Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 14
Knowledge and Practice on Diabetic Foot Self-Care and Associated Factors Among Diabetic Patients at Dessie Referral Hospital, Northeast Ethiopia: Mixed Method
Authors Tuha A , Getie Faris A , Andualem A , Ahmed Mohammed S
Received 3 January 2021
Accepted for publication 9 March 2021
Published 17 March 2021 Volume 2021:14 Pages 1203—1214
DOI https://doi.org/10.2147/DMSO.S300275
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Antonio Brunetti
Abdu Tuha, Abebe Getie Faris, Ababil Andualem, Solomon Ahmed Mohammed
Department of Pharmacy, College of Medicine and Health Science, Wollo University, Dessie, Ethiopia
Correspondence: Solomon Ahmed Mohammed P.O. Box: 1145 Tel +251910504378
Email [email protected]
Introduction: Diabetic foot ulcer is a serious and disabling complication of diabetes mellitus that consists of lesions in the deep tissues resulting in lower extremity amputations. The incidence of diabetic foot ulcers has increased due to an increased prevalence of diabetes mellitus. This study assessed the knowledge and practice of diabetic foot self-care among diabetic patients attending Dessie referral hospital.
Methods: A facility-based explanatory sequential mixed method was used from April 03/2019 to May 09/2019. The quantitative method employed an institutional-based cross-sectional study design using a structured questionnaire. A simple random sampling technique was employed to select the study participants. Descriptive and inferential statistics were made using the statistical package for social sciences version 20. Then, a phenomenological study was employed among key informants and content analysis was performed.
Results: Sixty-eight (19.8%) of patients with diabetes developed complication, among them 97 (28%) had foot ulcer. Nearly two-third (217 (61.3%): 95% CI (57.6– 68.3%)) and 134 (39%; 95% CI:34.3– 45.1%) of patients with diabetes had good knowledge and practice on diabetic foot self-care, respectively. Diabetic nephropathy ((AOR): 0.03, 95% CI (0.00– 0.27)) was associated with knowledge on foot self-care. Practice of foot self-care was associated with female ((AOR: 2.07, 95% CI (1.04– 4.12)), age 21– 30 ((AOR: 6.42, 95% CI (1.54– 26.8)), 31– 40 years ((AOR: 7.4, 95% CI (1.42– 39.05)), 41– 50 years ((AOR: 8.4, 95% CI (1.4– 50.6)), single ((AOR: 0.35, 95% CI (0.150– 0.81)), living in rural ((AOR: 0.31, 95% CI (0.18– 0.545)) and no comorbidity ((AOR: 0.406, 95% CI (0.18– 0.88)). Key informants were not compliant, confused and negligent to foot self-care.
Conclusion: Nearly two-third and more than one-third of patients with diabetes had good knowledge and practice on diabetic foot self-care, respectively. Patients lack proper knowledge regarding self-care and how it is practiced. Patient education and proper management of diabetic foot ulcers are recommended to reduce, delay, or prevent complications.
Keywords: diabetic foot self-care, knowledge, practice
Introduction
Diabetes mellitus (DM) is a group of chronic metabolic disorders characterized by elevated levels of blood glucose that is associated with significant morbidity, mortality, and increasing health care cost.1 Accordingly, there are two major types and treatment varieties according to the cause.2 The world prevalence of diabetes is 8.5% among adults aged over 18 years, affecting 422 million adults, in 2014 increased to 9.3% million people in 2019.3,4 The prevalence of DM has been steadily increasing for the past three decades and is growing most rapidly in low- and middle-income countries. According to the International Diabetes Federation Atlas guideline report, the prevalence of diabetes in Africa among adults aged 20–79 years was 4.2% in 2017.4,5 In Ethiopia, DM is emerging as one of the major chronic health problems, and the prevalence adjusted to the national population was 4.4% in 2013.6,7
Diabetes mellitus can result in blindness, renal failure, lower limb amputation, coronary artery disease, peripheral vascular disease, stroke, and other long-term consequences that impact significantly on the quality of life.1,3 Of these complications, diabetes-related foot problem affects the majority of patients with DM.8 A diabetic foot ulcer is the most frequently recognized complication of diabetes mellitus that consists of lesions in the deep tissues associated with neurological disorders and peripheral vascular disease in the lower limbs.9 It is a full-thickness wound penetrating through the dermis (the deep vascular and collagenous inner layer of the skin) located below the ankle in a DM patient.10 Wagner classified diabetic foot ulcers mainly based on wound depth. Accordingly, there are six wound grades. These include Grade 0 no ulcer, but the foot is at risk for ulceration, Grade 1 superficial ulceration, Grade 2 ulcer with deep infection, but without the involvement of the bone, Grade 3 ulcer with osteomyelitis, Grade 4 localized gangrene, and Grade 5 gangrene of the whole foot.11
The incidence of diabetic foot ulcers has increased due to the worldwide prevalence of DM and the prolonged life expectancy of patients with diabetes, poor knowledge and practice of diabetic foot self-care.9,12–14 The worldwide prevalence of diabetic foot ulceration is 6.3%. The lifetime risk of a person with diabetes developing a foot ulcer could be as high as 25%.9,15 Rates of foot ulceration in Africa vary between regions and have been estimated to be between 4% and 19%.16 In Ethiopia, the incidence and prevalence of diabetic foot ulcers are still unknown in the general population.7 The study conducted in North-West Ethiopia and South Ethiopia showed that the prevalence of diabetic foot ulcers among patients with diabetes was 13.6% and 14.8%, respectively.17,18
Diabetic foot complications are the most common cause of hospitalization in the patients with diabetes,19 and the risk of foot ulceration and limb amputation increases with older age, long duration of diabetes, poor glycemic control, peripheral neuropathy, cigarette smoking, foot deformities, peripheral arterial disease, history of foot ulcer, amputation, visual impairment and diabetic kidney disease (especially patients on dialysis).20,21 Factors that affect DM patients’ foot self-care include sex, educational status, residence, and non-attendant to diabetic educational program.11,12,22–25
Yet, early recognition and treatment of patients with diabetes and feet at risk for ulcers and amputations can delay or prevent adverse outcomes.26 Diabetic foot ulcer tends to affect not just physical but also psychosocial, economic, and overall quality of life of patients with DM.27,28 This study was conducted to assess knowledge and practice on diabetic foot self-care and associated factors among patients with diabetes at Dessie referral hospital, Northeast Ethiopia. This study is used to develop the strategies for counseling of vulnerable group on seriousness of the problem and help to develop effective prevention therapy.
Methods
Study Area and Period
This study was done in a hospital at Dessie town, Northeast Ethiopia, from 03/08/2019 to 09/09/2019. The city is located 401 kilometers north of Addis Ababa, Ethiopia, Amhara Region. The hospital is being provided with specialized health services to the town and the surrounding population. In the hospital, there are different wards and clinics within hospital at Dessie town, from which the diabetic ambulatory clinic is one of the services provided.
Study Design
An explanatory sequential mixed method was used. A facility-based cross-sectional study and phenomenological study were employed to assess knowledge and practice of diabetic foot self-care and associated factors among patients with DM at hospital in Dessie.
Population
All patients with DM attending hospital at Dessie during the study period were the source of the population while patients with diabetes attending hospital at Dessie diabetic ambulatory clinic during the study period and who fulfilled the inclusion criteria were the study population.
Inclusion and Exclusion Criteria
Patients with diabetes who had periodic regular follow-ups during the study period and willing to participate in the study were included. All DM patient who was unable to hear or communicate and with mental disabilities newly diagnosed patients with DM (≤1-month duration) and patients in serious clinical states who will not consent or communicate were excluded from the study.
Sample Size Determination and Sampling Technique
The sample size for the quantitative study was estimated by using a single population proportion formula using 54.6% prevalence,14 95% confidence level, and 5% tolerable sampling error. Since the source population was less than 10,000 (2000), the sample size was adjusted with a total of 352 participants. A simple random sampling technique was employed to select the study participants. The sample size for the phenomenological study was determined by the saturation of information concerning emerging themes. Thus, 12 key informants were included. Key informants were selected purposively by the DM clinic nurse.
Study Variables
The dependent variables were knowledge, and practice of patients with diabetes on diabetic foot self-care and independent variables were socio-demographic characteristics of DM patient, duration of DM, type of DM, previous history of foot ulcer, and presence of other complications (peripheral neuropathy, visual disturbance, and nephropathy).
Data Collection Procedure and Quality Assurance
Data collection instrument was adapted from similar study conducted before.24 The data collection instrument was pretested in five percent of the sample size at the Dessie health center. The pilot test was done prior to the actual study. After collecting the pilot data, the data were cleaned using principal component analysis. Knowledge and practice on patients with DM on diabetic foot self-care were assessed using a total of 16 questions (8 for each). The reliability of the questionnaire was also checked using Cronbach’s alpha test and a value of 79.8% was obtained. The data were collected using a structured interviewer-administered questionnaire by three nurses who had no working relation to the hospital after recruiting and training with the supervision of the principal investigators. Data completeness, accuracy, and consistency was checked after collection and appropriately arranged and kept in a secured place for compilation and analysis.
For the phenomenological study, the semi-structured interview guide was prepared in English initially and then translated to Amharic for interview, and finally back-translated into the English language for thematic analysis to maintain consistency and standardization of the instruments. The translation from Amharic to English was done through Google translator and edited with context. The principal investigators made the in-depth interview, which lasted 10–20 minutes. The interview was done in Amharic to fully explore in-depth about the knowledge and lived experience of patients with diabetes and all interviews were audio-recorded and transcribed verbatim.
To enhance the validity of an in-depth interview, the interview guide was tested for validity of contents by one expert from social and administrative pharmacy groups. Language translation was done to check message consistency. To assure the quality of the qualitative data, a standard questionnaire was used and four investigators were involved. Methodological triangulation (the data collected through field note, observations and individual in-depth interview) and investigator triangulation (four research team members participated in data coding and analysis) were also enhancing the validity. Multiple methods were used and the Amharic version of the transcript was also brought back to key informants and was signed. In this study, key informants were chosen carefully to be individuals who had all experience of DM. The researchers also separated themselves from the text by their understanding in a reflective way.
Issues of Reflexivity: The Principal Investigator Status as an Insider
The principal investigators being senior pharmacy professionals offer certain strengths and limitations for this study. They operated with an awareness of insider bias. They practiced non-judgment and with the awareness of professional relativity.
Data Processing and Analysis
Data entry and analysis were done using the Statistical Package for Social Sciences version 20. Variables with a p-value less than 0.25 were entered in the multivariate logistic regression after bivariate logistic regression analyses, and those with a p-value less than 0.05 were taken as statistically significant. Knowledge was measured using eight questions. Each “yes” answer carried one point and zero points for a “no and don’t know”. The points were then added up to provide a total knowledge score. The level of knowledge, whether good or poor, was determined based on the mean score. The practice was also determined in the same manner of knowledge.
All written transcripts were read several times to obtain their overall feelings and all sections of original transcripts were translated into English to facilitate coding line by line. The analysis was done manually using the principles of content analysis. A narrative strategy was employed for the presentation of qualitative findings. Key informants’ level of education, sex, and age were used to elucidate their verbatim portray.
Definition of Terms
Good knowledge: Patients with DM who score greater than or equal to mean score.
Poor knowledge: Patients with DM who score less than the mean score.
Good practice: Patients with DM who score greater than or equal to mean score.
Poor practice: Patients with DM who score less than the mean score.
Diabetes foot self-care: This is the care taken by patients with DM towards their foot health and wellbeing.
Sugar: A local nomenclature for DM.
Results
The response rate of the study was 344 (97.7%). Of the respondents, 78 (22.7%) were in the aged group of 41–50 years, 177 (51.5%) were female, and 243 (70.6%) were married. Moreover, 130 (37.8%) patients with DM were at the primary school level and 197 (57.3%) patients with DM were lived in the urban area. Most patients with DM 88 (25.6%) were housewives (Table 1).
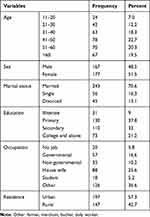 |
Table 1 Socio-Demographic Characteristics of Diabetic Patient at Dessie Referral Hospital, 2019 (N=344) |
More than one-third 244 (70.9%) of respondents were typed two DM patients, 65 (18.9%) had comorbidity. Sixty-eight (19.8%) of patients with DM developed a complication, among them 97 (28%) were developed foot ulcers (Table 2). The majority9 of key informants revealed that they had not developed diabetes foot ulcers. The key informant portrayed: “I have never admitted in health institutions and been treated. But I have kidney stones and liver fat. I still don’t have leg ulcers” (54, Male, Primary education). This finding was further substantiated by 50 years old female DM patient: “I have high blood pressure. I am still taking pressure pills. It hurts my heart too. I am also taking diuretics. I was examined and ordered to wear glasses. Occasionally it itches away at my feet. My leg was swollen and I was given medicine. All of this has to do with sugar’’ (50, Female, Secondary education). However, three of the key informants developed diabetes foot ulcer and one key informant described the scenario:
My leg was amputated because of the sugar. Iron pierced my feet. Although I went for treatment, I developed gangrene and had my leg amputated. A small wound causes a large wound. (70, Male, Illiterate)
 |
Table 2 Clinical Characteristics of DM Patient at Dessie Referral Hospital, 2019 (N=344) |
Knowledge of Patients with DM on Diabetic Foot Self-Care
Three hundred fourteen (91.3%) patients with DM knew the impact of regular taking of medication on the reduction of DM complications. Despite all key informants knew the role of appropriate usage of DM medication on the disease, the majority9 key informants were not using medications appropriately. One of the key informants explained the scenario:
I will take the pill. I have never quit. I was told to take it morning and night, so I took it as soon as I found it. I don’t wait time. I take it in the morning or I take it late. (64, Male, Illiterate)
It is substantiated by one of the key informants:
Because my home and place of work are far away; It is too late to take the medicine and I will take the next one. When I left the place where I was working, I fell asleep at night. (60, Male, Diploma)
Controlling blood sugar can reduce complications and nearly all10 key informants did not have a glucometer at their home. This was supported by one of the key informants:
I know my blood sugar level when going to a health facility. I do not have a home sugar meter. I only know when I come to my appointment. But when I feel ill, I go to a private clinic and get tested. (39, Female, Primary education)
Inspection of feet for foot ulcer knew by 250 (72.7%) patients with DM while 235 (68.3%) patients with DM knew wounds and infection may not heal quickly. This was further substantiated by one of the key informants: “I always see my feet. My legs are fine. I follow my feet and know that my feet will not be infected” (48, Male, Primary education).
Nearly half of 182 (52.9%) patients with DM knew the effect of smoking on the progression of DM. All of the key informants explained the health danger of smoking. One of the key informants portrayed that: “Drinking alcohol and smoking should not be used because it does not go well with sugar. These can cause further damage. They also aggravate the sugar”. (60, Male, Diploma)
The frequency of washing feet and wearing shoes and socks knew by 248 (72.1%) and 190 (55.2%) respectively. The temperature of water for washing feet was correctly answered by 57 (16.6%) patients with DM (Table 3). Nearly two-third (217 (61.3%): 95% confidence interval (CI) (57.6–68.3%)) of patients with DM had good knowledge of diabetic foot self-care. One key informant explained the frequency of washing feet as: “According to my religion, I wash my feet morning and night. I generally wash up to five times a day” (18, Female, Secondary education). Concerning the temperature of water for washing feet, five key informants used cold water. One key informant mentioned His experience: “I always wash my feet with cold water”. (55, Male, Illiterate) While four key informants reject this idea and one key informant mentioned: “I wash my feet with lukewarm water, not too cold or too hot. Because very cold water is not good, I wash my feet with lukewarm water”. (50, Male, Degree)
 |
Table 3 Knowledge of Diabetic Foot Self-Care Among DM Patient at Dessie Referral Hospital, 2019 (N=344) |
Multivariate logistic regression analysis revealed that knowledge regarding diabetic foot self-care was significantly associated with the type of complication. Patients with diabetic nephropathy had 97% less (Adjusted odds ratio (AOR): 0.03, 95% CI (0.00–0.27)) knowledge than patients with diabetic foot ulcers (Table 4).
 |
Table 4 Factors Associated with Knowledge of DM Patients on Diabetic Foot Self-Care at Dessie Referral Hospital, 2019 (N=344) |
The Practice of Patients with DM on Diabetic Foot Self-Care
Thirty (8.7%) of the patients with DM inspect their foot. Washing of feet more than once a day was done by 159 (46.2%) of patients with DM while 90 (26.2%) of patients with DM dry their feet after washing. Seven of the key informants mentioned regular inspection of their feet. One key informant described that: “I keep an eye on my feet. I see it day by day. After I wash my feet, I dry them. I thought it was sugar; it looks white”. (50, Male, Degree) Another key informant also mentioned the situation:
When I wash my feet and change my shoes, I see it. If I see anything from the change; I do not give time. I will consult a doctor. As a healthy person; I do not give time. The foot should dry out and the middle of your toe should be wiped. It should also be wiped with a towel. (55, Male, Illiterate)
While five key informants mentioned irregular inspection and a statement made by one key informant described: “Saying that my feet are fine; I did not always see my feet. I put on my shoes to keep me cool in the morning. I take off at night. I wash and sleep’’. (38, Male, Diploma)
The use of moisturizing cream on their feet was practiced by 269 (78.2%) patients with DM while 167 (48.5%) patients with DM were put moisturizing cream between their toes. Half of the key informants use moisturizing cream. One key informant stated:
… when my feet are white and I feel dry; I always use moisturizing cream. I often use it when my feet were rigid. I did not paint the middle of the fingers. I only paint the upper part of my foot. (54, Male, Primary education)
In this study wearing sandals/slippers and shoes without socks/stockings/tights was practiced by 121 (35.2%) 152 (44.2%) patients with DM, respectively (Table 5). One hundred thirty-four (39%; 95% CI: 34.3–45.1%) of patients with DM had a good practice on diabetic foot self-care. Nearly half of the key informants wear sandals/slippers in their house. The key informant stated:
When I leave home, I wear comfortable shoes. If I am at home, I use soft or non-slip sandals. I would not wear shoes without socks. I am careful not to wear tight shoes. I do not walk barefoot. Even at home, I move with socks. (50, Female, Primary education)
 |
Table 5 Practice DM Patient on Diabetic Foot Self-Care at Dessie Referral Hospital, 2019 (N=344) |
Another key informant also mentioned the scenario: “When the shoes are tight, I wear them without socks. And when it comes to wide shoes, I wear socks. I don’t always wear socks because I live in the countryside” (67, Male, Illiterate). However, five key informants mentioned walking barefoot without socks and sandals. One key informant described that:
When I walk home, I walk barefoot without socks. Because the carpet is in the house, I move barefoot. The women are angry since they are cleaning the house. I always take off my shoes when I go home. (70, Male, Primary education)
Multivariate logistic regression analysis revealed that the practice of patients with DM on diabetic foot self-care was significantly associated with sex, age, marital status, residence, and comorbidity. Females had 2.07 times ((AOR: 2.07, 95% CI (1.04–4.12)) good practice than males. Patients with age 21–30 years had 6.42 times ((AOR: 6.42, 95% CI (1.54–26.8)), patients with 31–40 years had 7.4 times ((AOR: 7.4, 95% CI (1.42–39.05)), and patients with 41–50 years had 8.4 times ((AOR: 8.4, 95% CI (1.4–50.6)) good practice than those greater than sixty years. Patient with single marital status were 65% less ((AOR: 0.35, 95% CI (0.150–0.81)) practice than married patients. Those patients living in the rural area were 69% less ((AOR: 0.319, 95% CI (0.18–0.545)) than those living in urban, and those patients with no comorbidity had 60% less ((AOR: 0.406, 95% CI (0.18–0.88)) practice than those with comorbidity (Table 6).
 |
Table 6 Factors Associated with Practice of Diabetic Foot Self-Care at Dessie Referral Hospital, 2019 (N=344) |
Discussion
A diabetic foot ulcer is one of the chronic complications of diabetes patient which lead to disability and death if it is not effectively prevented and controlled. To prevent this, the patient should have good knowledge and practice on diabetic foot self-care. This study was conducted to assess knowledge and practice on diabetic foot self-care and associated factors among patients with DM at Dessie referral hospital.
In this study, 217 (61.3%; 95% CI (57.6–68.3%)) of the patients with DM had good knowledge of diabetic foot self-care. This demonstrated a lower level of knowledge regarding diabetic foot self-care compared to the study done in China (70.38%).29 The possible reason might be china was a developed country and there may be enough access to information through health education, print, and electronic media that helps to enhance the public awareness of diabetes and its complications.24 The finding was higher than the studies done in Pakistan (29.3%),30 Saudi Arabia (53.6%),12 and Ethiopia (56.2%).24 This might be due to high illiteracy and low role of the physician in providing face-to-face health education program on diabetic foot self-care during admission including advice for diet, exercise and regular medication, and blood glucose checking.30,31 The qualitative study also identified the knowledge gap among key informants. A similar finding was reported from a qualitative study done at Addis Abeba where the overall self-care practices were not adequate.32
One hundred thirty-four (39%; 95% CI: 34.3–45.1%) of patients with DM had a good practice on diabetic foot self-care. This prevalence of diabetic foot self-care practice was lower than the study done at Bahirdar (54.6%),24 but this was higher than the study done in turkey.33 This might be attributed to health education on diabetic foot self-care. However, it was higher than the studies done in India (19.4%)31 and Pakistan (14%) due to the low educational status of the patient and low face-to-face health education by health care providers regarding diabetic foot self-care.29,31,34,35 The proper diabetes foot self-care is not adequately practiced by key informants. In Addis Abeba, most patients lack proper diabetes foot self-care is not adequately practiced. Foot self-care was also the list of recognized self-care practices and most have not even heard of what foot self-care is.32
Knowledge regarding diabetic foot self-care was significantly associated with the type of complication. Patients with diabetic nephropathy had 97% less (AOR: 0.03, 95% CI (0.00–0.27)) knowledge than patients with diabetic foot ulcers. There is a strong association between stage of chronic kidney disease and for diabetic foot ulcer or low extremity amputation; even individuals with moderate chronic kidney disease have an increased risk for a diabetic foot ulcer and low extremity amputation.36 Moreover, the risk of low extremity amputation is at least two to six times greater among those with both diabetes and chronic kidney disease than among those with diabetes alone.37
The finding of this study revealed that females had 2.07 times (AOR: 2.07, 95% CI (1.04–4.12)) good diabetic foot self-care practice than males. A similar finding was reported from Malaysia and Thailand.38,39 This was due to women often feel like victims of something outside of themselves in a position of restrictions and limitations when they do not make self-care a priority. Men presented with a lower prevalence of feet scaling and use of inappropriate shoes when compared to women. Moreover, men presented less healthy habits.40 Wearing open shoes increases the development of diabetic foot ulcer because it causes the foot sliding forward, foot exposed to external trauma and thus causing shear injury to the toes or plantar foot. It is important to encourage the use of suitable footwear, and to educate the patient to promote healthy foot care and footwear habits. Closed shoes create moisture on feet and prevent from drying.41
In this study, patients with DM with age 21–30 years had 6.42 times ((AOR: 6.42, 95% CI (1.54–26.8)), patients with 31–40 years had 7.4 times (AOR: 7.4, 95% CI (1.42–39.05)), and patients with 41–50 years had 8.4 times (AOR: 8.4, 95% CI (1.4–50.6)) good diabetic foot self-care practice than those greater than sixty years. This was due to elder patients where belief in God and do not use their treatment appropriately which reduce their daily diabetic foot self-care practice. Diabetic foot ulcers and lower extremity amputation are severe complications of older diabetic patients.35 The Elderly are not interested in increasing their longevity if the disease detracts their quality of life. Thus, family members and trusted caregivers should help them to identify, refine, and communicate their health goals.42 Cultural beliefs were portrayed as a barrier relating to subjective norms by key informants.43 Acknowledging and accepting the reality of diabetes is essential to living well with diabetes.44
Patients with single marital status had 65% less (AOR: 0.35, 95% CI (0.150–0.81)) diabetic foot self-care practice than married patients. The finding was similar to the study done in Malaysia where single marital status patients with DM were less (AOR: 0.75) diabetic foot self-care practice than married.38 This might be attributed to married patients with DM had emotional and instrumental social support, including activities related to attention or listening, accompanying medical visits than single, which had a positive influence on diabetic foot self-care practice.45 The qualitative study also identified that inadequate family support, and busy work schedules were the barriers related to diabetes foot self-care practice.43 Receiving support from family and care teams is essential to living well with diabetes.44
Those patients living in a rural area had 69% less (AOR: 0.31, 95% CI (0.18–0.545)) diabetic foot self-care practice than those who live in urban. This was similar to the study done at Bahirdar patients where patients with DM living in rural were less (AOR: 0.14)24 diabetic foot self-care practice than urban patients. This was because patients living in urban area had access to information through health education, print, and electronic media than these live in rural area.46 Key informants mentioned that poor income levels, lack of glucometers, and long distance to the hospital. This was in line with the qualitative study done in Ghana.43
Those patients with no comorbidity had 60% less (AOR: 0.40, 95% CI (0.18–0.88)) diabetic foot self-care practice than those with comorbidity. There was a significant relationship between self-care and the number of comorbidities.47 Patients with comorbid chronic diseases experience a wide range of barriers to self-care, including physical limitations, lack of knowledge, financial constraints, a need for social and emotional support, and aggravation of one condition by symptoms of or treatment of another, and polypharmacy.48 Since comorbidity influences the relationship between self-efficacy and self-care maintenance, self-management interventions need to address interactions between chronic conditions and individuals’ diseases condition.47,48
Despite the incidence of diabetic foot ulcers have increased due to the increased worldwide prevalence of diabetes mellitus, proper preventative measures, patient education, appropriate foot self-care, and proper management of diabetic foot ulcers can greatly reduce, delay or prevent complications infection, gangrene, and amputation. Adequate foot self-care practice of patients with diabetes can prevent the occurrence of foot ulcer. To improve diabetes foot self-care, health professionals should understand and acknowledge the effect of diabetes on patients’ health and communicate with patients openly with empathy and cultural consideration.44 This study helps for the hospital to design the strategy to improve the knowledge and practice of self-care practice of patient with diabetes by considering the impact of foot ulcers and consequent amputation on physical and psychosocial life domains.
Recall bias and social desirability bias was the limitation as patients were requested to respond based on their life experience, and the cross-sectional nature of the study made unable to establish cause-and-effect relationships. The nature of qualitative study alters confidence in the generalizability of the findings. This study is used to force urgent need of counseling for most of vulnerable group and public awareness regarding seriousness of the problem and developing effective prevention therapy to reduce its impact on general health of population. Moreover, is used to develop the strategy and the policy on the prevention of foot ulcer secondary to the uncontrolled diabetes, implementing and improving the self-care practice.
Conclusion
Nearly two-thirds of patients with DM had good knowledge of diabetes foot self-care and more than one-third of diabetic patients had good diabetic foot self-care practice. Diabetic nephropathy was associated with knowledge on foot self-care. The practice of foot self-care was associated with females, age 21–30 years, 31–40 years, 41–50 years, single marital status, living in the rural area, and no comorbidity. Key informants stated that they were not compliant with DM medication, confused by health education, and negligent to foot self-care. Generally, patients lack proper knowledge regarding self-care and how it is practiced. Health professionals should approach patients openly with empathy and emphasize the impact of foot ulcers and consequent amputation on physical and psychosocial life domains. Public awareness should be created regarding seriousness of the problem. Appropriate counseling and proper management of diabetic foot ulcers should be offered to reduce, delay, or prevent complications to vulnerable group.
Abbreviations
AOR, adjusted odds ratio; CI, confidence interval; DM, diabetes mellitus; COR, crude odds ratio.
Data Sharing Statement
The datasets are available from the corresponding author upon reasonable request.
Ethical Consideration
Ethical approval was obtained from the Ethics Review Committee of the Department of Pharmacy, College of Medicine and Health Sciences, Wollo University (WU Phar/078/11). Verbal informed consent was obtained from study participants while parental consent was obtained for adolescent patients after providing information regarding the purpose of the study, why and how they are selected to be involved in the study, and what will be expected of them. The participants’ informed consent included publication of anonymized responses. Verbal informed consent was approved by the Ethical committee. Confidentiality of patient-specific data was maintained throughout the study. The study was done with the consideration and compliance with declaration of Helsinki.
Acknowledgment
The authors would like to acknowledge Wollo University College of Medicine and Health Sciences Department of Pharmacy.
Authors’ Contribution
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no potential competing interests in this work.
References
1. Chisholm-Burns MA, Wells BG, Schwinghammer TL. Pharmacotherapy Principles and Practice. McGraw-Hill; 2016.
2. WHO. Definition of diabetes mellitus and prevalence. 2013.
3. World Health Organization. Global Report on Diabetes. World Health Organization; 2016.
4. Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas. Diabetes Res Clin Pract. 2019;157:107843. doi:10.1016/j.diabres.2019.107843
5. Cho N, Shaw J, Karuranga S, et al. IDF Diabetes Atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract. 2018;138:271–281. doi:10.1016/j.diabres.2018.02.023
6. Amogne W, Reja A, Amare A. Diabetic foot disease in Ethiopian patients: a hospital based study. Ethiop J Health Dev. 2011;25(1):17–21. doi:10.4314/ejhd.v25i1.69841
7. Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract. 2014;103(2):137–149. doi:10.1016/j.diabres.2013.11.002
8. Al-Rubeaan K, Al Derwish M, Ouizi S, et al. Diabetic foot complications and their risk factors from a large retrospective cohort study. PLoS One. 2015;10(5):e0124446. doi:10.1371/journal.pone.0124446
9. Zhang P, Lu J, Jing Y, Tang S, Zhu D, Bi Y. Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis. Ann Med. 2017;49(2):106–116. doi:10.1080/07853890.2016.1231932
10. Armstrong DG, Cohen K, Courric S, Bharara M, Marston W. Diabetic foot ulcers and vascular insufficiency: our population has changed, but our methods have not. J Diabetes Sci Technol. 2011;5(6):1591–1595. doi:10.1177/193229681100500636
11. Lavery LA, Armstrong DG, Harkless LB. Classification of diabetic foot wounds. J Foot Ankle Surg. 1996;35(6):528–531. doi:10.1016/S1067-2516(96)80125-6
12. Solan YM, Kheir HM, Mahfouz MS, et al. Diabetic foot care: knowledge and practice. J Endocrinol Metab. 2017;6(6):172–177. doi:10.14740/jem388e
13. Muhammad-Lutfi A, Zaraihah M, Anuar-Ramdhan I. Knowledge and practice of diabetic foot care in an in-patient setting at a tertiary medical center. Malay Orthopaed J. 2014;8(3):22. doi:10.5704/MOJ.1411.005
14. Amin N, Doupis J. Diabetic foot disease: from the evaluation of the “foot at risk” to the novel diabetic ulcer treatment modalities. World J Diabetes. 2016;7(7):153. doi:10.4239/wjd.v7.i7.153
15. Boulton AJ, Vileikyte L, Ragnarson-Tennvall G, Apelqvist J. The global burden of diabetic foot disease. Lancet. 2005;366(9498):1719–1724. doi:10.1016/S0140-6736(05)67698-2
16. Abbas Z, Archibald L. Epidemiology of the diabetic foot in Africa. Med Sci Monit. 2005;11(8):RA262–RA70.
17. Mariam TG, Alemayehu A, Tesfaye E, et al. Prevalence of diabetic foot ulcer and associated factors among adult diabetic patients who attend the diabetic follow-up clinic at the University of Gondar Referral Hospital, North West Ethiopia, 2016: institutional-based cross-sectional study. J Diabetes Res. 2017;2017:1–8. doi:10.1155/2017/2879249
18. Deribe B, Woldemichael K, Nemera G. Prevalence and factors influencing diabetic foot ulcer among diabetic patients attending Arbaminch Hospital, South Ethiopia. J Diabetes Metab. 2014;5(1):1–7. doi:10.4172/2155-6156.1000322
19. Gholap MC, Mohite VR. To assess the knowledge and practice regarding foot care among diabetes patients at Krishna hospital, Karad. Indian J Sci Res. 2013;4(2):69–75.
20. American Diabetic Association. Standards of medical care in diabetes—2018 abridged for primary care providers. Clin Diabetes. 2018;36(1):14.
21. Alexiadou K, Doupis J. Management of diabetic foot ulcers. Diabetes Ther. 2012;3(1):4. doi:10.1007/s13300-012-0004-9
22. American Diabetic Association. Lifestyle management: standards of medical care in diabetes—2018. Diabetes Care. 2018;41(Supplement 1):S38–S50. doi:10.2337/dc18-S004
23. Bago BJ Knowledge, practice and barriers of diabetic foot ulcer among diabetic patients in Hawassa University Comprehensive Specialized Hospital, Southern Ethiopia. 2017.
24. Seid A, Tsige Y. Knowledge, practice, and barriers of foot care among diabetic patients attending Felege Hiwot Referral Hospital, Bahir Dar, Northwest Ethiopia. Adv Nurs. 2015;2015:1–9. doi:10.1155/2015/934623
25. Schmidt S, Mayer H, Panfil EM. Diabetes foot self‐care practices in the German population. J Clin Nurs. 2008;17(21):2920–2926. doi:10.1111/j.1365-2702.2008.02352.x
26. Frykberg RG, Zgonis T, Armstrong DG, et al. Diabetic foot disorders: a clinical practice guideline (2006 revision). J Foot Ankle Surg. 2006;45(5):S1–S66.
27. Pitchai P, Joshi J. Knowledge and practice of foot care amongst diabetics in India: comparison between urban and rural setting-a reality check. Int J Health Sci Res. 2015;5(4):181–189.
28. Snyder RJ, Hanft JR. Diabetic foot ulcers–effects on QOL, costs, and mortality and the role of standard wound care and advanced-care therapies. Ostomy Wound Manage. 2009;55(11):28–38.
29. Li R, Yuan L, Guo X-H, et al. The current status of foot self-care knowledge, behaviours, and analysis of influencing factors in patients with type 2 diabetes mellitus in China. Int J Nurs Sci. 2014;1(3):266–271. doi:10.1016/j.ijnss.2014.05.023
30. Hasnain S, Sheikh NH. Knowledge and practices regarding foot care in diabetic patients visiting diabetic clinic in Jinnah Hospital, Lahore. J Pak Med Assoc. 2009;59(10):687.
31. Saurabh S, Sarkar S, Selvaraj K, Kar SS, Kumar SG, Roy G. Effectiveness of foot care education among people with type 2 diabetes in rural Puducherry, India. Indian J Endocrinol Metab. 2014;18(1):106. doi:10.4103/2230-8210.126587
32. Tewahido D, Berhane Y. Self-care practices among diabetes patients in Addis Ababa: a qualitative study. PLoS One. 2017;12(1):e0169062. doi:10.1371/journal.pone.0169062
33. Karadağ FY, Saltoğlu N, Ak Ö, et al. Foot self-care in diabetes mellitus: evaluation of patient awareness. Prim Care Diabetes. 2019;13(6):515–520. doi:10.1016/j.pcd.2019.06.003
34. Singh S, Jajoo S, Shukla S, Acharya S. Educating patients of diabetes mellitus for diabetic foot care. J Fam Med Prim Care. 2020;9(1):367. doi:10.4103/jfmpc.jfmpc_861_19
35. Beiranvand S, Fayazi S, Asadizaker M. Effect of educational programs on the knowledge, attitude, and practice of foot care in patients with diabetes. Jundishapur J Chronic Dis Care. 2015;4(2):e26540. doi:10.5812/jjcdc.26540
36. Margolis DJ, Hofstad O, Feldman HI. Association between renal failure and foot ulcer or lower-extremity amputation in patients with diabetes. Diabetes Care. 2008;31(7):1331–1336. doi:10.2337/dc07-2244
37. Morbach S, Quante C, Ochs HR, Gaschler F, Pallast J-M, Knevels U. Increased risk of lower-extremity amputation among Caucasian diabetic patients on dialysis. Diabetes Care. 2001;24(9):1689–1690. doi:10.2337/diacare.24.9.1689
38. Azmi N, Aris M, Nasreen H, Che-Ahmad A. Diabetic foot care practice and its associated factors among type 2 diabetes mellitus patients attending Primary Health Clinics in Kuantan, Malaysia: a cross sectional study. Med J Malaysia. 2020;19(2). doi:10.31436/imjm.v19i3.1660
39. Wankum N, Hongsranagon P, Sivina S. Assessment of knowledge, attitude, and practice of preventive self-care on diabetic foot ulcer in type II diabetic patients, Muang Roi-Et District, Roi-Et Province, Thailand. J Health Res. 2014;28(4):229–232.
40. Rossaneis MA, Haddad MDCFL, Mathias T, Marcon SS. Differences in foot self-care and lifestyle between men and women with diabetes mellitus. Rev Lat Am Enfermagem. 2016;24. doi:10.1590/1518-8345.1203.2761
41. Van Netten JJ, Lazzarini PA, Armstrong DG, et al. Diabetic Foot Australia guideline on footwear for people with diabetes. J Foot Ankle Res. 2018;11(1):1–4. doi:10.1186/s13047-017-0244-z
42. Groopman JE, Itri LM. Chemotherapy-induced anemia in adults: incidence and treatment. J Natl Cancer Inst. 1999;91(19):1616–1634. doi:10.1093/jnci/91.19.1616
43. Mogre V, Johnson NA, Tzelepis F, Paul C. Barriers to diabetic self‐care: a qualitative study of patients’ and healthcare providers’ perspectives. J Clin Nurs. 2019;28(11–12):2296–2308. doi:10.1111/jocn.14835
44. Ndjaboue R, Dansokho SC, Boudreault B, et al. Patients’ perspectives on how to improve diabetes care and self-management: qualitative study. BMJ Open. 2020;10(4):e032762. doi:10.1136/bmjopen-2019-032762
45. Brown DR, Gary LE. Social support network differentials among married and nonmarried Black females. Psychol Women Q. 1985;9(2):229–241. doi:10.1111/j.1471-6402.1985.tb00874.x
46. van der Hoeven M, Kruger A, Greeff M. Differences in health care seeking behaviour between rural and urban communities in South Africa. Int J Equity Health. 2012;11(1):31. doi:10.1186/1475-9276-11-31
47. Dickson VV, Buck H, Riegel B. Multiple comorbid conditions challenge heart failure self-care by decreasing self-efficacy. Nurs Res. 2013;62(1):2–9. doi:10.1097/NNR.0b013e31827337b3
48. Bayliss EA, Steiner JF, Fernald DH, Crane LA, Main DS. Descriptions of barriers to self-care by persons with comorbid chronic diseases. Ann Fam Med. 2003;1(1):15–21. doi:10.1370/afm.4
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
