Back to Journals » OncoTargets and Therapy » Volume 13
Lnc HAGLR Promotes Colon Cancer Progression Through Sponging miR‐185‐5p and Activating CDK4 and CDK6 in vitro and in vivo
Authors Sun W, Nie W , Wang Z, Zhang H, Li Y, Fang X
Received 15 January 2020
Accepted for publication 6 May 2020
Published 22 June 2020 Volume 2020:13 Pages 5913—5925
DOI https://doi.org/10.2147/OTT.S246092
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Federico Perche
Weixuan Sun,1 Wenting Nie,2 Zhaoyi Wang,1 Haolong Zhang,1 Yezhou Li,3 Xuedong Fang1
1Department of Gastrointestinal Colorectal and Anal Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China; 2Department of Plastic Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China; 3Department of Vascular Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China
Correspondence: Xuedong Fang
Department of Gastrointestinal Colorectal and Anal Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China
Email [email protected]
Yezhou Li
Department of Vascular Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China
Email [email protected]
Background/Aim: LncRNA plays a key role in tumor progression. HAGLR functions as an oncogene in many cancers. However, the molecular mechanism of HAGLR in colon cancer is still unclear.
Methods: qRT-PCR was used to measure the expression of HAGLR, miR-185-5p in colon cancer. The expression of CDK4 and CDK6 was detected by Western blot. CCK-8 assay, EdU staining, transwell and Annexin V-FITC/PI assay were used to analyze the effect of HAGLR and miR-185-5p on cell proliferation, invasion, migration and apoptosis. Bioinformatic analysis and luciferase were used to analyze the target genes of HAGLR and miR-185-5p. Nude mice were used to detect mouse tumor changes.
Results: Compared with normal colon cancer tissues and cells, the expression of HAGLR was increased in colon cancer tissues and cells. In addition, the expression of HAGLR down-regulation inhibited the growth, migration, and invasion of colon cancer cells. MiR-185-5p was reduced in colon cancer, and CDK4 and CDK6 acted as target genes of miR-185-5p to regulate the progress of colon cancer. And CDK4 and CDK6 were predicted as downstream targets of miR-185-5p. Finally, it was demonstrated that HAGLR regulated tumor progression in vivo.
Conclusion: Lnc HAGLR promoted the development of colon cancer by miR-185-5p/CDK4/CDK6 axis, and lnc HAGLR might be potential target for colon cancer.
Keywords: lnc HAGLR, miR-185-5p, CDK4/CDK6, colon cancer, proliferation, apoptosis
Introduction
Colon cancer is a frequently-occurring disease in the clinic.1,2 New cases of colon cancer have increased. It is worth noting that the incidence of colon cancer varies not only globally, but also its development trends in different countries and regions.3 The pathogenesis of colon cancer is not fully understood, and it is currently considered to be a combination of environmental factors and genetic factors.4 The main factors affecting the incidence of colon cancer include environmental factors, intestinal homeostasis, dietary structure, tobacco and alcohol habits, and physical exercise.5,6 At present, the principles for the treatment of colon cancer tumors are mainly comprehensive treatment based on surgical resection, chemotherapy, radiation therapy, and targeted therapy.7,8 From the perspective of the effect of treatment, individualized differences between patients with colon cancer tumors are obvious. The above treatments have obvious deficiencies and poor overall efficacy, which can only partially relieve clinical symptoms and have a poor prognosis.9,10 Therefore, finding new markers for early diagnosis of colon cancer is a hot topic of current research.
In the human genome sequence, during the transcription process, most products are non-coding transcripts. The length of long-chain non-coding RNA (lnc RNA) is generally greater than 200 nt.11,12 The regulation of LncRNA in cancer has been sought by many researchers.13,14 LncRNA is closely related to various tumor behaviors of malignant tumors, such as tumor autophagy, tumor resistance and tumor immunity.15,16 At present, it has been found that lncRNAs are dysregulated in colon cancer, and they can play a regulatory role in the tumor formation process, thereby affecting the occurrence, metastasis and prognosis of colon cancer.17,18 LncRNA HAGLR is a recently discovered lncRNA, which has been found abnormally expressed in many malignant tumors.19 For example, the study found that HAGLR in NSCLC was increase.20 But the mechanism of LncRNA HAGLR in colon cancer have not been studied.
The regulatory relationship of lncRNA-miRNAs has attracted the attention, and the interactive regulation of lncRNAs and miRNAs is the current research focus.21 MicroRNAs (miRNAs) are a single species derived from the organism’s own genome.22 miRNAs can play a carcinogenic role and promote the development of tumor cells. It can act as a proto-oncogene to inhibit tumor growth.23,24 In solid cancer patients, the expression of miRNAs in tumor tissue are dysregulated, which are related to tumorigenesis, invasion, metastasis, judgment of prognosis, and drug resistance.25,26 miRNAs have received more attention in the occurrence of colon cancer.27,28 miR-185-5p has been confirmed to be abnormally expressed in many tumor tissues.29 But its function in colon cancer is currently unknown. Cyclin-dependent kinases (CDKs) are the core part of the cell cycle regulation mechanism, and considered as important biomarkers for treatment of malignant tumors.30 CDK4 and CDK6 have been poorly studied in colon cancer.31,32 Therefore, it was speculated that LncRNA HAGLR can control the progression of colon cancer by CDK4 and CDK6 through miR-185-5p. The purpose of this study was to explore the mechanism of lncRNA HAGLR regulation of colon cancer.
Materials and Methods
Tissue Sample
The study was approved by the ethics committee of the First Affiliated Hospital of Jinzhou Medical University. A total of 25 colon cancer tissues and adjacent normal tissues were obtained from patients undergoing colon cancer surgery in our hospital. All patients have not received any radiation therapy or chemotherapy. Each patient signed an informed consent form.
Cell Culture
HCT-116, HCT-8, HT-29, DLD-1, SW620, and FHC cells were obtained from ATCC (Manassas, Virginia). The cells were cultured in RPMI 1640 medium containing 10% FBS (GIBCO, Carlsbad, CA) at 37 ° C in a cell incubator containing 5% CO2.
Cell Transfection
Lipofectamine 2000 reagent (Invitrogen) was used to transfect miR-185-5p mimic, inhibitor or its negative control into cells. A cDNA of human HAGLR was amplified from the mRNA of colon cancer cells. The sequence was designed using shRNA-luciferase (shLuc) as a negative control. The target product was subcloned into pcDNA3.1 (Invitrogen, Carlsbad, CA) and the lentiviral packaging vector pMD2.G was used to verify transfection by qRT-PCR.
Cell Proliferation Assay
SW480 cells were seeded in a 96-well plate at a density of 5,000 cells per well. 100 μL CCK-8 solution (Liji, Shanghai, China) was added. And absorbance was measured using a microplate reader (Potenov, Beijing, China) at 450nm.
Apoptosis Analysis
SW480 cells were plated into 6-well plates at a density of 5 × 10 5 cells/well, and the cells were collected and counted. The cells were centrifuged and 195 μL of Annexin V-FITC binding solution was added. After mixed with 5 μL Annexin V-FITC and 10 μL propidium iodide staining solution, the cells were incubated for 10–20 min, and then placed in an ice bath.
Transwell Assay
Transwell assays was used to detect cell invasion and metastasis. The upper basement membrane of the transwell chamber was pre-coated with 20 μg Matrigel and cultured overnight in a 24-well plate. After 12 hours of culture, it was washed for 3 times the PBS, fixed with 90% formaldehyde, and then stained in the crystal violet solution for 15 min. Then the photograph was taken under an inverted microscope. In the cell migration experiment, the upper chamber of the transwell chamber was free of matrigel coating, and the rest of the operation was the same as the invasion experiment.
EdU Analysis
After the SW480 cells were treated, the 10 µmol/L EdU was added to the culture medium and incubated in the incubator for 24 h. Then, 1 mL 0.5% triaxone and 0.5 mL of reaction solution were added, 0.5 mL of Hoechst 33342 solution was added for 30 min, and the observation and counting were performed under inverted fluorescence microscope.
Nude Mouse Tumor Xenograft Model
Male athymic BALB/c nude mice were purchased from the National Laboratory Animal Center (Beijing, China). Each mouse was injected subcutaneously with HCT-116 cells to establish a mouse xenograft model. A total of 5 × 106 shRNAHAGLR-HCT-116 and NC-HCT-116 cells were suspended in 100 μL phosphate buffer and injected subcutaneously into nude mice. Five weeks later, mice were euthanized. The study has been approved by the Ethics Committee of Beijing Friendship Hospital and is based on the the National Institutes of Health’s Guide to the Care and Use of Laboratory Animals.
RNA Pull-Down Assay
RNA immunoprecipitation assay was performed using EZMagna RIP kit (Millipore). Cells were lysed using complete RIP lysis buffer. Cell extracts were incubated with magnetic beads conjugated with anti AGO2 or anti IgG antibodies for 6 h. Then the RNA was purified by RT-qPCR analysis.
RNA Extraction and qRT-PCR
Total RNA in tissues and cells was extracted by TRIzol reagent (APPLYGEN, Beijing, China). And qRT-PCR was performed using ViiATM 7 real-time PCR system (Life Technologies, Grand Island, NY). 2-ΔΔCt Method was used to calculate the relative gene expression level. qRT-PCR methods were performed by the reference.33 β-actin was used as the internal control of HAGLR. U6 was used as the internal control of miR-185-5p. The primer sequence was as follows:
HAGLR
forward: 5′-GGCTCTTCCCTAATGTGTGG-3′,
reverse: 5′-CAGGTCCAGCATGAAACAGA-3′;
β-actin
forward 5′-CGCTCTCTGGTCCTCCTGTTC-3′,
reverse: 5′-ATCCGTTGGCTCCGACCTTCAC-3′;
miR-185-5p
forward: 5′–TGCGGTGGAGAGAAAGGCAG-3′,
reverse: 5‘-CTGCCTTTCTCTCCTCCGCA-3ʹ;
U6
forward: 5′-TGCGGGTGCTCGCTTCGGCAGC-3′,
reverse: 5′-TGCGGGTGCTCGCTTCGGCAGC-3′;
Determination of Luciferase Activity
Wild type (WT) or mutant (MUT) HAGLR binding miR-185-5p was subcloned into the pGL3 Basic vector. MiR-185-5p (RiboBio, Guangzhou, China) with 10 μg pLUC-WT-HAGLR or pLUC-MUT- HAGLR was co-transfected into HCT-116 cells. Wild-type (WT) or mutant (MUT) CDK4 and CDK6 combined with miR-185-5p were subcloned into pGL3 base vector (Promega). MiR-185-5p (RiboBio (Guangzhou, China) was co-transfected with 10 μg pLUC-WT-CDK4 and pLUC-WT-CDK6 or pLUC-MUT-CDK4 and pLUC-MUT-CDK6. After 48 h transfection, a dual luciferase assay system (Promega Corporation, Fitchburg, WI, USA) was used for luciferase activity.
Immunohistochemistry
Colon tissue were incubated with a primary antibody (Abcam) against Ki-67. Envision™ ABC kit (Gene-Tech Co,Ltd., Shanghai, China) was used for immunoassay. Leica DM4000B/M microscope (Leica Microsystems, Inc., Buffalo Grove, IL) was used for analysis.
Western Blot
The transfected cells were collected, total protein was extracted, and the protein concentration was quantified using the BCA Protein Assay Kit. It was incubated with anti-CDK4 (1: 1000, Xinyu, Shanghai, China) and anti-CDK6 (1: 1000, Xinyu, Shanghai, China) and anti-GAPDH antibody (1: 1000, Xinyu, Shanghai, China) at 4 ° C. Then it was incubated with anti-rabbit secondary antibody (1: 1000, Cell Signaling Technology, Boston, MA, USA) for 1 h.34
Statistical Method
The monitoring data were analyzed by SPSS19.0 statistical software. The results of data analysis were represented as mean ± standard deviation (mean ±SD). Multigroup data analysis was founded on one-way ANOVA. LSD test was leveraged for subsequent analysis. P < 0.05 indicated the difference was significant.
Results
HAGLR Was Upregulated and miR-185-5p Was Downregulated in Colon Cancer Tissues and Cell Lines
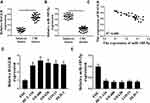
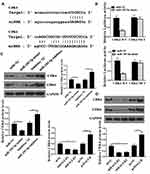
The results were shown in Figure 1A. Compared with adjacent normal tissues, the expression levels HAGLR in colon cancer tissues were significantly increased (P <0.05), while the expression levels of miR-185-5p were significantly reduced (P <0.05) (Figure 1B).There was a significant negative correlation between the expression levels of HAGLR and miR-185-5p in colon cancer tissues (Figure 1C). Compared with the normal colonic epithelial cell line FHC, the expression level of HAGLR in the colon cancer cell line (DLD-1, SW620 HCT-116, LOVO, SW480) was significantly increased (P <0.05, Figure 1D), and the expression level of miR-185-5p was also significantly reduced (P <0.05, Figure 1E). Because of the highest expression level of HCT-116 in SW480 cells, SW480 cells were selected for follow-up test.
HAGLR Expression Regulated Proliferation and Apoptosis in CC Cells
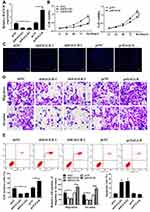
As shown in Figure 2A, in the SW480 cells, the expression level of HAGLR in the shHAGLR1 group and the shHAGLR2 group was significantly reduced, while the expression level of HAGLR in the pc HAGLR1 group was significantly increased, indicating that the transfection was successful (P <0.05). As shown in Figure 2B and C, compared with the sh-NC group, the cell viability in the shHAGLR1 group and the shHAGLR2 group was significantly reduced, and the positive rate of EdU staining was significantly reduced (P <0.05). As shown in Figure 2D, Tanswell results showed that compared with sh-NC group, the cell migration and invasion of the shHAGLR1 and the shHAGLR2 group were significantly reduced (P <0.05). As shown in Figure 2E, the shHAGLR1 group and the shHAGLR2 group could significantly induce apoptosis (P <0.05). The pcHAGLR2 result was the opposite of the shHAGLR1 result.
miR-185-5p Overexpression Inhibited Proliferation and Induced Apoptosis in CC Cells
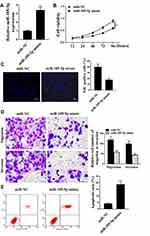
As shown in Figure 3A, compared with miR-NC group, the expression level of miR-185-5p in miR-185-5p mimic group was significantly increased in SW480 cells, indicating successful transfection (P <0.05). The cell viability was significantly reduced in the miR-185-5p mimic group, and the positive rate of EdU staining was significantly reduced (P <0.05, Figure 3B and C). Tanswell results showed that miR-185-5p mimic was significantly reduced cell migration and invasion (P <0.05), Figure 3D). MiR-185-5p mimic was significantly induced apoptosis (P <0.05, Figure 3E).
HAGLR Acted as a Sponge for miR-185-5p in CC Cells
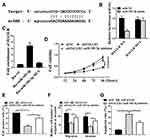
We used the Starbase 2.0 online prediction tool and miR-185-5p was identified as a potential target for HAGLR (Figure 4A). The WT-HAGLR or mutant (Mut) -HAGLR luciferase reporter plasmid was used for luciferin enzyme reporter assay. The results showed that the luciferase activity of pGL3-REPOR-HAGLR-WT was reduced by miR-185-5p mimics (P <0.05), but the luciferase activity of pGL3-REPOR-HAGLR-Mut did not change significantly (Figure 4B). In addition, the expression level of HAGLR using biotinylated miR-185-5p (miR-185-5p-bio) probes were significantly increased compared with control (NC-bio) or miR-185-5p probes (Figure 4C) (P <0.05). As shown in Figure 4D-G, compared with the shNC group, the cell viability, EdU staining positive rate, and cell invasion and migration rate of the shHAGLR1 group were significantly reduced (P <0.05). The apoptosis rate was significantly increased (P <0.05). And shHAGLR1 co-transfected with miR-185-5p inhibitor group could reverse the effects of shHAGLR1 on cell viability, invasion, migration and apoptosis (P <0.05). The results demonstrated that HAGLR exerted biological effects on colon cancer cells through miR-185-5p.
CDK6 and CDK4 Were the Target of miR‐185-5p
We used the Starbase 2.0 online prediction tool and CDK4 and CDK6 were identified as potential targets for miR-185-5p (Figure 5A). To verify the prediction results, WT-CDK4, WT-CDK6 or mutant (Mut) -CDK4 and (Mut)-CDK6 luciferase reporter plasmid were used for luciferase reporter gene assay. The luciferase activity of pGL3-REPOR-CDK4-WT and pGL3-REPOR-CDK6-WT was reduced by miR-185-5p mimic (P <0.05). However, the luciferase activity of pGL3-REPOR- CDK4-Mut and pGL3-REPOR- CDK6-Mut did not change significantly (Figure 5B). Compared with the control group, miR-185-5p mimic could significantly reduce the protein expression level of CDK4 and CDK6 (P <0.05), miR-185-5p inhibitor significantly increase the protein expression levels of CDK4 and CDK6 (P <0.05, Figure 5C). As shown in Figure 5D, shHAGLR1 and shHAGLR2 were significantly reduced the protein expression levels of CDK4 and CDK6 (P <0.05), while pcHAGLR was significantly increased the protein expression levels of CDK4 and CDK6 (P <0.05).
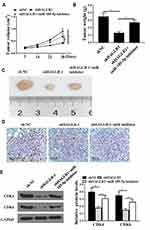
Downregulation of HAGLR Repressed Colon Cancer Progression in vivo
In vivo experiments were used to analyze whether HAGLR knockdown inhibited colon cancer progression. The results were shown in Figure 6A-C. Compared with the sh-NC group, shHAGLR1 was inhibited tumor growth, while co-transfection of shHAGLR1 with miR-185-5p inhibitor were significantly reversed the growth of shHAGLR1 on tumors. Ki-67 staining results showed that the number of positive cells in shHAGLR1 group was decreased, while co-transfection of shHAGLR1 with miR-185-5p inhibitor significantly reversed the effect of shHAGLR1 on the number of positive cells (Figure 6D). In addition, as shown in Figure 6E, the protein expression levels of CDK4 and CDK6 in the shHAGLR1 group were significantly reduced (P <0.05), and co-transfection of shHAGLR1 with miR-185-5p inhibitor significantly reversed the effect of shHAGLR1 on the protein expression levels of CDK4 and CDK6 (P <0.05, Figure 6E).
Discussion
Colon cancer is a common malignant tumor that originates in the colon and is the third most common gastrointestinal tumor.35 And 40–50 years old is the high incidence population.36 According to the pathological types, colon cancer can be divided into three major categories, adenocarcinoma, mucinous adenocarcinoma and undifferentiated carcinoma.37 However, invasive colon cancer is prone to metastasis in the early stage and easily relapses after treatment, which seriously affects the diagnosis and treatment and prognosis of colon cancer patients.38 The occurrence of colon cancer are the result of the interaction of multiple pathogenic factors, high-fat diet, insufficient food fiber and intestinal flora disturbance are the main environmental factors involved in colon cancer. Familial gonadal tumor polyposis and familial hereditary non-polyposis are typical hereditary factors that lead to colon cancer, and gene mutations are also important pathogenic factors that have been studied.39 The abnormal gene expression play a key role in the occurrence of colon cancer.40
Long-chain non-coding ribonucleic acid (lncRNA) is a type of RNA consisting of about 200–600 nucleotides. They generally do not encode proteins, but regulate the expression level of genes through RNA in the form of transcriptional regulation, epigenetic regulation and post transcriptional regulation.41 Studies have shown that lncRNA play a key role in many regulatory processes.42 LncRNAs were found to be abnormally expressed in colon cancer.43 The effect of lncRNA RP11- 462c24.1 on the clinicopathological characteristics of colon cancer, it has been found to be reduced in colorectal cancer tissues, and was significantly related to distant metastasis of tumors.44 In addition, it has been found that up-regulation of MALAT1 expression can promote the protein level of HIF-1αand endothelial cells, which affects the development of colon cancer.45 Studies have found that LncRNA HAGLR is abnormally expressed in many cancers. For example, The expression level of lncRNA HAGLR is increased in NSCLC.19 This study found that the expression level of LncRNA HAGLR was significantly increased in CRC. ShHAGLR could inhibited cell proliferation, invasion and migration, and induced apoptosis. shHAGLR could effectively inhibit the weight and volume of tumors in vivo. Therefore, HAGLR can control the development of colon cancer.
LncRNA regulates miRNA, thereby regulating protein translation and cell activity.46 At present, researchers have identified human miRNA, and miRNAs regulate the expression levels of various genes in the human body.23 The relationship between miRNAs and colon cancer has been obtained more attention. MiRNAs are expected to become targets for colon cancer treatment.47 For example, it has been found that the expression of miRNA-21 is abnormally increased in serum of colon cancer patients, and its expression level is related to TNM stage, chemotherapy effect and prognosis of patients.48 Screening results of colon cancer-related miRNA from a large sample showed that in clinical colon cancer samples and their corresponding cancer 21 miRNAs with up-regulated expression are found in tissues.49 MiR-185-5p is a miRNA with tumor growth inhibitory effect found. For example, studies have found that miR-185-5p suppresses prostate cancer proliferation.29 MiR-185-5p was screened as a target gene for HAGLR. MiR-185-5p was negatively correlated with HAGLR. The expression level of miR-185-5p was significantly reduced in colon cancer. And miR-185-5p mimic could inhibit cell proliferation, invasion and migration, and induce apoptosis. Co-transfection of shHAGLR with miR-185-5p inhibitor could reverse the effects of shHAGLR on cell proliferation, apoptosis and tumor weight and volume. These results suggested that HAGLR might promote colon cancer growth by regulating miR-185-5p.
miRNAs participate in tumor formation and progression by regulating genes during tumor formation.50 Cyclin-dependent kinases (CDKs) belong to the serine/threonine kinase family. The activities of CDK4 and CDK6 are considered to be closely related to the pathogenesis of cancer.51 CDK4 and CDK6 could promote tumorigenesis and development when over-activated.52 In this study, it was found that CDK4 and CDK6 were target gene of miR-185-5p. In addition, miR-185-5p mimic was reduced the expression levels of CDK4 and CDK6, miR-185-5p inhibitor was increased the expression levels of CDK4 and CDK6, while shHAGLR was reduced the expression levels of CDK4 and CDK6, pcHAGLR was increased the expression levels of CDK4 and CDK6. These data indicated that Lnc HAGLR promoted the development of colon cancer by targeting the miR-185-5p/CDK4/CDK6 axis.
There are several limitations in this research. Our study initially explained the mechanism of HAGLR/miR/CDK6 axis, while the deeper control mechanism has not been fully explained clearly. In addition, since our research focuses on CDK4 and CDK6, and the entire cell cycle regulation process involves multiple proteins, there may be more confounding factors in the real in vivo environment. Therefore, further experiments are necessary to provided more profound evidence in the future.
Conclusion
Lnc HAGLR promoted colon cancer progression by regulating the miR-185-5p/CDK4/CDK6 axis, suggesting that Lnc HAGLR might be a potential oncogene for colon cancer.
Disclosure
The authors declare that they have no competing interests.
References
1. Fadelu T, Zhang S, Niedzwiecki D, Ye X, Fuchs CS. Nut consumption and survival in patients with stage III colon cancer: results from CALGB 89803 (Alliance). J Clin Oncol off J Am Soc Clin Oncol. 2018;36:JCO2017755413. doi:10.1200/JCO.2017.75.5413
2. Hsiao KY, Lin YC, Gupta SK, et al. Noncoding effects of circular RNA CCDC66 promote colon cancer growth and metastasis. Cancer Res. 2017;77:2339.
3. Dolan RD, Mcsorley ST, Park JH, et al. The prognostic value of systemic inflammation in patients undergoing surgery for colon cancer: comparison of composite ratios and cumulative scores. Br J cancer. 2018;25:34.
4. Tylichová Z, Straková N, Vondráček J, Vaculová AH, Kozubík A, Hofmanová J. Activation of autophagy and PPARγ protect colon cancer cells against apoptosis induced by interactive effects of butyrate and DHA in a cell type-dependent manner: the role of cell differentiation. J Nutri Biochem. 2017;39:145–155.
5. O’Connell MJ, Laurie JA, Kahn M, et al. Prospectively randomized trial of postoperative adjuvant chemotherapy in patients with high-risk colon cancer. J Clin Oncol. 1998;16(1):295–300. doi:10.1200/JCO.1998.16.1.295
6. Thygesen LC, Grønbaek M, Johansen C, Fuchs CS, Giovannucci E. Prospective weight change and colon cancer risk in male US health professionals. Int J Cancer. 2008;123:1160–1165. doi:10.1002/ijc.23612
7. Gulack BC, Nussbaum DP, Keenan JE, et al. Surgical resection of the primary tumor in stage IV colorectal cancer without metastasectomy is associated with improved overall survival compared with chemotherapy/radiation therapy alone. Dis Colon Rectum. 2016;59.
8. Marur S, Forastiere AA. Challenges of integrating chemotherapy and targeted therapy with radiation in locally advanced head and neck squamous cell cancer. Curr Opinion Oncol. 2010;22:206–211.
9. Liu Q, Dakui L, Sanjun C, Qingguo L, Xinxiang L. P–TNM staging system for colon cancer: combination of P-stage and AJCC TNM staging system for improving prognostic prediction and clinical management. Cancer Manag Res. 2018;10:2303–2314. doi:10.2147/CMAR.S165188
10. Meyskens FL, Gerner EW. Development of difluoromethylornithine as a chemoprevention agent for the management of colon cancer. J Cell Biochem. 1995;59(S22):126–131. doi:10.1002/jcb.240590816
11. Chen W, Jia Y, Qi Z, Yueyong Z. Downregulation of lncRNA OGFRP1 inhibits hepatocellular carcinoma progression by AKT/mTOR and Wnt/β-catenin signaling pathways. Cancer Manag Res. 2018;10:1817–1826.
12. Geng C, Wang Z, Wang D, et al. LncRNADisease: a database for long-non-coding RNA-associated diseases. Nucl Acids Res. 2012.
13. Pieter-Jan V, Kenneth V, Gerben M, et al. An update on LNCipedia: a database for annotated human lncRNA sequences. Nucleic Acids Res. 2014;D1.
14. Qian L, Huang J, Zhou N, et al. LncRNA loc285194 is a p53-regulated tumor suppressor. Nucleic Acids Res. 2013;9.
15. Du P, Zhao H, Peng R, Liu Q, Liao Y. LncRNA-XIST interacts with miR-29c to modulate the chemoresistance of glioma cell to TMZ through DNA mismatch repair (MMR) pathway. Biosci Rep. 2017;37:BSR20170696. doi:10.1042/BSR20170696
16. Loewen G, Jayawickramarajah J, Zhuo Y, Shan B. Functions of lncRNA HOTAIR in lung cancer. J Hematol Oncol. 2014;7(1):90. doi:10.1186/s13045-014-0090-4
17. Huang JZ, Chen M, Chen D, et al. A peptide encoded by a putative lncRNA HOXB-AS3 suppresses colon cancer growth. 2017;68:171–184.
18. Sun L, Xue H, Jiang C, et al. LncRNA DQ786243 contributes to proliferation and metastasis of colorectal cancer both in vitro and in vivo. Biosci Rep. 2016;36(3):e00328–e00328. doi:10.1042/BSR20160048
19. Zakany J, Darbellay F, Mascrez B, Necsulea A, Duboule D. Control of growth and gut maturation by HoxD genes and the associated lncRNA Haglr. Proc Natl Acad Sci U S A. 2017;114:201712511.
20. Lu C, Ma J, Cai D. Increased HAGLR expression promotes non–small cell lung cancer proliferation and invasion via enhanced de novo lipogenesis. Tumour Biol J Int Soc Oncodevelop Biol Med. 2017;39:101042831769757.
21. Song J, Ye A, Jiang E, et al. Reconstruction and analysis of the aberrant lncRNA-miRNA-mRNA network based on competitive endogenous RNA in CESC. J Cell Biochem. 2018.
22. Adhami M, Haghdoost AA, Sadeghi B, Afshar RM. Candidate miRNAs in human breast cancer biomarkers: a systematic review. Breast Cancer. 2017;25(1):1–8. doi:10.1007/s12282-017-0793-9
23. Lei Y, Li C, Fang L, Fan Y, Zhang S. MiRNA-155 promotes proliferation by targeting caudal-type homeobox 1 (CDX1) in glioma cells. Biomed Pharmacother. 2017;95:1759–1764. doi:10.1016/j.biopha.2017.08.088
24. Pereira DM, Rodrigues PM, Borralho PM, Rodrigues CMP. Delivering the promise of miRNA cancer therapeutics. Drug Discov Today. 2012;18(5–6):282–289. doi:10.1016/j.drudis.2012.10.002
25. Naidu S, Magee P, Garofalo MM. MiRNA-based therapeutic intervention of cancer. J Hematol Oncol. 2015;8(1):68. doi:10.1186/s13045-015-0162-0
26. Srivastava K, Srivastava A. Comprehensive review of genetic association studies and meta-analyses on miRNA polymorphisms and cancer risk. PLoS One. 2012;7:e50966. doi:10.1371/journal.pone.0050966
27. Boni V, Zarate R, Villa JC, Bandrés E, Aranda E. Role of primary miRNA polymorphic variants in metastatic colon cancer patients treated with 5-fluorouracil and irinotecan. Pharmacogenomics J. 2011;11:429–436. doi:10.1038/tpj.2010.58
28. Yong M, Zou C, Meng F, Kong J, Dong H. The SNPs in pre-miRNA are related to the response of capecitabine-based therapy in advanced colon cancer patients. Oncotarget. 2017;9(6):6793–6799. doi:10.18632/oncotarget.23190
29. Tian C, Deng Y, Jin Y, Shi S, Bi H. Long non-coding RNA RNCR3 promotes prostate cancer progression through targeting miR-185-5p. Am J Transl Res. 2018;10(5):1562–1570.
30. Yun JL, Han HJ. Role of ATP in DNA synthesis of renal proximal tubule cells: involvement of calcium, MAPKs, and CDKs. Am J Physiol. 2006;291:F98–106. doi:10.1152/ajprenal.00486.2005
31. Wainberg ZA, Yufa A, Anghel A, Rogers AM, Slamon DJ. Abstract 4557: expression of p16 in colon cancer and cyclin D1 in gastric cancer predicts response to CDK4/6 inhibition in vitro. Cancer Res. 2014;74:4557.
32. Zhang J, Zhou L, Zhao S, Dicker DT, El-Deiry WS. The CDK4/6 inhibitor palbociclib synergizes with irinotecan to promote colorectal cancer cell death under hypoxia. Cell Cycle. 2017a;16. doi:10.1080/15384101.2017.1320005
33. Zhang L, Zhang Q, Wang X, Yang X, Li X, Yuan M. Selection of reference genes for qRT-PCR and expression analysis of high-altitude-related genes in grassland caterpillars (Lepidoptera: erebidae: gynaephora) along an altitude gradient. Ecol Evol. 2017b;7:9054–9065. doi:10.1002/ece3.3431
34. Sanchez-Campillo M, Bini L, Comanducci M, et al. Identification of immunoreactive proteins of Chlamydia trachomatis by Western blot analysis of a two-dimensional electrophoresis map with patient sera. Electrophoresis. 2015;20:2269–2279.
35. Xu B, Yu L, Zhao LZ. Curcumin up regulates T helper 1 cells in patients with colon cancer. Am J Transl Res. 2017;9(4):1866–1875.
36. Cusimano A, Balasus D, Azzolina A, Augello G, Cervello M. Oleocanthal exerts antitumor effects on human liver and colon cancer cells through ROS generation. Int J Oncol. 2017;51.
37. Fujikawa H, Toiyama Y, Inoue Y, Imaoka H, Kusunoki M. Prognostic impact of preoperative albumin-to-globulin ratio in patients with colon cancer undergoing surgery with curative intent. Anticancer Res. 2017;37:1335–1342.
38. Raimondo S, Saieva L, Cristaldi M, Monteleone F, Alessandro R. Label-free quantitative proteomic profiling of colon cancer cells identifies acetyl-CoA carboxylase alpha as antitumor target of Citrus limon-derived nanovesicles. J Proteomics. 2017;173.
39. Yun S, Han YS, Lee JH, Kim S, Sang HL. Enhanced susceptibility to 5-fluorouracil in human colon cancer cells by silencing of GRP78. Anticancer Res. 2017;37:2975–2984. doi:10.21873/anticanres.11651
40. Xiong Z, Fu Z, Shi J, Jiang X, Wan H. HtrA1 down-regulation induces cisplatin resistance in colon cancer by increasing XIAP and activating PI3K/Akt pathway. Ann Clin Lab Sci. 2017;47:264–270.
41. Li H, An J, Wu M, Zheng Q, Lu D. LncRNA HOTAIR promotes human liver cancer stem cell malignant growth through downregulation of SETD2. Oncotarget. 2014;6(29):27847–27864. doi:10.18632/oncotarget.4443
42. Xiao H, Tang K, Liu P, Chen K, Huang Q. LncRNA MALAT1 functions as a competing endogenous RNA to regulate ZEB2 expression by sponging miR-200s in clear cell kidney carcinoma. Oncotarget. 2015;6(35):38005–38015. doi:10.18632/oncotarget.5357
43. Huang J, Mo YY. Abstract 3537: suppression of miR-145 by long noncoding RNA RoR in colon cancer. Cancer Res. 2014;74:3537.
44. Wang C, Yu J, Han Y, Li L, Qi P. Long non-coding RNAs LOC285194, RP11-462C24.1 and Nbla12061 in serum provide a new approach for distinguishing patients with colorectal cancer from healthy controls. Oncotarget. 2016;7(43):70769–70778. doi:10.18632/oncotarget.12220
45. Hsu J, He G, Bhattacharjee G, et al. Abstract B23: selective inhibition of a long non-coding RNA (lncRNA), MALAT1 by antisense oligonucleotides results in significant anti-tumor effects in a variety of preclinical cancer models. Cancer Res. 2012;72(2 Supplement):B23–B23. doi:10.1158/1538-7445.NONRNA12-B23
46. Jalali S, Bhartiya D, Lalwani MK, Sivasubbu S, Scaria V. Systematic transcriptome wide analysis of lncRNA-miRNA interactions. PLoS One. 2013;8.
47. Cheng J, Deng R, Zhang P, Wu C, Tao K. MIR-219-5p plays a tumor suppressive role in colon cancer by targeting oncogene Sall4. Oncol Rep. 2015;34.
48. Liu TT, Liu X, Jiang-Feng HU, Zhu L, Centre HC. Serum content detection of miRNA-21 in colon cancer patients and its correlation with VEGF level. J Hainan Med Univ. 2014.
49. Kanematsu S, Tanimoto K, Suzuki Y, Sugano S. Screening for possible miRNA–mRNA associations in a colon cancer cell line. Gene. 2014;533(2):520–531. doi:10.1016/j.gene.2013.08.005
50. Guenther SM, Sinead A, Karen D, et al. Comparison of miRNA expression patterns using total RNA extracted from matched samples of formalin-fixed paraffin-embedded (FFPE) cells and snap frozen cells. BMC Biotechnol. 2007;7:36. doi:10.1186/1472-6750-7-36
51. Mateyak MK, Obaya AJ, Sedivy JM. c-Myc regulates cyclin D-Cdk4 and -Cdk6 activity but affects cell cycle progression at multiple independent points. Mol Cell Biol. 1999;19:4672–4683.
52. Kozar K, Sicinski P. Cell cycle progression without cyclin D-CDK4 and cyclin D-CDK6 complexes. Cell Cycle. 2005;4(3):388–391. doi:10.4161/cc.4.3.1551
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.