Back to Journals » Infection and Drug Resistance » Volume 12
Leprosy Reactions In Childhood: A Prospective Cohort Study In The Brazilian Amazon
Authors Bandeira SS, Pires CA, Quaresma JAS
Received 26 May 2019
Accepted for publication 8 August 2019
Published 17 October 2019 Volume 2019:12 Pages 3249—3257
DOI https://doi.org/10.2147/IDR.S217181
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Sabrina Sampaio Bandeira,1,2 Carla Avelar Pires,2,3 Juarez Antonio Simões Quaresma2,3
1Sanitary Dermatology Referral Unit “Dr. Marcello Cândia”, Secretary of State for Public Health, Marituba, PA, Brazil; 2Tropical Medicine Center, Federal University of Para, Belem, PA, Brazil; 3Center of Health and Biological Sciences, State University of Para, Belem, PA, Brazil
Correspondence: Juarez Antonio Simões Quaresma
Tropical Medicine Center, Federal University of Para, Av. Generalissimo Deodoro 92, Umarizal, Belem, PA 66055-240, Brazil
Email [email protected]
Background and objective: In highly endemic areas, severe multibacillary forms of leprosy and reactional episodes are not rare in children. The objective of the present study was to describe the clinical and epidemiological aspects of leprosy reactions in children from the Brazilian Amazon.
Methods: This was a prospective cohort study of 34 leprosy patients aged under 15 years diagnosed at a health referral unit in northern Brazil between April 2014 and June 2015. Follow-up medical consultations were performed during multidrug therapy (MDT) and one year after the end of treatment. Participants underwent a simple neurologic examination and answered a structured questionnaire.
Results: Of the 34 recruited patients, 18 (52.9%) had leprosy reactions and/or neuritis. Among these, 10 (55.6%) had reactions at diagnosis, 13 (72.2%) had reactions after MDT, and 14 (77.8%) had two or more reactional episodes. Type I reactions occurred in 14 (77.8%) cases. Complications, such as disabilities, necrotizing erythema nodosum, or Cushing’s syndrome, occurred in six (33.3%) patients. The following variables showed significant associations (p ≤ 0.05) with leprosy reactions: age 8–14 years, number of doctors seen (≥3), multibacillary classification, number of skin lesions (≥10), or borderline and lepromatous clinical forms. The high frequency of type I reactions resulted in prolonged corticosteroid therapy, which may cause deficient bone maturation in childhood.
Conclusion: Older age in children, consulting many physicians for diagnosis, severe clinical forms, and numerous skin lesions were positively associated with reaction development. Reactions after MDT highlight the need for continuity in healthcare of children with leprosy.
Keywords: Mycobacterium leprae, leprosy, children, clinical aspects, Brazil, Amazon
Introduction
Leprosy is a chronic infectious disease that affects the skin and peripheral nerve trunks.1 It was a long held belief that Mycobacterium leprae (M. leprae) was the sole microorganism that caused this disease; however, in 2008, a new species, Mycobacterium lepromatosis (M. lepromatosis), causing a unique form of diffuse lepromatous leprosy, was discovered.2–4 Owing to its long incubation period, leprosy has been considered a disease of adults; nevertheless, in highly endemic regions, many children are exposed at early ages to high bacillary loads in untreated patients, causing a large number of cases in childhood.5 In 2017 alone, 210,671 patients were diagnosed with leprosy worldwide. Brazil had the second largest number of new cases (26,875).6 The state of Pará, which is part of the Amazon region in northern Brazil, is highly endemic for leprosy. In 2017, Pará had 2562 newly diagnosed cases, of which 8.7% involved children.7
About 5–20% of children with leprosy will present with reaction episodes (exacerbation of inflammatory processes, either localized or systemic) at some point before, during, and/or after the end of MDT.8–10 Leprosy reactions can lead to permanent nerve damage, potentially resulting in disabilities and deformities.11,12 Severe nerve damage with musculoskeletal disorders and deformities can result in discrimination at school and difficulties in the social life of children with leprosy.13 Although physical and emotional impairments resulting from reactional episodes in childhood are evident, studies on this topic are scarce, especially among pediatric groups, both during MDT and after the end of treatment.10,14 This survey describes leprosy reactions and their associations with clinical and epidemiologic aspects of leprosy cases in children under 15 years old diagnosed at the Health Referral Unit “Marcello Candia” (HRUMC) in a highly endemic area of the Brazilian Amazon.
Methods
This prospective cohort, descriptive, analytical study was conducted among patients under the age of 15 years who were diagnosed with leprosy at the HRUMC located in the municipality of Marituba, in the state of Pará, in the Amazon region of northern Brazil. The HRUMC was built on the site of an old leper colony. Today, it is the only referral unit specializing in leprosy in the state of Pará.
Data were collected from April 2014 to June 2015 and all patients under the age of 15 years at the time of leprosy diagnosis at the HRUMC were invited to participate in the study (convenience sampling). Patients who lacked cognitive capacity or had difficulty understanding the instructions for the strength and sensory tests in the simple neurologic examination, who had a diagnosis of another associated neurologic disease, or who abandoned treatment and/or did not return for follow-up were excluded.
Patients were diagnosed by a committee of leprosy experts at the HRUMC. The diagnosis of leprosy was based on cardinal signs of leprosy and was supported by findings of histopathology and skin smear microscopy. All procedures for patients receiving drug treatment for leprosy and its reactional episodes were conducted according to the guidelines of the World Health Organization and Brazilian Ministry of Health.15
Because it is often used by HRUMC physicians, the leprosy clinical classification of Madrid16 was adopted in this study. It classifies leprosy into the following forms: indeterminate, tuberculoid, borderline, and lepromatous. For treatment purposes, all patients received an operational classification of paucibacillary (PB) or multibacillary (MB) leprosy, based on the World Health Organization’s definition of PB (patients with up to five skin lesions and/or with only one affected nerve trunk) and MB (patients with more than five skin lesions and/or more than one affected nerve trunk) leprosy.17 The reactional episodes were categorized as type I or type II,15 and neuritis was considered as isolated when it was not associated with other clinical signs and symptoms of reactions.18 Prednisone and thalidomide were used to treat type I and type II reactions, respectively. In cases of necrotizing erythema nodosum and/or type II reactions associated with neuritis, both of these drugs were used concomitantly.15
Patients aged under 15 years and their guardians were interviewed using a structured questionnaire. Clinical and epidemiological data were collected: clinical and operational classification, skin lesion number, age, sex, contact with individuals with leprosy (household contact or social contact), and diagnostic information (time to diagnosis, number of doctors consulted to obtain diagnosis, and other diagnoses). The time to diagnosis was defined as the interval between the perception of signs and symptoms and the diagnosis of leprosy.
The children also underwent an examination to assess the neural function of the main nerves affected by leprosy. This exam involved inspection, nerve palpation, muscle strength testing, and sensory evaluation of the eyes and upper and lower limbs; the degree of physical disability was also assessed, and all processes were conducted in accordance with the World Health Organization and the Brazilian Ministry of Health.15
Children studied underwent clinical follow-up evaluations at the HRUMC, in which they underwent further simple neurologic examinations six times in the 6 months of MDT and once a year after the end of treatment or upon any signs and/or symptoms of reactions. For patients with leprosy reactions, the following data were recorded: type, frequency, and time of the leprosy reaction.
Results were analyzed using GraphPad Prism version 5.0 software (GraphPad Software, Inc., San Diego, California). A descriptive analysis with calculation of the absolute and relative frequencies of the categorical variables was performed. To verify the degree of association between occurrence of leprosy reactions and clinical and epidemiologic variables, we used the Fisher’s exact test because of our small sample size. Leprosy reaction was considered as the dependent variable, whereas sex, age group, time to diagnosis, number of doctors consulted for diagnosis, other diagnoses, clinical form, operational classification, and skin lesion number were the independent variables. A value of p ≤ 0.05 was considered significant.
This study was approved by the Ethics Committee on Human Research of the Institute of Health Sciences, Federal University of Pará (Approval No. 1.059.013) and was conducted in accordance with the Declaration of Helsinki (1964) and its subsequent revisions. The anonymity of all participants was respected. An informed consent form was signed by the patient or legal representative as well as by children who were 12–14 years of age and agreed to participate in the study and the photo documentation publicity.
Results
There were 403 new cases of leprosy diagnosed at the HRUMC during the period of data collection; of these, 45 (11.2%) were children under the age of 15 years. However, only 34 (75.5%) were included in the study: three were not allowed to participate, seven were lost to follow-up (three abandoned treatment and four did not return after the end of MDT), and one did not have the cognitive capacity to understand the simple neurologic examination instructions.
Of the 34 participants, 20 (58.8%) were boys and 24 patients (70.6%) were in the 8–14 year age group. The youngest participant was only three years old and the mean age was 9.5 years. One-half of the participants (17 patients [50%]) had previous contact with individuals with leprosy; of these, 13 (76.5%) had household contact. The time from symptom onset to diagnosis was more than one year in 23 (67.6%) cases and 19 (55.9%) were seen by one or two doctors for the diagnosis of leprosy; however, 21 (61.8%) initially received other diagnoses, such as helminthiasis, allergy, or ringworm. Seventeen patients (50%) had the borderline clinical form, four (11.8%) had the lepromatous form, and only one had primarily neural leprosy with multiple nerve trunks affected. The most common type of disease was MB, affecting 22 (64.7%) patients, and 23 (67.6%) patients had up to nine skin lesions.
One of the major complications of leprosy is reactions. On analyzing the association between leprosy reaction occurrence and clinical and epidemiologic characteristics, the following variables showed significant associations (p≤0.05): age group 8–14 years, number of doctors seen for diagnosis (≥3), borderline and lepromatous clinical forms, MB operational classification, and number of skin lesions (≥10) (Table 1).
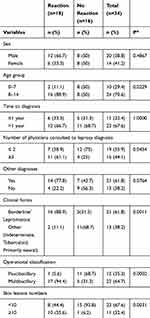 |
Table 1 Association Between Occurrence Of Leprosy Reactions And Clinical And Epidemiologic Characteristics |
Among participating children, 18 (52.9%) had leprosy reactions and/or isolated neuritis at some point during the course of the disease (at diagnosis, during treatment, and/or after the end of MDT). Of these, 14 (77.8%) had type I reactions and 14 had two or more reactional episodes. The majority of patients (13 [72.2%]) had reactions during MDT; nevertheless, the same number of participants had reactions after the end of treatment (Table 2). All children with leprosy reactions at the time of diagnosis had at least one more episode during the monitored period. Although type II reactions were the least frequent, occurring in only four (22.2%) cases, the episodes were recurrent (at intervals of 28 days or more after the reaction treatment), with a mean of three episodes per patient.
 |
Table 2 Type, Frequency And Moment That Leprosy Reactions Occurred In Patients Of Study |
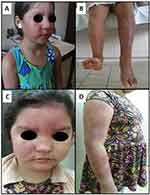
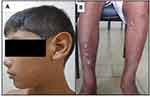
During the study period, six (33.3%) children had complications associated with reactional leprosy episodes or owing to prolonged use of corticosteroid therapy; these complications were necrotizing erythema nodosum, decreased muscle strength in the hands, ulnar claw, ulcers, foot drop, or Cushing’s syndrome (Figures 1A–D and 2A, B).
Discussion
This study revealed a higher proportion (52.9%) of children with reactions and/or isolated neuritis than many previous reports, in which only 5–20% of children with leprosy had reactional episodes.8,9,11,19–23 However, a survey in India showed that 30% of patients in a study of 306 children had leprosy reactions,14 and in a series of 18 cases in children under 15 years old from a referral center in south eastern Brazil, 50% presented with type I reactions.24
A potential explanation for this finding is the direct relationship between a high number of reactions and the predominance of MB forms in the patient population. There was a significant association between MB disease and borderline and lepromatous clinical forms with leprosy reaction occurrence. This is a consistent observation that has also been reported by other studies.18,25,26
Many studies on leprosy in childhood showed the disease in its initial PB forms and, owing to its long incubation period, complications of reactions and/or deformities were uncommon.9,19–21,27,28 However, this does not accurately reflect the situation in highly endemic areas, as some studies in these areas showed that the majority of children had MB forms and reactional episodes were not rare.8,22,29,30
Another fact that may have influenced the high number of reactional episodes is that most surveys on reactions in the pediatric age group are cross-sectional studies, with information only obtained at the time of diagnosis.9,13,18 In this study, children had follow-up medical evaluations during treatment and one year after discharge, which allowed us to observe the frequency of reactions for a longer time course (in line with the characteristics and course of the disease). Some authors also reported difficulties in monitoring patients; for example, in a study in India, only 60 (10.8%) out of 551 children under 18 years of age diagnosed with leprosy had follow-up examinations during MDT.10
As only patients under the age of 15 years were evaluated and monitored over time in this study, reactional episodes between adult and childhood patients could not be compared. Some authors described a high frequency (about 50–75%25,31–33) of leprosy reactions in adult patients during and after MDT, and a study, conducted in a reference center for leprosy in south eastern Brazil, reported that nearly 60% of adult patients had leprosy reactions, but only 23.5% of children and adolescents (age range 0–19 years) had this complication.25
Type I reactions were predominant in the children evaluated in this study, which is similar to reports in the literature.8,21,24,30 However, most patients (77.8%) developed two or more reactional episodes and this contradicts some studies conducted in all age groups, which reported type I reactions with single episodes or in only a minority of patients presenting with recurrence.8,34 In contrast, in a study in Thailand, 31.2% of the patients had several reactional episodes that continued for up to two years.35
A probable explanation for the high reaction recurrence rate in this study is the difficulty in determining whether a subsequent reaction event is a new episode or only a continuation of the first.36 Furthermore, underlying infections increase the risk of developing reactions and these should be investigated concurrently because underlying infections may be associated with the perpetuation of episodes.25
Successive episodes of type I reactions require high and prolonged corticosteroid doses. The use of corticoids in childhood is of great concern because of their effect on bone maturation and their potential to reduce growth.38 In addition, the occurrence of many reactional episodes, especially after discharge, prolongs the time of disease and treatment, which intensifies isolation and reinforces the idea that there is no cure for the patient.39
The majority of patients (72.2%) had leprosy reactions that occurred during MDT and the same percentage had reactions after the end of treatment. This contradicts the findings of several studies, which emphasize that reactional episodes are predominant only during MDT, with a reduction in incidence after discharge.26,34,36,37 However, a recent survey carried out in an endemic region of northeast Brazil, showed that leprosy reactions also occurred after discharge and had a close association with the onset of disabilities in this period.
The findings of this study emphasize the need for systematic and longitudinal care for patients even after the end of treatment; therefore, the period after discharge should not be neglected.12 Seven children diagnosed at the HRUMC could not be followed up. This situation is particularly complicated considering the severity of the sequelae associated with possible reactional episodes.11,12 Children are vulnerable members of society and cannot be expected to seek the help of health professionals; thus, it is necessary to inform parents and/or caregivers about the signs and symptoms of reactional episodes and nerve damage to ensure that the family is involved in the monitoring of children.
In this survey, a relationship was observed between the age group of 8–14 years or the number of skin lesions (≥10) with the occurrence of leprosy reactions. Older children have a longer disease progression, increasing the chances of developing severe forms and the associated complications, such as reactions and deformities.11 Some studies showed an increased risk of disabilities with increased age.8,11,40 The same principle applies to the number of skin lesions; thus, severe forms present with a greater number of skin lesions and a higher risk of reactions and disabilities.11
Another association was shown between more doctors consulted prior to disease diagnosis and the presence of reactional episodes. This finding may be due to late treatment. Most patients studied (67.6%) were diagnosed more than one year after the onset of disease. Long diagnostic delays increase the risk of peripheral nerve damage and the development of deformities.11,41–43
It should be emphasized that a leprosy diagnosis in childhood may be difficult and involve confusing sensory testing.19 This difficulty means that patients may initially receive other diagnoses, which contributes to increasing the time to treatment onset and therefore increases the risk of developing deformities.43 However, in the analysis of the association between the time of diagnosis or other diagnoses received and the occurrence of reactional episodes, there were no statistically significant associations. This finding might be because of the small sample size, which is a limitation of this study. It may also be because of an imprecise time of diagnosis since this was based on the individual’s (or caregiver’s) perception of when the signs and symptoms of the disease began.
Beyond the small sample size, another limitation of this study is that it was carried out in a referral unit for leprosy, where patients usually present with more severe forms of the disease and are difficult to diagnose. Therefore, it is not possible to generalize these findings to community populations; however, this does not decrease or invalidate the results obtained. It is also noteworthy that host genetic factors are known to predispose leprosy patients to developing reactions. Hence, studies using available technologies, such as genomics, epigenomics, transcriptomics, and proteomics, could help identify patients genetically predisposed to reactions, thereby allowing for prediction and prevention of reactions, especially in childhood.44,45
Disabilities like ulnar claw and foot drop were observed in children who had reactions in this study. There is evidence that reactions are important risk factors for the development of physical disabilities due to leprosy.11,46,47 Deformities that occur in childhood have devastating effects because children have to live the rest of their lives’ with the stigma of the disease, in addition to the skeletal muscle impairment that may also affect school and social activities.13,48
These results suggest that leprosy reactions are not rare in children in highly endemic areas and there is a predominance of recurrent type I reactions in pediatric age groups, which results in the use of corticosteroid therapy. Furthermore, children who are older, who are seen by a greater number of physicians, who have severe clinical forms, or who have a higher number of skin lesions have an increased risk of reactional episodes; therefore, these patients should be carefully monitored by the health team after completion of MDT. This also highlights the need for adequate education of patients and their parents regarding the signs and symptoms of reactions; this will help to diagnose and treat these leprosy complications early, minimizing disabilities and deformities in childhood.
Abbreviations
MDT, multidrug therapy; HRUMC, Health Referral Unit “Marcello Candia”; PB, paucibacillary; MB, multibacillary.
Acknowledgments
Program of support for qualified production: PAPQ/UFPA. This work was funded by the Council for Scientific and Technological Development (Grant number 305345/2018-4). The authors confirm they obtained the consent of the parent/guardian(s) of the patients in Figures 1 and 2 to include their images in this paper.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Walker SL, Lockwood DNJ. The clinical and immunological features of leprosy. Brit Med Bull. 2006;78:103–121. doi:10.1093/bmb/ldl010
2. Han XY, Sizer KC, Thompson EJ, et al. Comparative sequence analysis of Mycobacterium leprae and the new leprosy-causing Mycobacterium lepromatosis. J Bacteriol. 2009;191:6067–6074. doi:10.1128/JB.00762-09
3. Han XY, Sizer KC, Velarde-Félix JS, Frias-Castro LO, Vargas-Ocampo F. The Leprosy agents Mycobacterium lepromatosis and Mycobacterium leprae in Mexico. Int J Dermatol. 2012;51:952–959. doi:10.1111/j.1365-4632.2011.05414.x
4. Han XY, Zhang J, Li L. Leprosy agents Mycobacterium lepromatosis and Mycobacterium leprae in Mexico: a clarification. J Clin Microbiol. 2015;53:3387–3388. doi:10.1128/JCM.01588-15
5. Barreto JG, Bisanzio D, Guimarães LDS, et al. Spatial analysis spotlighting early childhood leprosy transmission in a hyperendemic municipality of the Brazilian Amazon Region. PLoS Negl Trop Dis. 2014;8:e2665. doi:10.1371/journal.pntd.0002665
6. World Health Organization (WHO). Global leprosy update, 2017: reducing the disease burden due to leprosy. Wkly Epidemiol Rec. 2018;93:445–456.
7. Hanseníase – DATASUS – Indicadores epidemiológicos e operacionais de hanseníase, por ano diagnóstico – Municípios/UF/Regiões/Brasil-2017. Brasil: Ministério da Saúde [Leprosy - DATASUS - Leprosy epidemiological and operational indicators, by diagnosis year - Municipalities / UF / Regions / Brazil-2017. Brazil: Ministry of Health.] Available from: http://tabnet.datasus.gov.br/cgi/sinannetbd/hanseniase/hansDB2017.htm.
8. Singal A, Sonthalia S, Pandhi D. Childhood leprosy in a tertiary-care hospital in Delhi, India: a reappraisal in the post-elimination era. Lepr Rev. 2011;82:259–269.
9. Chaitra P, Bhat RM. Post elimination status of childhood leprosy: report from tertiary-care hospital in South India. Biomed Res Int. 2013;2013:1–4. doi:10.1155/2013/328673
10. Gitte SV, Sabat RN, Kamble KM. Childhood leprosy in an endemic area of central India. Indian Pediatr. 2016;53:221–224. doi:10.1007/s13312-016-0824-1
11. Kar BR, Job CK. Visible deformity in childhood leprosy a 10-year study. Int J Lepr Other Mycobact Dis. 2005;73:243–248.
12. Raposo MT, Reis MC, Caminha AVDQ, et al. Grade 2 disabilities in leprosy patients from Brasil: need for follow-up after completion of multidrug therapy. PLoS Negl Trop Dis. 2018;12:1–12. doi:10.1371/journal.pntd.0006645
13. Ekeke N, Chukwu J, Nwafor C, et al. Children and leprosy in southern Nigeria: burden, challenges and prospects. Lepr Rev. 2014;85:111–117.
14. Jain S, Reddy RG, Osmani SN, Lockwood DNJ, Suneetha S. Childhood leprosy in an urban clinic, Hyderabad, India: clinical presentation and the role of household contacts. Lepr Rev. 2002;73:248–253.
15. Diretrizes para vigilância, atenção e eliminação da Hanseníase como problema de saúde pública: manual técnico-operacional [Guidelines for surveillance, care and elimination of Leprosy as a Public Health problem: technical and operational manual]. Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Brasília: Ministério da Saúde; 2016 [Health Surveillance Secretariat. Department of Communicable Disease Surveillance. Brasilia: Ministry of Health; 2016].
16. Madrid Congress. Technical resolutions. Classification of leprosy. Int J Lepr. 1953;21:504–516.
17. World Health Organization (WHO). Programme for the elimination of leprosy: a guide to eliminating leprosy as a public health problem. Technical documents. Geneva; 1995:60.
18. Scollard DM, Martelli CMT, Stefani MMA, et al. Risk factors for leprosy reactions in three endemic countries. Am J Trop Med Hyg. 2015;92:108–114. doi:10.4269/ajtmh.13-0221
19. Rao AG. Study of leprosy in children. Indian J Lepr. 2009;81:195–197.
20. Horo I, Rao PS, Nanda NK, Abraham S. Childhood leprosy: profiles from a leprosy referral hospital in West Bengal, India. Indian J Lepr. 2010;82:33–37.
21. Das L, Meher BK, Das NB, et al. A clinico-bacteriological study of leprosy in pediatric age group. Adv Infect Dis. 2013;3:269–273. doi:10.4236/aid.2013.34041
22. Palit A, Inamadar AC, Desai SS, et al. Childhood leprosy in post-elimination phase: data from a tertiary health care hospital in the Kamataka state of south India. Lepr Rev. 2014;85:85–92.
23. Nair SP. A clinico-epidemiological study of pediatric leprosy in the urban leprosy center of a tertiary care institute. Ind J Ped Dermatol. 2017;18:24–27. doi:10.4103/2319-7250.187890
24. Pinto ACVD, Wachholz PA, Silva GVD, et al. Profile of leprosy in children under 15 years of age monitored in a Brazilian referral center, 2004–2012. An Bras Dermatol. 2017;92:580–582. doi:10.1590/abd1806-4841.20175676
25. Antunes DE, Araujo S, Ferreira GP, et al. Identification of clinical, epidemiological and laboratory risk factors for leprosy reactions during and after multidrug therapy. Mem Inst Oswaldo Cruz. 2013;108:901–908. doi:10.1590/0074-0276130222
26. Queiroz TA, Carvalho FPBD, Simpson CA, et al. Clinical and epidemiological profile of patients with leprosy-related reactions. Rev Gaúcha Enferm. 2015;36:185–191. doi:10.1590/1983-1447.2015.esp.57405
27. Lana FCF, Amaral EP, Lanza FM, et al. Hansen’s disease in children under fifteen years-old in Jequitinhonha Valley, Minas Gerais, Brazil. Rev Bras Enferm. 2007;60:696–700. doi:10.1590/s0034-71672007000600014
28. Imbiriba EB, Guerrero JCH, Garnelo L, Levino A, Cunha MDG, Pedrosa V. Epidemiological profile of leprosy in children under 15 in Manaus (Northern Brazil), 1998–2005. Rev Saude Publ. 2008;42:1021–1026. doi:10.1590/s0034-89102008005000056
29. Ferreira IN, Alvarez RRA. Leprosy in patients under fifteen years of age in the city of Paracatu-MG (1994 to 2001). Rev Bras Epidemiol. 2005;8:41–49. doi:10.1590/S1415-790X2005000100006
30. Ramos JM, Reyes F, Lemma D, Tesfamariam A, Belinchón I, Górgolas M. The burden of leprosy in children and adolescents in rural southern Ethiopia. Pediatr Int Child Health. 2014;34:24–28. doi:10.1179/2046905513Y.0000000073
31. Motta ACF, Pereira KJ, Tarquínio DC, Vieira MB, Miyake K, Foss NT. Leprosy reactions: coinfections as a possible risk factor. Clinics. 2012;67:1145–1148. doi:10.6061/clinics/2012(10)05
32. Hungria EM, Oliveira RM, Penna GO, et al. Can baseline ML Flow test results predict leprosy reactions? An investigation in a cohort of patients enrolled in the uniform multidrug therapy clinical trial for leprosy patients in Brazil. Infect Dis Poverty. 2016;5:110. doi:10.1186/s40249-016-0203-0
33. Leon KE, Jacob JT, Franco-Paredes C, Kozarsky PE, Wu HM, Fairley JK. Delayed diagnosis, leprosy reactions, and nerve injury among individuals with Hansen’s disease seen at a United States clinic. OFID. 2016;ofw063. doi:10.1093/ofid/ofw063
34. Silva SFD, Griep RH. Leprosy reaction in patients of health centers from de Planning Area 3.2 of Rio de Janeiro Municipality. Hansen Int. 2007;32:155–162.
35. Suchonwanit P, Triamchaisri S, Wittayakornrerk S, et al. Leprosy reaction in Thai population: a 20-year restrospective study. Dermatol Res Pract. 2015;2015:253154. doi:10.1155/2015/253154
36. Richardus JH, Nicholls PG, Croft RP, Withington SG, Smith W, Cairns S. Incidence of acute nerve function impairment and reactions in leprosy: a prospective cohort analysis after 5 years of follow-up. Int J Epidemiol. 2004;33:337–343. doi:10.1093/ije/dyg225
37. Teixeira MAG, Silveira VMD, França ERD. Characteristics of leprosy reactions in paucibacillary and multibacillary individuals attended at two reference centers in Recife, Pernambuco. Rev Bras Med Trop. 2010;43:287–292. doi:10.1590/S0037-86822010000300015
38. Pitts CJD, Kearns AE. Update on medications with adverse skeletal effects. Mayo Clin Proc. 2011;86:338–343. doi:10.4065/mcp.2010.0636
39. Costa MD, Costa RD, Terra FDS, et al. Assessment of quality of life of patients with leprosy reactional states treated in a dermatology reference center. An Bras Dermatol. 2012;87:26–35. doi:10.1590/s0365-05962012000100003
40. Sethi M, Rao PS. Challenges in preventing disabilities among children affected by leprosy: findings from a referral hospital in North India. Lep Rev. 2015;86:296–297.
41. Nicholls PG, Wiens C, Smith WC. Delay in presentation in the context of local knowledge and attitude towards leprosy – the results of qualitative fieldwork in Paraguay. Int J Lepr Other Mycobact Dis. 2003;71:198–209. doi:10.1489/1544-581X(2003)71<198:DIPITC>2.0.CO;2
42. Van Veen NH, Meima A, Richardus JH. The relationship between detection delay and impairment in leprosy control: a comparison of patient cohorts from Bangladesh and Ethiopia. Lepr Rev. 2006;77:356–365.
43. Bandeira SS, Pires CA, Quaresma JAS. Nerve damage in young patients with leprosy diagnosed in a endemic area of the Brazilian Amazon: a cross-sectional study. J Pediatr. 2017;185:143–148. doi:10.1016/j.jpeds.2017.02.035
44. Lucena-Silva N, Teixeira MA, de Ramos AL, et al. The +3187A/G HLA-G polymorphic site is associated with polar forms and reactive reaction in leprosy. Mol Genet Genomic Med. 2013;1:123–130.
45. Fava VM, Manry J, Cobat A, et al. A missense LRRK2 variant is a risk factor for excessive inflammatory responses in leprosy. PLoS Negl Trop Dis. 2016;10:e0004412. doi:10.1371/journal.pntd.0004412
46. Van Brakel WH, Nicholls PG, Das L, et al. Cohort study: investigating prediction, detection and pathogenesis of neuropathy and reactions in leprosy. Methods and baseline results of a cohort of multibacillary leprosy patients in North India. Lepr Rev. 2005;76:264.
47. Oliveira DTD, Sherlock J, Melo EVD, et al. Clinical variables associated with leprosy reactions and persistence of physical impairment. Rev Soc Bras Med Trop. 2013;46:600–604. doi:10.1590/0037-8682-0100-2013
48. Neder L, Weelden MV, Viola GR, et al. Qualidade de vida relacionada à saúde avaliada pelo Inventário Pediátrico de Qualidade de Vida 4.0 em pacientes pediátricos com hanseníase e manifestações musculoesqueléticas [Health-related quality of life evaluated by Pediatric Quality of Life Inventory 4.0 in pediatric leprosy patients with musculoskeletal manifestations]. Rev Bras Reumatol. 2015;55:414–419. doi:10.1016/j.rbr.2014.12.013
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.