Back to Journals » Journal of Pain Research » Volume 12
Intraoperative administration of intravenous flurbiprofen axetil with nalbuphine reduces postoperative pain after orbital decompression: a single-center, prospective randomized controlled trial
Authors Ye H , Lian X, Chen R , Zhu Y , Chen H, Huang J, Xie L, Ma W, Yang H, Guo W
Received 16 September 2018
Accepted for publication 17 January 2019
Published 14 February 2019 Volume 2019:12 Pages 659—665
DOI https://doi.org/10.2147/JPR.S187020
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Michael A Ueberall
Huijing Ye,* Xiufen Lian,* Rongxin Chen, Yanling Zhu, Hongbin Chen, Jingxia Huang, Ling Xie, Wenfang Ma, Huasheng Yang, Wenjun Guo
State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou 510060, China
*These authors contributed equally to this work
Purpose: This study aimed to investigate postoperative analgesia achieved with intraoperative administration of intravenous flurbiprofen axetil and nalbuphine in patients undergoing orbital decompression.
Methods: Sixty-three patients undergoing orbital decompression under general anesthesia at the Zhongshan Ophthalmic Center at Sun Yat-sen University (Guangzhou, China) were randomly allocated into one of the following three groups (1:1:1): intraoperative flurbiprofen axetil 100 mg (Group 1); intraoperative nalbuphine 0.1 mg/kg (Group 2); or intraoperative flurbiprofen axetil 100 mg combined with nalbuphine 0.1 mg/kg (Group 3). The primary end point was mean postoperative pain intensity during the first 24 hours. The secondary efficacy end points were the intensity of pain and discomfort at 0, 2, 6, 10, and 24 hours after surgery and side effects at 24 hours after surgery.
Results: The demographic characteristics were similar among the three groups. Mean and peak postoperative pain scores during the first 24 hours in Group 3 were lower than those in Group 1 (P=0.007 and P=0.003, respectively) and Group 2 (P=0.001 and P=0.000, respectively). Additionally, the pain scores in Group 3 were significantly lower than those in Group 1 during the first 6 hours after surgery (P=0.003, 0.002, and 0.022 at 0, 2, and 6 hours, respectively) and those in Group 2 during the first 10 hours after surgery (P=0.008, 0.000, 0.001, and 0.019 at 0, 2, 6, and 10 hours, respectively). Discomfort scores were not significantly different among the three groups during the observation period, except at 2 hours after surgery, at which time the scores in Group 3 were significantly lower than those in Group 2 (P=0.033). Postoperative adverse effects and analgesic requirements were similar among the three groups.
Conclusion: Intraoperative administration of a combination of intravenous flurbiprofen axetil and nalbuphine is superior to single-dose flurbiprofen axetil or nalbuphine in patients undergoing orbital decompression.
Keywords: postoperative pain, flurbiprofen axetil, nalbuphine, orbital decompression
Introduction
Postoperative pain may result in patient discomfort and decrease patient satisfaction.1 Bone removal orbital decompression, being a complex ophthalmic surgery, can lead to severe pain, with significantly higher postoperative pain scores than other oculoplastic surgeries.2,3 Postoperative pain is a major problem following orbital decompression.
NSAIDs have been shown to effectively decrease postoperative pain.4 Flurbiprofen axetil is an injectable nonselective cyclooxygenase (COX) inhibitor that is mainly metabolized into flurbiprofen by hydrolysis; its analgesic effect is believed to result from the reversible inhibition of COX and the peripheral inhibition of prostaglandin synthesis. Moreover, the drug has shown no irreversible carcinogenic, teratogenic, or hepatotoxic effects. However, in our recent study,2 45.7% patients experienced significant postoperative pain with preoperative administration of intravenous flurbiprofen axetil.
Overall, opioids are important analgesic agents for postoperative pain management. Nalbuphine, a κ-receptor agonist and μ-receptor antagonist, is a semisynthetic opioid analgesic that belongs to the phenanthrene family. This drug may provide effective pain relief with fewer opioid-induced adverse effects than other opioids.5 Unfortunately, nalbuphine offers a duration of analgesia of only 4–5 hours, which is too short for postoperative pain management.6
It is clear that single-dose flurbiprofen axetil or nalbuphine is not effective enough for postoperative pain control following orbital decompression, and there is convincing evidence that multimodal analgesia is the best choice for pain management.7 Although little attention has been given to the postoperative analgesic efficacy of these drugs in patients undergoing orbital decompression, this combination may provide effective pain relief, reduce adverse effects, and increase patient acceptance and compliance with therapy.
In this randomized controlled clinical trial, we evaluated the postoperative analgesic efficacy and adverse effects of flurbiprofen axetil combined with nalbuphine in patients undergoing orbital decompression compared with those on a single dose of flurbiprofen axetil or nalbuphine.
Methods
Study design
This was a single-center, prospective, randomized, controlled, study (ClinicalTrials.gov identifier: NCT 03562611). The trial included 63 consecutive patients who underwent orbital decompression between June 8, 2018, and September 8, 2018, at the Zhongshan Ophthalmic Center at Sun Yat-sen University (Guangzhou, China). Approval was obtained in accordance with the Declaration of Helsinki and the Ethics Committee of the Zhongshan Ophthalmic Center at Sun Yat-sen University. Written informed consent was obtained from each patient.
Study visits were scheduled on Day 1 (Visit 1/baseline), Day 2 (Visit 2, day of surgery), and Day 3 (Visit 3, 24 hours after surgery).
Participants
Participants were screened before the trial by an experienced doctor, and 65 subjects agreed to participate in this study. The following inclusion criteria were applied: 1) bone removal orbital decompression because of thyroid eye disease; 2) age between 16 and 75 years; and 3) American Society of Anesthesiologists (ASA) physical status of I-II.
The exclusion criteria were as follows: 1) serious coexisting disease; 2) body mass index (BMI) <18.5 or >35; 3) contraindications or previous adverse reactions to any of the drugs used; 4) pregnancy; and 5) patients unable to cooperate.
Study protocol
At the baseline visit (Day 1), patients were randomly divided into one of three groups (1:1:1) by codes that were generated by a computer random number generator: Group 1, flurbiprofen axetil; Group 2, nalbuphine; and Group 3, flurbiprofen axetil combined with nalbuphine. The patients also completed the Self-rating Anxiety Scale (SAS) on Day 1. Both the anesthesiologists managing the postoperative courses and the patients were blinded to the treatment assignment for the duration of the study.
On Day 2 (day of surgery), general anesthesia was administered with induction via propofol (2 mg/kg) and fentanyl (2 µg/kg) for all patients, and anesthesia was maintained with propofol (6 mg/kg/h) and remifentanil (0.15 µg/kg/min). Group 1 received 100 mg of intravenous flurbiprofen axetil (Beijing Tide Pharmaceutical Co, Beijing, China) when the surgery was completed, and Group 2 received 0.1 mg/kg of intravenous nalbuphine (Yichang Humanwell Pharmaceutical Co, Hubei, China) after induction of anesthesia; Group 3 received both. Pain scores, discomfort scores, and analgesic medication use were recorded after surgery. Pain and discomfort levels were measured using a numerical rating scale (NRS) ranging from zero to ten, where zero represented no pain (or discomfort) and ten represented the worst pain (or discomfort) imaginable; this scale has been confirmed to be sensitive and reliable.8–11 Discomfort was defined as “an uncomfortable feeling of mental painfulness or distress”. Postoperative pain and discomfort intensity levels were measured at 0, 2, 6, 10, and 24 hours after recovery from general anesthesia. The patients were not restricted with respect to the receipt of any additional analgesic medication required. The most commonly used medication was 0.5 g of oral paracetamol.
Postoperative events, such as nausea, vomiting, dizziness, pruritus, respiratory depression, shivering, or hypotension, were reported for 24 hours (Day 3).
Statistical analyses
The primary outcome was the mean NRS score for pain for the first postoperative 24 hours (the average of 0, 2, 6, 10, and 24 hours). Secondary outcomes included the following: 1) NRS scores for pain and discomfort at 0, 2, 6, 10, and 24 hours postoperatively; 2) peak NRS scores for pain and discomfort during the first 24 hours postoperatively; and 3) side effects (nausea, vomiting, dizziness, respiratory depression, shivering, and hypotension) during the first 24 hours postoperatively.
The sample size was calculated after data were collected from 15 patients undergoing orbital decompression with the anesthetic technique described herein using flurbiprofen axetil, nalbuphine, or both. Mean NRS scores for pain during the first postoperative 24 hours were 3.53, 3.90, and 1.70 for Groups 1, 2, and 3, respectively, with an SD of 2.37. To maintain the power of the study at 80%, with a 5% alpha error and a sample size ratio of 1.0 between the three groups, a total sample size of 63 participants was needed, as determined using the SAS software (SAS Institute Inc, Cary, NC, USA). Thus, each group required at least 21 patients. The study was not powered for secondary end points.
All of the data were analyzed using SPSS for Windows (version 21.0, IBM Corporation, Armonk, NY, USA). ANOVA with Bonferroni’s or Tamhane’s test for multiple comparisons was used to evaluate differences in pain scores, discomfort scores, SAS scores, as well as demographic and perioperative data. Frequencies were calculated for qualitative variables, such as sex and ASA physical status in the three groups, using the chi-squared test or Fisher’s exact test. The level of significance was set at P<0.05.
Results
Participant flow
Figure 1 depicts the CONSORT flow of participants through the trial. Sixty-five individuals were assessed for eligibility for inclusion in the study, and two were disqualified based on the exclusion criteria. A total of 63 patients fulfilling the inclusion criteria and not meeting the exclusion criteria were included in the study. The median age was 47.8 (range: 26.0–69.0) years. There were 33 male and 30 female patients. The three groups were similar in terms of age, height, weight, BMI, sex, ASA physical status, SAS score, smoking history, and prior surgery on the operative eye (Table 1).
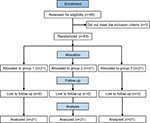  | Figure 1 Flowchart of patients in the study. Note: Group 1, flurbiprofen axetil; Group 2, nalbuphine; and Group 3, flurbiprofen axetil with nalbuphine. |
Postoperative pain and discomfort scores
Pain and discomfort scores were noted at 0, 2, 6, 10, and 24 hours (Table 2). There was a significant difference in the mean NRS pain score among the three groups during the first 24 hours (the average of 0, 2, 6, 10, and 24 hours) and at 0, 2, 6, and 10 hours postoperatively (P=0.001, 0.002, 0.023, 0.001, and 0.020) but not at 24 hours postoperatively (P=0.083). The mean NRS pain score in Group 3 for the first 24 hours was significantly lower than that in Groups 1 and 2 (P=0.007 and 0.001, respectively). Figure 2 shows that the Group 3 NRS pain scores were significantly lower than those in Group 1 during the first 6 hours after surgery (P=0.003, 0.002, and 0.022, respectively, at 0, 2, and 6 hours, respectively) and in Group 2 during the first 10 hours after surgery (P=0.008, 0.000, 0.001, and 0.019, at 0, 2, 6, and 10 hours, respectively). However, the NRS discomfort scores were always lower in Group 3 than in the other two groups but were not significantly different among the three groups during the observation period, except at 2 hours, at which time the NRS discomfort scores in Group 3 were significantly lower than those in Group 2 (P=0.033) (Figure 3).
The mean peak pain intensity scores for Groups 1 and 2 were 5.00 and 5.05, respectively, which were significantly greater than those in Group 3, with a mean of 2.14 (P=0.003 and 0.000) (Table 3). The mean time to reach the peak pain intensity was longer in Group 2 than in Groups 1 and 3 but was not significantly different among the three groups during the observation period. Overall, the peak NRS discomfort score and mean time to reach the peak discomfort intensity were not significantly different among the three groups during the observation period.
Postoperative adverse effects and analgesic requirements
The incidences of postoperative nausea and vomiting (PONV) and dizziness were not significantly different among the three groups (P=0.291 and 0.219, Table 1). No patient in this study experienced pruritus, respiratory depression, shivering, or hypotension.
Six patients (9.5%) requested an analgesic agent at an average of 1.5 (0.5–3) hours after surgery, and all received 0.5 g of oral paracetamol and experienced effective pain relief within 1 hour. The incidence of patients requiring rescue analgesics was not significantly different among the three groups (Table 1).
Discussion
The purpose of this study was to evaluate the effectiveness of postoperative analgesia after intraoperative administration of intravenous flurbiprofen axetil and nalbuphine in patients undergoing orbital decompression. Compared with other surgical disciplines, ophthalmic surgery is occasionally considered to cause severe postoperative pain with limited trauma,12 and there are accordingly only a few published studies on this topic.2 However, because bone removal ophthalmic surgery is a complex procedure, 45.7% patients undergoing orbital decompression experience significant postoperative pain even with preoperative administration of intravenous flurbiprofen axetil. Postoperative pain has been associated with smoking history, prior surgery on the operative eye, and anxiety,2 which were balanced in our study groups. In general, pain management following orbital decompression is challenging.
Multimodal analgesia is defined as combining analgesics with different mechanisms of action within the peripheral and central nervous system to maximize analgesic efficacy and minimize side effects;13 indeed, this is the best choice for pain management.7 A combination of opioids and NSAIDs is associated with greater patient satisfaction and fewer opioid-induced adverse effects than with single-agent analgesia.14 For example, flurbiprofen combined with nalbuphine is superior for patients undergoing open gastrointestinal surgery with efficient postoperative analgesia and decreases the severity of PONV.13 However, the postoperative analgesic efficacy of these drugs in patients undergoing orbital decompression is unknown. Our aim was to prospectively gather valid data on the intraoperative administration of intravenous nalbuphine and flurbiprofen axetil in patients undergoing orbital decompression.
In this study, patients who received a combination of flurbiprofen axetil and nalbuphine had lower pain scores than did those in the single-drug groups (flurbiprofen axetil or nalbuphine). The mean NRS pain score in the combined group for the first 24 hours was significantly lower than that in the single-drug groups (P=0.007 and 0.001, respectively). Furthermore, NRS pain scores in the combined group were also significantly lower than those in the flurbiprofen axetil group during the first 6 hours after surgery (P=0.003, 0.002, and 0.022) and in the nalbuphine group during the first 10 hours after surgery (P=0.008, 0.000, 0.001, and 0.019 at 0, 2, 6, and 10 hours, respectively). Similarly, the mean peak pain intensity score in the combined group was significantly lower than that in the single-drug groups (P=0.003 and 0.000). In the guidelines for the management of postoperative pain, it is strongly recommended that NSAIDs be used in conjunction with opioids because the use of this combination is associated with less postoperative pain and opioid consumption than with the use of opioids alone.7
Unlike the pain scores in the combined group and the flurbiprofen axetil group, which decreased with time, the pain intensity in the nalbuphine group peaked at 1.52 hours and not at 0 hour after surgery. This result may be because of the short duration of nalbuphine analgesia, which lasts ~4–5 hours and is too short for postoperative pain management.6 In contrast, the half-life of flurbiprofen axetil is 6 hours, offering a duration of analgesia of ~9 hours.15,16 Therefore, the combination of flurbiprofen axetil and nalbuphine led to a sustained decreased mean pain score <2 during the first postoperative 24 hours.
Our study also compared the side effect profiles of both drugs. Nalbuphine (36%) is known to cause more PONV than flurbiprofen axetil (0%).17 In our study, there were two and six cases of PONV in the flurbiprofen axetil and nalbuphine groups, respectively, and four cases in the combination group, but the differences were not statistically significant. Similarly, the combination of low-dose nalbuphine plus flurbiprofen has been reported to decrease the severity of PONV.13 We did not observe respiratory depression in any of the patients during the postoperative period.
Furthermore, the NRS discomfort score, which was defined as a subjective sensation involving “an uncomfortable feeling of mental painfulness or distress”, was higher in the nalbuphine group than in the other two groups, but no statistically significant differences were found, except at 2 hours postoperatively. This may be explained by discomfort being a subjective sensation of objective symptoms, including PONV and other side effects, which may cause patients an uncomfortable feeling in different parts of their body.
Conclusion
We demonstrate that the combination of flurbiprofen axetil with nalbuphine was optimal for analgesia in patients who underwent orbital decompression under general anesthesia. This combination minimized postoperative pain compared to flurbiprofen axetil or nalbuphine alone. This study may provide a novel and optimal option for analgesia, with potentially few side effects, for patients undergoing orbital decompression.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81870689; 81700875; 81670887; and 81470664) and the Sun Yat-Sen University Clinical Research 5010 Program, China (2014014). The sponsors had no role in the study design, survey process, data analysis, or manuscript preparation.
Disclosure
The authors report no conflicts of interest in this work.
References
Myles PS, Williams DL, Hendrata M, Anderson H, Weeks AM. Patient satisfaction after anaesthesia and surgery: results of a prospective survey of 10,811 patients. Br J Anaesth. 2000;84(1):6–10. | ||
Ye H, Chen R, Lian X, et al. Risk factors associated with postoperative pain and discomfort in oculoplastic surgery with general anesthesia: a prospective study. J Pain Res. 2018;11:407–415. | ||
Lesin M, Sundov ZD, Jukic M, Puljak L. Postoperative pain in complex ophthalmic surgical procedures: comparing practice with guidelines. Pain Med. 2014;15(6):1036–1042. | ||
Wang K, Luo J, Zheng L, Luo T. Preoperative flurbiprofen axetil administration for acute postoperative pain: a meta-analysis of randomized controlled trials. J Anesth. 2017;31(6):852–860. | ||
Yeh YC, Lin TF, Lin FS, Wang YP, Lin CJ, Sun WZ. Combination of opioid agonist and agonist–antagonist: patient-controlled analgesia requirement and adverse events among different-ratio morphine and nalbuphine admixtures for postoperative pain. Br J Anaesth. 2008;101(4):542–548. | ||
Schnabel A, Reichl SU, Zahn PK, Pogatzki-Zahn E. Nalbuphine for postoperative pain treatment in children. Cochrane Database Syst Rev. 2014;9(4):CD009583. | ||
Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131–157. | ||
Downie WW, Leatham PA, Rhind VM, Wright V, Branco JA, Anderson JA. Studies with pain rating scales. Ann Rheum Dis. 1978;37(4):378–381. | ||
Ferraz MB, Quaresma MR, Aquino LR, Atra E, Tugwell P, Goldsmith CH. Reliability of pain scales in the assessment of literate and illiterate patients with rheumatoid arthritis. Internet J Rheumatol. 1990;17(8):1022–1024. | ||
Jensen MP, McFarland CA. Increasing the reliability and validity of pain intensity measurement in chronic pain patients. Pain. 1993;55(2):195–203. | ||
Price DD, Bush FM, Long S, Harkins SW. A comparison of pain measurement characteristics of mechanical visual analogue and simple numerical rating scales. Pain. 1994;56(2):217–226. | ||
Henzler D, Kramer R, Steinhorst UH, Piepenbrock S, Rossaint R, Kuhlen R. Factors independently associated with increased risk of pain development after ophthalmic surgery. Eur J Anaesthesiol. 2004;21(2):101–106. | ||
Mao Y, Cao Y, Mei B, et al. Efficacy of nalbuphine with flurbiprofen on multimodal analgesia with transverse abdominis plane block in elderly patients undergoing open gastrointestinal surgery: a randomized, controlled, double-blinded trial. Pain Res Manag. 2018; 2018(2):1–9. | ||
Gupta A, Bah M. NSAIDs in the treatment of postoperative pain. Curr Pain Headache Rep. 2016;20(11):62. | ||
Sun M, Liao Q, Wen L, Yan X, Zhang F, Ouyang W. Effect of perioperative intravenous flurbiprofen axetil on chronic postmastectomy pain. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2013;38(7):653–660. | ||
Sultan A, McQuay HJ, Moore RA, Derry S. Single dose oral flurbiprofen for acute postoperative pain in adults. Cochrane Database Syst Rev Reviews. 2009;61(4):CD007358. | ||
van den Berg AA, Halliday E, Kisembo Lule E, Baloch MS. The effects of tramadol on postoperative nausea, vomiting and headache after ENT surgery. A placebo-controlled comparison with equipotent doses of nalbuphine and pethidine. Acta Anaesthesiol Scand. 1999;43(1):28–33. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





