Back to Journals » International Journal of Women's Health » Volume 8
Lipedema: diagnostic and management challenges
Authors Anne Warren Peled A, Kappos E
Received 11 February 2016
Accepted for publication 11 May 2016
Published 11 August 2016 Volume 2016:8 Pages 389—395
DOI https://doi.org/10.2147/IJWH.S106227
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Elie Al-Chaer
Anne Warren Peled,1 Elisabeth A Kappos2
1Private Plastic and Reconstructive Surgery Practice, San Francisco, CA, USA; 2Department of Plastic, Reconstructive, Aesthetic and Hand Surgery, University Hospital, Basel, Switzerland
Abstract: Lipedema is an uncommon disorder characterized by localized adiposity of the lower extremities, often occurring in females with a family history of the condition. The adiposity extends from hips to ankles and is typically unresponsive to weight loss. In addition to the aesthetic deformity, women also describe pain in the lower extremities, particularly with pressure, as well as easy bruising. Although the condition is well described, it is relatively rare and often misdiagnosed. The purpose of this review is to describe the initial evaluation and diagnosis of lipedema and discuss treatment options.
Keywords: lipedema, leg swelling, lipodystrophy, localized adiposity, lower extremity liposuction
Diagnosis
Initial evaluation
Clinical history
Initial evaluation for patients presenting with enlarged or swollen lower extremities should begin with a complete history of their condition. Clinical history should be focused on the onset and duration of symptoms, symptom progression, symptom exacerbation, and any prior attempts at treatment. If prior conservative treatment has been attempted, such as lymphatic massage/drainage or compression garment use, any response to treatment should be carefully evaluated and documented, as this may indicate a lymphatic component to the condition. Review of family history may also be illustrative, as patients with lipedema often have other female family members with similar symptoms.1
Physical examination
Physical examination should include assessment for other sites of swelling in consideration for systemic conditions, such as cardiac or renal disease.2 Focused lower extremity exam should include evaluation of asymmetry or other differences between the two legs as well as documentation of any prior surgical scars in the extremities or groin. The location, extent, and quality of leg enlargement should be carefully noted, with particular emphasis on the quality of the soft tissue and whether or not there appears to be a fluid component to the enlargement. This can be assessed by whether or not the tissue “pits” with pressure, applied by the examiner’s digit for a prolonged period of time, although some mild forms of swelling or advanced swelling with significant fibrosis may not demonstrate pitting.
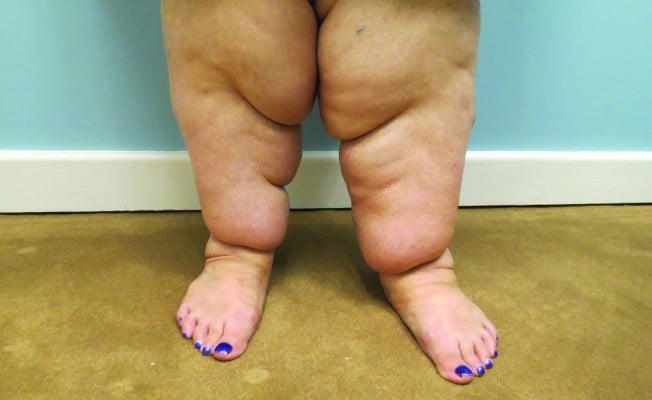
The fatty hypertrophy seen in patients with lipedema typically feels soft and doughy, though some areas of the fat may be firmer in patients with long-standing lipedema (Figure 1). Patients with lipedema may also have a history of easy bruising on their lower extremities, which can be assessed through the clinical history but may also be seen on examination.3
  | Figure 1 A 43-year-old woman with long-standing lipedema. |
The area of diffuse fatty hypertrophy typically starts at the hips and extends throughout the legs bilaterally and symmetrically.4 One of the major distinguishing features of lipedema is the sparing of the feet, which can create a distinct step-off at the ankle (Figure 2).5,6 A helpful diagnostic tool to distinguish lipedema from lymphedema is to pinch the skin over the dorsum of the base of the second toe (Stemmer sign); thickened skin that is difficult to lift off of the underlying tissue is considered diagnostic for lymphedema.7
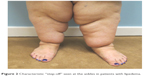  | Figure 2 Characteristic “step-off” seen at the ankles in patients with lipedema. |
The need for any further diagnostic workup is determined from the initial evaluation and performed as appropriate (Figure 3). Consideration for the following differential diagnosis for lipedema can be helpful when embarking upon further workup to determine proper clinical management.
  | Figure 3 Algorithm for workup in patients presenting with lower extremity enlargement. |
Differential diagnosis
The differential diagnosis for lipedema includes conditions causing lower extremity swelling as well as those causing lower extremity fatty excess (Table 1). Understanding the distinguishing features between these conditions and lipedema can help to accurately diagnose lipedema, which is often not considered as an initial diagnosis and thus may not be diagnosed until a potentially unnecessary workup has been done.8 Some of the main conditions in the differential diagnosis include the following.
  | Table 1 Differential diagnosis for lipedema |
Lymphedema
Lymphedema is the development of localized swelling due to lymphatic dysfunction. Lymphedema can be either primary or secondary. Primary causes for lower extremity lymphedema include Milroy’s disease, which is congenital hereditary lymphedema that is present at birth; familial lymphedema praecox, which presents during puberty; and lymphedema tarda, which arises in early adulthood.9 Secondary lymphedema typically occurs as a result of malignancy or its treatment or due to filariasis in areas where the disease is endemic.
In the typical presentation for lower extremity lymphedema, patients report a history of prior lymph node biopsy or dissection and/or radiation of the lymph node basins. Patients may also describe a history of cellulitis or other superficial infections in the affected extremity. Swelling is typically unilateral and often extends from the groin down the foot into the toes, with a positive Stemmer sign as described earlier. In the early stages, the swelling is typically soft and pitting (through deposition of adipose tissue) and can progress to fibrosis as the disease progresses.
The gold standard for diagnosing lymphedema is lymphoscintigraphy, which can demonstrate impaired lymphatic flow in the affected extremity following radiotracer injection. One caveat for using lymphoscintigraphy to distinguish lymphedema from lipedema is that patients with long-standing lipedema may develop secondary lymphedema as a result of progressive mechanical strain on the lymphatic system from the fatty hypertrophy.10–13 Long-standing lipedema in combination with morbid obesity can lead to edema due to immobility and subsequent decreased activation of the muscle pump. Further, the weight of the excess adipose tissue might decrease the lymphatic transport capacity as well increase the lymphatic load.
Other less invasive alternatives to lymphoscintigraphy to distinguish lymphedema from lipedema have recently been proposed, though their potential role in management has not yet been well studied.14,15 Computed tomography or magnetic resonance imaging (MRI) can also be helpful in differentiating the two, with MRI considered more helpful due to its higher sensitivity. With lymphedema, MRI can show dermal and subcutaneous tissue thickening, as well as a honeycomb appearance to the tissue due to fluid within the subcutaneous fat.16 Computed tomography and MRI images in patients with lipedema, on the other hand, typically demonstrate diffuse fatty hypertrophy throughout the bilateral lower extremities without skin abnormalities (Figure 4).
  | Figure 4 CT scan of a patient with lipedema demonstrating homogenous, circumferential fatty hypertrophy. |
Obesity/localized adiposity
Distinguishing lipedema from other conditions causing fatty excess in the lower extremities can be difficult. Making the diagnosis relies on some of the hallmark characteristics of lipedema, such as the easy bruising and pain with soft tissue pressure, as well as the step-off at the ankles. Women with generalized obesity typically do not have these history and physical exam characteristics. Additionally, although many patients with lipedema may also be overweight, those who have tried to lose weight through diet and exercise typically find that reducing the size of their legs is very challenging, which is often not the case in women with standard obesity.17
This distinction is less clear, however, in women who have localized adiposity in the lower extremities that is not due to lipedema. Similar to lipedema, localized adiposity in some areas of the legs, particularly the outer thighs, around the knees, and the lower calves, can be very resistant to diet and exercise in some women. Further, some women with lipedema may also have additional areas of localized adiposity,10 making the diagnosis even more challenging. It is also important to distinguish Dercum’s disease (adiposis dolorosa) from lipedema, which is characterized by multiple painful lipomatous tumors, differing from the diffuse soft fatty deposition seen with lipedema. In this scenario, physical examination is again essential to look for the characteristic symmetric enlargement and fatty hypertrophy extending from the hips to the ankles.
Venous disease
Another common condition that can present with bilateral lower extremity enlargement is chronic venous disease. Classic findings differentiating venous stasis from lipedema include pitting edema, improvement in symptoms, and leg swelling with elevation, and, in cases of long-standing venous disease, skin changes and soft tissue fibrosis. Varicose veins, however, are often seen in patients with lipedema and thus cannot be used as a distinguishing feature.1,17 Any history of lower extremity cellulitis or skin ulceration makes chronic venous stasis (or lymphedema) the likely etiology rather than lipedema, which rarely includes skin changes unless secondary lipo-lymphedema has developed. Additionally, in contrast to lipedema, swelling with venous stasis typically does not extend to the buttocks, though it does involve the ankles and feet.
Lower extremity ultrasound to assess venous competency can be helpful to confirm the diagnosis. If computed tomography or MRI studies are performed as part of the initial workup, the absence of circumferential fatty hypertrophy will exclude lipedema as a contributing factor.
Treatment
Due to the incomplete understanding of the pathophysiology of lipedema and the many unanswered questions regarding optimal therapeutic management, treatment options unfortunately still remain somewhat limited.18–20 Management of patient expectations through education regarding the condition and potential treatment outcomes is an essential part of lipedema management. While patients are often initially relieved to receive an accurate diagnosis for their condition, the lack of an easy solution can lead to disappointment and psychological support may be recommended.21–24
The goals of lipedema treatment include reducing related lower extremity symptoms and functional limitations and preventing progression of the disease. As an etiology-directed treatment for lipedema is currently unavailable, effective treatment should also target factors that negatively influence lipedema (such as obesity, the presence of lymphatic or venous edema, incorrect insight into the condition, and decreased physical activity).25,26
Conservative treatment
The standard conservative therapy for lipedema significantly differs from that of lymphedema.4 Some components, such as compression therapy, (re)developing an active lifestyle, and weight-loss programs, are, however, applicable to both.27,28
As lipedema is a chronic, incurable condition, it can be accompanied by lower extremity pain, loss of strength, and deconditioning; in more severe cases, lipedema can lead to deterioration in daily activity levels. Low levels of physical exercise are a risk factor for further deterioration of lipedema, especially in combination with an increase in body weight. The ultimate goal of therapeutic interventions is improving strength and fitness to enable an active lifestyle, which can help alleviate some symptoms, particularly in more mild cases. Obesity is also an important modifiable factor for which multidisciplinary cooperation with a dietician is necessary.25
Although dietary changes cannot prevent the disproportionate fat distribution in patients with lipedema, dietary modifications can improve prognosis and general well-being.24,29,30 The combination of early weight and diet changes may reduce local inflammation and thus prevent the exacerbation of clinical symptoms of lipedema.18
Before beginning treatment, the existence of a relevant edema component must be determined. If such a component exists, compression therapy may be useful. Compression therapy and therapeutic compression garments do not result in a decrease of fat deposition but can help to prevent further edema formation and stimulate the arterial, venous, and lymphatic flow, thereby reducing coinciding issues.31 Even patients with an edema component to their lipedema may not respond to lymphatic drainage, as the edema component in lipedema is often caused by a combination of mechanic and dynamic insufficiency, leaving the lymphatic system unaffected.24,25,29
The components of combined physiotherapy (complete decongestive therapy) have long been manual lymphatic massage, bandaging, and skin care. Several recent papers have shown that the effect of lymphatic drainage using manual lymphatic massage is minimal or none. This might be the beginning of a paradigm shift to only use bandaging and exclude manual lymphatic massage.
Skin hypersensitivity to palpation in lipedema patients is difficult to explain and treat. It is unknown if hypersensitivity is caused by nociceptive, neuropathic pain, or central sensitization, making optimal and effective treatment unclear. Pain-relieving interventions may nonetheless have a place in the initial treatment phase as pain relief may increase the patients’ capability to initiate a healthier and more active lifestyle.24
Early detection with concurrent weight and dietary changes through nutrition and exercise may be helpful in reducing nonlipedemic fat and reducing inflammation, which can also potentially reduce the debilitating effect of the condition. This approach may possibly prevent the lower body from enlarging as much as it would if the patient were to become obese. However, even with strict diet and exercise regimens, the disease may progress and further treatment may be necessary (Figure 5).
  | Figure 5 Algorithm for management of diagnosed lipedema. |
Surgical treatment
For patients with minimal or no improvement with conservative treatment, surgical options should be evaluated.8 While some studies have described that suction lipectomy with or without limited skin excision can improve the contour and size of the tissue in some cases, other reports do not recommend surgical treatments, such as lipectomy or liposuction, because of the possible risk of secondary lymphedema.4,18
Lipectomy and liposuction are nevertheless widely used to treat lipedema, with liposuction being the most commonly performed treatment. Previous studies have shown, however, that liposuction without preceding infiltration is contraindicated for lipedema because of the substantial risk of causing damage to the lymphatic system.27 With the introduction of tumescent local anesthesia and the subsequent use of supertumescent local anesthesia, the risk of damaging the lymphatic system during liposuction has dramatically decreased.32 Liposuction using supertumescent local anesthesia and vibrating cannulas is now considered an effective treatment for lipedema.20,27,33–35
Although noncurative, liposuction can improve functionality, pain, swelling, physical appearance, and the quality of life.23,27,33,34,36 Often, multiple sessions are necessary to adequately treat the extremities circumferentially and along their entire length. To prevent recurrence, as much as possible of the subcutaneous fat tissue should be removed while keeping in mind the limits of safety and cosmetic consequences. It is presumed that after surgery, no new fat cells will be formed or that, at worst, the few remaining cells will only cause minor visible tissue growth. Recent reports show that over a longer period of time, volume reduction occurs, and conservative treatment can be terminated or reduced in over 65% of treated patients. In the remaining patients, although conservative treatment remains necessary, quality of life, sensitivity, and mobility are notably improved.
If a lymphedema component exists prior to treatment, this component may still be present after treatment, making continuation of the appropriate lymphedema therapy necessary.34–37 In some patients, the lymphedema component may also improve or disappear after liposuction.
A second surgical treatment method involves excision of large localized deposits of lipedematous tissue as a debulking procedure.38 Those deposits can form in lipedematous areas on the legs and cause serious mechanical difficulties, such as valgus deformity of the knees or even a complete inability to walk. Even though the less invasive approach of liposuction should typically be the surgeon’s first choice, debulking surgery can be considered in more complicated and advanced cases. In cases of severe mechanical limitations, longitudinal debulking excision may be the only treatment.
To date, no correlation has been demonstrated between the amount of adipose tissue removal and extent of improvement. Treatment success does not seem to depend on the patient’s age, duration of the disease, or length of conservative therapy. While it has not yet been possible to identify the “ideal” timing for fat removal, liposuction should be considered in cases of progression of lipedema symptoms despite conservative therapy. Despite convincing short-term results,32–34,39 only very few long-term studies on liposuction for lipedema treatment exist to date.32–34,36,37,39,40 Future outcomes studies with longer-term results will help support the role of liposuction in the treatment of lipedema.
Conclusion
Lipedema is an underdiagnosed entity and often misdiagnosed as other similarly presenting conditions. Increased awareness of lipedema and its presentation may enable clinicians to diagnose and treat affected patients more efficiently. Diagnosis and treatment should be made as early as possible to prevent complications associated with increased functional and cosmetic morbidity. Exercise, weight-loss programs, reconditioning, and physiotherapy form important pillars of conservative lipedema treatment. Surgical options of liposuction and excisional lipectomy can be considered in patients who are resistant to conservative treatment. Liposuction appears to be the most effective and long-lasting treatment for lipedema to date, although many patients often require ongoing conservative treatment postoperatively to maintain results. Additionally, the avoidance of postoperative weight gain is essential in order to maintain the results of surgery.
Larger studies with longer-term follow-up will help to determine optimal treatment strategies.
Disclosure
The authors report no conflicts of interest in this work.
References
Wold LE, Hines EA, Allen EV. Lipedema of the legs: a syndrome characterized by fat legs and edema. Ann Intern Med. 1951;34(5):1243–1250. | ||
Garfein ES, Borud LJ, Warren AG, Slavin SA. Learning from a lymphedema clinic: an algorithm for the management of localized swelling. Plast Reconstr Surg. 2008;121(2):521–528. | ||
Allen EV, Hines EA Jr. Lipedema of the legs: a syndrome characterized by fat legs and orthostatic edema. Mayo Clin Proc. 1940;15:184–187. | ||
Rudkin GH, Miller TA. Lipedema: a clinical entity distinct from lymphedema. Plast Reconstr Surg. 1994;94(6):841–847. | ||
Harwood CA, Bull RH, Evans J, Mortimer PS. Lymphatic and venous function in lipoedema. Br J Dermatol. 1996;134(1):1–6. | ||
Beninson J, Edelglass JW. Lipedema – the non-lymphatic masquerader. Angiology. 1984;35(8):506–510. | ||
Stemmer R. [A clinical symptom for the early and differential diagnosis of lymphedema]. Vasa. 1976;5(3):261–262. | ||
Warren AG, Janz BA, Borud LJ, Slavin SA. Evaluation and management of the fat leg syndrome. Plast Reconstr Surg. 2007;119(1):9e–15e. | ||
Warren AG, Brorson H, Borud LJ, Slavin SA. Lymphedema: a comprehensive review. Ann Plast Surg. 2007;59(4):464–472. | ||
Bilancini S, Lucchi M, Tucci S, Eleuteri P. Functional lymphatic alterations in patients suffering from lipedema. Angiology. 1995;46(4):333–339. | ||
Jagtman BA, Kuiper JP, Brakkee AJ. [Measurements of skin elasticity in patients with lipedema of the Moncorps “rusticanus” type]. Phlebologie. 1984;37(3):315–319. | ||
Földi E, Földi M, Tischendorf F. [Adipositas, lipedema and lymphostasis.] Med Welt. 1983;34(7):198–200. | ||
Amann-Vesti BR, Franzeck UK, Bollinger A. Microlymphatic aneurysms in patients with lipedema. Lymphology. 2001;34(4):170–175. | ||
Birkballe S, Jensen MR, Noerregaard S, Gottrup F, Karlsmark T. Can tissue dielectric constant measurement aid in differentiating lymphoedema from lipoedema in women with swollen legs? Br J Dermatol. 2014;170(1):96–102. | ||
Lohrmann C, Foeldi E, Langer M. MR imaging of the lymphatic system in patients with lipedema and lipo-lymphedema. Microvasc Res. 2009;77(3):335–339. | ||
Duewell S, Hagspiel KD, Zuber J, von Schulthess GK, Bollinger A, Fuchs WA. Swollen lower extremity: role of MR imaging. Radiology. 1992;184:227–231. | ||
Child AH, Gordon KD, Sharpe P, et al. Lipedema: an inherited condition. Am J Med Genet A. 2010;152A(4):970–976. | ||
Suga H, Araki J, Aoi N, Kato H, Higashino T, Yoshimura K. Adipose tissue remodeling in lipedema: adipocyte death and concurrent regeneration. J Cutan Pathol. 2009;36(12):1293–1298. | ||
Szél E, Kemény L, Groma G, Szolnoky G. Pathophysiological dilemmas of lipedema. Med Hypotheses. 2014;83(5):599–606. | ||
Fife CE, Maus EA, Carter MJ. Lipedema: a frequently misdiagnosed and misunderstood fatty deposition syndrome. Adv Skin Wound Care. 2010;23(2):81–92. | ||
Fetzer A, Fetzer S. Early lipoedema diagnosis and the RCGP e-learning course. Br J Community Nurs. 2015;Suppl:S22, S24, S26–S28. | ||
Fetzer A, Wise C. Living with lipoedema: reviewing different self-management techniques. Br J Community Nurs. 2015;S14, S16–S19. | ||
Dudek JE, Białaszek W, Ostaszewski P. Quality of life in women with lipoedema: a contextual behavioral approach. Qual Life Res. 2016;25(2):401–408. | ||
Langendoen SI, Habbema L, Nijsten TE, Neumann HA. Lipoedema: from clinical presentation to therapy. A review of the literature. Br J Dermatol. 2009;161(5):980–986. | ||
Dutch Society for Dermatology and Venereology and the Dutch Academy of Medical Specialists. Lipedema Guidelines in the Netherlands 2014. Utrecht: Dutch Society for Dermatology and Venereology; 2014. | ||
Goodliffe JM, Ormerod JO, Beale A, Ramcharitar S. An under-diagnosed cause of leg swelling. BMJ Case Rep. Epub 2013 May 24. | ||
Forner-Cordero I, Szolnoky G, Forner-Cordero A, Kemény L. Lipedema: an overview of its clinical manifestations, diagnosis and treatment of the disproportional fatty deposition syndrome – systematic review. Clin Obes. 2012;2(3–4):86–95. | ||
Wagner S. Lymphedema and lipedema – an overview of conservative treatment. Vasa. 2011;40(4):271–279. | ||
Reich-Schupke S, Altmeyer P, Stücker M. Thick legs – not always lipedema. J Dtsch Dermatol Ges. 2013;11(3):225–233. | ||
Damstra RJ, Andrade M, Villavicencio JL. Diagnosis and treatment of primary lymphedema. Consensus document of the International Union of Phlebology (IUP)-2013. Int Angiol. 2013;32(6):541–574. | ||
Szolnoky G, Varga E, Varga M, Tuczai M, Dosa-Racz E, Kemeny L. Lymphedema treatment decreases pain intensity in lipedema. Lymphology. 2011;44(4):178–182. | ||
Stutz JJ, Krahl D. Water jet-assisted liposuction for patients with lipoedema: histologic and immunohistologic analysis of the aspirates of 30 lipoedema patients. Aesthetic Plast Surg. 2009;33(2):153–162. | ||
Rapprich S, Dingler A, Podda M. Liposuction is an effective treatment for lipedema-results of a study with 25 patients. J Dtsch Dermatol Ges. 2011;9(1):33–40. | ||
Schmeller W, Hueppe M, Meier-Vollrath I. Tumescent liposuction in lipoedema yields good long-term results. Br J Dermatol. 2012;166(1):161–168. | ||
Habbema L. Safety of liposuction using exclusively tumescent local anesthesia in 3,240 consecutive cases. Dermatol Surg. 2009;35(11):1728–1735. | ||
Warren Peled A, Slavin SA, Brorson H. Long-term outcome after surgical treatment of lipedema. Ann Plast Surg. 2012;68(3):303–307. | ||
Baumgartner A, Hueppe M, Schmeller W. Long-term benefit of liposuction in patients with lipoedema. A follow-up study after an average of 4 and 8 years. Br J Dermatol. 2016;174(5):1061–1067. | ||
Wollina U, Heinig B, Schönlebe J, Nowak A. Debulking surgery for elephantiasis nostras with large ectatic podoplanin-negative lymphatic vessels in patients with lipo-lymphedema. Eplasty. 2014;14:e11. | ||
Wollina U, Goldman A, Heinig B. Microcannular tumescent liposuction in advanced lipedema and Dercum’s disease. G Ital Dermatol Venereol. 2010;145(2):151–159. | ||
Okhovat JP, Alavi A. Lipedema: a review of the literature. Int J Low Extrem Wounds. 2015;14(3):262–267. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
