.
Keywords: LOC105372579, HCC, proliferation, EMT, progression
Corrigendum for this paper has been published.
Introduction
Hepatocellular carcinoma (HCC) is one of the most prevalent cancers and the leading cause of cancer-related deaths in China.1 In previous years, the number of newly diagnosed patients with HCC has been increased. Although improvements were achieved on therapeutic strategies, the outcomes of patients with HCC still remain unsatisfactory.2,3 The 5-year survival rate among patients with HCC is under 30%.4 Thus, understanding the underlying mechanism of HCC is an urgent requirement.
Long noncoding RNAs (lncRNAs) is a subgroup of noncoding RNAs with a length of over 200 nucleotides.5 LncRNAs cannot code proteins and exert functions through multiple mechanisms, such as miRNA sponges and remodeling DNA structure.6,7 Emerging evidence has shown that lncRNAs can work as oncogenes or tumor suppressors to regulate malignant behavior in cancers, including HCC.8,9 The aberrant expression of lncRNAs is often observed in various cancers.10,11 For example, lncRNA OIP5-AS1 is upregulated in bladder cancer, indicates poor prognosis, and promotes tumor cell growth while inhibiting apoptosis.12 LncRNA LINC00511 upregulation in breast cancer facilitates tumor cell proliferation through the miR-185-3p/E2F1/Nanog axis.13 In addition, increased expression of LINC00707 promotes HCC cell proliferation, migration, and invasion by enhancing the ERK/JNK/AKT pathway.14 Despite the essential roles of lncRNAs, how they regulate hepatocarcinogenesis has not been clearly elucidated.
LOC105372579 is a functionally undefined novel lncRNA. We found that LOC105372579 was upregulated in HCC tissues compared with normal controls through bioinformatics analysis, implying that it may be involved in HCC progression. Thus, the aim of our study is to determine the role of LOC105372579 in HCC and elucidate its functional mechanism. We performed loss-of-function assays and demonstrated that LOC105372579 promotes HCC progression through the miR-4316/FOXP4 axis.
Materials and methods
Tissue specimens
Fifty-four HCC samples and their adjacent normal tissues were obtained from the China-Japan Union Hospital of Jilin University. All these tissues were immediately stored in liquid nitrogen after collection. Association between the clinicopathologic characteristics and LOC105372579 expression in HCC was contained in Table 1. This study was approved by the ethics committee of the China-Japan Union Hospital of Jilin University. Written informed consent was collected from each patient. Experiments involving human tissues were conducted in accordance with the Declaration of Helsinki.
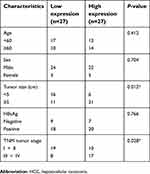 | Table 1 Association between the clinicopathologic characteristics and LOC105372579 expression in HCC |
Cell culture
HCC cell lines (SMCC7721, HepG2, Hep3B, and Huh7) and normal liver cell line (LO2) were purchased from the American Type Culture Collection (ATCC, Manassas, VA) or the cell bank of the Chinese Academy of Sciences (Shanghai, China). Cells were cultured using Roswell Park Memorial Institute 1640 medium complemented with 10% fetal bovine serum (Gibco, Carlsbad, CA), 100 U/ml penicillin, and 100 mg/ml streptomycin.
Cell transfection
To obtain stable LOC105372579-silenced HCC cell lines, LOC105372579 shRNA (#1: 5ʹ-AGCTAGGAATACCTT-TTACAT-3ʹ; #2: 5ʹ-AGCCATTCTGCCTTTAGATAT-3ʹ) or scramble control (5ʹ-AATTCTCCGAACGTGTCAC-GT-3ʹ) was synthesized by Auragene Bioscience of China and cloned into the pLVX-tdTomato-Puro vector (Biowit, Shenzhen, China). miR-4316 mimics, miR-4316 inhibitors, and negative controls were purchased from GeneCopoeia, Inc. (Rockville, MD, USA) and transfected into cells using Lipofectamine® 2000 transfection reagent (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer’s protocol.
Bioinformatics analysis
miRDB (
Cell counting kit-8 (CCK8) assay
Cells were plated into 96-well plates and cultured for indicated days. Then, CCK8 solution (Dojindo Molecular Technologies, Tokyo, Japan) was added and incubated for 2 h. The absorbance at 450 nm was measured using a microplate reader (Bio-Tek, Winooski, VT).
Transwell assay
A total of 2×105 cells were added into the upper chamber (precoated with Matrigel for invasion assay; BD Biosciences, New Jersey, USA) with 200 µl serum-free medium. The lower chamber was filled with 600 µl complete medium. After culturing for 48 h, the cells in the upper chamber were removed, and those in the lower chamber were fixed with 4% paraformaldehyde and stained with 0.5% crystal violet. Then, cell numbers were counted using a light microscope.
Quantitative reverse transcription polymerase chain reaction (qRT-PCR)
Total RNAs were extracted from HCC tissues and cell lines by using TRIzol reagent (Invitrogen) according to the manufacturer’s instruction.15 qRT-PCR was performed based on a previously reported method.15
Luciferase activity assay
The wild-type (WT) or mutant (MUT) sequences of LOC105372579 and FOXP4 were inserted into the pGL3 Basic Vector (Promega, Madison, WI). For luciferase reporter assay, the reporter vector and miR-4316 mimics were co-transfected into HCC cells. After 48 h, the luciferase activity was measured using the Dual-Luciferase Reporter Assay System (Promega).
RNA pull-down assay
The microRNA was labeled with biotin by using the Pierce RNA 3′ End Desthiobiotinylation Kit (Thermo Fisher Scientific, Waltham, MA). Then, WT or MUT biotin-miR-4316 was incubated with HCC cell lysates and added with magnetic beads. Subsequently, the beads were collected and washed. Precipitated RNAs were also eluted and analyzed using qRT-PCR analysis.
Animal study
Four-week-old male BALB/c nude mice (Shanghai Animal Laboratory Center, Shanghai, China) were used for xenograft assay. In brief, LOC105372579-silenced or control HCC cells were injected in the right flank of nude mouse (5×106 each). Tumor volumes were measured every week. After 4 weeks, tumor weight was determined. This study was approved by the ethics committee of the China-Japan Union Hospital of Jilin University. All animal operations were performed in accordance with the Animal Policy and Welfare Committee of our hospital.
Statistical analysis
GraphPad Prism 6.0 (GraphPad Software, Inc., La Jolla, CA) was used for statistical analysis. The results were collected from three independent experiments and expressed as mean± SD. Student’s t-test or one-way analysis of variance was used for analysis of significant differences. P<0.05 was considered statistically significant.
Results
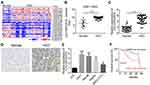
Increased expression of LOC105372579 in HCC tissues
To explore how lncRNA regulates HCC progression, we analyzed the GEO dataset (GSE113850) to search aberrantly expressed lncRNAs in HCC tissues (Figure 1A). As shown, LOC105372579 (probe ID: IVGNh09365) was an obviously upregulated lncRNA in HCC tissues (Figure 1A and B). To validate our results, we checked LOC105372579 expression in 54 HCC tissues and their matched normal controls by qRT-PCR. We confirmed that LOC105372579 expression was elevated in HCC tissues (Figure 1C), which was further validated by in situ hybridization (ISH) (Figure 1D). Consistently, the expression of LOC105372579 was also upregulated in HCC cell lines compared with LO2 cells (Figure 1E). To determine whether LOC105372579 can be a prognostic biomarker, we performed Kaplan–Meier curve analysis based on LOC105372579 expression. We divided these HCC samples into low and high expression groups according to the median value of LOC105372579. The results showed that LOC105372579 upregulation in patients with HCC indicated an unsatisfactory prognosis (Figure 1F). Thus, LOC105372579 might be involved in HCC progression.
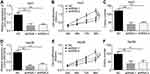
LOC105372579 silencing suppresses HCC cell proliferation
Next, we explored the function of LOC105372579 in HCC. We silenced LOC105372579 in Huh7 cells by using two independent shRNAs. qRT-PCR analysis validated the efficiency of LOC105372579 silencing in Huh7 cells (Figure 2A). CCK8 assay showed that LOC105372579 silencing suppressed the proliferation of Huh7 cells (Figure 2B). Moreover, colony formation assay indicated that LOC105372579 knockdown led to decreased colony number (Figure 2C). To further validate the role of LOC105372579, we utilized Hep3B cells. We knocked down LOC105372579 in Hep3B cells (Figure 2D). CCK8 and colony formation assay showed that LOC105372579 silencing inhibited the proliferation of Hep3B cells (Figure 2E and F).
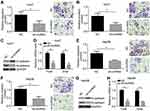
LOC105372579 knockdown inhibits migration, invasion, and epithelial–mese-nchymal transition (EMT)
Metastasis is a risk for HCC recurrence and malignance. Thus, we further evaluated the effects of LOC105372579 on HCC cells. Through Transwell assay, we found that LOC105372579 silencing significantly suppressed the proliferation and invasion of Huh7 cells (Figure 3A and B). Moreover, LOC105372579 knockdown impaired EMT in Huh7 cells (Figure 3C and D). Then, Hep3B cells were used to confirm our observation. We also found that the migration and invasion of Hep3B cells were attenuated by LOC105372579 silencing (Figure 3E and F). In addition, N-cadherin, Twist and Snail expression levels in Hep3B cells were decreased (Figure 3G and H), indicating that LOC105372579 silencing inhibits Hep3B EMT. Taken together, our results suggested that LOC105372579 knockdown suppresses HCC metastasis.
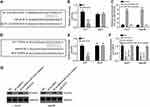
LOC105372579 regulates miR-4316/FOXP4 signaling in HCC cells
To analyze the mechanism of LOC105372579, we predicted the target miRNAs of LOC105372579 by bioinformatics analysis. miR-4316 was predicted as a potential candidate. We constructed WT and MUT luciferase reporters (Figure 4A). Luciferase reporter assay showed that miR-4316 mimics significantly suppressed the luciferase activity of WT-LOC105372579 reporter in Huh7 cells (Figure 4B). Moreover, RNA pulldown assay showed that biotin-labeled miR-4316 interacted with LOC105372579 in Hep3B and Huh7 cells (Figure 4C), demonstrating their direct interaction. FOXP4 was then predicted as a potential target of miR-4316 by bioinformatics method. Moreover, a previous study indicated that FOXP4 was targeted by miR-4316 in breast cancer.16 To determine whether miR-4316 targets FOXP in HCC cells, we constructed FOXP4 luciferase reporters (Figure 4D). Luciferase reporter assay showed that miR-4316 mimic dramatically suppressed the activity of FOXP4-WT reporter (Figure 4E), suggesting their interaction in HCC cells. To determine whether LOC105372579 can regulate FOXP4 expression by interacting with miR-4316, we performed qRT-PCR and Western blot. We found that LOC105372579 silencing or miR-4316 mimic transfection suppressed the expression of FOXP4 in Huh7 and Hep3B cells (Figure 4F and G). However, miR-4316 suppression could reverse the effect of LOC105372579 silencing (Figure 4F and G). In summary, our results suggest that LOC105372579 promotes FOXP4 expression by inhibiting miR-4316 in HCC.
LOC105372579/miR-4316/FOXP4 axis regulates proliferation, migration, and invasion of HCC cells
We further explored the effect of the LOC105372579/miR-4316/FOXP4 axis in HCC. CCK8, colony formation, and Transwell assays showed that miR-4316 suppression or FOXP4 restoration could reverse the effects of LOC105372579 silencing on Huh7 cell proliferation, migration, and invasion (Figure 5A–D). These observations were further validated by using Hep3B for experiments (Figure 5E–H). Taken together, our findings demonstrate that the LOC105372579/miR-4316/FOXP4 axis regulates HCC proliferation, migration, and invasion.
In vivo effect of LOC105372579 on HCC growth
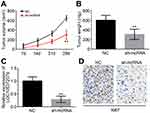
Finally, xenograft experiments were performed to test the effect of LOC105372579 on HCC growth. The tumor volumes and weights both decreased in the sh-LOC105372579 group compared with the control group (Figure 6A and B). Moreover, the expression of LOC105372579 was still reduced in the sh-LOC105372579 group of tumor tissues (Figure 6C), suggesting that the above observation was caused by LOC105372579 silencing. Then, we measured the expression of Ki67 in tumor tissues by immunohistochemistry (IHC) assay. We found that Ki67 expression was significantly decreased in the tumor tissues of the sh-LOC105372579 group (Figure 6D), suggesting that LOC105372579 silencing inhibited HCC proliferation in vivo.
Discussion
In recent years, a number of studies have recognized that lncRNAs are important regulators in human cancers including HCC.17 Hence, identifying novel lncRNAs involved in HCC progression will be of great significance. In this study, we found that LOC105372579 was highly expressed in HCC tissues and cell lines. Additionally, LOC105372579 suppression was able to inhibit HCC cell proliferation, migration, invasion, and EMT in vitro. Moreover, LOC105372579 silencing suppressed HCC growth in vivo. LOC105372579 was found to interact with miR-4316 and promote FOXP4 expression.
Previous studies have indicated that lncRNAs can act as oncogenes or tumor suppressors to regulate HCC progression.18,19 For instance, LINC01287 promotes tumor growth and EMT in HCC through miR-298/STAT3 signaling.20 LncRNA OGFRP1 promotes HCC growth and invasion by activating the AKT/mTOR and Wnt/β-catenin pathways.21 LncRNA HULC contributes to liver cancer progression by suppressing PTEN.22 As a novel lncRNA, the function of LOC105372579 is largely unknown. In our study, we for the first time defined LOC105372579 as an oncogene in HCC. We demonstrated that LOC105372579 is upregulated in HCC tissues and promotes tumor cell malignant behavior.
LncRNAs can exert roles through various mechanisms, including epigenetic regulation, post-transcriptional regulation, or miRNA sponge.6,23 For example, lncRNA NEAT1 sponges miR-139-5p to promote TGF-β1 expression and increase HCC progression.24 LncRNA HOTAIR enhances the malignant behavior of HCC by sponging miR-23b-3p from ZEB1 and inducing EMT.25 LncRNA CASC2 modulates the miR-362-5p/Nf-κB axis to initiate HCC development.26 In the current study, LOC105372579 was found to be located in the cytoplasm of HCC cells by ISH analysis. In addition, miR-4316 was identified to be a target of LOC105372579. miR-4316 was reported to inhibit tumor progression in breast cancer, papillary thyroid cancer, and bladder cancer.16,27,28 However, the role of miR-4316 in HCC needs further investigation. Here, we found that miR-4316 interacts with LOC105372579 in HCC cells. In addition, miR-4316 suppression could abolish the effect of LOC105372579 silencing in HCC cells, suggesting the anticancer role of miR-4316.
Several genes, including ZBTB2, E2F6, and FOXP4, have been reported to be the targets of miR-4316.16,27,28 In our study, we also predicted FOXP4 as the target of miR-4316 in HCC by bioinformatics analysis and confirmed their interaction in Huh7 and Hep3B cells. FOXP4 is a novel forkhead transcription factor involved in several cancers. For example, circRNA ZNF609 sponges miR-138-5p to promote FOXP4 expression, leading to renal carcinoma progression.29 FOXP4 is upregulated by the circMYO9B/miR-4316 axis and facilitates breast cancer progression.16 FOXP4 upregulation promotes the proliferation and inhibits the apoptosis of osteosarcoma cells.30 In our study, we demonstrated that FOXP4 was regulated by the LOC105372579/miR-4316 axis. We observed that FOXP4 expression was increased by LOC105372579-induced inhibition on miR-4316, resulting in HCC progression. However, how OC105372579/miR-4316/FOXP4 axis simultaneously contributes to HCC cell proliferation, migration, invasion and EMT remains unclear, which needs to be further investigated.
In conclusion, we found that LOC105372579 is a novel oncogenic lncRNA in HCC progression. We also elucidated the possible mechanism of the LOC105372579/miR-4316/FOXP4 axis in HCC development. Thus, our work provided new insights into the development of potential therapeutic targets for HCC treatment.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Maluccio M, Covey A. Recent progress in understanding, diagnosing, and treating hepatocellular carcinoma. CA Cancer J Clin. 2012;62(6):394–399. doi:10.3322/caac.21161
3. Colecchia A, Schiumerini R, Cucchetti A, et al. Prognostic factors for hepatocellular carcinoma recurrence. World J Gastroenterol. 2014;20(20):5935–5950. doi:10.3748/wjg.v20.i20.5935
4. Rahimi RS, Trotter JF. Liver transplantation for hepatocellular carcinoma: outcomes and treatment options for recurrence. Ann Gastroenterol. 2015;28(3):323–330.
5. Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136(4):629–641. doi:10.1016/j.cell.2009.02.006
6. Tu J, Zhao Z, Xu M, et al. LINC00707 contributes to hepatocellular carcinoma progression via sponging miR-206 to increase CDK14. J Cell Physiol. 2018. doi:10.1002/jcp.27737
7. Liu B, Ye B, Yang L, et al. Long noncoding RNA lncKdm2b is required for ILC3 maintenance by initiation of Zfp292 expression. Nat Immunol. 2017;18(5):499–508. doi:10.1038/ni.3712
8. Hu Z, Huang P, Yan Y, Zhou Z, Wang J, Wu G. Hepatitis B virus X protein related lncRNA WEE2-AS1 promotes hepatocellular carcinoma proliferation and invasion. Biochem Biophys Res Commun. 2019;508(1):79–86. doi:10.1016/j.bbrc.2018.11.091
9. Xu Y, Zhang G, Zou C, et al. Long noncoding RNA DGCR5 suppresses gastric cancer progression by acting as a competing endogenous RNA of PTEN and BTG1. J Cell Physiol. 2018. doi:10.1002/jcp.27861
10. Miao F, Chen J, Shi M, Song Y, Chen Z, Pang L. LncRNA HAND2-AS1 inhibits non-small cell lung cancer migration, invasion and maintain cell stemness through the interactions with TGF-beta1. Biosci Rep. 2019;39(1). pii:BSR20181525. doi:10.1042/BSR20181525
11. Wu J, Du M, Zhang Q, et al. Long noncoding RNA UCA1 promotes the proliferation, invasion, and migration of nasopharyngeal carcinoma cells via modulation of miR-145. Onco Targets Ther. 2018;11:7483–7492. doi:10.2147/OTT.S182290
12. Wang Y, Shi F, Xia Y, Zhao H. LncRNA OIP5-AS1 predicts poor prognosis and regulates cell proliferation and apoptosis in bladder cancer. J Cell Biochem. Epub 2018 Nov 28. doi:10.1002/jcb.28024
13. Lu G, Li Y, Ma Y, et al. Long noncoding RNA LINC00511 contributes to breast cancer tumourigenesis and stemness by inducing the miR-185-3p/E2F1/Nanog axis. J Exp Clin Cancer Res. 2018;37(1):289. doi:10.1186/s13046-018-0945-6
14. Wang J, Luo Z, Yao T, Li W, Pu J. LINC00707 promotes hepatocellular carcinoma progression through activating ERK/JNK/AKT pathway signaling pathway. J Cell Physiol. 2019;234(5):6908–6916. doi:10.1002/jcp.27449
15. Wang Y, Cheng Q, Liu J, Dong M. Leukemia stem cell-released microvesicles promote the survival and migration of myeloid leukemia cells and these effects can be inhibited by MicroRNA34a overexpression. Stem Cells Int. 2016;2016:9313425. doi:10.1155/2016/1243659
16. Wang N, Gu Y, Li L, et al. Circular RNA circMYO9B facilitates breast cancer cell proliferation and invasiveness via upregulating FOXP4 expression by sponging miR-4316. Arch Biochem Biophys. 2018;653:63–70. doi:10.1016/j.abb.2018.04.017
17. Xiao Z, Shen J, Zhang L, Li M, Hu W, Cho C. Therapeutic targeting of noncoding RNAs in hepatocellular carcinoma: recent progress and future prospects. Oncol Lett. 2018;15(3):3395–3402. doi:10.3892/ol.2018.7758
18. Zhang Z, Wang S, Liu W. EMT-related long non-coding RNA in hepatocellular carcinoma: A study with TCGA database. Biochem Biophys Res Commun. 2018;503(3):1530–1536. doi:10.1016/j.bbrc.2018.07.075
19. Ling ZA, Xiong DD, Meng RM, et al. LncRNA NEAT1 promotes deterioration of hepatocellular carcinoma based on in vitro experiments, data mining, and RT-qPCR analysis. Cell Physiol Biochem. 2018;48(2):540–555. doi:10.1159/000491811
20. Mo Y, He L, Lai Z, et al. LINC01287/miR-298/STAT3 feedback loop regulates growth and the epithelial-to-mesenchymal transition phenotype in hepatocellular carcinoma cells. J Exp Clin Cancer Res. 2018;37(1):149. doi:10.1186/s13046-018-0831-2
21. Chen W, You J, Zheng Q, Zhu YY. Downregulation of lncRNA OGFRP1 inhibits hepatocellular carcinoma progression by AKT/mTOR and Wnt/beta-catenin signaling pathways. Cancer Manag Res. 2018;10:1817–1826. doi:10.2147/CMAR.S164911
22. Xin X, Wu M, Meng Q, et al. Long noncoding RNA HULC accelerates liver cancer by inhibiting PTEN via autophagy cooperation to miR15a. Mol Cancer. 2018;17(1):94. doi:10.1186/s12943-018-0843-8
23. Zhu P, Wang Y, Wu J, et al. LncBRM initiates YAP1 signalling activation to drive self-renewal of liver cancer stem cells. Nat Commun. 2016;7:13608. doi:10.1038/ncomms13608
24. Tu J, Zhao Z, Xu M, Lu X, Chang L, Ji J. NEAT1 upregulates TGF-beta1 to induce hepatocellular carcinoma progression by sponging hsa-mir-139-5p. J Cell Physiol. 2018;233(11):8578–8587. doi:10.1002/jcp.26524
25. Yang T, He X, Chen A, Tan K, Du X. LncRNA HOTAIR contributes to the malignancy of hepatocellular carcinoma by enhancing epithelial-mesenchymal transition via sponging miR-23b-3p from ZEB1. Gene. 2018;670:114–122. doi:10.1016/j.gene.2018.05.061
26. Zhao L, Zhang Y, Zhang Y. Long noncoding RNA CASC2 regulates hepatocellular carcinoma cell oncogenesis through miR-362-5p/Nf-kappaB axis. J Cell Physiol. 2018;233(10):6661–6670. doi:10.1002/jcp.26446
27. Wu X, Yan Y, Li H, et al. DNA copy number gain-mediated lncRNA LINC01061 upregulation predicts poor prognosis and promotes papillary thyroid cancer progression. Biochem Biophys Res Commun. 2018;503(3):1247–1253. doi:10.1016/j.bbrc.2018.07.032
28. Wang F, Zu Y, Huang W, Chen H, Xie H, Yang Y. LncRNA CALML3-AS1 promotes tumorigenesis of bladder cancer via regulating ZBTB2 by suppression of microRNA-4316. Biochem Biophys Res Commun. 2018;504(1):171–176. doi:10.1016/j.bbrc.2018.08.150
29. Xiong Y, Zhang J, Song C. CircRNA ZNF609 functions as a competitive endogenous RNA to regulate FOXP4 expression by sponging miR-138-5p in renal carcinoma. J Cell Physiol. 2018. doi:10.1002/jcp.27744
30. Yin Z, Ding H, He E, Chen J, Li M. Up-regulation of microRNA-491-5p suppresses cell proliferation and promotes apoptosis by targeting FOXP4 in human osteosarcoma. Cell Prolif. 2017;50(1). doi:10.1111/cpr.12368
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.