Back to Journals » OncoTargets and Therapy » Volume 13
Knockdown of HMGB1 Suppresses Hypoxia-Induced Mitochondrial Biogenesis in Pancreatic Cancer Cells
Authors Yang L, Ye F, Zeng L, Li Y, Chai W
Received 15 October 2019
Accepted for publication 29 January 2020
Published 12 February 2020 Volume 2020:13 Pages 1187—1198
DOI https://doi.org/10.2147/OTT.S234530
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Sanjay Singh
Liangchun Yang,1 Fanghua Ye,1 Li Zeng,2 Yanling Li,2 Wenwen Chai2
1Department of Pediatrics, Xiangya Hospital, Central South University, Changsha, Hunan 410008, People’s Republic of China; 2Department of Nuclear Medicine, Hunan Cancer Hospital, Changsha, Hunan 410008, People’s Republic of China
Correspondence: Wenwen Chai
Department of Nuclear Medicine, Hunan Cancer Hospital, No. 283 Tongzipo Road, Changsha, Hunan 410008, People’s Republic of China
Tel +86 159 7425 5021
Email [email protected]
Purpose: To explore the regulatory effect of HMGB1 upon hypoxia-induced mitochondrial biogenesis in pancreatic cancer PANC1/CFPAC1 cells.
Methods: After a down-regulation of HMGB1 expression by lentivirus-mediated RNAi, the effect of knocking down HMGB1 on hypoxia-induced mitochondrial biogenesis was examined. NRF-1/TFAM expression, mtDNA copy number, ATP content and mitochondrial number/morphology in hypoxia-treated pancreatic cancer cells were detected by quantitative reverse transcription-polymerase chain reaction (qRT-PCR), Western blot, mtDNA and ATP assay kits and electron microscopy, respectively. Cell proliferation was measured by MTS assay. And protein and acetylation levels of PGC-1α and SIRT1 activity were detected by Western blot, immunoprecipitation (IP) and SIRT1 activity kit.
Results: Hypoxia enhanced the expressions of NRF-1/TFAM, boosted mtDNA copy number and ATP content and increased the number of mitochondria in pancreatic cancer cells while induction was suppressed by a knockdown of HMGB1. Knocking down HMGB1 expression lowered hypoxia-induced PGC-1α/SIRT1 expression and activity, phosphorylation of AMPK. PGC-1α over-expression by a plasmid transfection failed to boost mtDNA copy number or ATP content in HMGB1-knockdown cells. A knockdown of HMGB1 attenuated hypoxia with AICAR (an AMPK activator)-induced expression of NRF-1, TFAM, PGC-1α, SIRT1 and the proteins of complexes Ⅰ& Ⅲ and reduced the acetylation level of PGC-1α/SIRT1 activity. Additionally, SRT1720 (a SIRT1 activator)-induced elevation in SIRT1 activity boosted hypoxia-induced PGC-1α deacetylation, except in HMGB1-knockdown cells.
Conclusion: As a novel regulator of mitochondrial biogenesis via AMPK/SIRT1 pathway under hypoxia, HMGB1 may become a potential drug target for therapeutic interventions in pancreatic cancer.
Keywords: HMGB1, mitochondrial biogenesis, PGC-1α, AMPK/SIRT1 pathway, pancreatic cancer
Introduction
Pancreatic cancer has been known as a highly lethal gastrointestinal tumor with a 5-year survival rate of <5%.1 Nutritional deficiencies and cellular hypoxia result in their pathological manifestations of diffuse vascularized interstitial hyperplasia.2 A microenvironment characterized by hypoxic stress is a common feature of rapidly growing tumors. Tumor microenvironment has been gradually recognized as a key contributor to cancer progression and drug resistance.3 In tumor microenvironment, proliferative signaling induces angiogenesis, genomic instability and mutations so as to enable invasion, metastasis, resistance to apoptosis, escape from immune supervision and energy generation.3 An adequate energy supply is indispensable for cancer cells to proliferate, infiltrate and metastasize. Changes in mitochondrial energy metabolism determine the biological and clinical manifestations of cancer.4 Targeting mitochondrial energy metabolism has recently been attempted as a potential focus of cancer treatment. However, the role of mitochondrial energy metabolism in the proliferation and survival of pancreatic cancer cells and potential mechanisms has remained elusive.
As a predominant cellular site of energy production, mitochondria are regulated by the interactions of nucleus and mitochondrial genomes. Mitochondrial dysfunction and gene mutations have been implicated in shifting from a homeostasis of cellular metabolism to a state favoring tumor proliferation.5,6 Mitochondrial dysfunctions lead to excessive production of reactive oxygen species (ROS) promoting a rapid growth of pancreatic cancer cells.7 Loss of electron transport elements, energy-related dysfunctions and impaired protein synthesis due to mitochondrial abnormalities are also closely correlated with pancreatic carcinogenesis.8 Mitochondrial biogenesis is regulated by a diverse set of transcription factors and coactivators.9 PGC-1α has been identified as a key factor of associating external stimuli with modifications during mitochondrial biogenesis.10 The nuclear mitochondrial transcription factor gene (TFAM) is the first critical transcription factor of mitochondrial DNA (mtDNA) copy number and transcription. PGC-1α activates nuclear respiratory factor (NRF-1) and then up-regulates TFAM expression.11 All of these coactivator and transcription factors may transactivate various genes involved in mitochondrial functions such as oxidative phosphorylation, protein import and heme biosynthesis.12
High-mobility group box-1 (HMGB1), a DNA-binding protein, functions as a co-factor for proper transcriptional regulation in somatic cells.13 It is also translocated into cytosol and extracellular space by such multiple cellular stressors as protein aggregate, radiation, oxidation, chemotherapy & intracellular pathogen.14 During carcinogenesis and cancer therapy, HMGB1 is closely correlated with invasion, metastasis and drug resistance by regulating various signaling pathways of immunity, metabolism, genomic stability, apoptosis and autophagy.14 It was reported that endogenous HMGB1 protein expression in pancreatic tumors was lower than that in adjacent normal pancreatic tissue. In pancreatic cancer patients, a lower expression of HMGB1 was closely associated with worse survival.15 As a key regulator of mitochondrial function, HMGB1 maintains normal cellular functions and mitochondrial morphology. Tumors lacking HMGB1 had a markedly lower mitochondrial biogenesis and more mitochondrial dysfunctions.16 HMGB1 regulating heat shock protein beta-1 (HSPB1) expression provides a defense against mitochondrial abnormalities by sustaining OXPHOS in pancreatic cancer cells.17 Although the above studies helped us elucidate the role of HMGB1 in regulating mitochondrial energy metabolism in pancreatic cancer cells, the mechanism of this regulation in hypoxically stressful microenvironment is largely unclear.
Here mitochondrial biogenesis was examined in hypoxia-treated pancreatic cancer cell lines, HMGB1 involvement in mitochondrial biogenesis, the influence of HMGB1 on PGC-1α protein and acetylation levels and the relationship of HMGB1 and AMPK/SIRT1 pathway was also considered. The overall objective was to gain more insights into the regulatory function of HMGB1-mediated mitochondrial biogenesis during the survival of pancreatic cancer cells.
Materials and Methods
Reagents and Cell Culture
Aminoimidazole-4-carboxamide-1- β-D- ribofuranoside (AICAR) was supplied by Sigma Chemical Inc (St Louis, MO, USA); SRT1720 Tocris Bioscience (Bristol, UK); pancreatic cancer cell lines PANC1, CFPAC1, BXPC-3 and AsPC-1 American Type Culture Collection (Manassas, VA, USA); human bronchial epithelial cell HBE, acute myeloid leukemic cell HL-60 and chronic myelogenous leukemic cell K562 Xiangya School of Medicine Type Culture Collection (Changsha, China). HL-60 and K562 cells were cultured in RPMI-1640 medium (Invitrogen, San Diego, CA) and PANC1, CFPAC1, BXPC-3 and AsPC-1 cells in Dulbecco’s modified Eagle’s medium (DMEM; Hyclone, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Thermo Fisher Scientific Inc, Waltham, MA) and 1% antibiotics (100 U/mL penicillin G & 100 mg/mL streptomycin) at 37°C in a cell incubator with 5% CO2 (Thermo Fisher Scientific Inc, USA). Here PANC1 and CFPAC1 cells were divided into a normoxia group (a culture box containing 5%CO2) and a hypoxia group (a culture box containing 1%O2 +5%CO2 + 94%N2).
Lentivirus and Plasmid Transfection
Following the procedures of our previous study,18 HMGB1 small hairpin RNA (shRNA) lentiviral knockdown (GeneCopoeia, Guangzhou, China) or shRNA nonsilencing control was packaged with HIV-based packaging mix (GeneCopoeia) for infecting PANC1 and CFPAC1 cells and thus establishing cells constitutively suppressing HMGB1. Stable clones were selected by puromycin. The following HMGB1 shRNA oligonucleotide sequences were employed: HMGB1 shRNA: 5ʹ -CCGGGCAGATGACAAGCAGCCTTATCTCGAGATAAGGCTGCTTGTCATCTGCTTTTT-3ʹ. Nonsilencing shRNA (control shRNA) as mock-transfected control (target sequence: TTCTCCGAACGTGTCACGT). A lentivirus-PGC-1α vector system (Ubi-MCS-3FLAG- SV40-EGFP-IRES-puromycin) was supplied by GeneChem (Shanghai, China) for infecting PANC1/CFPAC1 cells for up-regulating HMGB1.
Quantitative Real-Time PCR (qRT-PCR)
Sample RNAs were extracted by Trizol (Invitrogen, USA) and cDNA synthesis via PrimeScript™ RT master mix (TaKaRa, Beijing, China). β-actin as a housekeeping gene for standardizing the expression of related genes. β-actin forward: 5′-TCGTGCGTGACATTAAAGAG-3′, β-actin reverse: 5′-ATTGCCGATAGTGATGACCT-3′. The primer sequences of related genes in this study are as follows: NRF-1 forward (5′-TGTCACCATGGCCCTTAACAGTGA-3′) and NRF-1 reverse (5′-TGAACTCCATCTGGGCCATTAGCA-3′), TFAM forward (5′-AAATGGCTGAAGTTGGGCGAAGTG-3′) and TFAM reverse (5′-AGCTTCTTGTGCCCAATCCCAATG-3′). Amplification was done by Opticon continuous fluorescence detection system (MJ Research Inc., Waltham, MA, USA). A total of 40 cycles were set up (denaturation for 15 s at 95°C and annealing for 30 s at 60°C). Fluorescent signal accumulation monitors the entire PCR process in real time and finally quantifies the unknown template by a standard curve.
Antibodies and Western Blot
Antibodies for PGC-1α (#4207), p-AMPKαT172 (#50081), AMPKα (#5832), NRF-1 (#46743), sirtuin1 (SIRT1) (#2496) and acetylated lysine (#MA1-2021) were supplied by Cell Signaling Technology (Beverly, MA, USA); antibodies for TFAM (#ab131607), OXPHOS complexesⅠ - Ⅴ (#ab110413) and β-actin (#ab3280) Abcam (Cambridge, MA, USA); antibodies against HMGB1 (#H00003146-M08) Novus (Littleton, CO, USA).
Treated cells were harvested, centrifuged and rinsed with PBS. After a treatment of lysis buffer (Beyotime, Beijing, China), the supernatant was harvested and stored on ice. The supernatant was centrifuged at 14,000 × g for 30 min at 4°C. Whole-cell lysate was separated by SDS polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred onto a polyvinylidene fluoride (PVDF) blotting membrane (Beyotime, Beijing, China). After 5% skim milk closure, the protein was detected by incubating with 4°C primary antibody overnight and secondary antibody for 1 h at room temperature. Immunoreactivity was determined and observed by enhanced chemiluminescent reagents (Pierce, Waltham, MA). Protein expression was quantified by Band Scan 5.0 system.
Cell Proliferation Assay
Five×103/well cells were inoculated into 24-well cell culture plates and incubated overnight in complete medium containing 0%, 10%, 20% and 40% propolis. After differential treatments, cell proliferation was measured by CellTiter96® aqueous nonradioactive (MTS) assay (Promega, Madison, WI) at 12/48/72 h. After an addition of detection reagent, cells were incubated at 37°C and 5% CO2 for 3 h. Absorbance was assayed at 490 nm by a spectrophotometer (BioTek, VT, USA). The cell-free wells containing the same media were used as blank controls.
Transmission Electron Microscopy
Based on the previously described methods,19 treated cells were collected and rinsed with precooled PBS. The samples were fixed for 3 h with 2.5% glutaraldehyde and 2% paraformaldehyde and 1.5 h with 1% osmium tetroxide. After dehydration and embedding, ultrathin sections were double-stained with 3% uranyl acetate and lead nitrate and later observed under a transmission electron microscope of H7500 (Hitachi, Tokyo, Japan).
Measurement of Intracellular ATP Levels
According to the instructions of luminescent ATP kit (Beyotime Biotechnology), first detection buffer was mixed with the substrate at room temperature. Then, 100 μL of the mixture was loaded into the cell culture plate and incubated for 15 min at room temperature. Finally, luminescent intensity was assayed by a microplate reader (Beckman Coulter Inc, Miami, USA). The incremental yield of ATP was expressed as a percentage of the control group.
Measurement of mtDNA Copy Number
According to the instructions of universal genome DNA kit (Zoman Biotech, Beijing, China), genomic DNA was isolated by a lysis buffer system and then purified by elution and adsorption. Finally, the sample was amplified by qRT-PCR using target gene primer of mitochondrial DNA and nuclear gene control primer. And the value of mtDNA copy number was shown as a relative number of Cyt B to genome β-actin.20
Immunoprecipitation (IP)
IP lysis buffer (Millipore, Billerica, MA, USA) containing protease inhibitor was added into harvested cells for lysis at 4°C for 30 min. After centrifugation at 12,000 g for 30 min, the supernatant was harvested and its protein concentration determined by the method of bicinchoninic acid (BCA). 1 μg anti-PGC-1α antibody and protein A/G beads were added into protein samples and incubated overnight at 4°C with gentle vortexing. After immunoprecipitation, protein A/G beads were centrifuged as pellet at 3000 g at 4°C and rinsed 3–4 times with 1 mL lysate. Finally, 5×SDS loading buffer was added into protein A/G beads and boiling water for 10 min for protein elution prior to SDS-PAGE electrophoresis. Protein was probed with anti-acetyl lysine antibody and visualized by Western blot.
SIRT1 Deacetylase Activity Assay
A commercial assay kit of Sigma Inc (St. Louis, MO, USA) was utilized. Collected cells were re-suspended in a lysate, centrifuged at 1300 × g for 10 min at 4°C and supernatant was removed. The nuclei were rinsed, sonicated for 30 s and placed on ice for later usage. After 10-min centrifugation at 20,000 × g, the supernatant was harvested and stored at −80°C or immediately utilized. Assay working buffer was added into 20 μl sample and agitated thoroughly to ensure sufficient reaction at room temperature. Fluorescent intensity was measured by a Full-Wavelength Microplate Reader at an emission wavelength of 460 nm and an excitation wavelength of 355 nm (measured every 2 min for 60 min). The activity of SIRT1 was expressed as protein concentration integrated by the change rate of fluorescent intensity.
Statistical Analysis
Data were analyzed using GraphPad Prism 6.0 software (San Diego, CA, USA). Quantitative variables were expressed as mean SD, and analyzed by Student´s t-test and one-way ANOVA. Experimental results are independently repeated 3 times and P<0.05 was considered statistically significant.
Results
Hypoxia-Induced Mitochondrial Biogenesis in PANC1/CFPAC1 Cells
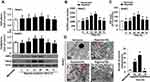
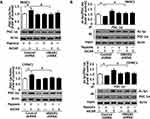
To determine whether hypoxia-induced mitochondrial biogenesis in pancreatic cancer cells, the cells were subjected to hypoxia for 3–72 h and NRF-1/TFAM expression was assayed by Western blot. As compared with the normoxia group, NRF-1/TFAM protein expression peaked at 12/24 h after hypoxic treatment (Figure 1A and S1A). Moreover, marked elevations of mtDNA and ATP levels occurred at 12/24 h but not at other timepoints (Figure 1B and C, Figure S1B and C). And ultrastructural analysis provided more supporting evidence of mitochondrial biogenesis. Hypoxia-treated cells had a significantly higher mitochondrial number than that of the normoxia group at 12/24 h (Figure 1D and S1D). In contrast, at 72 h, there was lower mitochondrial number; reduced matrix density; disorganized mitochondrial cristae and small swollen and irregular mitochondria (Figure 1D and S1D). Thus, 24 h hypoxic treatment was utilized for subsequent experiments. It suggested that hypoxic treatment induced mitochondrial biogenesis in a time-dependent manner in pancreatic cancer cells.
A Depletion of HMGB1 Suppressed Hypoxia-Induced Mitochondrial Biogenesis
Firstly the level of HMGB1 expression was determined by Western blot in different cell lines. Our previous studies have shown that HMGB1 was over-expressed in K562/HL-60 cells whereas it was noticeably lower in HBE cells.21 Similar to leukemia (HL-60 & K562) cells, HMGB1 expression was also elevated in all four pancreatic cancer cell lines (Figure S2A). Hypoxic environment of solid tumors induced the translocation of HMGB1 from the nucleus of tumor cells to cytoplasm.22 For further evaluating the relationship between HMGB1 expression and hypoxia, nuclear/cytosolic-HMGB1 expressions were detected by Western blot. Hypoxia lowered nuclear – HMGB1 expression and enhanced cytosolic-HMGB1 expression in a time-dependently manner, respectively (Figure S2B).
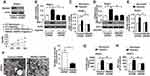
For elucidating the effect of HMGB1 on the viability of hypoxia-treated pancreatic cancer cells, an MTS assay was performed. Without a control or HMGB1 shRNA transfection, the proliferative capacity of hypoxia-treated cells was lower than that of cells under normoxia (Figure 2A and S3A). In addition, a depletion of HMGB1 expression significantly lowered the proliferative capacity of hypoxia-treated cells compared with those of control shRNA group (Figure 2A, Figure S3A). It suggested that knocking down HMGB1 expression suppressed the proliferation of PANC1/CFPAC1 cell under hypoxia. For further determining whether or not HMGB1 impacted mitochondrial biogenesis, PANC1/CFPAC1 cells were subjected to 24 h hypoxia and NRF-1/TFAM expression was assayed by Western blot and qRT-PCR. A depletion of HMGB1 expression significantly lowered the levels of NRF-1/TFAM protein and mRNA (Figure 2B–E, Figure S3B–E). It suggested that HMGB1 contributed to the expressions of mitochondrial biogenesis-related transcription factors.
Ultrastructural analysis provided more supporting evidence of HMGB1-regulated mitochondrial biogenesis. As compared with the control shRNA group, PANC1/CFPAC1 cells transfected with HMGB1 shRNA after 24 h hypoxia exhibited lower mitochondrial numbers; disorganized mitochondrial cristae and swollen & irregular morphologies (Figure 2F, Figure S3F). Moreover, a depletion of HMGB1 expression by target-specific shRNA significantly lowered mtDNA and ATP levels in hypoxia-treated cells as compared with the control shRNA group (Figure 2G and H, Figure S3G and H). It suggested that HMGB1 was sufficient for sustaining the biogenesis and mitochondrial function in pancreatic cancer cells.
Requirement of HMGB1 for PGC-1α-Regulated Mitochondrial Biogenesis
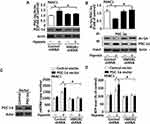
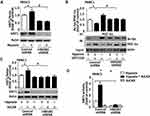
PGC-1α played a vital role in the regulation of mitochondrial biogenesis.10 However, the function of PGC-1α in response to HMGB1-mediated mitochondrial biogenesis has remained unknown. A depletion of HMGB1 expression significantly suppressed hypoxia-induced up-regulation of PGC-1α as compared with the control shRNA group (Figure 3A and S4A). Deacetylation by SIRT1 maintained the active form of PGC-1α for binding to chromatin and promoting the transcription of target genes.23 Hypoxic treatment significantly lowered PGC-1α acetylation level and thereby enhanced PGC-1α activity in the control shRNA group (Figure 3B and S4B). Knocking down HMGB1 expression reversed the decline of PGC-1α acetylation level in hypoxia-treated cells (Figure 3B and S4B). It suggested that HMGB1 may be a critical regulator of PGC-1α acetylation. For further exploring whether or not PGC-1α is necessary for HMGB1-regulated mitochondrial biogenesis, a PGC-1α plasmid was transfected into PANC1/CFPAC1 cells for up-regulating the expression of endogenous PGC-1α. And PGC-1α protein became markedly up-regulated in cells with transfected PGC-1α plasmid as compared with counterparts transfected with a control vector (Figure 3C and S4C). A pretreatment of pancreatic cancer cells with PGC-1α plasmid by gene transfection and 24 h hypoxic treatment significantly boosted mtDNA copy number and ATP content in the control shRNA group (Figure 3C and D, Figure S4C and D). In contrast, PGC-1α over-expression increased neither mtDNA copy number nor ATP content in HMGB1-knockdown cells (Figure 3C and D, Figure S4C and D). It suggested that HMGB1 was required for PGC-1α-regulated mitochondrial biogenesis.
Involvement of HMGB1 in the Regulation of AMPK-Mediated Mitochondrial Biogenesis
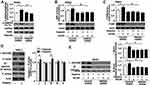
Functioning as a metabolic checkpoint, AMPK is normally activated by a high AMP/ATP ratio so that it maintains energy homeostasis.24 Activated AMPK is involved in the regulation of mitochondrial biogenesis.25 A role was also detected for AMPK in HMGB1-mediated mitochondrial biogenesis. AMPKα phosphorylation (p-AMPKα) was enhanced by 24 h hypoxic treatment in the control shRNA group. A depletion of HMGB1 expression significantly lowered the p-AMPKα level as compared with the control shRNA group (Figure 4A and S5A). Moreover, PANC1/CFPAC1 cells were pretreated for 2 h with AICAR (an AMPK activator) (0.5 mM) and then incubated for 24 h under hypoxia. Under hypoxic conditions, AICAR treatment significantly enhanced NRF-1/TFAM expression in the control shRNA group but not in the HMGB1-knockdown group (Figure 4B and C, Figure S5B and C). Mitochondrial OXPHOS system might generate cellular energy predominantly in the form of ATP.26 For further characterizing the role of hypoxia in regulating mitochondrial OXPHOS proteins, PANC1/CFPAC1 cells were subjected to 24 h hypoxia. Complexes Ⅰ/Ⅲ protein levels became markedly up-regulated whereas those of complexes Ⅱ/Ⅳ/Ⅴ showed no obvious change (Figure 4D and S5D). Furthermore, hypoxia with AICAR treatment could markedly boosted complexesⅠ/Ⅲ protein levels in the control shRNA group, while there was almost a total suppression in the HMGB1-knockdown group (Figure 4E and S5E). These results suggested that HMGB1 was involved in AMPK-mediated mitochondrial biogenesis in pancreatic cancer cells.
Involvement of HMGB1 in the Regulation of AMPK-Mediated PGC-1α Expression and Activity
To explore whether AMPK mediated the expression of PGC-1α by HMGB1, PANC1/CFPAC1 cells were pretreated with AICAR (0.5 mM) for 2 h and then incubated under 24 h hypoxia. The hypoxia-induced PGC-1α expression was enhanced by AICAR in the control shRNA group (Figure 5A). After transfection of HMGB1 shRNA, there was an obvious decline of AICAR-induced PGC-1α level as compared with the control shRNA group (Figure 5A). Furthermore, hypoxia with AICAR treatment significantly lowered the acetylation level of PGC-1α in the control group (Figure 5B). Knocking down HMGB1 expression suppressed the decline of PGC-1α acetylation level after hypoxia with AICAR treatment as compared with the control shRNA group (Figure 5B). It hinted at an important role of HMGB1 in AMPK-regulated PGC-1α expression and activity.
HMGB1-Regulated PGC-1α Deacetylation Through SIRT1
For further characterizing the role of SIRT1 in HMGB1-mediated activation of PGC-1α, SIRT1 expression was detected by Western blot. SIRT1 expression was up-regulated by 24 h hypoxic treatment in the control shRNA group (Figure 6A and S6A). A depletion of HMGB1 expression markedly lowered the SIRT1 level as compared with control shRNA groups (Figure 6A and S6A). Moreover, PANC1/CFPAC1 cells were pretreated for 1 h with SRT1720 (10 μM), an SIRT1 activator and then subjected to 24 h hypoxia. Hypoxia with SRT1720 treatment markedly lowered the acetylation level of PGC-1α in control shRNA groups (Figure 6B and S6B). Knocking down HMGB1 expression suppressed the decline of PGC-1α acetylation level under hypoxia and SRT1720 treatment as compared with the control shRNA group (Figure 6B and S6B). It hinted at the involvement of HMGB1 in SIRT1-mediated PGC-1α deacetylation. Furthermore, hypoxia-induced SIRT1 expression and activity were enhanced by AICAR treatment in the control shRNA group (Figure 6C and D, Figure S6C and D). After transfection of HMGB1 shRNA, the SIRT1 level and activity declined obviously as compared with the control shRNA group (Figure 6C and D, Figure S6C and D). These data suggested that HMGB1-regulated PGC-1α deacetylation through the AMPK-SIRT1 pathway.
Discussion
Here HMGB1-regulated hypoxia-induced mitochondrial biogenesis directly by affecting the expression and activity of PGC-1α in PANC1/CFPAC1 cells. HMGB1 is vital for AMPK/SIRT1-mediated PGC-1α modulation and mitochondrial biogenesis under hypoxia. It suggested that HMGB1 might play an important role in the regulation of mitochondrial homeostasis by AMPK/SIRT1 pathway under hypoxic conditions in pancreatic cancer.
Closely correlated with apoptosis, autophagy and aging of cells, mitochondrial energy metabolism plays an important role in the development of disease and subsequent treatment.27 It was reported that an ablation of K-Ras caused the demise of a large majority of malignant cells. Few surviving cells engaged in a high level of mitochondrial respiration and there were other characteristics of tumor initiation.28 Thus, cancer initiation/progression might be driven by or be dependent upon mitochondrial function. Targeting mitochondria and suppressing mitochondrial biogenesis are expected to be an effective therapeutic strategy for pancreatic cancer. Hypoxically stressful microenvironment is a feature of rapidly growing tumors inducing mitochondrial metabolic reprogramming and thereby promoting cancer survival.3 Here hypoxia could markedly boost the expression levels of NFR1, TFAM, mtDNA and ATP and mitochondria numbers in pancreatic cancer cells. It suggested that hypoxic treatment induced mitochondrial biogenesis and energy production in pancreatic cancer cells at the proper time to ensure their survival.
As an evolutionarily conserved chromatin-associated protein, HMGB1 maintains nuclear homeostasis and functions as a critical regulator of mitochondrial function and morphology.17 HMGB1 acts an important role during carcinogenesis and cancer therapy,21,29,30 and mitochondrial abnormalities are often predominant.31,32 Endogenous HMGB1 protein decreased and it was highly correlated with poor survival in pancreatic cancer patients.15 HMGB1 regulating heat shock protein beta-1 (HSPB1) expression serves as a defense against mitochondrial abnormalities, sustaining OXPHOS in pancreatic cancer cells.17 However, HMGB1 expression and function in regulating mitochondrial biogenesis in hypoxically stressful microenvironment of pancreatic cancer have been poorly elucidated. Here HMGB1 was required for hypoxia-induced mitochondrial biogenesis in pancreatic cancer cells. Knocking down HMGB1 expression lowered cell proliferation under hypoxic conditions. Additionally, knocking down HMGB1 expression reduced the NFR1/TFAM mRNA and protein levels, number of mitochondria, mtDNA copy number and ATP content. These observations provided rationales for developing novel therapies for modulating mitochondrial energy metabolism in pancreatic cancer.
Accumulating evidence has confirmed the involvement of PGC-1α in the regulation of cancer energy metabolism. PGC-1α is expressed abundantly in different cancers, including pancreatic cancer, melanoma, breast cancer, prostate cancer and hepatocarcinoma.33 In melanoma cells, activation of PGC-1α could enhance mitochondrial energy metabolism and ROS detoxification capacities and reprogram cell metabolism for maintaining cell growth and survival.34 In pancreatic cancer stem cells, highly expressed PGC-1α was vital for maintaining mitochondrial respiration.35 Overexpressed PGC-1α enabled pancreatic cancer stem cells more sensitive than differentiated counterparts to metformin.35 Additionally, PGC-1α is normally regulated by acetylation at the posttranslational level. Here HMGB1 not only up-regulated the protein expression of PGC-1α but also lowered the acetylation level of PGC-1α. Up-regulating PGC-1α expression in pancreatic cells by gene transfection significantly boosted mtDNA and ATP levels under hypoxia. In contrast, knocking down HMGB1 expression obviously suppressed PGC-1α-induced increases as compared with the control shRNA group. Together, these findings hinted at a role for HMGB1 in PGC-1α-regulated mitochondrial biogenesis in pancreatic cancer cells.
As a fuel sensor, AMPK plays important roles in regulating mitochondrial biogenesis and function in almost all cells under both physiological and pathological circumstances.25 Up-regulating or activating AMPK by AICAR treatment boosted the levels of mitochondrial biogenesis-related proteins and oxidative phosphorylation.36 AMPK could up-regulate SIRT1 and induce deacetylation of PGC-1α. AMPK-activated PGC-1α has attracted much attention in the regulation of mitochondrial biogenesis.37 Here a depletion of HMGB1 expression significantly suppressed hypoxia-induced phosphorylation of AMPK. Activating AMPK by AICAR treatment enhanced the hypoxia-induced expressions of NRF-1, TFAM, PGC-1α and proteins of complexes Ⅰ/Ⅲ and attenuated the acetylation level of PGC-1α, except in HMGB1-depleted cells. It suggested that HMGB1 was involved in AMPK-mediated mitochondrial biogenesis and related protein expressions in pancreatic cancer cells.
As a member of classⅢ histone deacetylase family of enzymes, SIRT1 plays some important functions in such a large variety of biological processes as inflammation, mitochondrial biogenesis, stress resistance and apoptosis.38 SIRT1 interacting with PGC-1α plays an important role during carcinogenesis. And a SIRT1/PGC-1α-dependent increase in OXPHOS and mitochondrial biogenesis promoted chemoresistance of colon cancer cell.39 Moreover, activating AMPK up-regulates SIRT1 expression and activity while suppressing AMPK markedly lowers the resveratrol-mediated SIRT1 level.40 It was reported that adiponectin up-regulated SIRT1 levels and mitochondrial gene expression for suppressing apoptosis through AMPK/SIRT1/PGC-1α pathway.41 Our study showed that hypoxia significantly up-regulated SIRT1 level and was suppressed by a depletion of HMGB1. SRT1720-induced elevation of SIRT1 activity boosted hypoxia-induced PGC-1α deacetylation, except in HMGB1-knockdown cells. It suggested that HMGB1 regulated the deacetylation of PGC-1α through SIRT1. Furthermore, an AMPK activator up-regulating SIRT1 expression and activity was also suppressed in HMGB1-knockdown cells. It hinted that HMGB1 was a critical regulator of SIRT1 expression and activity via the AMPK pathway.
Conclusions
HMGB1 functions as a positive regulator of mitochondrial biogenesis through the AMPK/PGC-1α pathway in pancreatic cancer cells. In hypoxia-treated cells, HMGB1-mediated mitochondrial biogenesis regulates NFR1/TFAM expression, mtDNA and ATP levels and PGC-1α/SIRT1 expression and activity. The above findings add novel insights into the role of HMGB1 during mitochondrial biogenesis. It provides rationales for utilizing HMGB1 as a potential therapeutic agent for mitochondrial dysfunction.
Acknowledgments
The present study was supported by National Natural Science Foundation of China (Grant numbers 81770178 & 81601528).
Author Contributions
All authors contributed to data analysis, drafting or revising the paper, gave final approval of the version to be published, and agreed to be accountable for all aspects of the paper.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Ilic M, Ilic I. Epidemiology of pancreatic cancer. World J Gastroenterol. 2016;22(44):9694–9705. doi:10.3748/wjg.v22.i44.9694
2. Apte M, Pirola RC, Wilson JS. Pancreatic stellate cell: physiologic role, role in fibrosis and cancer. Curr Opin Gastroenterol. 2015;31(5):416–423. doi:10.1097/MOG.0000000000000196
3. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi:10.1016/j.cell.2011.02.013
4. Kudryavtseva AV, Krasnov GS, Dmitriev AA, et al. Mitochondrial dysfunction and oxidative stress in aging and cancer. Oncotarget. 2016;7(29):44879–44905. doi:10.18632/oncotarget.v7i29
5. Gasparre G, Porcelli AM, Lenaz G, Romeo G. Relevance of mitochondrial genetics and metabolism in cancer development. Cold Spring Harb Perspect Biol. 2013;5(2). doi:10.1101/cshperspect.a011411
6. Ju YS, Alexandrov LB, Gerstung M, et al. Origins and functional consequences of somatic mitochondrial DNA mutations in human cancer. Elife. 2014;3. doi:10.7554/eLife.02935
7. Jeong Y, Lim JW, Kim H. Lycopene inhibits reactive oxygen species-mediated NF-kappaB signaling and induces apoptosis in pancreatic cancer cells. Nutrients. 2019;11(4). doi:10.3390/nu11040762
8. Hardie RA, van Dam E, Cowley M, et al. Mitochondrial mutations and metabolic adaptation in pancreatic cancer. Cancer Metab. 2017;5(2). doi:10.1186/s40170-017-0164-1
9. Scarpulla RC, Vega RB, Kelly DP. Transcriptional integration of mitochondrial biogenesis. Trends Endocrinol Metab. 2012;23(9):459–466. doi:10.1016/j.tem.2012.06.006
10. Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell. 1998;92(6):829–839. doi:10.1016/S0092-8674(00)81410-5
11. Taherzadeh-Fard E, Saft C, Akkad DA, et al. PGC-1alpha downstream transcription factors NRF-1 and TFAM are genetic modifiers of Huntington disease. Mol Neurodegener. 2011;6(1):32. doi:10.1186/1750-1326-6-32
12. Chau CM, Evans MJ, Scarpulla RC. Nuclear respiratory factor 1 activation sites in genes encoding the gamma-subunit of ATP synthase, eukaryotic initiation factor 2 alpha, and tyrosine aminotransferase. Specific interaction of purified NRF-1 with multiple target genes. J Biol Chem. 1992;267(10):6999–7006.
13. Lotze MT, Tracey KJ. High-mobility group box 1 protein (HMGB1): nuclear weapon in the immune arsenal. Nat Rev Immunol. 2005;5(4):331–342. doi:10.1038/nri1594
14. Tang D, Kang R, Zeh HJ
15. Kang R, Xie Y, Zhang Q, et al. Intracellular HMGB1 as a novel tumor suppressor of pancreatic cancer. Cell Res. 2017;27(7):916–932. doi:10.1038/cr.2017.51
16. Tohme S, Yazdani HO, Liu Y, et al. Hypoxia mediates mitochondrial biogenesis in hepatocellular carcinoma to promote tumor growth through HMGB1 and TLR9 interaction. Hepatology. 2017;66(1):182–197. doi:10.1002/hep.v66.1
17. Tang D, Kang R, Livesey KM, et al. High-mobility group box 1 is essential for mitochondrial quality control. Cell Metab. 2011;13(6):701–711. doi:10.1016/j.cmet.2011.04.008
18. Ye F, Chai W, Xie M, et al. HMGB1 regulates erastin-induced ferroptosis via RAS-JNK/p38 signaling in HL-60/NRAS(Q61L) cells. Am J Cancer Res. 2019;9(4):730–739.
19. Zhang Z, Wu B, Chai W, et al. Knockdown of WAVE1 enhances apoptosis of leukemia cells by downregulating autophagy. Int J Oncol. 2016;48(6):2647–2656. doi:10.3892/ijo.2016.3446
20. Venegas V, Halberg MC. Measurement of mitochondrial DNA copy number. Methods Mol Biol. 2012;837:327–335.
21. Yang L, Yu Y, Kang R, et al. Up-regulated autophagy by endogenous high mobility group box-1 promotes chemoresistance in leukemia cells. Leuk Lymphoma. 2012;53(2):315–322. doi:10.3109/10428194.2011.616962
22. Liu Y, Yan W, Tohme S, et al. Hypoxia induced HMGB1 and mitochondrial DNA interactions mediate tumor growth in hepatocellular carcinoma through Toll-like receptor 9. J Hepatol. 2015;63(1):114–121. doi:10.1016/j.jhep.2015.02.009
23. Ruderman NB, Xu XJ, Nelson L, et al. AMPK and SIRT1: a long-standing partnership? Am J Physiol Endocrinol Metab. 2010;298(4):E751–760. doi:10.1152/ajpendo.00745.2009
24. Hardie DG. Sensing of energy and nutrients by AMP-activated protein kinase. Am J Clin Nutr. 2011;93(4):891S–896. doi:10.3945/ajcn.110.001925
25. Bergeron R, Ren JM, Cadman KS, et al. Chronic activation of AMP kinase results in NRF-1 activation and mitochondrial biogenesis. Am J Physiol Endocrinol Metab. 2001;281(6):E1340–1346. doi:10.1152/ajpendo.2001.281.6.E1340
26. Komen JC, Thorburn DR. Turn up the power - pharmacological activation of mitochondrial biogenesis in mouse models. Br J Pharmacol. 2014;171(8):1818–1836. doi:10.1111/bph.12413
27. Wallace DC, Fan W, Procaccio V. Mitochondrial energetics and therapeutics. Annu Rev Pathol. 2010;5:297–348. doi:10.1146/annurev.pathol.4.110807.092314
28. Viale A, Pettazzoni P, Lyssiotis CA, et al. Oncogene ablation-resistant pancreatic cancer cells depend on mitochondrial function. Nature. 2014;514(7524):628–632. doi:10.1038/nature13611
29. Liu L, Yang M, Kang R, et al. HMGB1-induced autophagy promotes chemotherapy resistance in leukemia cells. Leukemia. 2011;25(1):23–31. doi:10.1038/leu.2010.225
30. Huang J, Ni J, Liu K, et al. HMGB1 promotes drug resistance in osteosarcoma. Cancer Res. 2012;72(1):230–238. doi:10.1158/0008-5472.CAN-11-2001
31. Chan DC. Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol. 2006;22:79–99. doi:10.1146/annurev.cellbio.22.010305.104638
32. Poyton RO, McEwen JE. Crosstalk between nuclear and mitochondrial genomes. Annu Rev Biochem. 1996;65:563–607. doi:10.1146/annurev.bi.65.070196.003023
33. Bost F, Kaminski L. The metabolic modulator PGC-1alpha in cancer. Am J Cancer Res. 2019;9(2):198–211.
34. Vazquez F, Lim JH, Chim H, et al. PGC1alpha expression defines a subset of human melanoma tumors with increased mitochondrial capacity and resistance to oxidative stress. Cancer Cell. 2013;23(3):287–301. doi:10.1016/j.ccr.2012.11.020
35. Sancho P, Burgos-Ramos E, Tavera A, et al. MYC/PGC-1alpha balance determines the metabolic phenotype and plasticity of pancreatic cancer stem cells. Cell Metab. 2015;22(4):590–605. doi:10.1016/j.cmet.2015.08.015
36. Jorgensen SB, Treebak JT, Viollet B, et al. Role of AMPKα2 in basal, training-, and AICAR-induced GLUT4, hexokinase II, and mitochondrial protein expression in mouse muscle. Am J Physiol Endocrinol Metab. 2007;292(1):E331–339. doi:10.1152/ajpendo.00243.2006
37. Kahn BB, Alquier T, Carling D, Hardie DG. AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 2005;1(1):15–25. doi:10.1016/j.cmet.2004.12.003
38. Vassilopoulos A, Fritz KS, Petersen DR, Gius D. The human sirtuin family: evolutionary divergences and functions. Hum Genomics. 2011;5(5):485–496. doi:10.1186/1479-7364-5-5-485
39. Vellinga TT, Borovski T, de Boer VC, et al. SIRT1/PGC1alpha-dependent increase in oxidative phosphorylation supports chemotherapy resistance of colon cancer. Clin Cancer Res. 2015;21(12):2870–2879. doi:10.1158/1078-0432.CCR-14-2290
40. Wan D, Zhou Y, Wang K, Hou Y, Hou R, Ye X. Resveratrol provides neuroprotection by inhibiting phosphodiesterases and regulating the cAMP/AMPK/SIRT1 pathway after stroke in rats. Brain Res Bull. 2016;121:255–262. doi:10.1016/j.brainresbull.2016.02.011
41. Huang B, Cheng X, Wang D, et al. Adiponectin promotes pancreatic cancer progression by inhibiting apoptosis via the activation of AMPK/Sirt1/PGC-1alpha signaling. Oncotarget. 2014;5(13):4732–4745. doi:10.18632/oncotarget.1963
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.