Back to Journals » International Journal of General Medicine » Volume 13
Intermittent Asymptomatic Fever in a Psoriasis Patient
Authors Zhang J, Huang H, Xiong XX, Lu Y , Kasyanju Carrero LM, Zhou B, Xu Y
Received 29 September 2020
Accepted for publication 20 November 2020
Published 8 December 2020 Volume 2020:13 Pages 1441—1443
DOI https://doi.org/10.2147/IJGM.S282160
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Jiawen Zhang,1,* Hequn Huang,2,* XiXi Xiong,1 Yan Lu,1 Lorna Martin Kasyanju Carrero,1 Bingrong Zhou,1 Yang Xu1
1Department of Dermatology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province 210029, People’s Republic of China; 2Department of Dermatology, No. 1 Hospital and Key Laboratory of Dermatology, Ministry of Education, Anhui Medical University, Hefei, Anhui Province 230032, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yang Xu
Department of Dermatology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province 210029, People’s Republic of China
Tel +86-13851856794
Email [email protected]
Abstract: Acitretin, an active metabolite of etretinate, is the most widely used systemic retinoid in the treatment of psoriasis. Several side effects of acitretin have been reported such as teratogenicity, cheilitis, xerosis, dyslipidemia, and photosensitivity. Here, we reported a case of acitretin-induced intermittent asymptomatic fever in a 79-year-old male psoriasis patient. To the best of our knowledge, only one such case has been reported in the literature so far. We report our case to draw clinical attention that acitretin may cause drug fever, which might not be a rare phenomenon.
Keywords: acitretin, drug fever, psoriasis
Introduction
Drug-induced fever is a febrile response after the administration of a drug without other underlying causes.1 Drug-induced fever might have four kinds of heat type, presenting as continuous, remittent, intermittent, or hectic.2 Body temperature during drug-induced fever is usually around 39°C–40°C.3
Acitretin, a metabolite of etretinate, has been used to treat psoriasis since the 1980s. Common side effects, which are generally dose-dependent, include cheilitis, desquamation, pruritus, alopecia, rhinitis, xerophthalmia, and xerosis. Hepatotoxicity, altered lipid profile, and pancreatitis are less common. Acitretin is limited by its teratogenicity and thus should be avoided during pregnancy.4
Up to now, only one case of acitretin-induced fever has been reported.2 Here, we reported the second case ever of acitretin-induced fever.
Case Report
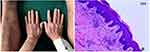
A 79-year-old man was presented with aggregated, erythematous, scaly plaques that appeared on his trunk and extremities 3 days ago. The patient had a 10-year history of psoriasis vulgaris, and his clinicopathological manifestations were consistent with psoriasis (Figure 1). He had a 20-year history of chronic obstructive pulmonary disease (treated using salmeterol and tiotropium bromide once daily), a 10-year history of hypertension (treated using nifedipine besylate 5 mg once daily), and a 5-year history of type-2 diabetes mellitus (treated using glimepiride 2 mg and acarbose 50 mg thrice daily). He was allergic to penicillin, cephalosporins, and magnesium isoglycyrrhizinate. The next day (day 2), acitretin 20 mg was administered at noon. The medications for chronic obstructive pulmonary disease, hypertension, and diabetes that he had been taking for the past 6 months were continued.
At 18:00 on day 3, 36 h after the first acitretin dose, the patient was found to have a fever (temperature, 38.5°C). The fever persisted for 4 h and disappeared after physical cooling at about 22:00. On admission, the chest CT scan showed signs of pulmonary infection. So daily 0.5g azithromycin was given intravenously. The patient did not feel any discomfort during this time. Further screening for infection, including bacterial cultures from the throat, skin, urine, and blood, serum antibody tests for Epstein-Barr virus and cytomegalovirus, and Aspergillus tests, yielded no positive results. Laboratory examinations indicated there might be no pathogenic infection. Intermittent fever persisted for the next 7 days (Figure 2). During the treatment of Acitretin, the patient’s skin manifestation is gradually getting better without any sign of aggravation. Meanwhile, the patient’s lips were dry which was alleviated by some moisturizer. On day 10, acitretin was discontinued, and pyrexia did not recur. The patient was administered methotrexate 7.5 mg/week and discharged after a marked therapeutic response. Follow-up at 1 and 3 months showed that the patient was well and afebrile.
Discussion
The key feature of this case is the sudden onset of fever after acitretin administration. The patient himself did not notice the fever. His heart rate was not elevated during pyrexia, and he was in a relatively good condition throughout. Although drug-induced fever is not listed as an adverse reaction in the package insert for acitretin, a sudden fever with a lack of self-discomfort and no evidence of relative bradycardia after the initiation of a medication should raise a high suspicion of drug-induced fever.
Besides high body temperature, other manifestations of drug fever include unawareness of the fever by the patient himself and disappearance of relative bradycardia. Patients with drug-induced fever may not have fatigue, chills, and discomfort, and their heart rate does not increase to the extent expected for the given temperature elevation. At 38.5°C, the appropriate pulse response is up to 113 beats per minute. The most sensible treatment for drug-induced fever is drug discontinuation. Challenge tests might re-induce fever and are controversial. Early diagnosis of drug fever and timely withdrawal of medicine play an essential role in maintaining efficient medical care.
Acitretin-induced fever is rare, with only one reported case.2 A 63-year-old man with psoriasis was administered acitretin 25 mg/day (0.3 mg/kg), and developed a sudden fever ~24 h later. The fever recurred every day thereafter (highest body temperature, 38.8°C), and subsided 24 h after acitretin discontinuation. Thus, the characteristics of acitretin-induced fever are as follows: (1) onset ~24 h after the first dose; (2) intermittent fever recurring every day (temperature < 39°C) and resolving spontaneously; (3) relative bradycardia and unawareness of the fever by the patient; and (4) complete resolution 1 day after acitretin withdrawal. Internal medical doctors and dermatologists should be aware of this side effect to avoid unnecessary screening.
Acitretin acts through retinoic acid receptors to decrease the expression of STAT1 and STAT3, and interfere with the JAK-STAT pathway. This may suppress keratinocyte proliferation and inhibit the expression of multiple cytokines.5 The mechanism of acitretin-induced fever needs further study.
Ethics Statement
The study was approved by the ethics committee of the First Affiliated Hospital of Nanjing Medical University. The patient also provided written informed consent for the case details and accompanying images to be published.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Patel RA, Gallagher JC. Pharmacotherapy. Drug Fever. 2010;30(1):57–69.
2. Rob F, Fialová J, Brejchová M. Drug fever as an adverse effect of acitretin in complicated psoriasis patient. Dermatol Ther. 2015;28(6):366–368. doi:10.1111/dth.12263
3. Labbus K, Junkmann JK, Perka C, Trampuz A, Renz N. Antibiotic-induced fever in orthopaedic patients-a diagnostic challenge. Int Orthop. 2018;42(8):1775–1781.
4. Kaushik SB, Lebwohl MG. Dermatology, psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80(1):43–53.
5. Heath MS, Sahni DR, Curry ZA, Feldman SR. Pharmacokinetics of tazarotene and acitretin in psoriasis. Expert Opin Drug Metab Toxicol. 2018;14(9):919–927.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.