Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Left Ventricular End-Diastolic Diameter and Cardiac Mortality in Duchenne Muscular Dystrophy
Authors Segawa K , Sugawara N , Maruo K , Kimura K, Komaki H, Takahashi Y, Sasaki M
Received 23 October 2019
Accepted for publication 7 December 2019
Published 16 January 2020 Volume 2020:16 Pages 171—178
DOI https://doi.org/10.2147/NDT.S235166
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Taro Kishi
Kazuhiko Segawa, 1 Norio Sugawara, 2 Kazushi Maruo, 3 Koichi Kimura, 4 Hirofumi Komaki, 5 Yuji Takahashi, 6 Masayuki Sasaki 7
1Department of Cardiology, National Center Hospital, National Center of Neurology and Psychiatry, Tokyo, Japan; 2Department of Psychiatry, Dokkyo Medical University School of Medicine, Tochigi, Japan; 3Department of Biostatistics, Faculty of Medicine, University of Tsukuba, Ibaraki, Japan; 4Department of General Medicine, The Institute of Medical Science, the University of Tokyo, Tokyo, Japan; 5Translational Medical Center, National Center of Neurology and Psychiatry, Tokyo, Japan; 6Department of Neurology, National Center Hospital, National Center of Neurology and Psychiatry, Tokyo, Japan; 7Department of Child Neurology, National Center Hospital, National Center of Neurology and Psychiatry, Tokyo, Japan
Correspondence: Kazuhiko Segawa
Department of Cardiology, National Center of Neurology and Psychiatry, 4-1-1 Ogawahigashimachi, Kodaira, Tokyo 187-8551, Japan
Tel +81-42-341-2711
Fax +81-42-344-6745
Email [email protected]
Purpose: This study aimed to examine weather left ventricular end-diastolic diameter (LVDd) could predict mortality from heart failure in patients with Duchenne muscular dystrophy (DMD) receiving standard cardio-protective therapies.
Patients and Methods: One hundred thirty-three patients with DMD aged ≥ 10 years who underwent echocardiography from 2011 to 2015 were included in this study and retrospectively followed until August 2018. Patients were divided into two groups according to LVDd at initial echocardiography: ≤ 54 mm (Group 1: n=119) and ≥ 55 mm (Group 2: n=14). To identify factors other than LVDd that may affect heart failure-related mortality, Group 2 patients who developed no left atrial (LA) enlargement, moderate mitral regurgitation (MR), or pulmonary hypertension (PH) during the observation period (Group 2A: n=5) were compared with those who newly developed one or more of those complications (Group 2B: n=7). Clinical outcomes were all-cause mortality and mortality from heart failure.
Results: Mean observation period was 5.5± 1.5 years in Group 1 and 4.4± 1.9 years in Group 2. A total of 14 patients (10.5%) died, including 6 of 119 (5.0%) patients in Group 1 and 8 of 14 (57.1%) patients in Group 2 (p< 0.001). Among these, 1 (0.8%) patient in Group 1 and 8 (57.1%) patients in Group 2 died from heart failure (p< 0.001). Group 2B patients had shorter survival compared to Group 2A patients (p=0.006).
Conclusion: LVDd ≥ 55 mm is a predictive factor for mortality from heart failure in patients with DMD. Complications including LA enlargement, moderate MR, and PH were found to be predictive factors for mortality from heart failure in a short period.
Keywords: cardiomyopathy, heart failure, echocardiography, left atrial enlargement, mitral regurgitation, pulmonary hypertension
Introduction
Duchenne muscular dystrophy (DMD) is a X-linked recessive disorder caused by mutations in the dystrophin gene. DMD is associated with progressive degeneration of skeletal and cardiac muscle tissue, which usually leads to cardiac and respiratory complications and ultimately, death.1 Multidisciplinary treatments including steroids, scoliosis surgery, respiratory support, and cardiac therapy have improved survival rates in patients with DMD.2 The use of non-invasive mechanical ventilation has also extended survival. Today, cardiomyopathy is the leading cause of death in these patients.3 The absence of cytoskeletal dystrophin protein makes cardiac myocytes vulnerable to mechanical damage.4 Clinically apparent cardiomyopathy, defined as left ventricular (LV) systolic dysfunction, is commonly diagnosed after age 10 years. Death from heart failure has been reported in some patients with cardiomyopathy.
Previous studies have described the effects of pharmacological interventions on cardiac mortality.5–7 However, despite standard cardio-protective therapies such as angiotensin converting enzyme inhibitors (ACEI), angiotensin receptor blockers (ARB), and beta blockers (BB), cardiac mortality is still prevalent in this patient population.
Although almost all patients with DMD develop cardiomyopathy, the age of onset, progression, and severity of the disease differ greatly among patients. As heart failure symptoms are masked by exercise inability due to skeletal muscle disorder, indices that lead to early therapeutic intervention for high risk patients are necessary. In the general population, left ventricular end-diastolic diameter (LVDd) and left ventricular ejection fraction (LVEF) are related to prognosis in dilated cardiomyopathy.8 While DMD patients who died of heart failure reportedly had significantly worse LV dilatation compared to those who died of respiratory failure,9 outcome indices that can be used to predict cardiac mortality have not been fully explored.
This study aimed to assess whether LVDd can predict heart failure-related deaths in patients with DMD receiving standard cardio-protective therapies.
Materials and Methods
Study Design and Participants
This retrospective cohort study targeted patients with DMD at the National Center of Neurology and Psychiatry. Patients aged ≥10 years who underwent echocardiography from January 2011 to December 2015 were included in this study and followed until August 2018. Clinical outcomes were all-cause mortality and mortality from heart failure. Exercise intolerance, a common symptom of heart failure, is not applicable in patients with DMD due to the inability to ambulate. Thus, heart failure was diagnosed according to symptoms such as sleep disturbance, loss of appetite, nausea, and abdominal pain or fullness, as well as supporting data including decreased LV systolic function and elevated brain natriuretic peptide.10 Sudden death without preceding signs or symptoms of heart failure was not included in deaths from heart failure.
This study was approved by the institutional review board of the National Center of Neurology and Psychiatry (A2015-068). All study participants were provided an opportunity to opt-out of participation at their free will. The data used in this study were obtained from a retrospective survey of clinical practice without any additional burden on patients. Accordingly, we considered that ethical issues could be solved by providing opt-out opportunities rather than written informed consent. This study was carried out in accordance with the principles of the Declaration of Helsinki.
Echocardiography
Echocardiography was performed by a single cardiologist (K.S.) for all patients. LVDd was measured using M-mode scan of the parasternal long-axis view with a 2D image. When the M-mode cursor could not be aligned perpendicularly to the LV long axis, LVDd was measured directly on 2D images. Left atrial (LA) enlargement was defined as a left atrial diameter (LAD) ≥40 mm. We did not correct LVDd and LAD for body surface area (BSA), as echocardiograms are often evaluated without correction for BSA in clinical practice. In addition, since non-ambulant DMD patients cannot keep the standing position, it is not easy to measure their height and body weight. Mitral regurgitation (MR) was evaluated by color Doppler flow image and was classified as none, mild, and moderate by regurgitation jet area (no severe MR). Pulmonary hypertension (PH) was defined as a ≥25 mmHg pressure gradient estimated from peak tricuspid regurgitation flow velocity.
We decided to use LVDd because it can be measured more easily and is more reproducible than other indices. Patients were divided into two groups according to LVDd at initial echocardiography: ≤54 mm (Group 1) and ≥55 mm (Group 2). We used a clinical relevant value of normal LVDd in men, which has been reported to be 50.2±4.1 mm,11 although the value may be influenced by age, physique, and race. Moreover, to clarify the predictive value of newly developed complications (i.e., LA enlargement, moderate MR, and PH), subgroup analysis was performed in Group 2 patients excluding those who had one or more complications at initial echocardiography. Specifically, patients in Group 2 who did not have these complications at initial echocardiography were divided further into two sub-groups: those who developed no new complications during the observation period (Group 2A), and those who developed one or more new complications during the observation period (Group 2B).
Survival curves were compared between Group 1 and Group 2, and between Group 2A and Group 2B. To assess the influence of LA dilatation, moderate MR, and PH on survival, the starting point of the survival curve in Group 2B was defined as when patients developed one or more new complications.
Statistical Analysis
Continuous and categorical variables are presented as minimum – maximum (median) and frequencies or percentages, respectively. Comparisons between Group 1 and Group 2, and between Group 2A and Group 2B, were performed using Fisher’s exact probability test and the Mann–Whitney U-test, respectively. Survival curves in patients with and without LV dilatation for all-cause mortality and mortality from heart failure were estimated by the Kaplan-Meier method. Differences in survival rate were evaluated using the Log rank test and the Cox proportional hazards model, including the following candidates of prognostic factors and covariates: age, mechanical ventilation, fractional shortening (FS), LAD, PH, moderate MR, ACEI/ARB, BB, aldosterone antagonist, diuretics, pimobendan, amiodarone, and steroids at initial cardiac evaluation. These variables were selected by the stepwise procedure (inclusion and exclusion criteria: p=0.1), with therapeutic agents treated as covariates. Survival curves for Group 2A and Group 2B were estimated by the Kaplan-Meier method. Differences in survival rate were evaluated using the Log rank test. P<0.05 was considered statistically significant.
Results
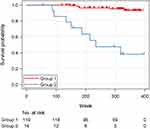
A total of 133 patients were included in this study. Patient clinical characteristics are summarized in Table 1. At initial echocardiography, LVDd was ≤54 mm in 119 of 133 (89.5%) patients (Group 1), and only the remaining 14 (10.5%) patients had LVDd  55 mm (Group 2). All patients with LVDd
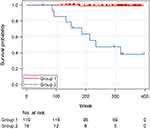
55 mm (Group 2). All patients with LVDd  55 mm showed reduced LV systolic function (FS<0.27) at the first echocardiography. Although median age at initial echocardiography was slightly higher in Group 2 compared to Group 1, there was no significant difference (p=0.051). The mean follow-up period was 5.5±1.5 years in Group 1 and 4.4±1.9 years in Group 2. Figures 1 and 2 show all-cause mortality and mortality from heart failure, respectively. A total of 14 (10.5%) patients died during the follow-up period, including 6 of 119 (5.0%) patients in Group 1 and 8 of 14 (57.1%) patients in Group 2. Overall survival during the 5-year follow-up period analyzed by the Log rank test revealed a significant difference between Group 1 (96.3%) and Group 2 (47.6%) (p<0.001). LVDd was not associated with all-cause mortality in the Cox proportional hazards model analysis (selected covariates: FS, PH, and steroids). One (0.8%) patient in Group 1 and 8 (57.1%) patients in Group 2 died from heart failure. Age range at death from heart failure was 14–38 (median, 23) years. Survival probability for the heart failure mortality during the 5-year follow-up period was estimated to be 99.0% for Group 1, which was significantly higher than 47.6% for Group 2 (Log rank test: p<0.001). LVDd was significantly associated with mortality from heart failure in the Cox proportional hazards model analysis (hazard ratio: 1.56, 95% confidence interval: 1.20–2.03; p=0.001; selected covariates: steroids, age, aldosterone antagonist, and LAD).
55 mm showed reduced LV systolic function (FS<0.27) at the first echocardiography. Although median age at initial echocardiography was slightly higher in Group 2 compared to Group 1, there was no significant difference (p=0.051). The mean follow-up period was 5.5±1.5 years in Group 1 and 4.4±1.9 years in Group 2. Figures 1 and 2 show all-cause mortality and mortality from heart failure, respectively. A total of 14 (10.5%) patients died during the follow-up period, including 6 of 119 (5.0%) patients in Group 1 and 8 of 14 (57.1%) patients in Group 2. Overall survival during the 5-year follow-up period analyzed by the Log rank test revealed a significant difference between Group 1 (96.3%) and Group 2 (47.6%) (p<0.001). LVDd was not associated with all-cause mortality in the Cox proportional hazards model analysis (selected covariates: FS, PH, and steroids). One (0.8%) patient in Group 1 and 8 (57.1%) patients in Group 2 died from heart failure. Age range at death from heart failure was 14–38 (median, 23) years. Survival probability for the heart failure mortality during the 5-year follow-up period was estimated to be 99.0% for Group 1, which was significantly higher than 47.6% for Group 2 (Log rank test: p<0.001). LVDd was significantly associated with mortality from heart failure in the Cox proportional hazards model analysis (hazard ratio: 1.56, 95% confidence interval: 1.20–2.03; p=0.001; selected covariates: steroids, age, aldosterone antagonist, and LAD).
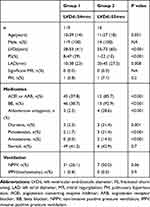 |
Table 1 Basic Patient Characteristics |
In Group 1, LV dilatation was noted only in 6 patients (0.5%) at final echocardiography. A decline in FS preceded LV dilatation in all 6 patients. None of the patients had complications (i.e., LA enlargement, moderate MR, or PH) at final echocardiography. One patient in Group 1 who died from heart failure without LV dilatation (LVDd: 50 mm) was hospitalized for fatigue, abdominal fullness, and dyspnea, and echocardiography at the time of death revealed severe hypokinesis of the LV wall and pericardial effusion.
Five patients died from causes other than heart failure; specifically, 1 died from sudden death, 1 from multiple organ failure after resuscitation from cardio-pulmonary arrest, 1 from renal failure and respiratory failure due to pneumonia, and 2 from unknown causes.
Table 2 summarizes the characteristics of patients in Group 2A (n=7) and Group 2B (n=5). Most patients in Group 2B had symptomatic heart failure, resulting in shorter survival compared to patients in Group 2A (Log rank test: p=0.006) (Figure 3).
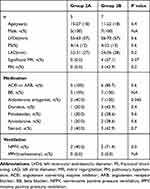 |
Table 2 Characteristics of Patients in Group 2A and Group 2B |
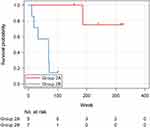 |
Figure 3 Kaplan-Meier estimate of survival from heart failure for Group 2A and Group 2B. Group 2B had a significantly shorter survival rate compared with Group 2A (p=0.006). |
Discussion
The present study investigated whether LVDd was a predictive factor for mortality from heart failure in patients with DMD aged  10 years. Patients with LVDd
10 years. Patients with LVDd  55 mm at initial echocardiography had a significantly higher mortality compared to those without. Moreover, most patients with LVDd
55 mm at initial echocardiography had a significantly higher mortality compared to those without. Moreover, most patients with LVDd  55 mm who newly developed one or more complications (i.e., LA enlargement, moderate MR, and PH) died in a short period after symptomatic heart failure. Vasan et al reported that, in the general population, an increase in LV internal dimension is a risk factor for congestive heart failure in individuals with no history of myocardial infarction.12 DMD results from a mutation in the dystrophin gene, which is responsible for the absence of the protein dystrophin. Dystrophin-deficient cardiomyocytes have an abnormally high membrane fragility. The loss of membrane integrity leads to increased permeability, thereby causing abnormal stress-induced influx of extracellular Ca2+ and membrane microrupture.13,14 In the dilated left ventricle, myocytes are stretched and this may facilitate further dilatation.
55 mm who newly developed one or more complications (i.e., LA enlargement, moderate MR, and PH) died in a short period after symptomatic heart failure. Vasan et al reported that, in the general population, an increase in LV internal dimension is a risk factor for congestive heart failure in individuals with no history of myocardial infarction.12 DMD results from a mutation in the dystrophin gene, which is responsible for the absence of the protein dystrophin. Dystrophin-deficient cardiomyocytes have an abnormally high membrane fragility. The loss of membrane integrity leads to increased permeability, thereby causing abnormal stress-induced influx of extracellular Ca2+ and membrane microrupture.13,14 In the dilated left ventricle, myocytes are stretched and this may facilitate further dilatation.
Previous studies demonstrated that ACEI/ARB and BB improve LV dysfunction,15–21 suggesting that interventions with these agents could improve FS or EF. However, for DMD patients with asymptomatic cardiomyopathy, a mild, or even significant improvement of LV function does not directly improve their quality of life. We emphasize the need for clinical research focused on not only improving LV function but also preventing symptomatic heart failure and resulting deaths. Duboc et al reported that an earlier intervention with perindopril was significantly associated with higher survival, free from all-cause death, over 10 years of follow-up in patients with preserved EF at baseline.5 The authors mentioned that depressed LV function (LVEF <45%) was a predictor of poor outcome, although they made no reference to LVDd.
The present study focused on LVDd because LV systolic dysfunction is common in DMD cardiomyopathy, whereas LV dilatation is not always observed. Most patients without LV dilatation escaped death from heart failure, even with LV systolic dysfunction. However, patients with LV dilatation had high mortality. Unlike LV dilatation that occurs independently of time (age), FS decline tends to increase with age. We considered the possibility that specific patients were more susceptible to death from heart failure regardless of age. Mekacini et al reported that no definite correlations were found between the clinical severity of myopathy and LV involvement.22 Wang et al reported that despite standard therapies including ACEI and BB, FS declined progressively in almost all patients, and that once heart failure deteriorated to New York Heart Association (NYHA) class 4, the survival period was short.23 On the other hand, Kwon et al reported that cardiac function in late-stage DMD patients (Swinyard-Deaver’s stage 7 or 8) showed stabilization of EF on adequate ventilatory support and optimal cardiac medication therapy until their mid-30s.24 Based on the results of the present study, we considered LVDd to be a critical marker for heart failure-related death. In patients with DMD cardiomyopathy, once LV dilates, it is generally irreversible. Although FS is also a potential predictive factor for heart failure-related mortality, it is reduced in a considerable number of DMD patients aged  10 years. Thus, cutoff FS that predicts death from heart failure is speculated to be much lower than the normal range (0.25–0.43). Standard cardio-protective therapies such as ACEI, ARB, and BB do not necessarily prevent LV dilatation. The mechanism underlying LV dilatation that affects only a portion of patients remains unknown. Neither the age of onset nor severity of LV dysfunction is correlated with the site of gene mutation.25
10 years. Thus, cutoff FS that predicts death from heart failure is speculated to be much lower than the normal range (0.25–0.43). Standard cardio-protective therapies such as ACEI, ARB, and BB do not necessarily prevent LV dilatation. The mechanism underlying LV dilatation that affects only a portion of patients remains unknown. Neither the age of onset nor severity of LV dysfunction is correlated with the site of gene mutation.25
At our institution, we have managed cardiomyopathy in patients with DMD for over 7 years according to recommendations summarized in a review article published by Bushby et al26. ACEI/ARB and BB are initiated at the onset of LV systolic dysfunction. Since no clinical evidence has been shown for other pharmacological therapies in DMD cardiomyopathy, the review recommends traditional treatment strategies for advanced heart failure. However, recent reports recommend that treatment with ACEI/ARB be initiated by 10 years of age.27,28 As such, since 2018, we have followed this recommendation when treating DMD patients with normal EF (these patients were not included in the present study). While the optimal timing for initiation of treatment with aldosterone antagonists remains unknown, early initiation has been shown to offer effective and safe cardioprotection in mice29 and DMD patients.30–32 However, it is unclear whether the attenuation of cardiac systolic dysfunction by aldosterone antagonists reduces mortality from heart failure, and there are no specific treatment policies for DMD patients with advanced heart failure. Thus, mortality from heart failure is still a major problem despite the availability of contemporary cardio-protective therapies. We use aldosterone antagonists when LVDd exceeds 54 mm based on studies on heart failure in the general population.33,34 Aldosterone antagonist use was significantly associated with survival free from heart failure in the Cox proportional hazards model analysis (hazard ratio: 0.00, 95% confidence interval: 0.00–0.17; p=0.007). Although survival is affected by the extent of aggressive therapy for end-stage heart failure, patients with end-stage heart failure generally do not recover even with intensive care.
Echocardiography is the standard modality for evaluating cardiac function in DMD patients.9 For non-ambulant patients and their caregivers, transferring the patient from a wheelchair to an examination table, and into a lateral decubitus position, is stressful. Therefore, we often carry out echocardiography in a seated position for obese non-ambulant patients. In addition, scoliosis and thoracic deformity make echocardiography more difficult. LVDd can be measured more easily and is more reproducible than other indices.
In a recent study, DMD patients with normal EF were shown to have reduced circumferential strain on cardiac magnetic resonance imaging (MRI).35 Another study reported that DMD patients with EF >55% were positive for late gadolinium enhancement in 30% of cases.36 Although ventricular strain rate and late gadolinium enhancement can predict early myocardial damage, cardiac MRI is costly and time-consuming compared with echocardiography. Furthermore, the relationship between early abnormal findings and heart failure-related death is not well understood.
In the general population, 2013 ACCF/AHA guidelines for the management of heart failure classify heart failure into Stages A, B, C, and D.37 Stage C, which corresponds to NYHA classes 2 and 3, is defined as structural heart disease with prior or current symptoms of heart failure. Due to loss of exercise capability, DMD patients do not complain of dyspnea on exertion, and thus, Stage C heart failure cannot be applied to DMD cardiomyopathy. For non-ambulant DMD patients, symptomatic heart failure is equivalent to NHYA class 4. From this perspective, asymptomatic patients with LV dilatation can be classified as Stage C. We suggest that the timing of additional therapeutic strategies should be identified to prevent NYHA class 4 heart failure in DMD patients with LV dilatation. For this reason, we propose the following stage classification for DMD cardiomyopathy based on the results of the present study (Figure 4): Stages 0, 1, and 3 corresponding to Stages A, B, and D according to ACCF/AHA guidelines, respectively. We classify asymptomatic DMD patients with LV dilatation as Stage 2, rather than Stage C. Furthermore, we suggest that stage variation is clinically more important than FS (or EF) variation in individual patients.Since death from heart failure occurs even in young patients undergoing ACEI/ARB and BB therapies, additional therapeutic strategies based on LVDd are needed in order to prevent heart failure-related death.
Limitations
This study was carried out with a retrospective cohort study design at a single institution. The observation period was only a part of the entire course of DMD cardiomyopathy. Some patients dropped out and could not be followed up to the end of the observation period. Finally, characteristics of patients in our hospital may not be representative of those of the entire DMD population. Therefore, there is concern as to whether the findings of the present study can be applied to other facilities or regions.
Prospective long-term trials with a large sample size will be necessary to determine the impact of LVDd on survival in DMD patients.
Conclusion
LVDd is a predictive factor for mortality from heart failure in patients with DMD. Patients who developed symptomatic heart failure frequently exhibited LA enlargement, moderate MR, and PH in addition to LV dilatation and had poor prognoses. Strategies aimed at preventing LV dilatation are necessary to improve survival from heart failure.
Disclosure
Dr Hirofumi Komaki reports grants, personal fees from Pfizer Japan Inc., Sanofi Aventis, Taiho Pharmaceutical Co., Ltd., Nippon Shinyaku Co., Ltd., Daiichi Sankyo Co. Ltd, Chugai Pharma, Sumitomo Dainippon Pharma Co., PTC Therapeutics, Takeda Pharmaceutical Co., Ltd., Biogen, Alexion Pharmaceuticals Inc., and Sarepta therapeutics, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Van Ruiten H, Bushby K, Guglieri M. State of the art advances in Duchenne muscular dystrophy. EMJ. 2017;2(1):90–99.
2. Strehle E-M, Straub V. Recent advances in the management of Duchenne muscular dystrophy. Arch Dis Child. 2015;100(12):1173–1177. doi:10.1136/archdischild-2014-307962
3. Eagle M, Baudouin SV, Chandler C, Giddings DR, Bullock R, Bushby K. Survival in Duchenne muscular dystrophy: improvements in life expectancy since 1967 and the impact of home nocturnal ventilation. Neuromuscular Disord. 2002;12(10):926–929. doi:10.1016/S0960-8966(02)00140-2
4. Townsend D, Yasuda S, McNally E, Metzger JM. Distinct pathophysiological mechanisms of cardiomyopathy in hearts lacking dystrophin or the sarcoglycan complex. FASEB J. 2011;25(9):3106–3114. doi:10.1096/fj.10-178913
5. Duboc D, Meune C, Pierre B, et al. Perindopril preventive treatment on mortality in Duchenne muscular dystrophy: 10 years’ follow-up. Am Heart J. 2007;154(3):596–602. doi:10.1016/j.ahj.2007.05.014
6. Matsumura T, Tamura T, Kuru S, Kikuchi Y, Kawai M. Carvedilol can prevent cardiac events in Duchenne muscular dystrophy. Intern Med. 2010;49(14):1357–1363. doi:10.2169/internalmedicine.49.3259
7. Ogata H, Ishikawa Y, Ishikawa Y, Minami R. Beneficial effects of beta-blockers and angiotensin-converting enzyme inhibitors in Duchenne muscular dystrophy. J Cardiol. 2009;53(1):72–78. doi:10.1016/j.jjcc.2008.08.013
8. Armstrong WF, Ryan T. Feigenbaum’s Echocardiography.
9. Sasaki K, Sakata K, Kachi E, Hirata S, Ishihara T, Ishikawa K. Sequential changes in cardiac structure and function in patients with Duchenne type muscular dystrophy: a two-dimensional echocardiographic study. Am Heart J. 1998;135(6):937–944. doi:10.1016/S0002-8703(98)70057-2
10. Spurney CF. Cardiomyopathy of Duchenne muscular dystrophy: current understanding and future directions. Muscle Nerve. 2011;44(1):8–19. doi:10.1002/mus.v44.1
11. Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16(3):233–271. doi:10.1093/ehjci/jev014
12. Vasan RS, Larson MG, Benjamin EJ, Evans JC, Levy D. Left ventricular dilatation and the risk of congestive heart failure in people without myocardial infarction. N Engl J Med. 1997;336(19):1350–1355. doi:10.1056/NEJM199705083361903
13. Allen DG, Whitehead NP. Duchenne muscular dystrophy–what causes the increased membrane permeability in skeletal muscle? Int J Biochem Cell Biol. 2011;43(3):290–294. doi:10.1016/j.biocel.2010.11.005
14. Williams IA, Allen DG. Intracellular calcium handling in ventricular myocytes from mdx mice. Am J Physiol Heart Circ Physiol. 2007;292(2):H846–H855. doi:10.1152/ajpheart.00688.2006
15. Allen HD, Flanigan KM, Thrush PT, et al. A randomized, double-blind trial of lisinopril and losartan for the treatment of cardiomyopathy in duchenne muscular dystrophy. PLoS Curr. 2013;5. doi:10.1371/currents.md.2cc69a1dae4be7dfe2bcb420024ea865
16. Viollet L, Thrush PT, Flanigan KM, Mendell JR, Allen HD. Effects of angiotensin-converting enzyme inhibitors and/or beta blockers on the cardiomyopathy in Duchenne muscular dystrophy. Am J Cardiol. 2012;110(1):98–102. doi:10.1016/j.amjcard.2012.02.064
17. Kwon HW, Kwon BS, Kim GB, et al. The effect of enalapril and carvedilol on left ventricular dysfunction in middle childhood and adolescent patients with muscular dystrophy. Korean Circ J. 2012;42(3):184–191. doi:10.4070/kcj.2012.42.3.184
18. Kajimoto H, Ishigaki K, Okumura K, et al. Beta-blocker therapy for cardiac dysfunction in patients with muscular dystrophy. Circ J. 2006;70(8):991–994. doi:10.1253/circj.70.991
19. Ramaciotti C, Heistein LC, Coursey M, et al. Left ventricular function and response to enalapril in patients with Duchenne muscular dystrophy during the second decade of life. Am J Cardiol. 2006;98(6):825–827. doi:10.1016/j.amjcard.2006.04.020
20. Jefferies JL, Eidem BW, Belmont JW, et al. Genetic predictors and remodeling of dilated cardiomyopathy in muscular dystrophy. Circulation. 2005;112(18):2799–2804. doi:10.1161/CIRCULATIONAHA.104.528281
21. Ishikawa Y, Bach JR, Minami R. Cardioprotection for Duchenne’s muscular dystrophy. Am Heart J. 1999;137(5):895–902. doi:10.1016/S0002-8703(99)70414-X
22. Melacini P, Vianello A, Villanova C, et al. Cardiac and respiratory involvement in advanced stage Duchenne muscular dystrophy. Neuromuscular Disord. 1996;6(5):367–376. doi:10.1016/0960-8966(96)00357-4
23. Wang M, Birnkrant DJ, Super DM, Jacobs IB, Bahler RC. Progressive left ventricular dysfunction and long-term outcomes in patients with Duchenne muscular dystrophy receiving cardiopulmonary therapies. Open Heart. 2018;5(1):e000783. doi:10.1136/openhrt-2018-000783
24. Kwon SW, Kang S-W, Kim J-Y, et al. Outcomes of cardiac involvement in patients with late-stage Duchenne muscular dystrophy under management in the pulmonary rehabilitation center of a tertiary referral hospital. Cardiology. 2012;121(3):186–193. doi:10.1159/000336810
25. Ashwath ML, Jacobs IB, Crowe CA, Ashwath RC, Super DM, Bahler RC. Left ventricular dysfunction in Duchenne muscular dystrophy and genotype. Am J Cardiol. 2014;114(2):284–289. doi:10.1016/j.amjcard.2014.04.038
26. Bushby K, Finkel R, Birnkrant DJ, et al. Diagnosis and management of Duchenne muscular dystrophy, part 2: implementation of multidisciplinary care. Lancet Neurol. 2010;9(2):177–189. doi:10.1016/S1474-4422(09)70272-8
27. McNally EM, Kaltman JR, Benson DW, et al. Contemporary cardiac issues in Duchenne muscular dystrophy. Circulation. 2015;131(18):1590–1598. doi:10.1161/CIRCULATIONAHA.114.015151
28. Birnkrant DJ, Bushby K, Bann CM, et al. Diagnosis and management of Duchenne muscular dystrophy, part 2: respiratory, cardiac, bone health, and orthopaedic management. Lancet Neurol. 2018;17(4):347–361. doi:10.1016/S1474-4422(18)30025-5
29. Rafael-Fortney JA, Chimanji NS, Schill KE, et al. Early treatment with lisinopril and spironolactone preserves cardiac and skeletal muscle in duchenne muscular dystrophy mice. Circulation. 2011;124(5):582–588. doi:10.1161/CIRCULATIONAHA.111.031716
30. Raman SV, Hor KN, Mazur W, et al. Eplerenone for early cardiomyopathy in Duchenne muscular dystrophy: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2015;14(2):153–161. doi:10.1016/S1474-4422(14)70318-7
31. Raman SV, Hor KN, Mazur W, et al. Eplerenone for early cardiomyopathy in Duchenne muscular dystrophy: results of a two-year open-label extension trial. Orphanet J Rare Dis. 2017;12(1):39. doi:10.1186/s13023-017-0590-8
32. Raman SV, Hor KN, Mazur W, et al. Stabilization of early Duchenne cardiomyopathy with aldosterone inhibition: results of the multicenter AIDMD trial. J Am Heart Assoc. 2019;8(19):e013501. doi:10.1161/JAHA.119.013501
33. Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341(10):709–717. doi:10.1056/NEJM199909023411001
34. Zannad F, McMurray JJ, Krum H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364(1):11–21. doi:10.1056/NEJMoa1009492
35. Hor KN, Wansapura J, Markham LW, et al. Circumferential strain analysis identifies strata of cardiomyopathy in Duchenne muscular dystrophy: a cardiac magnetic resonance tagging study. J Am Coll Cardiol. 2009;53(14):1204–1210. doi:10.1016/j.jacc.2008.12.032
36. Hor KN, Taylor MD, Al-Khalidi HR, et al. Prevalence and distribution of late gadolinium enhancement in a large population of patients with Duchenne muscular dystrophy: effect of age and left ventricular systolic function. J Cardiovasc Magn Reson. 2013;15(1):107. doi:10.1186/1532-429X-15-107
37. Yancy CW, Jessup M, Bozkurt B, et al. ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):e147–e239. doi:10.1016/j.jacc.2013.05.019
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.