Back to Journals » Journal of Multidisciplinary Healthcare » Volume 13
Intelligent Telehealth System To Support Epilepsy Diagnosis
Authors Molina E ![]() , Sarmiento Torres CE
, Sarmiento Torres CE ![]() , Salazar-Cabrera R
, Salazar-Cabrera R ![]() , López DM
, López DM ![]() , Vargas-Cañas R
, Vargas-Cañas R
Received 31 January 2020
Accepted for publication 24 April 2020
Published 15 May 2020 Volume 2020:13 Pages 433—445
DOI https://doi.org/10.2147/JMDH.S247878
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Edward Molina,1 Camilo Ernesto Sarmiento Torres,2 Ricardo Salazar-Cabrera,1 Diego M López,1 Rubiel Vargas-Cañas2
1Telematics Department, Universidad del Cauca, Popayán, Cauca, Colombia; 2Department of Physics, Universidad del Cauca, Popayán, Cauca, Colombia
Correspondence: Diego M López Tel +57 3015819362 Email [email protected]
Background: Availability and opportunity of epilepsy diagnostic services is a significant challenge, especially in developing countries with a low number of neurologists. The most commonly used test to diagnose epilepsy is electroencephalogram (EEG). A typical EEG recording lasts for 20 to 30 minutes; however, a specialist requires much more time to read it. Furthermore, no evidence was found in the literature on open-source systems for the cost-effective management of patient information using electronic health records (EHR) that adequately integrate EEG analysis for automatic identification of abnormal signals.
Objective: To develop an integrated open-source EHR system for the management of the patients’ personal, clinical, and EEG data, and for automatic identification of abnormal EEG signals.
Methods: The core of the system is an EHR and telehealth service based on the OpenMRS platform. On top of that, we developed an intelligent component to automatically detect abnormal segments of EEG tests using machine learning algorithms, as well as a service to annotate and visualize abnormal segments in EEG signals. Finally, we evaluated the intelligent component and the integrated system using precision, recall, and accuracy metrics.
Results: The system allowed to manage patients’ information properly, store and manage the EEG tests recorded with a medical EEG device, and to detect abnormal segments of signals with a precision of 85.10%, a recall of 97.16%, and an accuracy of 99.92%.
Conclusion: Digital health is a multidisciplinary field of research in which artificial intelligence is playing a significant role in boosting traditional health services. Notably, the developed system could significantly reduce the time a neurologist spends in the reading of an EEG for the diagnosis of epilepsy, saving approximately 65– 75% of the time consumed. It can be used in a telehealth environment. In this way, the availability and provision of diagnostic services for epilepsy management could be improved, especially in developing countries where the number of neurologists is low.
Keywords: electroencephalogram, electronic health record, machine learning, diagnostic support system, EEG, EHR
Introduction
Around 50 million people worldwide suffer from epilepsy, thus becoming one of the most common neurological diseases around the globe.1 Nearly 80% of people with epilepsy live in low and middle-income countries (LMIC).1 Higher incidence in LMIC is likely due to exposure to the disease risk factors, eg, endemic infections, injuries, socioeconomic factors, as well as lack of access to health care.2
The low availability of neurologists in LMIC causes some consequences such as the low coverage of health services for epilepsy.3 In Colombia, for instance, only 231 neurologists were available in 2011 (around one neurologist for every 200,000 inhabitants),4 while in the USA, the number of neurologists in 2012 was 16,366 (around one neurologist per 19,000 inhabitants).5 The lack of available specialists in LMIC affects the epilepsy diagnosis due to the complexity and the required time to analyze the diagnostic test.
Epilepsy is more frequently in children than in adults. The incidence of the disease in children under 18 years is 85.9 per 100,000, while in adults over 19 years is 64.81 per 100,000.6 Generally, adults and children present the same types of seizure, though some may be more common in childhood than adulthood.7 In addition, child-focused detection of epilepsy allows early detection to carry out adequate patient treatment during adulthood. Despite of this, there is less research efforts focused on epilepsy in children. Due to the above reasons we consider important to focus our research on pediatric patients.
EEG is the most used test to confirm epilepsy cases. Nevertheless, it has been demonstrated that it is possibly managing epilepsy without EEG. Examples include guidelines from the National Institute for Health and Care Excellence (NICE) covering diagnosis, treatment and management of epilepsy and seizures in children, young people and adults in primary and secondary care.8 Another example is the classification of chronic epilepsy into focal and generalized cases reliably made in most patients based upon their clinical information alone.9 EEG is required to determine seizure type enabling the correct prognosis, also to assess the risk of seizure recurrence. Likewise, EEG is helpful when epilepsy diagnosis is unclear after the clinical assessment is performed or in case when surgery is necessary.8
EEG signals are collected by placing electrodes on the scalp in order to record brain electrical activity. Commonly, the specialist visually inspects the EEG signal by pages (a page of EEG usually contains 10 seconds recording), trying to identify abnormal segments in the signal. The American Clinical Neurophysiology Society recommends a minimum of 20 minutes of artifact-free recording to evaluate baseline wakefulness, and the International League Against Epilepsy recommends recording at least 30 minutes. In line with these recommendations, many routine EEG last 20–30 minutes.10
In Colombia, routine EEG records last around 30 minutes, and these are interpreted by a neurologist in approximately 30–60 minutes because the specialist must read each page carefully. In such a way, an EEG record has around 180 pages, and each page contains the information of several channels. Neurologists must determine any abnormality in any of the channels. This reading must be rigorous because some abnormalities such as spikes are only 20–70 milliseconds in length and cannot get unnoticed. Commonly, an EEG test can contain several pages without any abnormality. Hence, a neurologist spends a great deal of time reading rigorously pages of the EEG test that have no abnormalities. A system that prevents the neurologist reviewing EEG pages without any apparent abnormality, and that also alerts to the neurologist only in the specific seconds in which there is possibly an abnormality, is called a decision support system. That system could support the diagnosis of epilepsy without eliminating the essential role of a neurologist in reading, revising, and providing a diagnosis based on the EEG records.
Epileptic signals from EEG records are highly dynamic and non-linear, which makes the visual inspection of specialists more complex. However, artificial intelligence (AI) technologies, such as machine learning, are becoming useful for identifying abnormal segments of EEG signals.
EEG waveform is considered abnormal when irregular brain activity is detected on continuous intervals. The abnormal EEG waveform is differentiated as epileptic and non-epileptic seizures.11 Epileptiform transients such as spikes and sharp waves are the inter-ictal (period between seizures) marker of a patient with epilepsy and are the EEG sign of a seizure focus.12 Therefore, the identification of spikes in the EEG tests is relevant for the diagnosis of epilepsy.
In addition to the EEG waveform, a patient’s information management is significant to perform a more efficient and accurate diagnosis and treatment of epilepsy or any other disease. EHR systems facilitate proper management of patient´s information.
The term EHR is widely used in many countries with variations in its definition and scope. In order to adequately describe the system proposed in this study, which is constrained to clinical settings use, we use the generally accepted definition of EHR as a longitudinal health record managed by healthcare professionals during patients visits to multiple healthcare facilities.13 In other words, EHRs contain the medical history of the patient, which can be retrieved at any time to support a clinical decision. It might include information about patient’s illnesses, operations, medications, allergies, and lab results.14 EHR systems have some benefits such as:13,14
- Uniquely identified patients,
- Clinical documentation at the point of care,
- Use of standard terminologies and vocabulary to guaranty interoperability,
- Decision support and secondary data analysis,
- The efficiency of healthcare processes, including increased cost-effectivity of clinical services.
There are a variety of platforms on the market that allow the use of EHRs. However, especially in the context of LMIC, “open-source” platforms must be available. In this way, health care institutions dealing with EEG and epilepsy management would not have to invest large amounts of money in using private EHRs.
Lack of access to epilepsy health care in LMIC occurs mainly in isolated rural areas, so telehealth and patient scheduling are also essential aspects. Telehealth refers to the use of medical (health) information that is exchanged from one site to another through electronic communication to improve a patient’s health.15 The objective of combining telehealth and patient scheduling is that trained personnel using certified EEG devices can reach specific rural locations for taking diagnostic EEG tests. At the same time, trained personnel can register EEG recordings and patient information in an EHR system. Later, the medical specialists (neurologists) can review the tests (usually in medical centers of capital cities distributed throughout the territory) and provide diagnostic results. Therefore, the EEG tests and patient´s information must be appropriately stored for secure remote access and management.
Currently, there are medical assistance systems, mainly used in developed countries, which are responsible for performing the analysis of an EEG in a short time and issue an automatic diagnosis for the verification of the specialist in neurology. The aim of our approach is not to diagnose epilepsy automatically but to support the medical specialist in the diagnostic process. Therefore, to eliminate the time spent in the revision of EEG pages without abnormal signals and directing the neurologist’s attention to EEG pages where there are probable abnormalities to be confirmed.
A variety of systems have been proposed in the literature that works as remote or telehealth platforms to connect health care workers with medical doctors and to transmit data from the patients to physicians.16,17 These approaches also incorporate EHR systems but are not focused on epilepsy management, so they do not provide an EEG repository either allow automatic detection of abnormalities in EEG records.
On the other hand, some approaches address the automated EEG-based epilepsy diagnosis or the automated detection of epileptic seizures.18–20 However, these approaches do not use EHR for the patient´s information management. Additionally, some of them are not diagnostic support systems. In the context of our work, specialists consider that the contribution of neurologists in the diagnosis of epilepsy is not expendable. We highlight that the procedure that we use for the feature extraction varies concerning the three mentioned works. Besides, our work is focused on the detection of ictal and inter-ictal spikes in EEG records, not only on seizure detection.
Some other authors have analyzed epileptic EEG signals applying different features to differentiate normal signals of ictal and inter-ictal signals.21–23 Still, they did not consider abnormalities associated with epileptic spikes. Our research explores these signals in the time and time-frequency domains, with features based on energy, entropy, and chaotic dynamic.
Finally, to the best of our knowledge, no system in the literature integrates both a telehealth EHR system for epilepsy management and an intelligent component to identify epileptiform EEG events automatically.
This paper describes the implementation of an open-source EHR and telehealth system for the management of the patients´ personal, clinical, and EEG data, appointment scheduling, and the automatic identification of abnormal EEG signals based on an intelligent component. This system would allow the neurologist to perform epilepsy diagnosis more efficiently and accurately, and it could operate in a telehealth environment.
In the following sections, the methods and procedures followed in system development are described; then, the obtained results with the use of the system are shown; finally, discussions, conclusions, and acknowledgments are included.
Methods and Procedures
We performed the development of the system in three stages. In the first stage, we developed the component responsible for the management of patients’ personal and clinical information, ie, the electronic health record (EHR) system. This system was based on OpenMRS, open-source software for building EHR systems. This first component was developed, taking into account the type of EEG recording device to be used and the file format of the EEG record generated by the device.
In the second stage, we developed an intelligent component that allows the detection of abnormal signals in EEG records using machine learning algorithms. Finally, in the third stage, we integrated the intelligent component into the EHR system, so the specialist could adequately observe the detected abnormal segments.
The dataset used in the implementation of the intelligent component was collected from 100 pediatric patients. The human subjects study (collection of EEG records) was reviewed and approved by the Ethical Committee of the University of Cauca, Colombia (http://vri.unicauca.edu.co/index.php/comite-de-etica), and the subjects signed informed consent forms.
This article principally details what it is described in stages two and three, because most of what it is described in stage one, corresponds to previous work.24 Once the system was developed, it was evaluated using EEG records collected from some patients.
The system development and evaluation stages are described below.
Development of the EHR System
The development of this component is described in a previous paper by some of the authors;24 however, some relevant details are given hereafter. To identify software requirements, we did in-depth interviews with a neuro-pediatrician and an EEG technician, as well as a visit to an EEG clinic. The objective of the interview was to understand the conventional procedure of collecting and managing both clinical and EEG data in the context of the diagnosis of epilepsy, as well as to know the needs of the neuro-pediatrician concerning the diagnosis of epilepsy in a telehealth environment. As a result of the in-depth interviews, system requirements were identified, such as the patients’ personal (Figure 1) and clinical (Figure 2) information to be collected.
 |
Figure 1 Registration of patient personal data in the EHR system (fictitious data). |
 |
Figure 2 Registration of EEG and patient clinical data in the EHR system (fictitious data). |
Bahmni (https://www.bahmni.org) was used for the EHR implementation; it is an open-source distribution of the OpenMRS (http://www.openmrs.org) framework.
EHR Requirements
Functional requirements were described in detail using “user stories”. System requirements include: management of patients’ personal and clinical data, EEG records registration, results reporting of EEG tests, and appointments scheduling and management. The system should allow health workers (EEG technicians) to take EEG tests on patients in different rural areas, and it also should allow neurologists to review the patients’ personal, clinical, and EEG data. In such a way, the system should allow a telehealth service deployment.
The system’s user interface should be in Spanish because this work is part of the NeuroMoTIC project, developed by the University of Cauca, Colombia. The project aims to propose and develop a system that allows the collection, management, and classification of patients’ clinical information and EEG signals to support the diagnosis of epilepsy in rural areas in Colombia.
The system should differentiate three users. The first user is the EEG technician (health care worker), who is responsible for recording patients’ personal, clinical, and EEG data, and for appointments management. The second user is the neurologist, who inspects patients’ clinical data and EEG tests to provide test results. The third user is the system administrator, who creates and manages user accounts for users to can access and use the system.
The EEG device chosen for signals recording was Brain Wave II (BWII-EEG) from the provider Neurovirtual (Ft. Lauderdale, FL, USA). This device record signals at a sampling frequency of 200 Hz in 18 channels, and it can export EEG records to EDF (European Data Format) file format.
EHR Platform
This component was developed using the OpenMRS framework. OpenMRS is an EHR, open-source framework written in Java programming language.25 It provides a data model and an API (Application Programming Interface) to access it. This system allows add-on modules created by users to add or remove functionality from the system. OpenMRS is the most used open-source EHR, and it also has an active community of developers and support. This system was selected as the EHR base system for implementation for the reasons mentioned above.
The open-source distribution of OpenMRS that we used specifically was Bahmni. This distribution was built as a JavaScript application on top of the OpenMRS data model and API.
Development of the Intelligent Component for Abnormal EEG Signals Detection
The development of this component was divided into two phases: training and testing. In the training phase, a model was constructed using a training dataset, which allows binary classification of EEG signals. In the testing phase, the performance of the model was evaluated using a testing dataset, ie, unseen data. Each of the two phases had the following steps: dataset configuration, feature extraction, training/testing, and evaluation. The mentioned steps are detailed below.
Dataset Configuration
The EEG dataset was collected from 100 children, between one month and 17 years old, with suspected epilepsy. Following bioethics standards, informed consent was obtained from patients’ carer before the EEG signals collection. The Ethics Committee approved the consent mentioned above of the Universidad of Cauca, Colombia. The acquisition device was the BWII-EEG, and the used software was BWAnalysis.26 A pediatric neurologist reviewed each EEG test, and results were registered according to events of interest (epileptiform abnormalities).
The abnormalities to be detected by the intelligent component were spikes, poly-spikes, and spikes with slow-waves. We could not take into account other abnormalities (such as acute waves) because the dataset did not include them.
Each EEG test was segmented in windows of one second (each window includes 200 samples, according to the sampling frequency of the device).27 This window duration was selected because the spikes are approximated 70ms time long. Besides, the window resolution allowed that the signal processing could be performed in less than three minutes (larger windows sizes could represent more time in signal processing). Extracted segments were divided into two groups:
- Training dataset (50 EEGs) with 1477 EEG segments, 845 normal, and 632 abnormal.
- Testing dataset (50 EEGs) with 370 EEG segments, 220 normal, and 150 abnormal.
Feature Extraction
The feature extraction was performed in the time domain, frequency domain, and time-frequency domain. In the time domain, the signals were analyzed only by its amplitude. In the frequency domain, the Fast Fourier Transform (FFT) was applied to the signals. Additionally, in the time-frequency domain, the Discrete Wavelet Transform (DWT) was used.
In total, 32 features were selected and used by the described method (more details are presented in another article by some of the authors).28 The principal selected features were:
- In Time Domain: minimum, maximum, kurtosis, energy, and Shannon entropy.
- In Frequency Domain: median.
- In Time-Frequency Domain: variance (cA4, cD4, cD3), energy (cA4, cD4, cD3, cD2), Shannon entropy (cA4, cD4, cD3, cD2), activity (cA4, cD4), Henry entropy (cD4, cD3, cD2, cD1), Hurst exponent (cD4), standard deviation (cD3, cD2), approximate entropy (cD3), minimum (cD2, cD1), maximum (cD2, cD1) and kurtosis (cD2). Note that cA and cD stand for approximation coefficient and detail coefficient from DWT, respectively.
Training
Five different supervised classification algorithms were implemented using the training dataset for constructing the model for EEG signals classification. The implemented algorithms were: multinomial logistic regression (MLR), K-nearest neighbors (K-NN), random forest (RF), support vector machine (SVM) and multilayer perceptron neural network (MLP). The parameters were adjusted for each algorithm, and the classification model was selected by the 10-folds cross-validation method.
Evaluation
The performance of the model was evaluated through the confusion matrix and accuracy (Acc), precision (Pr), and recall (r) metrics. As acceptance criterion, the evaluated model must have a high recall, to reduce the probability that the component does not detect some signals in the EEG records that are effectively abnormalities.
Integration of the Intelligent Component into the EHR System
The developed intelligent component receives as input (sent from the EHR system) the EEG test file in EDF format, this file has a duration of 30 minutes. Then, it is processed for approximately three and a half minutes. Finally, the intelligent component delivers, as a result, an EDF file with annotations containing information about the EEG abnormalities found, detailing the channel, and the exact second in which they were presented.
The intelligent component was developed using the Python programming language, and it was deployed as a web service. This web service can be consumed by the EHR system to analyze an EDF file (EEG record) and get this file annotated with the abnormalities found. In such a way, the EHR system can graphically present to the neurologist the segments of the EEG record that were identified as abnormalities.
Evaluation of the Integrated System
Fifty EEG tests were used to evaluate the integrated system. These tests are different from those utilized in the training and evaluation phases of the development of the intelligent component.
First, the automatic analysis of EEG records was performed by the intelligent component. Then, the abnormalities identified by the component were visualized and confirmed or rejected by a neurologist.
It should be taken into account that some of the identified abnormalities may correspond to the same instant of time (in different channels); therefore, the number of signal segments that the neurologist should check is commonly less than the number of identified abnormalities.
Results
Next, the EHR and telehealth system for the management of clinical data and automatic detection of abnormalities in EEG tests is presented, as well as the results obtained in the training and evaluation phases of the development of the intelligent component. Finally, we report the integrated system evaluation results.
EHR System for Clinical Data Management and Automatic Detection of EEG Abnormalities
System Architecture
The architecture of the entire developed system is presented in Figure 3. The main functionalities of the system are described in the next sections.
 |
Figure 3 Architecture of the entire system. |
Registration of Patient Data and EEG Tests
Initially, a technician (health care worker) registers the patient personal data in the EHR system (Figure 1). Afterward, the technician records the EEG signals from the patient using the BWII-EEG medical device and the BWAnalysis software. After the EEG signals have been collected, the technician registers the patient clinical information, and upload the EDF file (EEG record) in the system (Figure 2). At this stage, the clinical data and the EEG test is available in the system for revision by a neurologist in a telehealth context if needed.
Reading and Analysis of EEG Tests
Once the technician has recorded an EEG test, the neurologist can access the patient’s personal, clinical, and EEG data through the EHR system. The neurologist can read the EEG record page by page. However, instead of doing that, the neurologist also can use the analysis feature, which is performed by the intelligent component.
When using the analysis feature, the system automatically identifies abnormalities in the EEG record. The analysis of a 30-minutes EEG test lasts approximately 3.5 minutes. When the analysis is finished, the system presents graphically to the neurologist a list of each abnormality identified, detailing the second and channels where it occurred (Figure 4). The EEG analysis feature allows a neurologist to visualize EEG abnormality alerts in order to confirm or reject them.
 |
Figure 4 Visualization of abnormal EEG signals found by the intelligent component. |
The neurologist can register annotations in the EHR system such as normal body movements, blinking, or abnormal signals related to epilepsy, while he/she is reading the EEG test.
EEG Reporting
Once the EEG test revision is completed (page by page revision or alerts revision) the neurologist can register an EEG results report. This report describes the neurologist’s revision concept of the EEG test (normal or abnormal EEG) and whether or not the patient has suffered epileptic seizures during the test. The neurologist can download the report as a PDF file.
Appointments Management
The system allows a technician to define the services provided by the health care institution, ie, dates, locations, and opening and closing hours for EEG tests service. It also allows a technician to schedule an appointment for EEG tests, registering in the system appointment data such as patient information, location, date, and time. This feature is especially useful if the system is intended to be used in a telehealth environment.
Training and Evaluation Results of the Intelligent Component for Abnormal EEG Signals Detection
Through the method of 10-fold cross-validation applied to the training dataset, it was obtained the recall (r), accuracy (Acc), and precision (Pr) metrics of the trained models. The best model was obtained with the training of the Multilayer Perceptron Neural Network (MLP). The results are presented in Table 1. According to Table 1, the MLP model presented the highest levels of precision, accuracy, and recall in the training phase.
 |
Table 1 Training Metrics of Machine Learning Algorithms by 10-Fold Cross-Validation Method |
Furthermore, the evaluation of the models was performed using the testing dataset, calculating the confusion matrix (Figure 5), and the same performance metrics as the training phase (Table 2).
 |
Table 2 Evaluation Metrics of Machine Learning Algorithms |
 |
Figure 5 Confusion matrix of MLP model. |
Multi-layer Perceptron (MLP) is a supervised learning algorithm that learns the function shown in Equation 1 by training on a dataset, where m is the number of dimensions for input and o is the number of dimensions for output.
(1)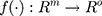
Given the set of features shown in Equation 2 and a target Y, the MLP algorithm can learn a non-linear function approximator for either classification.
(2)
The MLP algorithm is made of an input layer with several nodes equal to the number of features, and a hidden layer where the values of the previous layer are transformed into a weighted linear sum, followed by a non-linear activation function. It also contains an output layer, which receives the values of the hidden layer and transforms the output values.29
In this work, the Limited-Memory-Broyden–Fletcher–Goldfarb–Shanno (LBFGS) method was applied for training of MLP algorithm, with the Relu activation function, a tolerance of 1 x 10−3, one hidden layer, and 29 neurons.
Evaluation of the Integrated System
The system performance was evaluated by comparing the EEG abnormalities detected by the component versus the abnormalities found by the neuro-pediatrician. For this evaluation, 50 EEG tests that were not used in the training and evaluation phases of the classification model were used. 49 of 50 tests were normal, only 1 of them was abnormal. The results are presented in Tables 3 and 4.
 |
Table 3 Evaluation of the Integrated System Concerning Neuro-Pediatrician Revision of EEGs (Normal Tests) |
 |
Table 4 Evaluation of the Integrated System for Neuro-Pediatrician Revision of EEGs (Abnormal Test) |
The intelligent component was evaluated using only the accuracy metric on the normal EEG tests because they did not have abnormalities. The average accuracy was 99.24%, and the average error was 0.077% (Table 3). On the other hand, the precision, recall, and accuracy metrics were calculated on the abnormal EEG test. As a result, the system was able to identify 588 abnormal segments due to the high recall design. Only 39 false negatives were detected, representing an error of 0.08% (Table 4).
Additionally, the time spent by the neurologist at reading the abnormal signals detected in EEG records by the intelligent component was measured. This time was compared to the time a neurologist takes to read a complete 30-minute EEG test, ie, 30–60 minutes.
The time the neurologist spent to read an EEG test using the system described is 10–15 minutes. The comparison between the two revision approaches showed an average saving of approximately 65–75% of the time when the EEG analysis feature of the EHR system is used. This saving of time is significant enough to achieve improvements in the availability and opportunity of the epilepsy diagnostic service.
Discussion
We have described the implementation process of an open-source EHR system for the management of patients’ personal and clinical information related to epilepsy, appointment scheduling, EEG data, and the intelligent detection of abnormal segments in EEG records. This system has been proved to allow neurologists to perform the identification of abnormal signals with an average accuracy of 99.24% in normal EEG tests and 99.92% in abnormal EEG tests. Some preliminary results demonstrate that the neurologist could reduce the time to read an EEG by 65%–75%. This reduction in time occurred because the neurologist did not have to invest time reviewing large sections of an EEG test that had no apparent abnormality. Instead, the neurologist revision could be focused on the abnormal segments automatically detected by the analysis feature of the EHR system.
The developed system has relevant functionalities that facilitate the management of epileptic patients’ information, and it also supports the diagnostic process making it more efficient. However, we consider that the system has some aspects of being improved, which are discussed below.
It is crucial that the system can identify all types of abnormalities, which requires a dataset that includes abnormalities such as sharp waves and other types of abnormalities that have not been analyzed so far. With the dataset mentioned above, it would be possible to adjust the intelligent component model and allow the detection of all types of abnormalities. However, the inclusion of new types of abnormalities could affect the percentage of system detection errors.
Although the percentage of error in the detection of abnormalities can be considered relatively low (0.08%), it is essential to identify some aspects that could improve accuracy, such as a change in the features extraction process. The detailed analysis of the false positives in the detection, in a considerable number of EEG, can help to identify these aspects to improve.
It is also important to mention a technical problem identified in the operation of the system. Occasionally, the neurologist may request the technician to take an EEG with different configurations (regarding the quantity and location of the sensors on the patient). The information on the configuration of the used assembly at the time of taking the EEG is not included in the EDF file (EEG record) that is generated through BWAnalysis, the recording software of the EEG device. For this reason, to achieve proper functioning of the intelligent component, it is necessary that before generating the EDF file in the recording software, the technician previously selects the type of assembly called “banana.” This restriction is necessary to take it into account when operating the system and try to solve it in another way later; it is an integration problem that will arise in any specific EEG device configuration.
It would also be worthwhile to be able to carry out subsequent experiments with a higher number of neurologists. This because the reading of the EEG tests was made only with the participation of a neuro-pediatrician, which can skew the obtained results. The participation of a more significant number of neurologists in another experiment would give the system greater reliability.
The multidisciplinary approach that we used in our work involved the collaboration between a computer scientist and a neurologist in the process of epilepsy diagnosis. We were able to understand that the system was not focused on trying to perform an automatic diagnosis of epilepsy because this requires a much higher scope than the one proposed in this work. The developed system allows the neurologist to focus her attention on the sections where there is an abnormality to confirm or rule it out.
We consider it is essential to mention that the automatic, online, and real-time detection of the abnormalities of an EEG in our system could be possible, because the detection made by the system is done with short segments of the signal, lasting one second. The restriction for this functionality is because the user interface of the EEG device generates the EDF file once the EEG exam is finished. Possibilities should be evaluated, with a new version of the selected EEG device, or with other devices, or with another integration process, to ensure that the signal can be analyzed segment by segment in real-time.
The developed system was designed to operate in a telehealth context, more precisely in a Tele-EEG service. Tele-EEG refers to the transmission of EEG recordings to a distant center for remote interpretation; this transmission could be done off-line (transmission of EEG as complete file) or on-line (transmission of EEG in real-time).30,31 In such a way, the proposed system also fits into a Tele-EEG system category.
As mentioned earlier, the developed system could be extended to include real-time transmission and analysis of EEG segments supported on Artificial Intelligence (AI) algorithms. This extension could enable additional Tele-EEG applications and services, ie, to monitor seizures in a natural environment, operating theatre, or intensive care unit (ICU).31 The real-time analysis could improve the decision-making process of neurologists to identify abnormal segments.
There are many works describing approaches for automatic seizure detection using AI. Some of them state that their approaches are enabled to support tele-EEG systems.32 These approaches can analyze EEG records off-line to provide automatic seizure detection. In another way, several semi-automatic detection algorithms are available on continuous EEG monitoring devices to facilitate the interpretation of EEG.33 These devices and algorithms can be used in a tele-EEG context for on-line analysis. Some works confirm the relevance of using automatic seizure detection in tele-EEG systems for seizure recognition and classification, seizure prediction, pathological pattern detection, and automated alerts.30,34-36 Bearing in mind what described above, it can be concluded that AI can play an essential role in the improvement of tele-EEG services.
As future work, we propose the possibility of training health workers in rural areas to record the EEG tests. Training in basic computer skills is also necessary.37,38 Additionally, we propose carrying out campaigns in rural areas to move an EEG device temporarily, since due to the high costs of the device, it is not possible to keep it in the health centers of these areas. In this way, when the EEG device is transferred to rural areas, trained personnel will be available to take the EEG tests to patients in the area, making possible the operation of telehealth services; these tests will be scheduled in advance.
Conclusion
The developed system allows the adequate management of patients’ personal, clinical, and EEG information (using open-source EHR technology). It also performs automatic detection of abnormalities in EEG exams with a precision of 85.10%, a recall of 97.16%, and an accuracy of 99.92%, which helps to speed up the diagnostic process of epilepsy disease, by reducing the required time by neurologists to review EEG exams.
The selected algorithm for the intelligent component of the system was MLP, which produced the best results.
The evaluation of the integrated system made it possible to identify that a neurologist could significantly reduce the reading time of an EEG for the diagnosis of epilepsy, saving approximately 65–75% of the time. The above, considering that he/she should no longer read all the segments of the EEG, but only the abnormal segments that our system identifies automatically.
The use of our system to support the diagnostic process, not only improves the efficiency in the performed process by neurologists (reducing the time required to review an EEG and giving them the possibility to review a higher number of EEG exams in a specific time). It also provides the possibility that neurologists can review some EEG exams in other locations at regional, national or international levels. Therefore, the system can be used in a telehealth environment where it is possible to access remotely to the clinical and the EEG data.
Acknowledgments
This work was funded by a grant from COLCIENCIAS - Colombian Agency for Science, Technology, and Innovation – under Call 715-2015, “Convocatoria para Proyectos de Investigación y Desarrollo en Ingenierias,” project “NeuroMoTIC: Sistema móvil para el Apoyo Diagnóstico de la Epilepsia,” contract number FP44842-154-2016.
The authors thank to Axon PED SAS for their collaboration in taking the EEG tests and to all the patients who collaborated by allowing the recording of EEG tests and clinical data.
Disclosure
Mr Edward Molina reports grants from Colombian Agency for Science, Technology, and Innovation Colciencias, during the conduct of the study. The authors declare no other conflicts of interest.
References
1. WHO (World Health Organization). Epilepsy. Available from: https://www.who.int/news-room/fact-sheets/detail/epilepsy.
2. WHO (World Health Organization). Epilepsy. A public health imperative. ISBN 978-92-4-151593-1. 2019.
3. Méndez-Ayala A, Nariño D, Rosselli D. Burden of epilepsy in Colombia. Neuroepidemiology. 2015;44:144–148. doi:10.1159/000381424
4. Amaya J, Beltrán A, Chavarro D, et al. Estudio de disponibilidad y distribución de la oferta de médicos especialistas, en servicios de alta y mediana complejidad en Colombia. Doc. técnico GPES/1682C–13.
5. American A. Neurology. The doctor won’t see you now? Study: US facing a neurologist shortage. Available from: https://www.aan.com/PressRoom/Home/PressRelease/1178#targetText=By%202025%2C%20that%20number%20will,to%2021%2C440%20during%20that%20time.
6. Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88(3):296–303. doi:10.1212/WNL.0000000000003509
7. Epilepsy Society. Epilepsy in Childhood. Available from: https://www.epilepsysociety.org.uk/epilepsy-childhood.
8. NICE National Institute for Health and Care Excellence. Epilepsies: diagnosis and management. Available from: https://www.nice.org.uk/guidance/cg137/resources/epilepsies-diagnosis-and-managementpdf-35109515407813.
9. Kumar S, Singh MB, Shukla G, et al. Effective clinical classification of chronic epilepsy into focal and generalized: a cross sectional study. Seizure. 2017;53:81–85. doi:10.1016/j.seizure.2017.11.002
10. Burkholder DB, Britton JW, Rajasekaran V, et al. Routine vs extended outpatient EEG for the detection of interictal epileptiform discharges. Neurology. 2016;86(16):1524–1530. doi:10.1212/WNL.0000000000002592
11. Medithe JWC, Nelakuditi UR, Study of normal and abnormal EEG.
12. Boro AD, Haut S. Focal EEG Waveform Abnormalities. Medscape. Available from: https://emedicine.medscape.com/article/1139025-overview., 2017.
13. WHO (World Health Organization) Western Pacific Region 2013. Electronic Health Records. Manual for Developing Countries. 2006. ISBN 92 9061 2177.
14. Lopez DM, Blobel BG. Connecting public health and clinical information systems by using a standardized methodology. Stud Health Technol Inform. 2007;129(Pt 1):132–136.
15. Tuckson RV, Edmunds M, Hodgkins ML. Telehealth. N Engl J Med. 2017;377:1585–1592. doi:10.1056/NEJMsr1503323
16. Celi LA, Sarmenta L, Rotberg J, Marcelo A, Clifford G. Mobile care (Moca) for remote diagnosis and screening. J Health Inform Dev Ctries. 2009;3(1):17.
17. Townsend B, Abawajy J, Kim TH. SMS-based medical diagnostic telemetry data transmission protocol for medical sensors. Sensors. 2011;4231–4243. doi:10.3390/s110404231
18. Peker M, Sen B, Delen D. A novel method for automated diagnosis of epilepsy using complex-valued classifiers. IEEE J Biomed Health Inform. 2016;20(1):108–118. doi:10.1109/JBHI.2014.2387795
19. Tiwari AK, Pachori RB, Kanhangad V, Panigrahi BK. Automated diagnosis of epilepsy using key-point-based local binary pattern of EEG signals. IEEE J Biomed Health Inform. 2017;21(4):888–896. doi:10.1109/JBHI.2016.2589971
20. Wang G, Ren D, Li K, Wang D, Wang M, Yan X. EEG based detection of epileptic seizures through the use of a directed transfer function method. IEEE Access. 2018;6:47189–47198. doi:10.1109/ACCESS.2018.2867008
21. Ibrahim S, Djemal R, Alsuwailem A. Electroencephalography (EEG) signal processing for epilepsy and autism spectrum disorder diagnosis. Biocybern Biomed Eng. 2018;38:16–26. doi:10.1016/j.bbe.2017.08.006
22. Alickovic E, Kevric J, Subasi A. Performance evaluation of empirical mode decomposition, discrete wavelet transform, and wavelet packed decomposition for automated epileptic seizure detection and prediction. Biomed Signal Process Control. 2018;39:94–102. doi:10.1016/j.bspc.2017.07.022
23. Kalbkhani H, Shayesteh MG. Stockwell transform for epileptic seizure detection from EEG signals. Biomed Signal Process Control. 2017. doi:10.1016/j.bspc.2017.05.008
24. Molina E, Salazar-Cabrera R, López DM NeuroEHR: open source telehealth system for the management of clinical data, EEG and remote diagnosis of epilepsy.
25. Wolfe BA, Mamlin BW, Biondich PG, et al. The OpenMRS system: collaborating toward an open source EMR for developing countries. Proc AMIA Annu Symp. 2006;2006:1146.
26. Neurovirtual. Available from: https://neurovirtual.com/.
27. Samiee K, Kovács P, Gabbouj M. Epileptic seizure detection in long-term EEG records using sparse rational decomposition and local Gabor binary patterns feature extraction. Knowl-Based Syst. 2017;118:228–240. doi:10.1016/j.knosys.2016.11.023
28. Mera-Gaona M, Vargas-Canas R, Lopez DM. Towards a selection mechanism of relevant features for automatic epileptic seizures detection. Stud Health Technol Inform. 2016;228:722–726.
29. Rumelhart D, Hinton G, Williams R. Learning representations by back-propagating errors. Nature. 1986;323:533–536. doi:10.1038/323533a0
30. Lemesle M, Kubis N, Sauleau P, Tich SG, Touzery-de Villepin A. Tele-transmission of EEG recordings. Neurophysiol Clin. 2015;45(1):121–130. doi:10.1016/j.neucli.2014.12.001
31. Holder D, Cameron J, Binnie C. Tele-EEG in epilepsy: review and initial experience with software to enable EEG review over a telephone link. Seizure. 2003;12(issue 2):85–91. doi:10.1016/S1059131102002297
32. Zeng K, Yan J, Wang Y, Sik A, Ouyang G, Li X. Automatic detection of absence seizures with compressive sensing EEG. Neurocomputing. 2016;171:497–502. doi:10.1016/j.neucom.2015.06.076
33. André-Obadia N, Parain D, Szurhaj W. Continuous EEG monitoring in adults in the intensive care unit (ICU). Clin Neurophysiol. 2015;45(1):39–46. doi:10.1016/j.neucli.2014.11.003
34. Kissani N, Modeste Y, Patterson V, et al. Telemedicine in epilepsy: how can we improve care, teaching, and awareness? Epilepsy Behav. 2020;103(A):106854. doi:10.1016/j.yebeh.2019.106854
35. Meisel C, Loddenkemper T. Seizure prediction and intervention. Neuropharmacology. 2002;107898.
36. Schramm P, Luczak J, Engelhard K, El Shazly J, Juenemann M, Tschernatsch M. Continuous electroencephalography in a mixed non-neurological intensive care population, an observational study. J Crit Care. 2017;39:62–65. doi:10.1016/j.jcrc.2017.01.015
37. Blas MM, Curioso WH, Garcia PJ, et al. Training the biomedical informatics workforce in Latin America: results of a needs assessment. BMJ Open. 2011;1(2):e000233. doi:10.1136/bmjopen-2011-000233
38. Lopez DM, Blobel B. mHealth in low- and middle-income countries: status, requirements and strategies. Stud Health Technol Inform. 2015;79–87.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
