Back to Journals » Cancer Management and Research » Volume 12
Is There a Survival Benefit for Patients with Advanced Ovarian Clear Cell Carcinoma Who Complete More Than 6 Cycles of Postoperative Chemotherapy?
Authors Wang J, Shi Y , Liu Y, Li W, Jiang H, Cai H
Received 3 September 2020
Accepted for publication 12 October 2020
Published 13 November 2020 Volume 2020:12 Pages 11631—11638
DOI https://doi.org/10.2147/CMAR.S280141
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chien-Feng Li
Jing Wang,1– 4,* Yuying Shi,1,3,4,* Yan Liu,2 Wei Li,5 Hong Jiang,2 Hongbing Cai1,3,4
1Department of Gynecological Oncology, Zhongnan Hospital of Wuhan University, Wuhan, Hubei 430071, People’s Republic of China; 2Department of Obstetrics and Gynecology, Xiangyang Central Hospital, Affiliated Hospital of Hubei University of Arts and Science, Xiangyang, Hubei, People’s Republic of China; 3Hubei Key Laboratory of Tumor Biological Behaviors, Wuhan, Hubei, People’s Republic of China; 4Hubei Cancer Clinical Study Center, Wuhan, Hubei, People’s Republic of China; 5Department of Obstetrics and Gynecology, Xiangyang No. 1 People’s Hospital, Hubei University of Medicine, Xiangyang, Hubei 441000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Hongbing Cai Department of Gynecological Oncology
Zhongnan Hospital of Wuhan University, NO. 169 Donghu Road, Wuchang District, Wuhan, Hubei 430071, People’s Republic of China
Tel +86 15090961340
Email [email protected]
Hong Jiang
Department of Obstetrics and Gynecology, Xiangyang Central Hospital, Affiliated Hospital of Hubei University of Arts and Science, NO. 136 Jinzhou Road, Xiangcheng District, Xiangyang, Hubei 441000, People’s Republic of China
Email [email protected]
Purpose: To provide a reference for clinicians, whether patients with advanced ovarian clear cell carcinoma (OCCC) require chemotherapy (CT) for more than 6 cycles after tumor debulking.
Patients and Methods: A retrospective review was performed on 85 women diagnosed and treated for advanced OCCC. Outcomes of patients who underwent > 6 vs ≤ 6 cycles of CT were analyzed based on clinicopathological factors.
Results: Among the 85 patients with advanced OCCC, 47 patients underwent ≤ 6 cycles of CT, and 38 patients underwent CT for over 6 cycles. Out of these, 49 patients had disease recurrence, and 35 died. The 2-year progression-free survival (PFS) for patients in the two groups was 51.5% and 42.2% (P>0.05), respectively. The 2-year overall survival (OS) was 59.7% and 64.5%, respectively (P>0.05), and the difference was not statistically significant. Multivariate analysis showed that residual tumor diameter was an independent risk factor for prognosis (PFS and OS). We divided the patients into three groups according to residual tumor diameter as 0 (R0), ≤ 1cm (R1), and > 1cm (R2). The prognosis of R0 was better than R1 and R2. Further studies found that patients who received postoperative adjuvant chemotherapy for over 6 cycles showed no difference in improved prognosis, regardless of residual tumor diameter.
Conclusion: Patients with advanced OCCC who received more than 6 courses of adjuvant chemotherapy after surgery did not show improved prognosis. The residual tumor diameter is an independent indicator of prognosis in patients with advanced OCCC. Complete staging improves the prognosis of patients compared to the ideal or non-ideal cytoreductive surgery.
Keywords: ovarian clear cell carcinoma, chemotherapy, residual tumor, prognosis, overall survival
Introduction
Epithelial ovarian cancer (EOC) is the most common cause of gynecological malignancy deaths. Each year, approximately 230,000 women are diagnosed with EOC globally, and 150,000 women die of the disease.1 Ovarian clear cell carcinoma (OCCC) is a rare subtype of EOC, accounting for only 5–10% cases. However, it is of great concern because of its distinctive clinical features, such as concurrent endometriosis and poor prognosis.2–5 The incidence of OCCC varies regionally and is significantly higher in Asia than in other regions. Reports show that OCCC accounts for approximately 4.8% of patients with EOC in whites, 3.1% in blacks, 11.1% in Asians, and 25% in Japanese patients.6,7 A recent Japanese study reported that OCCC increased significantly, accounting for up to 30% of EOC.1
In endometriosis patients, the risk of OCCC is significantly increased (relative risk = 12.4).8,9 The majority of women (56.3%) who are diagnosed with OCCC are in the early stage. According to the Federation of Gynecology and Obstetrics (FIGO) staging criteria (2014), the 5-year survival rate of OCCC patients staged I–IV is 85.3%, 60.3%, 31.5%, and 17.5%, respectively, which is significantly lower than serous ovarian adenocarcinoma at corresponding stages.10,11
Currently, the standard treatment for ovarian cancer is maximum tumor cell reduction combined with platinum-based CT.12 However, compared with other subtypes, OCCC is less sensitive to platinum-based CT, and patients in the advanced stage of the disease are more likely to develop drug resistance to CT, which results in a high recurrence rate and poor prognosis.13–16 The 2019 National Comprehensive Cancer Network (NCCN) guidelines recommended 6 cycles of CT after surgery for patients with advanced ovarian cancer (stages II–IV), and 3–6 cycles of intravenous chemotherapy for treating early-stage disease.17,18 There is no evidence confirming the requirement of more than 6 cycles of combination chemotherapy as initial chemotherapy for ovarian cancer patients.19 For patients with OCCC, there is no consensus on the sufficient number of CT cycles, and whether over 6 cycles of CT are beneficial. We, therefore, retrospectively analyzed the clinicopathological data of 85 patients with advanced OCCC, evaluated the effect of different CT cycles on disease prognosis, and provided a reference for clinicians to select appropriate CT cycles.
Patients and Methods
Ethical Statement for Collecting Clinical Information
This was a retrospective study. The study was carried out according to the Declaration of Helsinki, and written informed consent was obtained from all patients or their legal guardians. Ethical approval was obtained from the Institutional Review Board of the Zhongnan Hospital of Wuhan University.
Study Design and Patient Selection
In a retrospective study, medical records were reviewed for 134 women diagnosed and treated for OCCC at Zhongnan Hospital of Wuhan University, Hubei Cancer Hospital, Chongqing Cancer Hospital, and Xiangyang Central Hospital from January 2012 to December 2017. Following the FIGO staging criteria (2014), stage I was considered an early-stage disease, while stages greater than stage I were classified as advanced disease. Experienced pathologists reviewed the pathological diagnoses. Patients who underwent surgical staging and were confirmed as OCCC, staged II–IV, along with complete follow-up information, were included for the study. Based on the study entry criteria, 85 cases were selected (Figure 1). Progression-free survival (PFS) was calculated as the number of months from the time of diagnosis to tumor progression or patient’s death, regardless of cause. Tumor progression was defined as tumor marker elevation, radiological or clinical recurrence, or pathological evidence after reoperation. Overall survival (OS) was calculated from the time of diagnosis to the patient’s death or the end of follow-up. The study was approved by the Institutional Review Board of Zhongnan Hospital of Wuhan University.
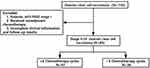 |
Figure 1 Flow chart of research object selection. |
Follow-up data were collected from August 2017, with a median follow-up of 19 months (range, 3–75 months). Data on the clinical and pathologic characteristics of study patients were collected, including age, preoperative serum cancer antigen 125 (CA-125) level, ascitis, residual tumor size, stage, lymph node (LN) excision metastasis, and CT cycles underwent after cytoreductive surgery. For evaluation of the surgical effect, the ideal reduction was a diameter of ≤1 cm for postoperative residual tumor (R1), whereas a residual tumor size >1 cm (R2) was a non-ideal reduction, and residual tumor size of zero (R0) was considered complete reduction. Progression-free survival (PFS) was defined in months from the time of diagnosis to that of tumor progression, patient death, or the last follow-up. The criteria for tumor progression was to meet at least one of the following conditions: elevation of tumor markers, imaging recurrence, clinical symptoms, and pathological evidence after reoperation. Overall survival (OS) was defined as the time from diagnosis to the patient’s death or the end of follow-up.
Since the NCCN guidelines (2019) stated that “There is no evidence confirming that more than 6 cycles of combination chemotherapy are required for initial chemotherapy”, the outcomes of patients who underwent >6 versus ≤6 cycles of CT were compared.
Statistical Analysis
Data were analyzed using SPSS version 18.0. Kaplan-Meier survival analyses were performed to estimate PFS and OS, and the Log rank test was employed to compare the differences in survival functions. All P-values <0.05 were regarded as statistically significant.
Results
All 85 patients with advanced OCCC between January 2012 and December 2017 were included in this analysis. Subgroup analyses of the relative risk of disease recurrence for patients receiving more than six versus less than six cycles of CT based on clinicopathological prognostic factors are shown in Table 1. The median age at diagnosis was 50 years. In 57 patients (67.1%), the level of CA125 was high before treatment, and 31 patients (36.5%) had ascites. There were 17 patients (20%) with stage II, 58 patients (68.2%) with stage III, and 10 patients (11.8%) with stage IV disease. All patients were treated with varying degrees of tumor cell destruction, with lymph node resection in 53 (62.4%) patients. Metastasis was observed in 19 (35.9%) patients. Forty (40) patients (47.1%) achieved complete tumor reduction, 26 patients (30.6%) achieved ideal tumor reduction, and 19 patients (22.4%) achieved non-ideal tumor reduction. The tumor histopathological type was pure in 76 cases (89.4%). All patients were treated with postoperative adjuvant platinum-based CT; of these, 38 patients (44.7%) underwent more than six cycles of CT. According to the number of CT cycles, we divided the cases into two groups: >6 cycle group and ≤6 cycle group. The two groups were compared for age, preoperative CA125 levels, FIGO stage, lymph node resection, metastasis, residual tumor, histopathological type, ascites, and other clinical features. The differences were not statistically significant (P > 0.05).
 |
Table 1 Subgroup Analyses of Relative Risk of Disease Recurrence for Patients Receiving More Than Six versus Less Than Six Cycles of CT Based on Clinicopathological Prognostic Factors (N= 85) |
The median follow-up time for the cases was 19 months (range, 3–75 months). Among the 85 patients, 49 had disease recurrence, and 35 died. Recurrence occurred in 31 of 35 patients who died, and in 18 of 50 patients who survived. According to the Kaplan-Meier survival curve, the 2-year PFS of the ≤6 cycle group and the >6 cycle group were estimated to be 51.5% and 42.2% (P>0.05), and OS was 59.7% and 64.5% (P>0.05), respectively. The effect of the number of CT cycles on PFS and OS was not statistically significant (Figures 2 and 3). The hazard ratio (HR) of disease recurrence in patients who underwent ≤6 versus >6 cycles of CT was 0.86 (95% CI: 0.49–1.52; p=0.605). The HRs of recurrence based on clinicopathological prognostic factors are shown in Table 1. Univariate analysis showed that there was no significant difference in recurrence risk between the two groups.
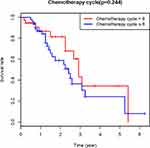 |
Figure 2 Effects of different chemotherapy cycles on PFS (P > 0.05). |
 |
Figure 3 Effects of different chemotherapy cycles on OS (P > 0.05). |
Multivariate analysis showed that residual tumor diameter was an independent risk factor for prognosis. In the three groups divided according to their residual tumor diameter (R0, R1 and R2), thorough tumor reduction (R0) significantly improved the prognosis, and the 2-year PFS and OS were significantly higher than those in the other two groups (Table 2). The prognosis of the group with residual tumor ≤1cm (R1) showed no significant difference compared with the >1cm group (R2).
 |
Table 2 Multivariate Survival Analysis of the Influence of Residual Tumor on Prognosis |
According to the subgroup analysis of residual tumor diameter, there were 40 cases in the R0 group, 26 cases in the R1 group, and 19 cases in the R2 group. Forty-seven (47) patients underwent ≤6 cycles of CT, and 38 patients underwent >6 cycles of CT. For the R0 group, the 2-year PFS of the ≤6 and >6 were 77.7% and 76.0%, respectively, and OS was 81.6% and 87.5%, respectively, with no significant difference in prognosis (P > 0.05). For the R1 group, the 2-year PFS of the ≤6 and >6 cycles groups were 26.3% and 13.9%, respectively, and OS was 41.0% and 55.6%, respectively, with no significant difference in prognosis (P > 0.05). For the R2 group, the 2-year PFS and OS were 33.3% and 0%, respectively, with no significant difference in prognosis (P > 0.05). In conclusion, more than 6 courses of CT did not improve the prognosis, regardless of residual tumor diameter. The results were presented in forest maps (Figures 4 and 5) using the R language.
 |
Figure 4 According to the diameter of residual tumor, the patients were divided into three subgroups, and the effects of chemotherapy cycle on OS were compared (P > 0.05). |
 |
Figure 5 According to the diameter of residual tumor, the patients were divided into three subgroups, and the effects of chemotherapy cycle on PFS were compared (P > 0.05). |
Discussion
The GOG18 (157) compared the prognosis between 3-cycle CT and 6-cycle CT in early high-risk ovarian cancer and concluded that 6-cycle chemotherapy could not significantly reduce the risk of recurrence, but increased toxic reactions (such as neurotoxicity, granulocytopenia, and anemia). Prendergast et al20 also proposed that the recurrence rate and survival rate of 3-cycle CT corresponded to 6-cycle CT. We speculated that early OCCC patients could benefit from up to 6 CT cycles, and the indications of CT should be closely monitored to reduce toxic reactions. Therefore, the subjects included in this study were mainly stage II–IV patients with advanced OCCC.
Current FIGO and NCCN guidelines recommend adjuvant chemotherapy for all stages of ovarian cancer. For advanced OCCC, the number of chemotherapy cycles is still controversial. In clinical practice, doctors perform over 6 cycles of CT for some patients according to their personal experience as well as the patients’ condition (ie elevated CA125 levels, persistent disease, etc). Bertelsen et al21 conducted three randomized trials comparing the survival benefits of 5–6, 8, 10, and 12 cycles of CT, respectively, showing that the optimal duration of first-line chemotherapy for advanced ovarian cancer was no more than 6 cycles. Therefore, it is generally believed that stage II–IV patients with ovarian cancer can get an objective remission after surgery with 6 cycles of CT. However, it is not clear whether it is suitable for OCCC patients with high platinum resistance. Pectasides et al22 showed that women with clear cell tumors have significantly lower response rates to platinum-based first-line CT compared to those with serous ovarian cancers. Similarly, Sugiyama et al14 compared patients with OCCC versus serous ovarian cancers, reporting that OCCC patients had a significantly lower response rate to CT (11.1%), versus serous ovarian cancer patients (72.5%).
The results of this study showed that, compared with the >6 cycle group, the 2-year overall survival rate of patients in the ≤6 cycle group, as well as the risk of recurrence, was not significantly different. Multivariate analyses also showed that the duration of chemotherapy was not an independent prognostic factor.
Many studies have shown that ideal reductive surgery can prolong the survival time of ovarian cancer patients,23,24 and compared with patients who have achieved the ideal reduction, the risk of death increased by 1.89 times for women who have not.25 However, this study showed that complete tumor reduction could significantly improve OS and PFS in patients with advanced OCCC. On the other hand, an ideal reduction did not improve OS and PFS compared with non-ideal reduction (Table 2). The reason for the significantly poor prognosis of patients without complete reduction surgery may be that patients in this group will relapse within a short period, combined with the cytotoxicity of chemotherapy drugs and the high resistance to platinum. Eventually, patients without complete reduction surgery develop progressive or refractory ovarian cancer, leading to a worse prognosis.
Patients with advanced OCCC often have a complex condition, and the primary staging operation cannot achieve complete tumor cell reduction for all patients. Therefore, it is not clear whether postoperative chemotherapy with different cycles can further improve the prognosis. Suidan et al23 evaluated the prognosis of epithelial ovarian cancer patients receiving CT 6 cycles after ideal tumor cell reduction in stage III/IV, finding no significant statistical difference between OS and PFS at 5 years after 1–2, 3–4, and 5–6 cycles of CT. The literature reported that the overall response rate of patients with OCCC to chemotherapeutic drugs was less than 50% and decreased with increasing stages.26,27 In this study, patients were divided into three groups based on the diameter of their postoperative residual tumor, and the effect of the chemotherapy cycle on prognosis was compared. The results showed no significant difference in PFS and OS between the ≤6-cycle and >6-cycle groups. Moreover, prognosis in patients with advanced OCCC did not improve after >6 cycles of chemotherapy, regardless of whether they underwent complete tumor reduction.
The present study is retrospective in nature. Due to many limitations, inherent selection bias and recall bias may affect the accuracy of the results. Large-scale clinical studies are needed in the future to further clarify the clinicopathological characteristics of patients with advanced OCCC to guide clinical treatment and to predict the survival outcome in such patients. However, due to the low incidence of OCCC, the implementation of large-scale prospective clinical trials is very challenging.
Conclusion
In summary, for patients with advanced OCCC, the diameter of postoperative residual tumor is an independent factor affecting prognosis. Gynecologic oncologists should improve their surgical skills as much as possible because the initial staging of ovarian cancer significantly affects the prognosis. However, regardless of whether the initial staging operation is complete, the appropriate cycle of postoperative adjuvant chemotherapy should be carefully selected. More than six cycles of chemotherapy do not improve prognosis but may increase toxicity and drug resistance.
Funding
This work was supported by the National Natural Science Foundation of China (No. 8197103302).
Disclosure
The authors report no competing financial interests or conflicts of interest in this work.
References
1. Machida H, Matsuo K, Yamagami W, et al. Trends and characteristics of epithelial ovarian cancer in Japan between 2002 and 2015: a JSGO-JSOG joint study. Gynecol Oncol. 2019;153:589–596. doi:10.1016/j.ygyno.2019.03.243
2. Kim SI, Lee JW, Lee M, et al. Genomic landscape of ovarian clear cell carcinoma via whole exome sequencing. Gynecol Oncol. 2018;148:375–382. doi:10.1016/j.ygyno.2017.12.005
3. Sugiyama T, Okamoto A, Enomoto T, et al. Randomized Phase III trial of irinotecan plus cisplatin compared with paclitaxel plus carboplatin as first-line chemotherapy for ovarian clear cell carcinoma: JGOG3017/GCIG trial. J Clin Oncol. 2016;34:2881–2887. doi:10.1200/JCO.2016.66.9010
4. Schnack TH, Høgdall E, Nedergaard L, Høgdall C. Demographic clinical and prognostic factors of primary ovarian adenocarcinomas of serous and clear cell histology-a comparative study. Int J Gynecol Cancer. 2016;26:82–90. doi:10.1097/IGC.0000000000000585
5. Köbel M, Kalloger SE, Huntsman DG, et al. Differences in tumor type in low-stage versus high-stage ovarian carcinomas. Int J Gynecol Pathol. 2010;29:203–211. doi:10.1097/PGP.0b013e3181c042b6
6. Anglesio MS, Carey MS, Köbel M, Mackay H, Huntsman DG. Vancouver ovarian clear cell symposium speakers. clear cell carcinoma of the ovary: a report from the first ovarian clear cell symposium, June 24th, 2010. Gynecol Oncol. 2011;121:407–415. doi:10.1016/j.ygyno.2011.01.005
7. Wang S, Qiu L, Lang JH, et al. Clinical analysis of ovarian epithelial carcinoma with coexisting pelvic endometriosis. Am J Obstet Gynecol. 2013;208:
8. Kobayashi H, Sumimoto K, Moniwa N, et al. Risk of developing ovarian cancer among women with ovarian endometrioma: a cohort study in Shizuoka, Japan. Int J Gynecol Cancer. 2007;17:37–43. doi:10.1111/j.1525-1438.2006.00754.x
9. Fujiwara K, Shintani D, Nishikawa T. Clear-cell carcinoma of the ovary. Ann Oncol. 2016;27(Suppl 1):i50–50i52. doi:10.1093/annonc/mdw086
10. Kaur S, Kerkar RA, Maheshwari A, Shylasree TS, Gupta S, Deodhar K. Clinical characteristics with patterns of relapse and survival analysis of ovarian clear cell carcinoma. Indian J Cancer. 2016;53:288–291. doi:10.4103/0019-509X.197719
11. Okamoto A, Sugiyama T, Hamano T, et al. Randomized phase III trial of paclitaxel/carboplatin (PC) versus cisplatin/irinotecan (CPT-P) as first-line chemotherapy in patients with clear cell carcinoma (CCC) of the ovary: a Japanese gynecologic oncology group (JGOG)/GCIG study. J Clin Oncol. 2014;32(suppl 5):5507. doi:10.1200/jco.2014.32.15_suppl.5507
12. Ozols RF, Bundy BN, Greer BE, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a gynecologic oncology group study. J Clin Oncol. 2003;21:3194–3200. doi:10.1200/JCO.2003.02.153
13. Itamochi H, Kigawa J, Terakawa N. Mechanisms of chemoresistance and poor prognosis in ovarian clear cell carcinoma. Cancer Sci. 2008;99:653–658. doi:10.1111/j.1349-7006.2008.00747.x
14. Sugiyama T, Kamura T, Kigawa J, et al. Clinical characteristics of clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer. 2000;88:2584–2589. doi:10.1002/1097-0142(20000601)88:11<2584::AID-CNCR22>3.0.CO;2-5
15. Oliver KE, Brady WE, Birrer M, et al. An evaluation of progression free survival and overall survival of ovarian cancer patients with clear cell carcinoma versus serous carcinoma treated with platinum therapy: an NRG oncology/gynecologic oncology group experience. Gynecol Oncol. 2017;147:243–249. doi:10.1016/j.ygyno.2017.08.004
16. Al-Barrak J, Santos JL, Tinker A, et al. Exploring palliative treatment outcomes in women with advanced or recurrent ovarian clear cell carcinoma. Gynecol Oncol. 2011;122:107–110. doi:10.1016/j.ygyno.2011.03.011
17. Pignata S, Scambia G, Ferrandina G, et al. Carboplatin plus paclitaxel versus carboplatin plus pegylated liposomal doxorubicin as first-line treatment for patients with ovarian cancer: the MITO-2 randomized phase III trial. J Clin Oncol. 2011;29:3628–3635. doi:10.1200/JCO.2010.33.8566
18. Bell J, Brady MF, Young RC, et al. Randomized phase III trial of three versus six cycles of adjuvant carboplatin and paclitaxel in early stage epithelial ovarian carcinoma: a gynecologic oncology group study. Gynecol Oncol. 2006;102:432–439. doi:10.1016/j.ygyno.2006.06.013
19. Bookman MA, Brady MF, McGuire WP, et al. Evaluation of new platinum-based treatment regimens in advanced-stage ovarian cancer: a Phase III trial of the gynecologic cancer intergroup. J Clin Oncol. 2009;27:1419–1425. doi:10.1200/JCO.2008.19.1684
20. Prendergast EN, Holzapfel M, Mueller JJ, et al. Three versus six cycles of adjuvant platinum-based chemotherapy in early stage clear cell ovarian carcinoma - a multi-institutional cohort. Gynecol Oncol. 2017;144:274–278. doi:10.1016/j.ygyno.2016.12.004
21. Bertelsen K, Grenman S, Rustin GJ. How long should first-line chemotherapy continue. Ann Oncol. 1999;10(Suppl 1):17–20. doi:10.1016/S0923-7534(20)31479-4
22. Pectasides D, Fountzilas G, Aravantinos G, et al. Advanced stage clear-cell epithelial ovarian cancer: the Hellenic Cooperative Oncology Group experience. Gynecol Oncol. 2006;102:285–291. doi:10.1016/j.ygyno.2005.12.038
23. Suidan RS, Zhou Q, Iasonos A, et al. Prognostic significance of the number of postoperative intraperitoneal chemotherapy cycles for patients with advanced epithelial ovarian cancer. Int J Gynecol Cancer. 2015;25:599–606. doi:10.1097/IGC.0000000000000389
24. Winter WE
25. Tewari D, Java JJ, Salani R, et al. Long-term survival advantage and prognostic factors associated with intraperitoneal chemotherapy treatment in advanced ovarian cancer: a gynecologic oncology group study. J Clin Oncol. 2015;33:1460–1466. doi:10.1200/JCO.2014.55.9898
26. Takano M, Sugiyama T, Yaegashi N, et al. Less impact of adjuvant chemotherapy for stage I clear cell carcinoma of the ovary: a retrospective Japan Clear Cell Carcinoma Study. Int J Gynecol Cancer. 2010;20:1506–1510.
27. Utsunomiya H, Akahira J, Tanno S, et al. Paclitaxel-platinum combination chemotherapy for advanced or recurrent ovarian clear cell adenocarcinoma: a multicenter trial. Int J Gynecol Cancer. 2006;16:52–56. doi:10.1111/j.1525-1438.2006.00289.x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
