Back to Journals » Patient Preference and Adherence » Volume 14
Insufficient Knowledge and Vaccination Practice of Inflammatory Bowel Disease Patients in the People’s Republic of China
Authors Feng S, Lin S, Ma L, Xu S, Chen Y
Received 30 May 2020
Accepted for publication 30 July 2020
Published 24 August 2020 Volume 2020:14 Pages 1513—1521
DOI https://doi.org/10.2147/PPA.S265346
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Naifeng Liu
Shuyi Feng,1 Saizheng Lin,2 Lingling Ma,3 Song Xu,4 Yan Chen5
1The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China; 2Department of Gastroenterology, Ningbo Zhenhai People’s Hospital, Ningbo, Zhejiang Province, People’s Republic of China; 3Department of Gastroenterology, Dongying People’s Hospital, Dongying, Shandong Province, People’s Republic of China; 4PhD Center for Inflammatory Bowel Disease, Department of Gastroenterology, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China; 5Center for Inflammatory Bowel Disease, Department of Gastroenterology, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China
Correspondence: Yan Chen
Center for Inflammatory Bowel Disease, Department of Gastroenterology, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province 310009, People’s Republic of China
Email [email protected]
Background: Inflammatory bowel disease (IBD) patients are at risk for HBV and varicella, which makes vaccination an important role.
Aim: To assess knowledge and vaccination rate of Chinese IBD patients and find the influencing factors.
Methods: An online anonymous questionnaire was distributed to IBD patients through www.wjx.cn in the People’s Republic of China during October 2019 to February 2020. The Kappa value and multivariate analysis were used to analyze the data using SPSS 25.0.
Results: Among 900 participants, 746 (82.9%) hadreceived immunosuppressive treatment. Only 25 (2.78%) patients thought that they had sufficient information on vaccination, and 311 (34.6%) patients remembered being reminded about vaccination by their doctors before. The self-reported vaccination rate for hepatitis B was 125/666 (18.8%), and for varicella 44/671 (6.6%). Information from doctors was positively associated with vaccination status of hepatitis B (odds ratio [OR]=4.61, 95% confidence interval [Cl: 2.73– 7.76], p< 0.001) and varicella (OR=4.93, 95%Cl :2.02– 12.05, p< 0.001). The patients whose duration of IBD diagnosis was more than 2 years (OR=0.37, 95% Cl: 0.16– 0.87, p=0.022) and those older than 38 years (OR=0.37, 95% Cl: 0.14– 1.00, p=0.0497) were negatively related with the status of varicella vaccination.
Conclusion: Vaccination-related knowledge level in IBD patients appeared to be low and the rate of vaccination of both hepatitis and varicella was way below the recommended level. Proper education of both IBD patients and gastroenterologists in the People’s Republic of China is needed.
Keywords: inflammatory bowel disease, vaccination, hepatitis B, varicella
Introduction
Inflammatory bowel disease (IBD) is a chronic inflammatory disease of the gastrointestinal tract including ulcerative colitis (UC) and Crohn’s disease (CD). IBD has become a global disease with increasing incidence and prevalence1 and poses important challenges for the IBD community.2 The incidence of IBD is still low in the People's Republic of China when compared to that in Western countries with a reported rate of 1.80/100,000.3 However recent epidemiological data revealed a dramatic rise in the prevalence and incidence of IBD in the People's Republic of China.4
It has been recognized that IBD patients are at an increased risk of infectious complications compared to the non-IBD population.5 Patients with IBD are often treated with long-term immune-suppressive therapies, including various biological agents, which may thus further increase the chance of infection. Many of these infections are preventable with proper vaccinations.6,7
Hepatitis B prevalence is highest in the Western Pacific Region where 6.2% of the adult population is infected,8 and reactivation of hepatitis B was reported in immunosuppressed IBD patients with severe consequences.9 All adult patients with IBD should receive non-live hepatitis B vaccine.10 Despite these recommendations, vaccination rates of hepatitis B for IBD patients remain low in Canada.11 In a study of 500 IBD patients in the United States, only 254 (51%) had screening for hepatitis B virus (HBV), and 95 (49%) were vaccinated for HBV.12 In another study of 2,076 IBD patients in Spain, only 12% of patients were vaccinated for hepatitis B.13 However, the data of IBD patients’ hepatitis B vaccination rates in China have not been reported yet. An Asian study also demonstrated that the IBD patients exhibited a higher risk of varicella infection than non-IBD patients,14 and it has been recommended that adults with IBD should be assessed and those without history of disease or evidence of seropositivity should receive the vaccine.10 Thus, it is significant to access the immunization rates and knowledge for varicella in IBD patients.
China Crohn’s and Colitis Foundation (CCCF) is a non-profit, voluntary organization that aims to help patients affected by IBD through various educational activities carried out in the People's Republic of China.15 This survey was performed to: 1) access the IBD patients’ overall knowledge and attitudes towards vaccination; 2) find the vaccination rates for hepatitis B, varicella and tendency to rabies vaccination; and 3) identify the factors affecting IBD patients’ vaccination to discover strategies for improvement in China.
Methods
Participants
The participants were IBD patients mainly from six provinces (Zhejiang, Jiangsu, Chongqing, Sichuan, Hubei, Anhui), and informed consent was received from all participants who completed the questionnaire. The questionnaire was online and the data were collected entirely in a web-based format; no special software or APP was required; patients could access the questionnaire easily by any networked electronic devices. This study was a retrospective research. The inclusion criteria were IBD patients who met WHO standards and had the ability to read and answer questionnaires independently. The exclusion criteria included patients with total colectomy, stoma patients and patients who were unable to read. The feedback rate was 36.2% (905/2500). Participants had to be able read and understand Chinese.
Study Questionnaire
We deployed a 37-question electronic questionnaire between October 2019 and February 2020 through www.wjx.cn (see full questionnaire in Supplementary data). The questionnaire was designed by three IBD specialists. This anonymous questionnaire included five sections with open and close-ended questions: 1) social demographic data; 2) patients’ treatment status; 3) source of IBD-related vaccine information; and patients’ preferences, 4) patients’ knowledge of the vaccination; and 5) type of vaccines and disease status.
This study was approved by the Ethics Committee at the Second Affiliated Hospital of Zhejiang University School of Medicine. (No. Yan2019-225) and this study was conducted in accordance with the Declaration of Helsinki.
Statistical Analysis
All statistical analyses were conducted using SPSS software (version 25.0; IBM Corp., Armonk, NY, USA). Descriptive statistics were used to characterize the population and outcome measures. The Kappa value was used to explore whether a relationship exists between notification and immunosuppressive therapy. Multivariate analysis was used to determine the factors influencing patients’ knowledge on recommended and contradicted vaccination and vaccination rates. A p-value < 0.05 was considered significant.
Results
We received 905 returned questionnaires online, of which four questionnaires were incompletely filled and one was a repeated questionnaire. Below are the results of these questionnaires.
Demographics of the Responding Patients
Analysis was performed using 900 valid questionnaires from patients diagnosed with IBD as shown in Table 1. Among them, 534 (59.3%) were male, 303 (33.7%) were younger than 28 years old, 509 (56.6%) had high education (> high school diploma), 415 (46.1%) were registered rural residents and 167 (18.6%) were CCCF patient volunteers.
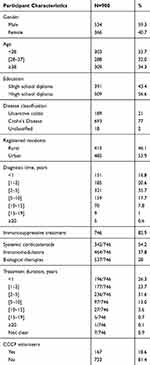 |
Table 1 Participant Characteristics |
Among the 900 patients, 693 (77.0%) were diagnosed as CD, 189 (21.0%) were diagnosed as UC, 18 (2.0%) were unclassified IBD. Most patients (657, 73.0%) were diagnosed less than 5 years. A total of 746 (82.9%) patients received immunosuppressive treatment including systemic corticosteroids (342/746, 54.2%), immunomodulators (464/746, 37.8%), or biological therapies (537/746, 28.0%). Among those treated, 373 (50.0%) had started the treatment less than 2 years previously and 130 (17.4%) had started more than 5 years previously.
Knowledge on and Attitudes Towards the Vaccination
Only 25 (2.8%) patients thought that they had sufficient information on vaccination that applied to their disease conditions, and 483 (53.7%) patients thought that they knew nothing about that. Moreover, 723 (80.3%) patients did not realize the recommended vaccination and 779 (86.6%) patients did not know the contraindicated vaccines for their diseases. However, 498 (55.3%) patients felt that they more susceptible to infections than healthy individuals.
In a multivariate analysis (Table 2), we found that patients with CD knew more about recommended vaccination than patients with UC (odds ratio [OR]=1.74, 95% confidence interval [Cl: 1.07–2.83], p=0.026), and patients under 28 were better informed on vaccination than those over 37 (OR=1.81, 95% Cl: 1.19–2.76, p=0.006), but there was no significant difference in the knowledge between the age group of 28–37 and the age group > 37. Then we found that factors associated with patient’s knowledge of contraindicated vaccines were the diagnosis Crohn’s disease (OR=2.18, 95% Cl: 1.18–4.04, p=0.013) and being CCCF volunteers (OR=2.04, 95% Cl: 1.31–3.16, p=0.001) (Table 3).
 |
Table 2 Multivariate Analysis of Factors Associated with Patient’s Knowledge of Recommended Vaccination |
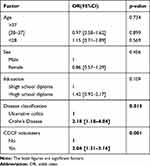 |
Table 3 Multivariate Analysis of Factors Associated with Patient’s Knowledge of Contraindicated Vaccines |
When asked about patient’s trust in domestic vaccines in general, 377/900 (41.9%) patients chose “basically safe,” 337/900 (37.4%) patients chose “mostly safe,” and 141/900 (15.7%) patients are not sure about that, only 45/900 (5.0%) chose “only part of vaccines are safe” or “all vaccines are unsafe.”
Sources of Information on IBD-Related Vaccination
We investigated 417 patients who were self-assessed to have knowledge of IBD-related vaccination, the most common sources of information were an IBD specialist (86.6%), a patient association (34.3%) and other patients (26.6%) as shown in Figure 1. Next, we investigated patient’s trust in information from various sources. Among 900 participants, 862/900 (95.8%) patients thought information from an IBD-specialist was “trustable” or “rather trustable,” and the top three sources after that were general gastroenterologists (665/900, 73.8%), patient associations (600/900, 66.7%) and public health agencies (545/900, 60.6%). (See full table in Supplementary data.)
 |
Figure 1 Sources of IBD-related vaccination knowledge. |
Notification from Clinicians
When we investigated the notification of vaccination during the illness by doctors, we found that only 311/900 (34.6%) patients said they have been informed before, 337/900 (37.4%) patients clearly remembered not being told about vaccine-related matters and 252/900 (28.0%) could not remember or were not clear about that. Then, we analyzed the relationship between vaccination notification and immunosuppressive treatment. Deleting the patients who were not clear or could not remember whether they have been notified or treated with immunosuppressive therapy, we found notification rates are significantly higher in patients who have received immunosuppressive therapy before (p<0.001). (See full table in Supplementary data.)
We then focused on the factors associated with notification rates at the time of the patient’s first immunosuppressive treatment (Table 4). We found that patients who used biological therapies as their first immunosuppressive therapy are more likely to be informed about vaccination (OR=2.07, 95%Cl: 1.27–3.38, p=0.003). Compared with IBD specialists, non-IBD physicians have a lower notification rate (OR=0.38, 95% Cl: 0.22–0.66, p=0.001). Time since first used immunosuppressive therapy is also a significant factor, patients treated for more than 5 years had a lower notification rate than patients treated less than 2 years (OR=0.26, 95% Cl: 0.15–0.46, p<0.001), but there was no significant difference between patients treated for less than 2 years and patients treated less than 5 years but greater than 2 years.
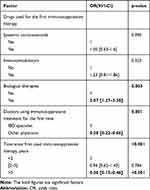 |
Table 4 Multivariate Analysis of Factors Associated with Notification Rate at Patients’ Initial Immunosuppressive Treatment |
The Rate of Hepatitis B Vaccination and Associated Factors
Among 900 participants, 72 (8.0%) patients could not remember whether they had been vaccinated against hepatitis B, and 162 (18.0%) patients were either infected with hepatitis B or immune against HBV. Deleting these data, the number of patients was 666, and the self-reported vaccination rate against hepatitis B was 18.8% (125/666).
The main motivators for vaccination were “Vaccination has been recommended by doctors or nurses” (68.8%) and “I felt that I am at a risk of getting hepatitis B” (25.6%). The main reason for not receiving vaccination was “No one has recommended me to vaccinate” (74.3%).
Then, we tried to find out the factors that influence the patient’s vaccination rate. Because the number of unclassified IBD patients was too small, we deleted the data of them when analyzing to avoid accidents. We also deleted the patients who could not remember whether they had been informed about the vaccine. In the multivariate analysis (Table 5), notification from doctors (OR=4.61, 95% Cl:2.73–7.76, p<0.001) and patient trust on hepatitis B vaccine safety (OR=2.68, 95% Cl :1.08–6.65, p=0.034) were positively associated with hepatitis B vaccination. Patients with 2–5 years’ diagnosis time were less likely to be vaccinated when compared with patients with < 2 years’ diagnosis time (OR=0.33, 95% Cl :0.18–0.61, p<0.001), but there was no significant difference between patients with a diagnosis longer than 5 years and patients with a diagnosis less than 2 years.
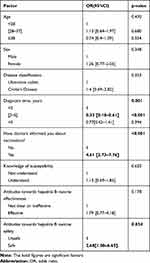 |
Table 5 Multivariate Analysis of Factors Associated with Hepatitis B Vaccination |
The Rate of Varicella Vaccination and Associated Factors
Deleting the patients who are not sure whether they have been vaccinated against varicella (92/900, 10.2%) and the patients already immune to varicella (137/900, 15.2%), the amount of the rest of the patients was 671. We found the reported rate of vaccination against varicella was only 6.56% (44/671).
The main motivator for vaccination was also “Vaccination has been recommended by doctors or nurses” (63.6%) and the main reason for not receiving vaccination was still “No one has recommended me to vaccinate” (61.6%).
We also analyzed the factors that may influence patient’s vaccination rate, and still deleted the untyped patients and the patients who could not recall whether they had been notified (Table 6). Doctor’s notification was still a positive factor (OR=4.93, 95% Cl: 2.02–12.05, p<0.001) in patient varicella vaccination. Both patients with >5 years’ diagnosis time (OR=0.25, 95% Cl: 0.08–0.8, p=0.020) and patients with 2–5 years of diagnosis (OR=0.37, 95% Cl:0.16–0.87, p=0.022) were less likely to be vaccinated against varicella than a patient with <2 years’ diagnosis time. Another factor related with varicella vaccination is the age of patients. We found that people over 38 were negatively associated with varicella vaccination (OR=0.37, 95% Cl: 0.14–1.00, p=0.0497), but there was no significant difference between patients under 28 and 28–37-year-old patients.
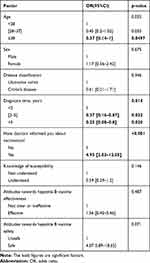 |
Table 6 Multivariate Analysis of Factors Associated with Varicella Vaccination |
Discussion
Our study reports a low knowledge level of IBD patients on vaccination and most patients did not know the recommended (80.3%) and the contraindicated (86.6%) vaccines. In addition, most IBD patients (92.1%) expressed their desire to learn more about vaccine-related information. Inadequate knowledge contributes to low vaccination rates among IBD patients, which put patients with IBD at an increased risk of developing avoidable, and potentially life-threatening infections. Many of these infections are preventable due to the development and availability of effective vaccines.16,17 With insufficient knowledge of contraindicated vaccines for disease, IBD patients might be injected with some live attenuated vaccines by mistake, which is dangerous since they are more susceptible to infections than healthy individuals and are more likely to get serious infectious complications than non-IBD patients.
Patients with IBD greater than 37 years of age got less vaccine-related information compared to patients less than 28. Studies from Finland and Spain showed that younger patients used the Internet as a source of medicine information more often, which may explain this result.18,19 In the People's Republic of China, internet and WeChat has been widely used in disease education for IBD patients,20 and young patients were better at using social software to get vaccine-related information. Therefore, this result suggests that we should pay more attention to the promotion of middle-aged and elderly patients, such as through hospital brochures. Compared with the ulcerative colitis patients, the Crohn’s disease patients knew more about recommended and contraindicated vaccination, which may be related to the average age of Crohn’s disease patients (31.81±10.69) being younger than those with ulcerative colitis (40.84±13.38). Whether the patient was a CCCF volunteer made a significant difference in the knowledge level of contraindicated vaccination, indicating that the patient organization has more publicity in this regard, but there was no relevance for the recommendation vaccines, probably because patients still rely more on doctors for recommended vaccination.
We then investigated the sources of vaccination information, and most of the information was still from IBD specialists, followed by patient groups and other patients, which is consistent with a Spanish study.21 However, the most trusted resources were IBD specialists, general gastroenterologists, patient associations, and public health agencies. Therefore, it is necessary to strengthen the education of general gastroenterologists and the release of relevant information from the public health agencies. When being asked about the interest in professional and specific information about the vaccination, most patients (92.11%) chose “Yes,” which suggests that the patients were very eager for vaccination knowledge.
Patients with IBD, particularly those on immunosuppressive medications, are at risk for the development of HBV.17 The prevalence of HBV infection in IBD patients in the People's Republic of China was higher than that in Europe or USA.22 HBV vaccination is recommended in all HBV anti-HBcAb seronegative patients with IBD.5 However, our study reports a low vaccination coverage for hepatitis B (18.78%), which is lower than the data reported in the USA (49%).12 Varicella is a highly contagious disease, sometimes with serious complications and can be fatal in IBD patients, but can be effectively prevented by vaccination.13 The risk of VZV infection is increased with all immunosuppressants used in IBD and 5 of 20 cases of varicella proved fatal in a review.23 Our study reported a very low rate of vaccination against varicella (6.6%) among Chinese patients with IBD. In contrast, the rate in IBD patients from the USA (20.96%)24 and Korea (34.9%)25 were much higher.
Our study found that the low rate of HBV vaccination is related to the IBD patients’ concerns about the safety of hepatitis B vaccine. There was no such correlation between varicella vaccination coverage and patients’ concerns about the safety of varicella vaccine, which may be explained by the fact that the patients were less familiar with varicella vaccine. The hepatitis B vaccine belongs to type I vaccines in the People's Republic of China. It is a vaccine that the government provides to citizens free of charge and every Chinese child must be vaccinated after birth. However, the varicella vaccine is a type II vaccine, which is a self-funded and voluntary vaccine. Therefore, it is not surprising that Chinese patients were more familiar with hepatitis B vaccine and had more concerns about the safety of hepatitis B vaccine. That observation is not specific to IBD patients. A study in the People's Republic of China suggested that doubts about the effectiveness or safety is one of the top three reasons for missing HBV vaccination among migrant workers.26 In fact, the hepatitis B vaccine is very safe in the People's Republic of China and the Global Alliance on Vaccine and Immunization (GAVI) partnered with the Government of the People's Republic of China to provide hepatitis B vaccine in 2002,27 which emphasized the need for a correct cognition of hepatitis B vaccine.
Longer diagnosis time and older age were both negative factors for varicella vaccination. Since varicella is a type of live vaccine, the recommendations are for a minimum interval of 3 weeks between vaccination and start of immunosuppression.5 This result may reflect the increasing awareness of Chinese doctors about varicella vaccination in IBD patients in recent years, which is consistent with a previous study in the People's Republic of China.28 Older age was a negative factor for varicella vaccination but not for HBV vaccine, which may reflect the information online or new media was more important in varicella than HBV vaccine.
Interestingly, we found that patients with <2 years’ diagnosis time was associated with higher vaccination rate compared with 2–5 years’ diagnosis time, but not with >5 years’ diagnosis time. This result may be explained that hepatitis B is a type of inactivated vaccine, which can be replanted during immunosuppression.
Notification from doctors was a strong influencing factor both in hepatitis B and varicella vaccination. “No one has recommended me to vaccinate” is also the most common reason for not receiving vaccination in both hepatitis B and varicella. We further investigated the relationship between doctors’ notification and immunosuppressive treatment, and we found that notification rates are significantly higher in patients with immunosuppressive treatment. Previous studies in the USA and the People's Republic of China have suggested that gastroenterologist knowledge of the appropriate immunizations to recommend to the IBD patient is poor,28,29 which may explain this result. Various guidelines have emphasized the importance in vaccinating IBD patients. However, in most cases, clinicians reminded patients of the risk of infection and the need of updated vaccination when the latter were receiving immunosuppressive therapy. Fewer physicians provided the information at the time of diagnosis.
We further investigated the factors that influenced the notification rates at the time of patients’ initial immunosuppressive treatment. In line with previous publications, we found that IBD specialists more often deliver the reminder than non-IBD physicians. There is a need for improving awareness about vaccination in IBD patients among other physicians in the People's Republic of China. The duration of diagnosis was also a factor impacting the notification rate at the time of patient’s initial immunosuppressive treatment. Patients with shorter diagnosis time got higher notification rates, which also reflects the progress of gastroenterologist’s awareness of vaccination in IBD patients in recent years. Using biological therapies as first immunosuppressive treatment positively influenced the notification rates, which may be related to the fact that the People's Republic of China has only begun to use biological therapies in recent years.
Despite the development awareness about vaccination in IBD patients among Chinese physicians, our study calls for a better education improvement for patients and doctors, especially non-IBD physicians. In fact, a previous study suggested that patients’ adherence to vaccination guidelines increased significantly after a period of systematic information from health-care professionals.30 As for patients’ education, patient association propaganda is a very effective means. We should pay more attention to the elderly patients who may get less access to the Internet by using the traditional media such as hospital brochures.
Our study is the first to evaluate influencing factors in hepatitis B and varicella vaccination in Chinese IBD patients. However, there were some limitations which should be acknowledged. First, our participants were mainly from six provinces of China, and the questionnaire was online, thus our sample may not be representative of all IBD patients in China. Secondly, only a small number of participants kept their vaccination records, which could lead to recall desirability biases. Third, only adult IBD patients were included in this study, thus the vaccination of child patients should be investigated in a further study. However, these limitations do not question the low vaccination rate of IBD patients in China.
In conclusion, our study reports a low knowledge level of vaccination in IBD patients in the People's Republic of China and a low self-reported vaccination rate of hepatitis B and varicella in this population. The results of our study emphasize that education, both of IBD patients and gastroenterologists in the People's Republic of China is urgently needed.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142(1):46–54 e42. doi:10.1053/j.gastro.2011.10.001
2. Kaplan GG. The global burden of IBD: from 2015 to 2025. Nat Rev Gastroenterol Hepatol. 2015;12(12):720–727. doi:10.1038/nrgastro.2015.150
3. Cui G, Yuan A. A systematic review of epidemiology and risk factors associated with Chinese inflammatory bowel disease. Front Med. 2018;5:183. doi:10.3389/fmed.2018.00183
4. Ng SC, Shi HY, Hamidi N, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2018;390(10114):2769–2778. doi:10.1016/S0140-6736(17)32448-0
5. Rahier JF, Magro F, Abreu C, et al. Second European evidence-based consensus on the prevention, diagnosis and management of opportunistic infections in inflammatory bowel disease. J Crohns Colitis. 2014;8(6):443–468. doi:10.1016/j.crohns.2013.12.013
6. Ford AC, Peyrin-Biroulet L. Opportunistic infections with anti-tumor necrosis factor-α therapy in inflammatory bowel disease: meta-analysis of randomized controlled trials. Am J Gastroenterol. 2013;108(8):1268–1276. doi:10.1038/ajg.2013.138
7. Melmed GY, Ippoliti AF, Papadakis KA, et al. Patients with inflammatory bowel disease are at risk for vaccine-preventable illnesses. Am J Gastroenterol. 2006;101(8):1834–1840. doi:10.1111/j.1572-0241.2006.00646.x
8. World Health Organization. Hepatitis B. Available from: https://www.who.int/news-room/fact-sheets/detail/hepatitis-b.
9. Loras C, Gisbert JP, Mínguez M, et al. Liver dysfunction related to hepatitis B and C in patients with inflammatory bowel disease treated with immunosuppressive therapy. Gut. 2010;59(10):1340–1346. doi:10.1136/gut.2010.208413
10. Farraye FA, Melmed GY, Lichtenstein GR, Kane SV. ACG clinical guideline: preventive care in inflammatory bowel disease. Am J Gastroenterol. 2017;112(2):241–258. doi:10.1038/ajg.2016.537
11. Malhi G, Rumman A, Thanabalan R, et al. Vaccination in inflammatory bowel disease patients: attitudes, knowledge, and uptake. J Crohns Colitis. 2015;9(6):439–444. doi:10.1093/ecco-jcc/jjv064
12. Ben Musa R, Gampa A, Basu S, et al. Hepatitis B vaccination in patients with inflammatory bowel disease. World J Gastroenterol. 2014;20(41):15358–15366. doi:10.3748/wjg.v20.i41.15358
13. Loras C, Saro C, Gonzalez-Huix F, et al. Prevalence and factors related to hepatitis B and C in inflammatory bowel disease patients in Spain: a nationwide, multicenter study. Am J Gastroenterol. 2009;104(1):57–63. doi:10.1038/ajg.2008.4
14. Tsai SY, Yang TY, Lin CL, Tsai YH, Kuo CF, Kao CH. Increased risk of varicella zoster virus infection in inflammatory bowel disease in an Asian population: a nationwide population-based cohort study. Int J Clin Pract. 2015;69(2):228–234. doi:10.1111/ijcp.12508
15. Chen Y. Perspectives of IBD China: is Crohn’s and colitis foundation model a solution to health care issues for the country? Inflamm Bowel Dis. 2018;24(5):925–929. doi:10.1093/ibd/izy056
16. Wasan SK, Baker SE, Skolnik PR, Farraye FA. A practical guide to vaccinating the inflammatory bowel disease patient. Am J Gastroenterol. 2010;105(6):1231–1238. doi:10.1038/ajg.2009.733
17. Dezfoli S, Melmed GY. Vaccination issues in patients with inflammatory bowel disease receiving immunosuppression. Gastroenterol Hepatol (N Y). 2012;8(8):504–512.
18. Catalán-Serra I, Huguet-Malavés JM, Mínguez M, et al. Information resources used by patients with inflammatory bowel disease: satisfaction, expectations and information gaps. Gastroenterol Hepatol. 2015;38(6):355–363. doi:10.1016/j.gastrohep.2014.09.003
19. Närhi U. Sources of medicine information and their reliability evaluated by medicine users. Pharm World Sci. 2007;29(6):688–694. doi:10.1007/s11096-007-9131-1
20. Yu Q, Xu L, Li L, et al. Internet and WeChat used by patients with Crohn’s disease in China: a multi-center questionnaire survey. BMC Gastroenterol. 2019;19(1):97. doi:10.1186/s12876-019-1011-3
21. Martín Fernández C, Maroto Martín C, Fernández Salazar L. Using the internet to evaluate the opinion of patients with inflammatory bowel disease with regard to the available information. Rev Esp Enferm Apar Dig. 2018;110(5):274–284. doi:10.17235/reed.2018.5331/2017
22. Chen D, Luo S, Ben Q, Lu L, Wan X, Wu J. Prevalence of hepatitis B and C and factors for infection and nonimmune in inflammatory bowel disease patients in China. Eur J Gastroenterol Hepatol. 2017;29(5):509–515. doi:10.1097/MEG.0000000000000838
23. Cullen G, Baden RP, Cheifetz AS. Varicella zoster virus infection in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18(12):2392–2403. doi:10.1002/ibd.22950
24. Khan N, Trivedi C, Kavani H, Lewis J, Yang YX. Frequency of herpes zoster vaccination among inflammatory bowel disease patients. Inflamm Bowel Dis. 2019;25(2):345–351. doi:10.1093/ibd/izy232
25. Yun HS, Min YW, Chang DK, et al. Factors associated with vaccination among inflammatory bowel disease patients in Korea. Korean J Gastroenterol. 2013;61(4):203–208. doi:10.4166/kjg.2013.61.4.203
26. Xiang H, Tang XJ, Xiao M, et al. Study on status and willingness towards hepatitis B vaccination among migrant workers in chongqing, china: a cross-sectional study. Int J Env Res Pub He. 2019;16(20). doi:10.3390/ijerph16204046.
27. Liang X, Cui F, Hadler S, et al. Origins, design and implementation of the China GAVI project. Vaccine. 2013;31(Suppl 9):J8–14. doi:10.1016/j.vaccine.2012.12.019
28. Li SY, Li PW, Shen B, et al. Insufficient awareness and vaccination practices for inflammatory bowel disease patients in China: a multi-center survey of Chinese gastroenterologists. J Dig Dis. 2020;21(1):46–51. doi:10.1111/1751-2980.12834
29. Wasan SK, Coukos JA, Farraye FA. Vaccinating the inflammatory bowel disease patient: deficiencies in gastroenterologists knowledge. Inflamm Bowel Dis. 2011;17(12):2536–2540. doi:10.1002/ibd.21667
30. Christensen KR, Steenholdt C, Buhl SS, Ainsworth MA, Thomsen O, Brynskov J. Systematic information to health-care professionals about vaccination guidelines improves adherence in patients with inflammatory bowel disease in anti-TNFα therapy. Am J Gastroenterol. 2015;110(11):1526–1532. doi:10.1038/ajg.2015.162
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
