Back to Journals » Journal of Pain Research » Volume 8
Is chronic post-herniorrhaphy pain always chronic? A literature review
Authors Sandblom G
Received 12 February 2015
Accepted for publication 5 March 2015
Published 22 May 2015 Volume 2015:8 Pages 241—245
DOI https://doi.org/10.2147/JPR.S82708
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Michael Schatman
Gabriel Sandblom
Department of Clinical Science, Intervention and Technology, Karolinska Institute, Huddinge, Sweden
Introduction: Chronic post-surgery pain (CPSP) has gained increased recognition as a major factor influencing health-related quality-of-life following most surgical procedures, in particular following surgery for benign conditions. The natural course of CPSP, however, is not well-known.
Methods: A literature review was undertaken, searching for studies with repeated estimates of post-herniorrhaphy pain. The hypothetical halvation time was calculated from the repeat estimates.
Results: Eight studies fulfilling the criteria were identified. With one exception, the extrapolated halvation times ranged from 1.3 to 9.2 years.
Discussion: Even if CPSP is generally very treatment-resistant, in many cases it eventually dissipates with time. Further studies are required to evaluate the prevalence of pain beyond the first decade.
Keywords: hernia, pain, chronic post-surgery pain, neuropathic pain, halvation time
Background
The trauma inflicted by a surgical intervention sometimes results in persistent pain. In contrast to acute postoperative pain, which is a physiological reaction to tissue trauma, chronic post-surgery pain (CPSP) persists beyond the usual course of natural healing. Pain persisting at the sight of surgery beyond the usual course of natural postoperative pain usually differs in character from that suffered during the first weeks after an operation. It may be burning, tingling, or electric shock-like, and may be associated with paresthesia, hyperalgesia or allodynia. It is often neuropathic in nature,1,2 but may also be caused by chronic inflammation, tension or local compression from a dislocated mesh (“meshoma”).3
CPSP is a condition that has gained increased recognition in recent years.4 Several reports have focused on chronic pain, in particular after hernia repair.5–7 Since the main indication for performing a hernia repair is discomfort or pain from the hernia, the risk of developing iatrogenic pain is something that must be taken into account when deciding on a procedure that could cause more harm than good. Studies on CPSP after hernia surgery have reported prevalence ranging from 0% to 54%.8–11 There are probably several explanations for this spread such as the lack of a uniformly accepted instrument for the assessment of pain.12 Another factor that may have had a large influence on outcome is the timing of follow-up. Pain persisting longer than 3 months is often considered a criterion for CPSP. In most cases, however, pain gradually decreases with time.8,13
Measurement of pain prevalence with adequate external validity requires standardized patient-related outcome measures. Standardized tools for assessment of postoperative pain include the Brief Pain Inventory14 and Short Form McGill Pain Questionnaire.15 There are also instruments specifically aimed at assessing post-herniorrhaphy pain, including the Carolina Comfort Scale16 and Inguinal Pain Questionnaire.17
In most cases, CPSP is not constant. With time, it may progress as well as regress.18 Fortunately, however, there is a tendency to decrease with time.8,13 There are few studies that have explored the natural course of CPSP. An understanding of the natural course can, however, be achieved by considering the prevalence of pain at two different points in time following surgery, and in the same population. By assuming an exponential course, the halvation time can be determined using the following formula:
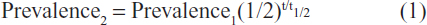
The formula defines the estimates, ie, the prevalence of pain, at two separate occasions (prevalence1 and prevalence2) at a certain time interval (t). Assuming that the course is exponential during the whole period of observation, the halvation time (t1/2) can be determined from prevalence1, prevalence2, and t. Although a complex and multifactorial process such as the natural course of postoperative pain cannot be represented by a mathematical model, assuming an exponential course may help to compare outcomes from several studies and provide a basis for exploring factors that could influence the duration of postoperative pain.
Literature review
A PubMed search from January 1980 to June 2014 was carried out with the search terms “Postherniorrhaphy pain” or “Long-term post-surgery pain and Hernia” or “Chronic postoperative pain and Hernia”. Inclusion criteria were studies including assessment of persisting pain on at least two defined occasions postoperatively at defined intervals, the second occasion being at least 1 year after surgery. Pain estimates within 3 months after surgery were not included in the present review since pain present during the first postoperative months is usually not considered as chronic and may be related to other factors than those that cause pain of longer persistence. Studies describing the same study group in two separate publications were also included. In the case of randomized controlled trials, the randomization arms were considered as separate groups. The following studies were excluded:
- studies with only one estimate of pain prevalence;
- studies with poorly or undefined follow-up intervals;
- studies with incomplete follow-up;
- studies not including pain as endpoint or defining pain in a way that made it impossible to dichotomize it.
Outcome
Altogether 889 publications fulfilling the criteria were identified. After reviewing all abstracts and the full papers of all publications suggesting a follow-up of a previously reported study, eight study groups presented in eleven publications with estimates of pain on at least two occasions were identified (Table 1). The table shows the two estimates, starting from the first time when at least 3 months had elapsed since surgery. With the exception of one of the arms in one study,28 the extrapolated t1/2s ranged from 2.9 to 9.2 years.
  | Table 1 Studies with repeated estimates of pain prevalence following groin hernia surgery |
Discussion
All studies reviewed found a gradual decrease in pain with time. Even if t1/2 is a very simplified way of describing the course of CPSP, it may help to understand the slow but gradual healing process.
None of the studies included in the present review had a follow-up time exceeding 10 years. It cannot, therefore, be excluded that the gradual decrease seen during the first 10 years ceases later on. Since it is only in the last 10 years that the problem of CPSP has gained focus, it is difficult to identify patient cohorts dating further back in time. Nevertheless, there are no obvious reasons to believe that the natural course of CPSP has changed over history.
The natural course of persisting pain should be considered before advocating aggressive measures to deal with it, such as a reoperation. It cannot be ruled out that the favorable long-term outcome after reoperation is, at least partly, regression to the mean phenomenon. Even without re-intervention, time itself would have led to improvement.19
An exponential model is only fully adequate if reduction in pain is constant over time. Needless to say, there are several factors that do not fit into such a model. The course of post-herniorrhaphy pain is probably dependent on a number of factors that vary with time, including nerve entrapment, inflammatory reaction, nerve regeneration, psychological assimilation, and decreasing demands on physical performance with age. The pathogenesis of CPSP varies and depends on the intervention. Nerve lesions play a major role in the development of persistent pain following hernia repair.1,2 The ilioinguinal, iliohypgastric, and genital branch of the genitofemoral nerve are the nerves most at risk for being damaged. Preoperative pain,6 inflammatory reaction, and genetic susceptibility,20 may also contribute. Neuropathic pain seems to be more persistent,21 whereas nociceptive pain and pain caused by mechanical compression or tension may gradually decrease with time as a result of tissue rearrangement. Psychological adaptation and coping may also influence the long-term course. Whatever, it must be a solace for these patients to know that the pain is likely to gradually decrease with time.
Since the main indication for hernia surgery is to reduce pain, great care in decision-making should be taken in order not to cause iatrogenic pain following a procedure undertaken on vague indications. Patients who are at risk for developing CPSP (young patients,7 women,7 patients perceiving intense preoperative pain,1 patients with tendency to develop anxiety and depression,6 and patients with previous history of high sensibility to nociceptive pain) should be identified before the decision to perform surgery is taken. Even if some of the symptoms may resolve with time, great care should be taken not to cause iatrogenic pain that may persist for many years.
Disclosure
The author has no conflicts of interest to declare.
References
Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. 2006;367(9522):1618–1625. | |
Magnusson N, Hedberg M, österberg J, Sandblom G. Sensory disturbances and neuropathic pain after inguinal hernia surgery. Scand J Pain. 2010;1:108–111. | |
Amid PK. Radiologic images of meshoma: a new phenomenon causing chronic pain after prosthetic repair of abdominal wall hernias. Arch Surg. 2004;139(12):1297–1298. | |
Alfieri S, Amid PK, Campanelli G, et al. International guidelines for prevention and management of post-operative chronic pain following inguinal hernia surgery. Hernia. 2011;15(3):239–249. | |
Dahlstrand U, Sandblom G, Nordin P, Wollert S, Gunnarsson U. Chronic pain after femoral hernia repair: a cross-sectional study. Ann Surg. 2011;254(6):1017–1021. | |
Kalliomäki ML, Meyerson J, Gunnarsson U, Gordh T, Sandblom G. Long-term pain after inguinal hernia repair in a population-based cohort; risk factors and interference with daily activities. Eur J Pain. 2008;12(2):214–225. | |
Fränneby U, Sandblom G, Nordin P, Nyrén O, Gunnarsson U. Risk factors for long-term pain after hernia surgery. Ann Surg. 2006;244(2):212–219. | |
van Veen RN, Wijsmuller AR, Vrijland WW, Hop WC, Lange JF, Jeekel J. Randomized clinical trial of mesh versus non-mesh primary inguinal hernia repair: long-term chronic pain at 10 years. Surgery. 2007;142(5):695–698. | |
Nienhuijs S, Staal E, Strobbe L, Rosman C, Groenewoud H, Bleichrodt R. Chronic pain after mesh repair of inguinal hernia: a systematic review. Am J Surg. 2007;194(3):394–400. | |
Aasvang EK, Bay-Nielsen M, Kehlet H. Pain and functional impairment 6 years after inguinal herniorrhaphy. Hernia. 2006;10(4):316–321. | |
Poobalan AS, Bruce J, Smith WC, King PM, Krukowski ZH, Chambers WA. A review of chronic pain after inguinal herniorrhaphy. Clin J Pain. 2003;19(1):48–54. | |
VanDenKerkhof EG, Peters ML, Bruce J. Chronic pain after surgery: time for standardization? A framework to establish core risk factor and outcome domains for epidemiological studies. Clin J Pain. 2013;29(1):2–8. | |
Sandblom G, Kalliomäli M L, Gordh T, Gunnarsson U. Natural course of persistent pain after groin hernia surgery. Scand J Pain. 2010;1:55–59. | |
Cleeland CS. Pain assessment in cancer. In: Osoba D, editor. Effect of Cancer on Quality of Life. Boca Raton: CRC Press, Inc.; 1991:293–305. | |
Melzack R. The short-form McGill Pain Questionnaire. Pain. 1987; 30(2):191–197. | |
Zaborszky A, Gyanti R, Barry JA, Saxby BK, Bhattacharya P, Hasan FA. Measurement issues when assessing quality of life outcomes for different types of hernia mesh repair. Ann R Coll Surg Engl. 2011;93(4):281–285. | |
Fränneby U, Gunnarsson U, Andersson M, et al. Validation of an Inguinal Pain Questionnaire for assessment of chronic pain after groin hernia repair. Br J Surg. 2008;95(4):488–493. | |
Galer BS, Henderson J, Perander J, Jensen MP. Course of symptoms and quality of life measurement in Complex Regional Pain Syndrome: a pilot survey. J Pain Symptom Manage. 2000;20(4):286–292. | |
Valvekens E, Nijs Y, Miserez M. Long-term outcome of surgical treatment of chronic postoperative groin pain: a word of caution. Hernia. Epub June 19, 2013. | |
Dominguez CA, Kalliomäki A, Gunnarsson U, et al. The DRB1*04- DQB1*03:02 HLA haplotype is associated with increased risk of chronic pain after inguinal hernia surgery. Pain. 2013;154(3):427–433. | |
Schwartzman RJ, Erwin KL, Alexander GM. The natural history of complex regional pain syndrome. Clin J Pain. 2009;25(4):273–280. | |
Bay-Nielsen M, Perkins FM, Kehlet H; Danish Hernia Database. Pain and functional impairment 1 year after inguinal herniorrhaphy: a nationwide questionnaire study. Ann Surg. 2001;233(1):1–7. | |
Eklund A, Rudberg C, Smedberg S, et al. Short-term results of a randomized clinical trial comparing Lichtenstein open repair with totally extraperitoneal laparoscopic inguinal hernia repair. Br J Surg. 2006; 93(9):1060–1068. | |
Eklund A, Montgomery A, Bergkvist L, Rudberg C; Swedish Multicentre Trial of Inguinal Hernia Repair by Laparoscopy (SMIL) study group. Chronic pain 5 years after randomized comparison of laparoscopic and Lichtenstein inguinal hernia repair. Br J Surg. 2010;97(4):600–608. | |
Grant AM, Scott NW, O’Dwyer PJ; MRC Laparoscopic Groin Hernia Trial Group. Five-year follow-up of a randomized trial to assess pain and numbness after laparoscopic or open repair of groin hernia. Br J Surg. 2004;91(12):1570–1574. | |
Andersson B, Hallén M, Leveau P, Bergenfelz A, Westerdahl J. Laparoscopic extraperitoneal inguinal hernia repair versus open mesh repair: a prospective randomized controlled trial. Surgery. 2003;133(5):464–472. | |
Hallén M, Bergenfelz A, Westerdahl J. Laparoscopic extraperitoneal inguinal hernia repair versus open mesh repair: long-term follow-up of a randomized controlled trial. Surgery. 2008;143(3):313–317. | |
Eker HH, Langeveld HR, Klitsie PJ, et al. Randomized clinical trial of total extraperitoneal inguinal hernioplasty vs Lichtenstein repair: a long-term follow-up study. Arch Surg. 2012;147(3):256–260. | |
Polish Hernia Study Group, Smietanski M. Randomized clinical trial comparing a polypropylene with a poliglecaprone and polypropylene composite mesh for inguinal hernioplasty. Br J Surg. 2008;95(12):1462–1468. | |
Bury K, Śmietański M; Polish Hernia Study Group. Five-year results of a randomized clinical trial comparing a polypropylene mesh with a poliglecaprone and polypropylene composite mesh for inguinal hernioplasty. Hernia. 2012;16(5):549–553. | |
Nikkolo C, Lepner U, Murruste M, Vaasna T, Seepter H, Tikk T. Randomized clinical trial comparing lightweight mesh with heavyweight mesh for inguinal hernioplasty. Hernia. 2010;14(3):253–258. | |
Nikkolo C, Murruste M, Vaasna T, Seepter H, Tikk T, Lepner U. Three-year results of randomized clinical trial comparing lightweight mesh with heavyweight mesh for inguinal hernioplasty. Hernia. 2012;16(5):555–559. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
