Back to Journals » International Journal of Nanomedicine » Volume 14
Krill Oil-Incorporated Liposomes As An Effective Nanovehicle To Ameliorate The Inflammatory Responses Of DSS-Induced Colitis
Authors Kim JH, Hong SS, Lee M, Lee EH, Rhee I, Chang SY, Lim SJ
Received 19 June 2019
Accepted for publication 23 August 2019
Published 6 November 2019 Volume 2019:14 Pages 8305—8320
DOI https://doi.org/10.2147/IJN.S220053
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Thomas Webster
Jin-Hee Kim,1,* Soon-Seok Hong,1,* Myoungsoo Lee,2 Eun-Hye Lee,1 Inmoo Rhee,1 Sun-Young Chang,2 Soo-Jeong Lim1
1Department of Integrated Bioscience and Biotechnology, Sejong University, Seoul 05006, Republic of Korea; 2Laboratory of Microbiology, College of Pharmacy, and Research Institute of Pharmaceutical Science and Technology (RIPST), Ajou University, Suwon 16499, Republic of Korea
*These authors contributed equally to this work
Correspondence: Sun-Young Chang
Laboratory of Microbiology, College of Pharmacy, and Research Institute of Pharmaceutical Science and Technology (RIPST), Ajou University, Suwon 16499, Republic of Korea
Tel +82 31 219 3454
Fax +82 31 219 3435
Email [email protected]
Soo-Jeong Lim
Department of Integrated Bioscience and Biotechnology, Sejong University, 209 Neungdong-ro, Gwangjin-gu, Seoul 05006, Republic of Korea
Tel +82 2 3408 3767
Fax +82 2 3408 4334
Email [email protected]
Background: Phosphatidylcholine (PC) and Omega-3 fatty acid (Omega-3) are promising therapeutic molecules for treating inflammatory bowel disease (IBD).
Purpose: Based on the IBD therapeutic potential of nanoparticles, we herein sought to develop Omega-3-incorporated PC nanoparticles (liposomes) as an orally administrable vehicle for treating IBD.
Methods: Liposomes prepared with or without Omega-3 incorporation were compared in terms of colloidal stability and anitiinflammatory effects.
Results: The incorporation of free Omega-3 (alpha-linolenic acid, eicosapentaenoic acid or docosahexaenoic acid) into liposomes induced time-dependent membrane fusion, resulting in particle size increase from nm to μm during storage. In contrast, krill oil incorporation into liposomes (KO liposomes) did not induce the fusion and the particle size maintained <250 nm during storage. KO liposomes also maintained colloidal stability in simulated gastrointestinal conditions and exhibited a high capacity to entrap the IBD drug, budesonide (BDS). KO liposomes greatly suppressed the lipopolysaccharide-induced production of pro-inflammatory cytokines in cultured macrophages and completely restored inflammation-impaired membrane barrier function in an intestinal barrier model. In mice subjected to dextran sulfate sodium-induced colitis, oral administration of BDS-entrapped KO liposomes suppressed tumor necrosis factor-α production (by 84.1%), interleukin-6 production (by 35.3%), and the systemic level of endotoxin (by 96.8%), and slightly reduced the macroscopic signs of the disease.
Conclusion: Taken together, KO liposomes may have great potential as a nanovehicle for oral delivery of IBD drugs.
Keywords: inflammatory bowel disease, phosphatidylcholine, liposomes, krill oil, Omega-3 fatty acid
Introduction
Inflammatory bowel diseases (IBD) are chronic inflammatory conditions that affect the gastrointestinal (GI) tract. Ulcerative colitis (UC) and Crohn’s disease, the two major types of IBD, share some common symptoms, such as bleeding, diarrhea, and weight loss. The precise etiology of IBD is not fully understood yet, but it appears to involve genetic and environmental factors.1 No curative treatment is available; most of the current therapies use the frequent administration of anti-inflammatory and/or immunosuppressive drugs with the goal of suppressing abnormal immune activation, achieving remission, and preventing relapse.2 These therapies, however, often lead to serious adverse effects and exhibit incomplete clinical efficacy. Thus, a major goal in IBD research is the development of new therapeutic approaches.
Accumulating evidence suggests that the intestinal mucosal barrier plays a central role in the pathogenesis of IBD.3 The major component of this barrier is the epithelial layer, the luminal surface of which is covered by mucus. Phosphatidylcholine (PC) binds to mucins of the colonic mucus to establish a protective surface that prevents potentially toxic antigens, including bacteria, from adhering and penetrating. PC is also integrated into the plasma membrane of enterocytes, and participates in various signaling pathways associated with inflammation.4 The level of PC in the colonic mucus is markedly reduced in UC patients, allowing colonic bacteria to penetrate through the mucosal barrier and trigger nonspecific but aggressive immune responses.2 Based on these findings, it has been hypothesized that the local supplementation of PC could help normalize the colonic mucus PC content, re-constitute the mucosal barrier, and suppress inflammation in UC. Indeed, PC supplementation studies in animal models and patients have yielded very encouraging results4,5 and PC supplementation is currently considered to be a potential approach for treating IBD without substantial adverse effects.2
Omega‐3 fatty acids (Omega-3) are long chain polyunsaturated fatty acids that range from 18 to 22 carbon atoms in chain length and present the first of many double bonds at the third carbon. Dietary supplement with Omega-3 provides multiple health benefits, including the inhibition of various aspects of inflammation. The mechanisms underlying these effects involve the incorporation of fatty acid into cell membrane and the decreased activation of pro-inflammatory transcription factors. Omega-3 intake has been shown to attenuate colonic damage and inflammation in animal models of IBD.6 Similarly, krill oil (KO), which is a marine-derived oil that is rich in Omega-3, was found to reduce intestinal inflammation in association with improved epithelial integrity.7,8 Collectively, these results strongly suggest the potential of Omega-3 in ameliorating the inflammatory responses of IBD. However, Omega-3 exhibits chemical instability due to its intrinsic susceptibility to oxidative degradation, potentially limiting its health benefits.9
For IBD therapy, researchers have sought to develop formulations that can increase the local drug concentration and residence time in the inflamed tissue, leading to enhanced therapeutic activity. Intestinal transit times are frequently accelerated in IBD patients as a consequence of diarrhea, but nanoparticle formulations have been recognized as an efficient means to prolong the colonic passage of drugs.10 The mucus layer tends to be increased in inflamed/ulcerated regions, allowing mucus-adherent nanoparticles to localize in these regions.11 Following this adherence, the increased permeability of the mucosa can allow nanoparticles to passively accumulate in the gaps between the cells; the nanoparticles may then be taken up by phagocytic cells that populate the inflamed region.11 Given these tendencies, nanoparticles are considered to be an ideal formulation for IBD therapy.
Liposomes formed of PC bilayers represent first-generation nanoparticle drug carriers that are already in clinical use.12 The liposome encapsulation of chemically unstable compounds, such as Omega-3, can offer a physical barrier that prevents the oxidative degradation of these compounds.9 This, together with the anti-inflammatory activity and intestinal barrier activity of PC, suggests that Omega-3-incorporated liposomes may be a promising nanovehicle for effective IBD therapy.
Fatty acids are amphipathic, and are thus rapidly incorporated into PC membranes.13 The monounsaturated fatty acid, oleic acid, was intercalated between PC molecules at up to 33 mol% and oleic acid-incorporated membranes retained the physical stability of conventional liposomes.14 Furthermore, oleic acid or its 2-hydroxyl derivative-incorporated liposomes demonstrated multiple benefits as drug carriers, such as improved colloidal stability and hydrophobic drug-loading capacity.14,15 The hydrocarbon tails of Omega-3, due to the presence of multiple double bonds in the cis configuration, are much more curved and flexible than those of monounsaturated fatty acid, suggesting that it may affect the liposome structure differently.16 With this regard, researchers studying the cellular membrane structure from a biophysical perspective reported that the insertion of docosahexaenoic acid (DHA) causes phase separation and fusion of membranes, leading to marked structural destabilization.17 Assuming that the effect of Omega-3 on the liposomal membrane may differ depending on the chemical structure and insertion content of Omega-3, we herein explored the impact of Omega-3 on the physical stability, the anti-inflammatory activity, and intestinal barrier activity of liposomes in order to develop an effective nanovehicle platform for IBD therapy.
Materials And Methods
Reagents And Materials
1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC), 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC), and dioleoyltrimethylaminopropane (DOTAP) were purchased from Avanti Polar Lipid Inc. (Alabaster, AL, USA). Budesonide (BDS), alpha-linolenic acid (ALA, ≥98% purity), and eicosapentaenoic acid (EPA, ≥98% purity) were obtained from Cayman Chemical (Ann Arbor, MI, USA). DHA (≥98% purity), cholesterol (CHOL), α-tocopherol, mucin (from porcine stomach), and lysozyme (from chicken egg white) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Lipopolysaccharide (LPS) was purchased from Invivogen (San Diego, CA, USA). Dextran sulfate sodium (DSS) was obtained from MP Biomedicals (Solon, OH, USA). Krill oil (NKO®; specific composition, 42% w/w phospholipids, ≥26.5 w/w% total Omega-3, ≥8.5% w/w DHA, ≥14.5% w/w EPA, and 0.125±0.025 w/w% astaxanthin) was kindly provided by Neptune Technologies & Bioresources Inc. (Quebec, Canada).
Cell Lines And Culture Conditions
RAW 264.7 (murine macrophage) and CaCO-2 (human intestinal epithelial) cell lines were obtained from American Type Culture Collection (Manassas, VA, USA). Cells were cultured in DMEM (Welgene, Daegu, Korea) supplemented with 10% thermal-inactivated FBS and 100 units/mL penicillin/streptomycin (Gibco, Grand Island, NY, USA). Cells were grown in incubators in a humid atmosphere of 95% air and 5% CO2.
Preparation Of Liposomes
Liposomes were prepared by freeze-drying method as reported earlier.18 Lipid mixtures were dissolved in tertiary butyl alcohol. When the incorporation of Omega-3 and/or BDS was required, appropriate amounts of each were dissolved together with the lipid mixture. The mixtures were freeze dried (FDU-1200, EYELA, Tohoku, Japan). The obtained lipid cakes were hydrated with 5% dextrose dissolved in distilled water. The liposome dispersions were shortly vortexed and sonicated for 1 hr at above the known phase-transition temperature of PC by using ultrasonic cleaning bath (3510-DTH Ultrasonic cleaner, Branson, Danbury, CT, USA). To obtain a liposomal dispersion with improved homogeneity, additional sonication for 7 mins was performed by using a cell disruptor (Bioruptor®, UCD-200 T, Cosmo Bio, Tokyo, Japan) set at 250 W. The prepared liposomes were stored at 4°C until use.
Physicochemical Characterization Of Liposomes
Particle sizes of dispersed liposomes were measured by the dynamic light scattering method using a fiber-optic particle analyzer (FPAR-1000, Otsuka Electronics, Japan). Prior to measurement, liposomes were appropriately diluted with 5% dextrose solution. System was used in the auto-measuring mode and the particle size analysis data were assessed using CONTIN program provided by the manufacturer. Zeta potentials, the electrical potential at the shear plane of the liposomal particles, were determined using zetasizer (Zetasizer Nano ZSP, Malvern Instruments, Northampton, MA). Prior to measurement, samples were diluted 50-fold with deionized water to reach the analytical measurement range. Default instrument settings and automatic analysis were used for all measurements. Each measurement was carried out in duplicate.
To perform negative-stain transmission electron microscopy, liposome samples were dropped on 200 mesh copper grid coated with carbon and negatively stained with 2% uranyl acetate for 1 min. Excess stain was removed, allowed to air-dry completely and dried samples were examined by using Tecnai G2 spirit (FEI Company, Hillsboro, OR, USA) operating at 120 keV.
With liposome samples diluted to 23 mM of total lipid concentration, differential scanning calorimetry (DSC) was performed using a NanoDSC® (TA instrument, DE, USA) at the scan rate of 1°C/min and 35–65°C temperature range. Five percent dextrose solution was used as a reference. Data were analyzed using Nano Analyze software provided by manufacturer.
Storage stability of liposomes was assessed by monitoring changes in their sizes and morphology when incubated at room temperature over time. The GI stability of liposomes was evaluated by monitoring changes in their sizes when 50 μL of liposomes were incubated with 950 μL of phosphate-buffered saline (pH 7.4), simulated intestinal fluid (SIF, 6.8 g KH2PO4/1 L water, pH 7.5, with 10 g/L pancreatin), or simulated gastric fluid (SGF, 2 g NaCl/1 L water, pH 1.2, with pepsin 3.2 g/L).19
To evaluate the adhesive properties of the liposomes, their adsorption onto charged proteins was determined by using mucin and lysozyme as negatively and positively charged model proteins, respectively.20,21 Lysozyme or mucin solution (4 mg/mL in distilled water) was incubated with liposome dispersion (1:1, v/v, 40 μmole lipid per 1 mL) for 1 hr at 37°C. Then, non-adsorbed lysozymes were separated by dialysis using a Franz-type vertical diffusion system in which dialysis membrane (molecular weight cutoff 100 kDa for lysozyme and 1000 kDa for mucin) was placed between donor chamber and receptor chamber. After 4 hrs, samples containing non-adsorbed proteins were withdrawn from the receptor chamber and the protein concentration was quantified by the UV-Vis spectrophotometer (Beckman Coulter, CA, USA) at 280 nm. 4 mg/mL of each protein solution pre-incubated with 5% dextrose (liposome-free) underwent the same procedure and the dialyzed amount was used as a 100% control of free/non-adsorbed proteins.
Determination Of BDS Concentration Encapsulated In Liposomes
Prepared liposome dispersions were immediately filtered through a 0.8-μm syringe membrane filter to remove the non-encapsulated/precipitated BDS. The concentration of encapsulated BDS was then determined by HPLC analysis.22 Fifty microliter of BDS-entrapped liposome dispersion was freeze-dried, dissolved in 0.5 mL of methanol, and then centrifuged at 10,000 rpm for 10 mins. Twenty microliter of the supernatant BDS sample was injected into the Nanospace SI-2 HPLC system (Shiseido Co., Ltd, Tokyo, Japan) equipped a mobile phase delivery pump (SP 3201) and UV-visible detector (SP 3002). A Capcellpak C18 column (UG120, Shiseido, Tokyo, Japan) was used to perform the isocratic elution with a mobile phase consisting of methanol and distilled water (72:28, v/v), the flow rate of 0.8 mL/min, and column temperature of 40°C. The detection wavelength of BDS was 243 nm.
In Vitro Effect Of KO Liposomes On Epithelial Barrier Function
Epithelial barrier integrity was assessed by measuring transepithelial electrical resistance (TEER) with an epithelial volt-ohm meter (EVOM; World Precision Instruments, Berlin, Germany). Decreased TEER is a significant indicator of impaired barrier function.
Caco-2 cells, an intestinal epithelial cell line widely used as a model to study intestinal barrier function in vitro, were placed at a density of 3.75×105 cells per well onto a 24-mm polycarbonate transwell insert plates (0.4 μm pore size and a surface area of 4.67 cm2 (Corning, NY, USA)). The filter was placed in 6-well cell culture plates. The cell culture medium was changed every 3 days until the cells were fully differentiated (TEER value >1200 Ω ‧ cm2). A steady-state of TEER was achieved after about 3 weeks, indicating that the barrier function model was established. RAW 264.7 macrophages were separately seeded at 8.5×105 cells/well into 6-well tissue culture plate. After 2 days, fresh, serum-free DMEM media containing 100 ng/mL of LPS were given to cells to mimic the inflammation. After 5-hr LPS stimulation, the culture supernatants were collected and then applied to fully differentiate Caco-2 monolayer (1 mL per well) to induce the disruption of the monolayer. When to test the effect of liposomes on the barrier function impaired by the inflammation, liposome dispersions or an equal volume of vehicle were given to Caco-2 monolayer cells right after supernatant stimulation. After an additional incubation for 3 hrs, the monolayer cells were rinsed with Hank’s Balanced Salts Solution (HBSS; Welgene, Daegu, Korea) for 10 mins and then TEER was measured. Measurements were carried out at a constant temperature at three different locations within each well. Wells containing only culture medium were used as a blank control.
TEER (Ω⋅cm2) = (measured TEER – control TEER) × effective membrane area of the cell culture well
In Vitro Effect Of KO Liposomes On The Level Of Pro-Inflammatory Cytokines
For IL-6 detection, RAW 264.7 cells were seeded at 2.5×105 cells/well into 24-well tissue culture plate and incubated overnight. And then, the cells were given fresh DMEM without FBS. To evaluate the anti-inflammatory effect of liposomes or BDS solution (in DMSO), appropriate amount of each was applied to the cells. At 2-hr post-incubation, fresh media were given again and the cells were stimulated with 100 ng/mL of LPS for 6 hrs. Concentration of IL-6 in the culture supernatants was quantified by murine IL-6 Quantikine® ELISA kit (R&D Systems Inc., MN, USA) according to the manufacturer’s instructions. For tumor necrosis factor-alpha (TNF-α) detection, RAW 264.7 cells were seeded at 1×106 cells/well into 6-well plate with or without BDS solution or liposomes. After 2-hr incubation, cells were harvested and washed with DMEM media. The cells were re-plated at 1×104 cells/well into 96-well and stimulated with the indicated concentration of 100 ng/mL of LPS for 24 hrs. TNF-α in the supernatant was then quantified using ELISA kit, as detailed by the manufacturer (R&D Systems Inc., MN, USA).
Induction Of DSS-Mediated Colitis In Mice
All experiments were approved by the Institutional Animal Care and Use Committee of Ajou University (IACUC approval number 2017–0021) and followed by the guidelines of AAALAC international and the Laboratory Animal Act of Korean Food and Drug Administration. Six-week-old female C57BL/6NCrljOri mice purchased from Orient Bio (Seongnam-Si, Korea) were housed at 24±1°C with a 12-hr light/dark cycle and food/water available ad libitum. Colitis was induced by administration of autoclaved drinking water containing 2% DSS for 5 consecutive days. To evaluate the progression of colitis, a disease activity index (DAI) score was assessed during the duration of experiment. The DAI was determined by combining scores of weight loss compared to initial weight, stool consistency, and bleeding (divided by three). Scores were defined as follows: weight loss: 0 (no loss), 1 (1–5%), 2 (5–10%), 3 (10–20%), and 4 (>20%); stool consistency: 0 (normal), 2 (loose stool), and 4 (diarrhea); and bleeding: 0 (no bleeding), 2 (slight bleeding), 4 (gross bleeding).
In Vivo Effects Of Liposomes On DSS-Induced Colitis
Prior to the induction of colitis, mice were randomized into five groups: the negative control group (no DSS, n=4), the vehicle-treated DSS group (positive control, n=6), the KO liposome-treated DSS group (KO liposome group, n=6), the BDS-treated DSS group (BDS group, n=8), and the BDS-encapsulated KO liposome-treated DSS group (BDS(+) KO liposome group, n=8). Free BDS was prepared by dispersing BDS at a concentration of 53 μg/mL in oil-in-water emulsion containing 0.5% soybean oil (Sigma-Aldrich, St. Louis, MO, USA) inner core stabilized by 1% carboxymethylcellulose (Sigma-Aldrich, St. Louis, MO, USA). KO liposomes and BDS(+) KO liposomes were prepared with the same composition of lipid:KO mixture in the presence of BDS (53 μg per 40 μmole of lipid mixture). BDS was given to mice at a dose of 0.4 mg/kg BDS and liposomes were given to mice at a dose corresponding to 300 μmole/kg as a total lipid mixture. Positive control group received vehicle solution instead of treatment. Each treatment was given to mice by oral gavage at the scheduled time points.
Body weight and DAI were checked daily to assess the severity of colitis. At day 9 after administration of DSS, serum and colon tissue samples were collected. Colon length and weight were measured to assess disease severity. The distal colons were washed with PBS and fixed in 4% formaldehyde at 4°C. The tissues were dehydrated by gradually soaking them in alcohol and xylene and then embedded in paraffin. The paraffin-embedded specimens were cut into 5-μm sections, stained with hematoxylin and eosin, and viewed with a digital light microscope DS-Fi2 (Nikon, Tokyo, Japan). Colon tissues were subjected to pathological scoring, which was performed in a blinded manner by a skilled pathologist.23
To assess the effect of liposomes on inflammation in vivo, cytokine (TNF-α, IL-6 and IFN-γ) level in serum collected from experimental groups was measured using murine inflammatory cytokine beads array kit (CBA kit, BD biosciences, USA) according to the manufacturer’s instructions. Briefly, serum was diluted twofold with dilution buffer in CBA kit. Diluted samples were incubated with cytokine anti-beads at room temperature for 2 hrs. Then, CBA data were acquired on BD FACSARIA Ⅲ (BD bioscience, San Jose, CA, USA) and analyzed using FCAP array software for quantifying cytokine level of samples.
Serum level of circulating endotoxin LPS was determined by Pierce LAL chromogenic endotoxin quantitation kit (Thermo scientific, USA). Briefly, pooled serum diluted 50-fold with endotoxin-free water was dispensed into microplate well. After 5-min incubation at 37°C, limulus amebocyte lysate was added, followed by addition of chromogenic substrate for detecting LPS. LPS concentration was calculated by measurement of absorbance of samples and standards at 405 nm on a plate reader.
Statistical Analysis
Statistically significant differences for results obtained under different experimental conditions were determined using the two-tailed unpaired Student’s t-test or one-way ANOVA, Bonferroni’s Multiple Comparison Test.
Results
Effect Of Omega-3 Incorporation On The Physicochemical Properties Of Liposomes
Liposomes with varying compositions were prepared as described in Table 1. ALA (three double bonds), EPA (five double bonds), or DHA (six double bonds) were incorporated in liposomes in which DOPC (C18:1) with a ≤0°C gel-to-liquid crystalline phase-transition temperature or DSPC (C18:0) with a >50°C phase-transition temperature was used as the main “fluid” and “rigid” PC components, which enabled us to also examine the impact of PC fluidity in our system. Alpha-tocopherol was supplemented as a protectant to inhibit the oxidative degradation of Omega-3.
 |
Table 1 Lipid Composition Used To Prepare Various Liposome Formulations. The Indicated Amount Of Each Component Was Used To Produce 1 mL Of Liposomal Dispersions |
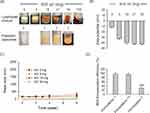
Omega-3-free lipid mixture produced very stable nano-sized liposomes (Formulations 1 and 2) whose size remained unchanged up to 6-week incubation (Figure 1). Incorporation of ALA, EPA, or DHA (Formulations 3–8) produced larger vesicles that tended to grow in size over time: the mean sizes of DHA-, EPA-, and ALA-incorporated DSPC-based liposomes (Formulations 4, 6, and 8) increased by 9.3-, 6.3-, and 4.0-fold after 6-weeks, reaching ≥2 μm. DOPC-based liposomes (Formulations 3, 5, and 7) also tended to grow or aggregate in a time-dependent manner (Figure 1). In contrast, no significant size change was found during incubation of KO-incorporated liposomes (Formulations 9 and 10). Collectively, these data indicate the time-dependent size increases of liposomes incorporated with free Omega-3 (ALA, EPA, and DHA), but not those with KO.
To determine the highest KO content that could be stably incorporated into liposomes, we next prepared liposomes with varying contents of KO. After lyophilization of lipid mixture mixed with varying KO content (the first step to prepare liposomes), a lipid powder cake wet with KO was obtained with KO up to ~27 mg while the lipid mixture was found as dissolved in KO at ≥54 mg KO. Consequently, a homogeneous dispersion of liposomes could be produced with a lipid mixture containing ≤27 mg KO (Figure 2A). Increasing the KO in the liposomes increased the negativity of the zeta potential of particles until a saturation point was reached at 27 mg KO (Figure 2B). KO-incorporated liposomes had a zeta potential lower than −40 mV, indicating that KO incorporation yielded a partial electrostatic stabilization of the liposomes. Particle sizes of liposomes incorporated with 0–18 mg KO (Formulations 1, 9 and 11) did not significantly change during storage while those with 36 mg KO slightly increased at the final time point (Figure 2C). BDS, an IBD drug, could be encapsulated with high efficiency (94–96%) in both Omega-3-free (conventional) and KO liposomes (Formulations 1 and 11), but not in DHA-incorporated liposomes (Formulation 7) (Figure 2D). Together, our results show that KO-incorporated liposomes exhibited a high physical stability and drug-loading capacity, and 18 mg KO per 40 μmol lipids were maximal to exhibit these characteristics.
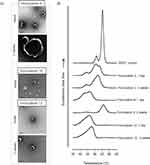
The time-dependent changes in liposome size and morphology were visualized by TEM. Liposomes incorporated with 6 or 18 mg KO (Formulations 10 and 12) were mostly spherical with dimensions of ≤150 nm. In contrast, most of the EPA-incorporated liposomes (Formulation 6) were not smooth spheres, instead exhibiting edges (Figure 3A, top). After a 3-week incubation, the KO-incorporated liposomes retained their initial size and shape while the EPA liposomes were visualized as fused vesicles with diameters >1 μm (Figure 3A). Time-dependent fusion was also observed with the ALA- and DHA-incorporated liposomes (data not shown). Thus, the higher stability of KO-incorporated liposomes could reflect that these liposomes are more able to resist time-dependent fusion.
To further examine the mechanisms that might underlie the differences in liposomes incorporated with free Omega-3 and KO, we performed DSC studies. DSPC alone showed pre- and major transition peaks at 51.0°C and 55.4°C, respectively (Figure 3B), which was in good agreement with the literature.24 Previous studies showed that mixing CHOL and α-tocopherol with rigid PC lowered the pre- and main transition temperatures and enthalpy, and reduced the packing order of gel-phase membranes, as shown in our Formulation 2.25 Here, we found that the incorporation of EPA (Formulation 6) significantly lowered and widened the major transition peak, which appeared split into two overlapping peaks, possibly indicating a phase separation in the PC membrane. In contrast, Formulation 12 showed a single phase-transition peak centering at 45.8°C without splitting (Figure 3B), suggesting that the lipid distribution in the KO-incorporated membrane was more homogeneous.
When Formulation 2 was incubated for 2 weeks, the pre- and major transition peaks of DSPC appeared at close to the original temperatures, although the major peak was somewhat sharper (Figure 3B). Marked changes were seen in the thermogram of Formulation 6 for the same duration: the original peak was abolished nearly to baseline and a new symmetrical peak appeared near a temperature of 56.0°C, which appears to the major transition of DSPC. This suggests the extensive, time-dependent membrane rearrangement in EPA-incorporated liposomes, including the local formation of EPA-free DSPC membranes. Following storage, Formulation 12 did not exhibit any change in the phase-transition temperature range, except that the center of the major transition peak was slightly shifted to the right. Taken together, these data indicate that drastic, time-dependent PC membrane rearrangement is induced by EPA, but not by KO.
In Vitro Anti-Inflammatory Effects Of KO Liposomes
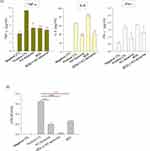
Formulation 11, renamed as KO liposomes from now on, was used for subsequent studies. When RAW264.7 macrophages were pretreated with KO liposomes prior to LPS stimulation, the productions of the proinflammatory cytokines, IL-6 and TNF-α, were significantly decreased to an extent comparable to that seen in BDS-pretreated cells. Moreover, combined treatment with BDS and KO liposomes suppressed the levels of these cytokines even further (Figure 4A). Omega-3-free liposomes did not affect the production of proinflammatory cytokines (Figure 4A), suggesting that the potent anti-inflammatory effects of KO liposomes are mainly due to the incorporated KO. Cell viability assay showed that KO liposome did not induce evident cell death in macrophages, excluding the possibility that the anti-inflammatory effects of KO liposomes are mediated by cytotoxicity (data not shown).
We next investigated the effect of our liposome formulations on the barrier function of CaCo-2 cell monolayer. The application of the cell culture supernatant obtained from LPS-treated RAW264.7 cells reduced the TEER value of the monolayer to 39.9% of the control value (Figure 4B), suggesting that the membrane barrier was injured by components of the supernatant (eg, pro-inflammatory cytokines). When the impaired layer was post-treated with liposomes, the TEER value was recovered. Notably, 2.8 μL/mL of KO liposomes almost completely recovered the TER value (~98.0%), while the same volume of conventional liposomes yielded a recovery of only ~64.7% (Figure 4B). Under the same conditions, 1 μM BDS did not affect the TEER value of the monolayer (data not shown). These findings indicate that supplementation with a liposomal vehicle, particularly one incorporating KO, is very effective in restoring barrier function impaired by inflammatory factors.
Physical Stability And Adsorption Properties Of KO Liposomes Under Simulated In Vivo Conditions
Since the GI stability of a liposomal formulation is important for its ability to be administered orally, we assessed changes in the sizes of liposomes incubated for 24 hrs with PBS, SGF, and SIF at 37°C, to mimic the conditions in various regions of the GI tract. No size change was observed among the Omega-3-free liposomes, regardless of the incubation medium (Figure 5A). The KO liposomes maintained their initial size under all tested conditions, with the exception of incubation in SGF for 24 hrs. KO emulsions can be produced without the need for additional emulsifiers, given that Omega-3 is present in both its PC-bound and free forms.26 Thus, we prepared KO emulsions and tested their GI stability for comparison. The particle size of KO emulsion started to increase after as little as 1 hr of incubation in SGF (4.3-fold); after that time point, the size could not be determined due to extensive aggregation (Figure 5A). Considering that the gastric emptying time is generally 3–4 hrs after oral administration,27 R5 these data indicate that KO liposomes may serve as an orally administrable therapeutic vehicle for treating IBD.
As anionic nanoparticles may exhibit stronger adherence to inflamed colon regions due to their strong electrostatic interaction with cationic proteins prevalent on cells commonly found in an inflamed region (eg, eosinophils),11 we investigated the protein adsorption properties of KO liposomes. As shown in Figure 5B, when KO liposomes were incubated with lysozyme as a cationic model protein, ~93.5% of the lysozyme adsorbed to the liposomes. In contrast, we observed minimal adsorption of lysozyme to Omeg-3-free liposomes. Mucin, which is an anionic protein that coats the GI epithelium, exhibited higher protein adsorption on conventional liposomes compared to KO liposomes; however, the level of adsorption was below 10% in both cases whereas it was >55% in case of DOTAP liposomes whose surface is highly positive (Figure 5B).28 These data suggest that the incorporation of KO into liposomes greatly increases their ability to adsorb cationic protein.
In Vivo Anti-Inflammatory Effects Of KO Liposomes
Mice in the DSS group showed increased DAI compared with healthy controls, indicating that the DSS treatment successfully induced colitis. Treatment with BDS suspension, KO liposomes, or BDS-entrapped KO liposomes slightly decreased the DSS-induced level of DAI, although statistically not significant (Figure 6A). The colon length was shortened in DSS group, in accordance with the findings of other studies.29,30 BDS or KO liposome treatment slightly but significantly ameliorated DSS-induced colon shortening (Figure 6B). Pathological analysis of colon sections from DSS mice revealed signs of severe colitis, including crypt loss, submucosal edema, and infiltration of inflammatory cells. These parameters appeared to be reduced in the BDS-entrapped KO liposome group, but the pathologic scoring results did not significantly differ among the treatment groups (Figure 6C).
DSS administration significantly increased the serum levels of TNF-α and IL-6 in mice, and tended to non-significantly increase the serum level of IFN-γ (Figure 7A). BDS significantly decreased the DSS-induced production of TNF-α but increased that of IL-6. KO liposomes and BDS(+) KO liposomes significantly decreased the serum levels of both TNF-α and IL-6 (Figure 7A), suggesting that orally administered KO liposomes exerted a potent anti-inflammatory activity.
Serum endotoxin levels are known to be correlated with both the permeability of the intestinal mucosa and disease activity in IBD.31 As shown in Figure 7B, serum LPS was detectable in the positive control groups but not in the negative control groups, suggesting that LPS permeated through the impaired intestinal barrier in our DSS mouse model. KO liposomes, BDS(+) KO liposomes, or BDS suspension significantly reduced the serum level of LPS. Among them, BDS(+) KO liposomes exhibited the strongest decreasing effect: the serum LPS level was decreased by 3.3-, 31.2-, and 2.5-fold by KO liposomes, BDS(+) KO liposomes, and BDS suspension, respectively. These findings suggest that KO liposomes, particularly BDS-entrapped KO liposomes, can improve intestinal barrier function in a mouse model of colitis.
Discussion
As a first step in developing Omega-3-incorporated liposomes as an effective vehicle for IBD therapy, we set out to ensure that they exhibited physical stability before and after administration. However, we found that embedding free Omega-3 within the membrane induced the physical destabilization (fusion) of liposomes, as revealed by a drastic size increase (Figure 1 and Figure 3A) and alteration of the DSC thermogram (Figure 3B). Other groups had previously noted the physical instability of Omega-3-incorporated liposomes.17,32 Notably, Omega-3 has a non-cylindrical, inverted conical molecular shape reflecting that the diameter of the head group is much smaller than that of the hydrocarbon tail, and thus its incorporation between cylindrical PC molecules increases the built-in curvature stress of the membrane.33,34 This can cause marked structural changes of the lipid bilayer, and can be associated with phase separation and membrane fusion.32,35 Omega-3-induced edge formation may provide closer contact regions between membranes, thereby driving vesicle fusion (Figure 3A).36 The more rapid destabilization of vesicles incorporating Omega-3 with higher numbers of cis double bonds (eg, DHA and EPA compared to ALA) may reflect the higher degree of curvature stress induced by the increased hydrocarbon tail chain bending of the fatty acids. It is not yet clear how KO liposomes retain a degree of stability comparable to that of Omeg-3-free liposomes despite having a high Omega-3 content. It is plausible that the incorporation of KO may cause less curvature stress due to the presence of Omega-3 mainly as phospholipid bound forms in KO. However, we found that the incorporation of docosahexaenoyl PC induced the time-dependent fusion of DSPC liposomes comparable to that of DHA (unpublished data), and these data suggest that the difference cannot be solely attributed to the different forms of Omega-3. KO has multiple components, including astaxanthin and vitamin A, that may be fitted into spaces between Omega-3 and PC molecules to help lessen the curvature stress, and/or which could increase the electrostatic stabilization of particles by providing highly negative surface charges (Figure 2B).
In addition to being nanosized and electrostatically stable vehicles, KO liposomes exhibited high GI stability, high drug loading capacity, and strong adherence to cationic proteins, emphasizing their potential as an orally administrable IBD drug-carrying vehicle. These results indicate that using KO liposomes as an IBD drug carrier may yield increased drug accumulation in the inflamed intestinal region.
Our in vitro data demonstrate that KO liposomes themselves have potent anti-inflammatory effects. It seems likely that the liposome-incorporated KO blocked the production of pro-inflammatory cytokines by suppressing the LPS-induced expression of genes encoding inflammatory factors, such as NF-κB.37 Pro-inflammatory cytokines produced by macrophages are known to act directly on intestinal epithelial cells to increase membrane permeability.38 Thus, the ability of KO liposomes to restore impaired barrier function may be associated with their suppressive effect on cytokine production. The administration of Omega-3-free liposomes or post-treatment with KO liposomes also induced recovery, suggesting that underlying mechanism involves the physical interaction of liposomes with the impaired barrier, presumably via the incorporation of PC into impaired membranes. The higher recovery effect of KO liposomes compared to Omeg-3-free liposomes seems likely to reflect both the supplementation of additional PC and the anti-inflammatory activity of KO.
The induction of colitis in DSS-treated mice was confirmed by monitoring increases in macroscopic (DAI, colon weight/length ratio, colonic inflammation) and biochemical (serum levels of TNF-α, IL-6, IFN-γ, and LPS) parameters. Oral administration of KO liposomes greatly reduced the biochemical signs of colitis (Figure 7A and B) but only slightly reduced the macroscopic signs of the disease (Figure 6). Similarly, BDS, which is widely used to manage IBD, greatly reduced the biochemical parameters (serum levels of LPS and TNF-α) but not the macroscopic signs of the disease under our experimental conditions. In this context, there are conflicting reports as to whether BDS improves the overall disease status in DSS-induced colitis, even though it shows anti-inflammatory activity in this model.39,40 It is likely that certain experimental details, including the treatment schedule, may affect the effect of a treatment on the overall disease status.
The systemic levels of endotoxins (eg, LPS), which contribute to the inflammatory cascade in IBD patients,41 are positively correlated with clinical activity of UC.31 Here, we show that KO liposomes can potently suppress the serum levels of LPS and proinflammatory cytokines, suggesting that the clinical activity of colitis is decreased in these mice. We speculate that the Omega-3 and PC provided by KO liposomes are incorporated into the impaired enterocyte membrane and/or bind to mucin and thereby re-establish the hydrophobic protective barrier in the inflamed/impaired region and decrease the permeation of LPS. BDS-entrapped KO liposomes suppressed LPS more than KO liposomes, further supporting the potential of KO liposomes as an effective drug carrier for IBD.
Additional studies, including efforts to reduce early intestinal absorption of the vesicles, are warranted to support the development of our KO liposomes as a clinically useful drug carrier for treating human IBD patients. It may be helpful to additionally coat the liposomes to allow time- or pH-controlled drug release and support the accumulation of drugs beyond the duodenum.42 However, for such efforts, it will be necessary to address the potential variation of intestinal transit times and intraluminal pH depending on the patient’s disease state.
 |
Figure 1 Effect of Omega-3 fatty acid incorporation on the time-dependent changes of mean particle size of liposomes. Notes: Liposomes (Formulations 1–10) were prepared from lipid mixtures as described in Table 1. Prepared liposomes were incubated at room temperature for up to 6 weeks. Data are expressed as means±SD (n=3; *p<0.01; ***p<0.001, compared to the initial condition). Abbreviations: ALA, alpha-linolenic acid; EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid. |
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) and funded by the Ministry of Science, ICT and Future Planning (2015R1A2A2A01005783 and 2018R1A2B6003390 to S.J. Lim; 2017R1A2B4002419 to S.Y. Chang).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
Jin-Hee Kim and Soo-Jeong Lim report a patent 10-2018-0079467 pending to Republic of Korea. The authors report no other conflicts of interest in this work.
References
1. Cohen LJ, Cho JH, Gevers D, Chu H. Genetic factors and the intestinal microbiome guide development of microbe-based therapies for inflammatory bowel diseases. Gastroenterology. 2019;156(8):2174–2189. doi:10.1053/j.gastro.2019.03.017
2. Torres J, Danese S, Colombel JF. New therapeutic avenues in ulcerative colitis: thinking out of the box. Gut. 2013;62(11):1642–1652. doi:10.1136/gutjnl-2012-303959
3. Salim SY, Soderholm JD. Importance of disrupted intestinal barrier in inflammatory bowel diseases. Inflamm Bowel Dis. 2011;17(1):362–381. doi:10.1002/ibd.21403
4. Stremmel W, Ehehalt R, Staffer S, et al. Mucosal protection by phosphatidylcholine. Dig Dis. 2012;30(Suppl 3):85–91. doi:10.1159/000342729
5. Karner M, Kocjan A, Stein J, et al. First multicenter study of modified release phosphatidylcholine “LT-02” in ulcerative colitis: a randomized, placebo-controlled trial in mesalazine-refractory courses. Am J Gastroenterol. 2014;109(7):1041–1051. doi:10.1038/ajg.2014.104
6. Calder PC. Marine omega-3 fatty acids and inflammatory processes: effects, mechanisms and clinical relevance. Biochim Biophys Acta. 2015;1851(4):469–484. doi:10.1016/j.bbalip.2014.08.010
7. Costanzo M, Cesi V, Prete E, et al. Krill oil reduces intestinal inflammation by improving epithelial integrity and impairing adherent-invasive Escherichia coli pathogenicity. Dig Liver Dis. 2016;48(1):34–42. doi:10.1016/j.dld.2015.09.012
8. Grimstad T, Bjorndal B, Cacabelos D, et al. Dietary supplementation of krill oil attenuates inflammation and oxidative stress in experimental ulcerative colitis in rats. Scand J Gastroenterol. 2012;47(1):49–58. doi:10.3109/00365521.2011.634025
9. Wang Q, Lv S, Lu J, Jiang S, Lin L. Characterization, stability, and in vitro release evaluation of carboxymethyl chitosan coated liposomes containing fish oil. J Food Sci. 2015;80(7):C1460–C1467. doi:10.1111/1750-3841.12929
10. Beloqui A, Coco R, Alhouayek M, et al. Budesonide-loaded nanostructured lipid carriers reduce inflammation in murine DSS-induced colitis. Int J Pharm. 2013;454(2):775–783. doi:10.1016/j.ijpharm.2013.05.017
11. Collnot EM, Ali H, Lehr CM. Nano- and microparticulate drug carriers for targeting of the inflamed intestinal mucosa. J Control Release. 2012;161(2):235–246. doi:10.1016/j.jconrel.2012.01.028
12. Choi YH, Han HK. Nanomedicines: current status and future perspectives in aspect of drug delivery and pharmacokinetics. J Pharm Investig. 2018;48(1):43–60. doi:10.1007/s40005-017-0370-4
13. Leekumjorn S, Cho HJ, Wu Y, Wright NT, Sum AK, Chan C. The role of fatty acid unsaturation in minimizing biophysical changes on the structure and local effects of bilayer membranes. Biochim Biophys Acta. 2009;1788(7):1508–1516. doi:10.1016/j.bbamem.2009.04.002
14. Notman R, Noro MG, Anwar J. Interaction of oleic acid with dipalmitoylphosphatidylcholine (DPPC) bilayers simulated by molecular dynamics. J Phys Chem B. 2007;111(44):12748–12755. doi:10.1021/jp0723564
15. Jang E-J, Choi WR, Kim S-Y, et al. 2-Hydroxyoleic acid-inserted liposomes as a multifunctional carrier of anticancer drugs. Drug Deliv. 2017;24(1):1587–1597. doi:10.1080/10717544.2017.1388452
16. Arouri A, Lauritsen KE, Nielsen HL, Mouritsen OG. Effect of fatty acids on the permeability barrier of model and biological membranes. Chem Phys Lipids. 2016;200:139–146. doi:10.1016/j.chemphyslip.2016.10.001
17. Ehringer W, Belcher D, Wassall SR, Stillwell W. A comparison of the effects of linolenic (18:3 omega 3) and docosahexaenoic (22:6 omega 3) acids on phospholipid bilayers. Chem Phys Lipids. 1990;54(2):79–88.
18. Hong SS, Kim SH, Lim SJ. Effects of triglycerides on the hydrophobic drug loading capacity of saturated phosphatidylcholine-based liposomes. Int J Pharm. 2015;483(1–2):142–150. doi:10.1016/j.ijpharm.2015.02.013
19. Tapal A, Tiku PK. Complexation of curcumin with soy protein isolate and its implications on solubility and stability of curcumin. Food Chem. 2012;130(4):960–965. doi:10.1016/j.foodchem.2011.08.025
20. Jung IW, Han HK. Effective mucoadhesive liposomal delivery system for risedronate: preparation and in vitro/in vivo characterization. Int J Nanomedicine. 2014;9:2299–2306. doi:10.2147/IJN.S61181
21. Sciscione F, Pucci C, La Mesa C. Binding of a protein or a small polyelectrolyte onto synthetic vesicles. Langmuir. 2014;30(10):2810–2819. doi:10.1021/la500199w
22. Toropainen T, Velaga S, Heikkila T, et al. Preparation of budesonide/gamma-cyclodextrin complexes in supercritical fluids with a novel SEDS method. J Pharm Sci. 2006;95(10):2235–2245. doi:10.1002/jps.20702
23. Dieleman LA, Palmen MJ, Akol H, et al. Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. Clin Exp Immunol. 1998;114(3):385–391. doi:10.1046/j.1365-2249.1998.00728.x
24. Sade A, Banerjee S, Severcan F. Concentration-dependent differing actions of the nonsteroidal anti-inflammatory drug, celecoxib, in distearoyl phosphatidylcholine multilamellar vesicles. J Liposome Res. 2010;20(2):168–177. doi:10.3109/08982100903244492
25. Serro AP, Galante R, Kozica A, et al. Effect of tetracaine on DMPC and DMPC+cholesterol biomembrane models: liposomes and monolayers. Colloids Surf B Biointerfaces. 2014;116:63–71. doi:10.1016/j.colsurfb.2013.12.042
26. Wu Q, Uluata S, Cui L, et al. Physical and oxidation stability of self-emulsifying krill oil-in-water emulsions. Food Funct. 2016;7(8):3590–3598. doi:10.1039/c6fo00045b
27. Minekus M, Alminger M, Alvito P, et al. A standardised static in vitro digestion method suitable for food - an international consensus. Food Funct. 2014;5(6):1113–1124. doi:10.1039/c3fo60702j
28. Kim SY, Lee SJ, Kim JK, Choi HG, Lim SJ. Optimization and physicochemical characterization of a cationic lipid-phosphatidylcholine mixed emulsion formulated as a highly efficient vehicle that facilitates adenoviral gene transfer. Int J Nanomedicine. 2017;12:7323–7335. doi:10.2147/IJN.S146785
29. Kwon SH, Seo EB, Lee SH, et al. T cell-specific knockout of STAT3 ameliorates dextran sulfate sodium-induced colitis by reducing the inflammatory response. Immune Netw. 2018;18(4):e30. doi:10.4110/in.2018.18.e30
30. Yang JY, Kim MS, Kim E, et al. Enteric viruses ameliorate gut inflammation via toll-like receptor 3 and toll-like receptor 7-mediated interferon-beta production. Immunity. 2016;44(4):889–900. doi:10.1016/j.immuni.2016.03.009
31. Gardiner KR, Halliday MI, Barclay GR, et al. Significance of systemic endotoxaemia in inflammatory bowel disease. Gut. 1995;36(6):897–901. doi:10.1136/gut.36.6.897
32. Ibarguren M, Lopez DJ, Escriba PV. The effect of natural and synthetic fatty acids on membrane structure, microdomain organization, cellular functions and human health. Biochim Biophys Acta. 2014;1838(6):1518–1528. doi:10.1016/j.bbamem.2013.12.021
33. Arouri A, Mouritsen OG. Membrane-perturbing effect of fatty acids and lysolipids. Prog Lipid Res. 2013;52(1):130–140. doi:10.1016/j.plipres.2012.09.002
34. Mouritsen OG. Lipids, curvature, and nano-medicine. Eur J Lipid Sci Technol. 2011;113(10):1174–1187. doi:10.1002/ejlt.201100050
35. Onuki Y, Morishita M, Chiba Y, Tokiwa S, Takayama K. Docosahexaenoic acid and eicosapentaenoic acid induce changes in the physical properties of a lipid bilayer model membrane. Chem Pharm Bull (Tokyo). 2006;54(1):68–71. doi:10.1248/cpb.54.68
36. Malinin VS, Frederik P, Lentz BR. Osmotic and curvature stress affect PEG-induced fusion of lipid vesicles but not mixing of their lipids. Biophys J. 2002;82(4):2090–2100. doi:10.1016/S0006-3495(02)75556-2
37. Liu T, Zhang L, Joo D, Sun SC. NF-kappaB signaling in inflammation. Signal Transduct Target Ther. 2017;2. doi:10.1038/sigtrans.2017.23
38. Coskun M. Intestinal epithelium in inflammatory bowel disease. Front Med (Lausanne). 2014;1:24.
39. Naeem M, Choi M, Cao J, et al. Colon-targeted delivery of budesonide using dual pH- and time-dependent polymeric nanoparticles for colitis therapy. Drug Des Devel Ther. 2015;9:3789–3799. doi:10.2147/DDDT.S88672
40. Ocon B, Aranda CJ, Gamez-Belmonte R, et al. The glucocorticoid budesonide has protective and deleterious effects in experimental colitis in mice. Biochem Pharmacol. 2016;116:73–88. doi:10.1016/j.bcp.2016.07.010
41. Fukui H. Increased intestinal permeability and decreased barrier function: does it really influence the risk of inflammation? Inflamm Intest Dis. 2016;1(3):135–145. doi:10.1159/000447252
42. Patole VC, Pandit AP. Mesalamine-loaded alginate microspheres filled in enteric coated HPMC capsules for local treatment of ulcerative colitis: in vitro and in vivo characterization. J Pharm Investig. 2018;48(3):257–267. doi:10.1007/s40005-017-0304-1
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.