Back to Journals » Clinical Ophthalmology » Volume 16
Insights into Current Management Strategies for Dysthyroid Optic Neuropathy: A Review
Authors Tagami M, Honda S, Azumi A
Received 7 January 2022
Accepted for publication 10 March 2022
Published 18 March 2022 Volume 2022:16 Pages 841—850
DOI https://doi.org/10.2147/OPTH.S284609
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Mizuki Tagami,1 Shigeru Honda,1 Atsushi Azumi2
1Department of Ophthalmology and Visual Sciences, Graduate School of Medicine, Osaka City University, Osaka, Japan; 2Ophthalmology Department and Eye Center, Kobe Kaisei Hospital, Kobe, Hyogo, Japan
Correspondence: Mizuki Tagami, Department of Ophthalmology and Visual Science, Graduate School of Medicine, Osaka City University, 1-5-7 Asahimachi, Abeno-ku, Osaka-shi, 545-8586, Japan, Tel/Fax +81-6-6645-3867, Email [email protected]
Abstract: Dysthyroid optic neuropathy (DON) is a potentially sight-threatening eye disease associated with Graves’ orbitopathy (GO). DON is not common in GO patients, reportedly occurring in only about 5% of patients. The pathogenesis of severe DON is considered to involve both muscular nerve strangulation and impaired blood flow. There is some objective grading of physical examination findings and the severity of GO, including a clinical activity score (CAS) and EUropean Group On Graves’ Orbitopathy (EUGOGO), but no specialized protocol completely characterizes DON. Most clinicians have decided that the combination of clinical activity findings, including visual acuity, color vision, and central critical fusion frequency, and radiological findings, including magnetic resonance imaging (MRI), can be used to diagnose DON. MRI has the most useful findings, with T2-weighted and fat-suppressed images using short-tau inversion recovery (STIR) sequences enabling detection of extraocular changes including muscle and/orbital fat tissue swelling and inflammation and, therefore, disease activity. The first-choice treatment for DON is intravenous administration of steroids, with or without radiotherapy. Unfortunately, refractoriness to this medical treatment may indicate the need for immediate orbital decompression within 2 weeks. Especially in the acute phase of DON, thyroid function is often unstable, and the surgeon must always assume the risk of general anesthesia and intra- and post-operative management. In addition, there are currently many possible therapeutic options, including molecular-targeted drugs. The early introduction and combination of these immunomodulators, including Janus kinase inhibitors and insulin-like growth factor-1 receptor antibody (teprotumumab), may be effective for GO with DON. However, this is still under investigation, and the number of case reports is small. It is possible that these options could reduce systemic adverse events due to unfocused glucocorticoid administration. The pathophysiology of DON is not yet fully understood, and further studies of its treatment and long-term visual function prognosis are needed.
Keywords: Graves’ orbitopathy, orbital decompression, dysthyroid optic neuropathy, magnetic resonance imaging, MRI, visual outcome, teprotumumab, Janus kinase inhibitors
Introduction
Graves’ orbitopathy (GO) is a potentially sight-threatening eye disease, particularly with the associated dysthyroid optic neuropathy (DON).1–3 DON may also cause loss of vision in patients with GO.4–6 DON is not common in GO patients, having been reported to occur in only about 5% of patients.7 The 2016 European Thyroid Association/European Group on Graves’ Orbitopathy Guidelines for the Management of Graves’ Orbitopathy (EUGOGO) recommended treatment with high-dose (0.5–1.0 g/day) intravenous steroids for 1 or 2 weeks. If there is poor treatment response, prompt orbital decompression is required.8 Regarding orbital decompression, various methods and options have become available in the last 20 years, and there are reports that the long-term prognosis is also acceptable.9–12 Currently, there is no consensus on the most appropriate evaluation, but new reports on evaluations of GO are being published regularly.13–15
Detailed evaluation of the use of the insulin-like growth factor-1 receptor antibody (IGF-IR) for DON has not yet been performed, but clinical trials and reports have shown better outcomes with respect to proptosis, Clinical Activity Score (CAS), and quality of life.16–18
In this paper, the current management strategies for DON are presented and reviewed.
Epidemiology of DON
DON is not common in GO patients, and reportedly only occurs in about 5% of patients.19 In addition, the DON diagnosis might be missed in patients without proptosis. GO occurs before, during, and after the onset of hyperthyroidism, but less often in patients with hyperthyroidism or hypothyroidism.19 As an example of this complexity, the median CAS for this series was 4/7, compared to 3 or less in more than 25% of DON patients. Indeed, the diagnosis is often unclear.20 In previous series, only moderate soft tissue signs were reported in DON patients. The visual acuity of 12 eyes with a CAS of 4 or less was 0.5 or less.21 The sex distribution of GO patients shows a female-to-male ratio of 5:1. In other words, patients with DON are more likely to be male and over 60 years of age.22,23
Risk Factors for DON
Several genes involved in immunomodulation, thyroid metabolism, and other mechanisms are involved in the etiology of GO, but none have been found to be necessary, sufficient, or specific for the development of DON.24
In particular, there are many reports that human leucocyte antigen (HLA) gene polymorphisms and, specifically, amino-acid signatures of HLA are associated with the development of thyroid autoimmune diseases and GO.25,26 Okada et al reported HLA imputation to genome-wide association study data for Graves’ disease in Japanese patients (n = 9003) and found that amino acid polymorphisms of multiple class I and class II HLA genes contributed independently to disease risk (HLA-DPB1, HLA-A, HLA-B, and HLA-DRB1; P < 2.3×10−6); HLA-DPB1 may have a prominent impact on disease development (P = 1.6×10−42).25 The relationship between HLA-DPB1 and GO and DON could be a meaningful direction to explore in the future.
Other risk factors are associated with GO, including age, sex, genetic background, smoking, and thyroid dysfunction including hyperthyroidism. These risk factors might also be associated with DON. Smoking, particularly being a current smoker, may be a risk factor for DON as well, although the evidence is not as overwhelming. A recent review of 604 patients with GO reported an odds ratio of 1.5 for current smokers developing DON, but it was not statistically significant.27 However, another retrospective study reported that smoking could be an important predictor in the development of both severe GO (OR = 6.5) and DON (OR = 10.0).28 There are multiple reports of high levels of thyroid autoantibodies, including thyroid stimulating hormone receptor antibody (TRAb) or thyroid stimulating antibody(TSAb), and the severity of DON, but the numbers were small in case series.9,29
Diabetes mellitus (DM) is considered one of the risk factors for GO. In one study, though only 3.1% of patients with GO had DM, and 3.9% developed DON, 33.3% of GO patients with DM eventually developed DON.30 Of course, it is considered that there are many cases that need to be excluded, such as patients with diabetic optic neuropathy and those with secondary glaucoma, such as neovascular glaucoma.31,32
Radioisotope (RI) therapy may increase the risk of GO progression compared to medication or surgical thyroidectomy.22 In other words, RI could increase DON risk, although this still requires further consideration.
Age and sex also appear to affect severe GO, including DON. Increased age correlates with increased severity of thyroid eye disease, and age may be one of the greatest risk factors for DON.23,25 Patients with DON are significantly older than patients with GO without DON, with an average age of 61 years. In another study, patients with GO developed DON, and patients with DON were older than those without DON (54 vs 46 years).33 Several other studies also showed a strong correlation between age and DON.34,35 For every decade increase in age of onset of GO, the odds of developing DON could increase by over about 60%.27 Aging might also affect the response to DON treatment, with younger age predicting a better visual outcome.36 Male sex has also been correlated with the development of DON, especially with aging.22,24,33
Pathological Condition of DON
The pathogenesis of DON has been considered to involve mechanical and inflammatory aspects. The most widely accepted mechanism is secondary to an apex compartment syndrome due to orbital extraocular tissue with orbital fibroblasts increasing and extraocular muscle swelling compressing the optic nerve around the common tendinous ring.33,37
Orbital fibroblasts mainly affect GO and DON.27 TSAb and TRAb serve as the autoimmune targets in GO, and antibody levels correlate with disease activity.38 Orbital fibroblasts in GO express higher hormone receptor levels compared to those in healthy subjects.39
The IGF-IR is strongly expressed in GO orbital tissue.13,14 It forms a complex with the TSH receptor and takes control of downstream signaling.15 Teprotumumab is a fully humanized immunoglobulin G1 monoclonal inhibitory antibody that binds to the extracellular portion of IGF-IR and blocks its activation and signaling by endogenous ligands. The binding of teprotumumab also draws IGF-IR into the cell and induces its degradation. It also stimulates production of more than 90% of the receptor proteins present on the cell surface.15 Doppler ultrasonography shows the superior ophthalmic vein as the most susceptible to changes, with decreased flow in severe orbitopathy.40 This mechanism suggests that one trigger for DON may be ischemia due to impaired blood flow. The pathogenesis of severe DON is considered to involve both muscular nerve strangulation and impaired blood flow.
Clinical Diagnosis of DON
There is some objective grading of physical examination findings and the severity of GO, including CAS and EUGOGO (Tables 1 and 2), but no specialized protocol completely characterizes DON.8,41
 |
Table 1 Clinical Activity Score (CAS) |
 |
Table 2 Classification of the Severity of Graves’ Orbitopathy (GO) |
Most clinicians have decided that the combination of clinical activity findings, including visual acuity, color vision, and central critical fusion frequency (CFF), and radiological findings, including magnetic resonance imaging (MRI), can be used to diagnose DON.9 Tissue enlargement in patients with DON often results in a local compartment phenomenon at the orbital apex, rather than severe proptosis, and patients with DON may not demonstrate clear orbital inflammation.42 Decreasing visual acuity is nonspecific, but it is decreased more in eyes with DON than in those with simple GO. Previous studies have shown less than 20/40 visual acuity in patients with DON compared to 97% in patients without DON.33 In addition, afferent pupillary defects in GO patients with DON are very specific.33 However, they may be absent in bilateral DON, or they may occur for reasons other than DON.
Color vision changes represent an early sign of optic nerve compression. A recent article reported that blue-yellow (tritan) deficiency is a sensitive indicator of dysthyroid optic neuropathy. Thus, in cases with suspected DON, a color vision test for tritan deficiency is an essential tool for prompt and correct assessment of DON.43
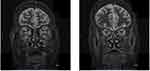
MRI has the most useful findings. T2-weighted and fat-suppressed images using STIR sequences enable detection of extraocular muscle/orbital fat and stromal tissue inflammation, and, therefore, STIR-MRI reflects disease activity.44–47 This makes MRI ideal for distinguishing active inflammatory GO from fibrotic late-stage disease and is critical for determining the type and the timing for those needing immediate treatment (Figure 1).44–47 Computed tomography (CT) is better at evaluating bony orbital anatomy, so it may be critically useful for surgical planning, including urgent orbital decompression.
Intraocular Optical Coherence Tomography (OCT) Changes of Visual Function in DON
Patients with DON have some variational scotomata and visual field defects.22,48 In recent years, OCT imaging findings of the retinal nerve fiber layer in eye diseases including glaucoma have been actively compared with visual fields and visual function.49–52 Some studies investigated the effects of optic nerve compression on the peripapillary retinal nerve fiber layer (RNFL) thickness in eyes with DON.53,54 The visual prognosis of orbital decompression surgery in patients with DON has been studied. A previous article reported that RNFL thickness can be used as a prognostic factor for DON before decompression surgery.54 Another article reported that RNFL thickness in each quadrant was negatively correlated with postoperative changes in best corrected visual acuity (BCVA) and visual field defects and positively correlated with changes of MD and P100 amplitude at 60 arcmin. These results suggest that OCT findings are correlated with visual function in DON.55
The advantage of OCT findings for DON is that it can predict future visual acuity from the inside of the eye noninvasively, meaning it is likely to be used more often in the future.
Treatment Options for DON: Overview
The majority of GO patients have mild or moderate symptoms that can be improved with conservative treatment or less invasive treatment.56 For patients with moderate-to-severe and active GO, the first-choice treatment is intravenous administration of steroids, with or without radiotherapy. Unfortunately, refractoriness to this medical treatment may indicate the immediate need for orbital decompression within 2 weeks, especially in the acute phase of DON.19 Based on previous studies, the first-line treatment of DON is high-dose (500 or 1000 mg/day) intravenous methylprednisolone for three consecutive days or during the first week, which can be repeated for another one week (Figure 2).5,19,57 When considering treatment, hyperthyroidism needs to be taken into account, since dysthyroidism may precipitate disease progression, and the treatment range or intensity may affect disease activity, though this might still be unclear.19 Other immunosuppressive and biologic options, such as methotrexate, rapamycin, adalimumab, rituximab, and teprotumumab have been discussed, but they are generally not considered first-line therapies.1–3,6,58 Rehabilitative surgery including strabismus surgery is best performed in the inactive disease phase, but it may be considered an urgent surgical treatment option for cases of DON refractory to medical treatment.59
 |
Figure 2 Algorithm for the management of sight-threatening Graves’ orbitopathy including DON. The first-line treatment for optic neuropathy is high-dose IV methylprednisolone or steroid pulse therapy (single doses of 500 to 1000 mg) for three consecutive days, or preferably and for safety reasons, every second day (alternate days) during the first week, which can be repeated for a second week. When the response is absent or poor, with deterioration in visual acuity or CFF, urgent orbital decompression surgery will be needed. Data from Bartalena L, Kahaly GJ, Baldeschi L, et al. The 2021 European Group on Graves’ orbitopathy (EUGOGO) clinical practice guidelines for the medical management of Graves’ orbitopathy. Eur J Endocrinol. 2021;185(4):G43–G67.19 |
There are currently some suggested guidelines for treating DON. The American Society of Ophthalmic and Reconstructive Surgery (ASOPRS) members chose both oral (about 40% of members) and IV (about 50% of members) steroids as first-line treatments for severe GO.60 In contrast, Japanese physicians prefer IV over oral steroids as first-line therapy.9,61 ASOPRS members also chose orbital decompression (83%), RT (70%), biologic agents (33%), and intra-orbital local steroid injections (28%). The treatment of sight-threatening GO could consist of a combination of these treatments, including urgent orbital decompression.
Medical Treatment
Steroid pulse or high-dose IV 0.5–1.0 g per day glucocorticoid is standard first-line treatment of GO with DON, based on objective ophthalmic examination (visual acuity, CFF, visual fields, color vision).5,19,43,57 A higher cumulative dose of over 7.5 g (starting with 0.5–1.0 g as a single IV dose) is only considered for the most severe cases within the spectrum of severe active-phase GO including DON, as the higher dose bears a higher risk of drug-induced adverse events.62
At present, no significant difference between 0.5 g and 1 g has been confirmed, but it is necessary to first give 1 g of steroid pulse for 1 week and confirm the result. If there is an improvement in visual function in one week, a steroid pulse in the second week may be acceptable management. However, there should be no hesitation to perform urgent surgical treatment, even if the patient is not refractory to medical treatment. In addition, the authors argue that 3 courses (9 g) of steroid pulse therapy, 1.0 g per day, should be given very carefully due to the problems of systemic adverse events.
Orbital RT for GO has been reported in several randomized, controlled trials (RCTs), which found it to be more effective than sham irradiation in improving diplopia and ductions, although its efficacy was limited in two other RCTs.63–66 In another RCT, orbital RT was found to be as effective as oral prednisone, and other RCTs have shown that orbital RT synergistically potentiates the effects of oral steroids.67–69 Even in RCTs, the synergistic effect of the combined use of steroids and RT is not clear. Future studies might address the question of whether the combination of steroid pulse or high-dose IV 0.5–1.0 g per day of glucocorticoids and RT might represent a first-line, combined treatment for active GO.
We have a possible strategy of adding orbital RT as much as possible in acute GO with or without DON (Figure 2).
Surgical Management
The essence of urgent surgery for DON is to decrease orbital soft tissue compression and/or expand bony volume to decompress the optic nerve at the orbital apex. Orbital fat can be removed from any location within the orbit, and bony expansion can occur along any orbital wall. The inferomedial wall extends deeper to the apex, so that it is usually considered the first approach to decompress apex compression; in addition, lateral wall bone removal and/or fat-only removal may also decompress the apex.9 Several approaches to the medial wall have been described, including transantral, transcutaneous, endonasal, and transcaruncular approaches.70–72 A previous report showed that, in patients with DON, an enlarged orbital fat compartment may be one of the causes of optic neuropathy, and fat decompression could be an alternative surgical procedure to bony decompression.73
However, a small RCT showed that DON patients who underwent immediate decompression did not have a better outcome than those given IV glucocorticoids as first-line treatment.5 In addition, this previous report discussed the complications of orbital surgery; a total of 1665 patients underwent orbital surgery, with 14 patients sustaining severe vision loss (overall risk, 0.84%), and the risk of a blinding surgical complication was significantly higher for patients with optic canal decompression surgery (risk, 15%).74 The endoscopic endonasal approach effectively treats DON, but it may induce new diplopia in about 70% of cases, and endoscopic endonasal decompression might be difficult in inferior-medial hard bony decompression.75,76
Especially in the acute phase of DON, thyroid function is often unstable, and the surgeon must always consider the risk of general anesthesia and intraoperative and postoperative management.77 Considering this, it may be a necessary or useful treatment option to perform medial (transcaruncular) decompression without deep lateral decompression within a few hours, since it requires a relatively short operation time, and to prevent thyroid storm by intensive management in a high care unit (HCU) for several days after urgent surgery.
Possible New Treatment Strategy for DON
In recent years, strong evidence has implicated the IGF-IR in the pathogenesis of GO.13–15 The safety and efficacy of teprotumumab were evaluated sequentially in two RCTs, including 170 patients with active GO.16,17 Both trials had similar designs, and patients were randomly assigned, with 83 patients receiving teprotumumab every 3 weeks IV for eight doses over 24 weeks and 87 patients receiving placebo. Seventy-three percent in the teprotumumab group (vs 14% in the placebo group) were overall responders, with both CAS and proptosis improvement. Individually, the proptosis response was much more common in the teprotumumab group. Recently, systematic analyses and off-treatment follow-up results from the two RCTs were published.18 Although the clinical use of teprotumumab for DON has not been sufficiently evaluated, it is considered to have become a very important future treatment option.
Janus kinase (JAK) inhibitors are molecular agents that inhibit the JAK receptors.78 JAK-mediated pathways are involved in the pathogenesis of several autoimmune diseases including rheumatoid arthritis, inflammatory bowel disease, Graves’ disease, and other immune-mediated inflammatory diseases and infectious disease with cytokine storm including COVID-19.78–81 There are some members of the tyrosine kinase family of protein kinases. Tofacitinib and baricitinib are first-generation JAK inhibitors.82,83 Tofacitinib inhibits JAK 1 and JAK 3, and baricitinib inhibits JAK 1 and JAK 2. In combination with teprotumumab, it may suppress much of the inflammatory cascade in GO. The method of administration is also oral, which is considered to be a highly acceptable option. It may be a possible treatment by inhibiting IGF-IR and the JAK-STAT pathway, instead of only IV glucocorticoid therapies in the future.
Conclusion
There is no doubt that DON is the most severe degree of GO. Various treatment options have been considered. In particular, high levels of evidence support rapid treatment with IV glucocorticoids, including steroid pulse therapy. It is also important that there is the possibility of prompt surgical intervention in cases of non-response. Fortunately, if the response to IV steroid therapy is good, the combined use of RT may reduce the possibility of recurrence. Unfortunately, refractoriness to this medical treatment may indicate the need for immediate orbital decompression within 2 weeks.
Current therapeutic options also include molecular-targeted drugs. Early introduction and use in combination with these immunomodulators including JAK inhibitors and teprotumumab may provide efficacy, reduce systemic adverse events due to unfocussed glucocorticoid administration, and contribute to patient quality of life.
As many patients with DON have unstable thyroid function, it is considered essential that an ophthalmologist and an endocrinologist work together in both critical care and general care settings (Figure 3).
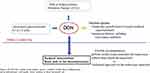 |
Figure 3 Schema of this review: Summary. |
Data Sharing Statement
All data included in this study are available from the corresponding author on reasonable request.
Acknowledgments
The first author would like to express thanks to Sentaro Kusuhara, M.D., Ph.D., for his writing advice.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bahn RS, Heufelder AE, Heufelder AE. Pathogenesis of Graves’ ophthalmopathy. N Engl J Med. 1993;329(20):1468–1475. doi:10.1056/NEJM199311113292007
2. Bartalena L, Tanda ML. Graves’ ophthalmopathy. N Engl J Med. 2009;360(10):994–1001. doi:10.1056/NEJMcp0806317
3. Bahn RS. Graves’ ophthalmopathy. N Engl J Med. 2010;362(8):726–738. doi:10.1056/NEJMra0905750
4. Currò N, Covelli D, Vannucchi G, et al. Therapeutic outcomes of high-dose intravenous steroids in the treatment of dysthyroid optic neuropathy. Thyroid. 2014;24(5):897–905. doi:10.1089/thy.2013.0445
5. Wakelkamp IM, Baldeschi L, Saeed P, et al. Surgical or medical decompression as a first-line treatment of optic neuropathy in Graves’ ophthalmopathy? A randomized controlled trial. Clin Endocrinol (Oxf). 2005;63(3):323–328. doi:10.1111/j.1365-2265.2005.02345.x
6. Saeed P, Rad ST, Bisschop PH. Dysthyroid optic neuropathy. Ophthal Plast Reconstr Surg. 2018;34(4S):S60–S7. doi:10.1097/IOP.0000000000001146
7. Bartley GB, Fatourechi V, Kadrmas EF, et al. The treatment of Graves’ ophthalmopathy in an incidence cohort. Am J Ophthalmol. 1996;121(2):200–206. doi:10.1016/S0002-9394(14)70585-9
8. Bartalena L, Baldeschi L, Boboridis K, et al. The 2016 European Thyroid Association/European Group on Graves’ orbitopathy guidelines for the management of Graves’ orbitopathy. Eur Thyroid J. 2016;5(1):9–26. doi:10.1159/000443828
9. Tagami M, Honda S, Azumi A. Preoperative clinical factors and visual outcomes following orbital decompression with dysthyroid optic neuropathy. BMC Ophthalmol. 2020;20(1):30. doi:10.1186/s12886-020-1314-8
10. Tu Y, Wu S, Pan Z, et al. Endoscopic transconjunctival deep lateral wall decompression for thyroid-associated orbitopathy: a minimally invasive alternative: transconjunctival endoscopic with wall decompression for TAO. Am J Ophthalmol. 2021;235:71–79. doi:10.1016/j.ajo.2021.08.013
11. Singh S, Curragh DS, Selva D. Augmented endoscopic orbital apex decompression in dysthyroid optic neuropathy. Eye (Lond). 2019;33(10):1613–1618. doi:10.1038/s41433-019-0464-5
12. Carter KD, Frueh BR, Hessburg TP, Musch DC. Long-term efficacy of orbital decompression for compressive optic neuropathy of Graves’ eye disease. Ophthalmology. 1991;98(9):1435–1442. doi:10.1016/S0161-6420(91)32115-8
13. Tsui S, Naik V, Hoa N, et al. Evidence for an association between thyroid-stimulating hormone and insulin-like growth factor 1 receptors: a tale of two antigens implicated in Graves’ disease. J Immunol. 2008;181:4397–4405. doi:10.4049/jimmunol.181.6.4397
14. Douglas R, Naik V, Hwang C, et al. B cells from patients with Graves’ disease aberrantly express the IGF-1 receptor: implications for disease pathogenesis. J Immunol. 2008;181:5768–5774. doi:10.4049/jimmunol.181.8.5768
15. Smith T, Janssen J. Insulin-like growth factor-I receptor and thyroid-associated ophthalmopathy. Endocr Rev. 2019;40:236–267. doi:10.1210/er.2018-00066
16. Smith T, Kahaly G, Ezra D, et al. Teprotumumab for thyroid-associated ophthalmopathy. N Engl J Med. 2017;376:1748–1761. doi:10.1056/NEJMoa1614949
17. Douglas R, Kahaly G, Patel A, et al. Teprotumumab for the treatment of active thyroid eye disease. N Engl J Med. 2020;382:341–352. doi:10.1056/NEJMoa1910434
18. Kahaly G, Douglas R, Holt R, et al. Teprotumumab for patients with active thyroid eye disease: a pooled data analysis, subgroup analyses, and off-treatment follow-up results from two randomised, double-masked, placebo-controlled, multicentre trials. Lancet Diabetes Endocrinol. 2021;9:360–372.
19. Bartalena L, Kahaly GJ, Baldeschi L, et al. The 2021 European Group on Graves’ orbitopathy (EUGOGO) clinical practice guidelines for the medical management of Graves’ orbitopathy. Eur J Endocrinol. 2021;185(4):G43–g67. doi:10.1530/EJE-21-0479
20. McKeag D, Lane C, Lazarus JH, et al. Clinical features of dysthyroid optic neuropathy: a European Group on Graves’ Orbitopathy (EUGOGO) survey. Br J Ophthalmol. 2007;91(4):455–458. doi:10.1136/bjo.2006.094607
21. Ohtsuka K, Nakamura Y. Results of transmedial-canthal ethmoidal decompression for severe dysthyroid optic neuropathy. Jpn J Ophthalmol. 1999;43(5):426–432. doi:10.1016/S0021-5155(99)00091-X
22. Trobe JD, Glaser JS, Laflamme P. Dysthyroid optic neuropathy. Clinical profile and rationale for management. Arch Ophthalmol. 1978;96(7):1199–1209. doi:10.1001/archopht.1978.03910060033007
23. Lazarus JH. Epidemiology of Graves’ orbitopathy (GO) and relationship with thyroid disease. Best Pract Res Clin Endocrinol Metab. 2012;26(3):273–279. doi:10.1016/j.beem.2011.10.005
24. Stan MN, Bahn RS. Risk factors for development or deterioration of Graves’ ophthalmopathy. Thyroid. 2010;20(7):777–783. doi:10.1089/thy.2010.1634
25. Okada Y, Momozawa Y, Ashikawa K, et al. Construction of a population-specific HLA imputation reference panel and its application to Graves’ disease risk in Japanese. Nat Genet. 2015;47(7):798–802. doi:10.1038/ng.3310
26. Huang X, Liu G, Mei S, et al. Human leucocyte antigen alleles confer susceptibility and progression to Graves’ ophthalmopathy in a Southern Chinese population. Br J Ophthalmol. 2021;105(10):1462–1468. doi:10.1136/bjophthalmol-2020-317091
27. Khong JJ, Finch S, De Silva C, et al. Risk factors for Graves’ orbitopathy; the Australian Thyroid-Associated Orbitopathy Research (ATOR) study. J Clin Endocrinol Metab. 2016;101(7):2711–2720. doi:10.1210/jc.2015-4294
28. Lee JH, Lee SY, Yoon JS. Risk factors associated with the severity of thyroid-associated orbitopathy in Korean patients. Korean J Ophthalmol. 2010;24(5):267–273. doi:10.3341/kjo.2010.24.5.267
29. Deng Z, Chen L, Tan J, et al. Combination model of thyrotrophin receptor antibody and volumetric orbital apex crowding index as an indicator of dysthyroid optic neuropathy. Dis Markers. 2021;2021:9964232. doi:10.1155/2021/9964232
30. Kalmann R, Mourits MP. Diabetes mellitus: a risk factor in patients with Graves’ orbitopathy. Br J Ophthalmol. 1999;83(4):463–465. doi:10.1136/bjo.83.4.463
31. Hayreh SS, Zimmerman MB. Nonarteritic anterior ischemic optic neuropathy: clinical characteristics in diabetic patients versus nondiabetic patients. Ophthalmology. 2008;115(10):1818–1825. doi:10.1016/j.ophtha.2008.03.032
32. Hayreh SS. Neovascular glaucoma. Prog Retin Eye Res. 2007;26(5):470–485. doi:10.1016/j.preteyeres.2007.06.001
33. Neigel JM, Rootman J, Belkin RI, et al. Dysthyroid optic neuropathy. The crowded orbital apex syndrome. Ophthalmology. 1988;95(11):1515–1521. doi:10.1016/S0161-6420(88)32978-7
34. Mensah A, Vignal-Clermont C, Mehanna C, et al. Dysthyroid optic neuropathy: atypical initial presentation and persistent visual loss. Orbit. 2009;28(6):354–362. doi:10.3109/01676830903104728
35. Kendler DL, Lippa J, Rootman J. The initial clinical characteristics of Graves’ orbitopathy vary with age and sex. Arch Ophthalmol. 1993;111(2):197–201. doi:10.1001/archopht.1993.01090020051022
36. Miśkiewicz P, Rutkowska B, Jabłońska A, et al. Complete recovery of visual acuity as the main goal of treatment in patients with dysthyroid optic neuropathy. Endokrynol Pol. 2016;67(2):166–173. doi:10.5603/EP.a2016.0018
37. Bahn RS. Current insights into the pathogenesis of Graves’ ophthalmopathy. Horm Metab Res. 2015;47(10):773–778. doi:10.1055/s-0035-1555762
38. Ponto KA, Diana T, Binder H, et al. Thyroid-stimulating immunoglobulins indicate the onset of dysthyroid optic neuropathy. J Endocrinol Invest. 2015;38(7):769–777. doi:10.1007/s40618-015-0254-2
39. Iyer S, Bahn R. Immunopathogenesis of Graves’ ophthalmopathy: the role of the TSH receptor. Best Pract Res Clin Endocrinol Metab. 2012;26(3):281–289. doi:10.1016/j.beem.2011.10.003
40. Walasik-Szemplińska D, Pauk-Domańska M, Sanocka U, Sudoł-Szopińska I. Doppler imaging of orbital vessels in the assessment of the activity and severity of thyroid-associated orbitopathy. J Ultrason. 2015;15(63):388–397. doi:10.15557/JoU.2015.0036
41. Mourits MP, Prummel MF, Wiersinga WM, Koornneef L. Clinical activity score as a guide in the management of patients with Graves’ ophthalmopathy. Clin Endocrinol (Oxf). 1997;47(1):9–14. doi:10.1046/j.1365-2265.1997.2331047.x
42. Soares-Welch CV, Fatourechi V, Bartley GB, et al. Optic neuropathy of Graves disease: results of transantral orbital decompression and long-term follow-up in 215 patients. Am J Ophthalmol. 2003;136(3):433–441. doi:10.1016/S0002-9394(03)00233-2
43. Garip Kuebler A, Halfter K, Reznicek L, et al. A pathological indicator for dysthyroid optic neuropathy: tritan color vision deficiency. Graefes Arch Clin Exp Ophthalmol. 2021;259(11):3421–3426. doi:10.1007/s00417-021-05227-8
44. Kirsch E, von Arx G, Hammer B. Imaging in Graves’ orbitopathy. Orbit. 2009;28(4):219–225.
45. Hu H, Xu XQ, Wu FY, et al. Diagnosis and stage of Graves’ ophthalmopathy: efficacy of quantitative measurements of the lacrimal gland based on 3-T magnetic resonance imaging. Exp Ther Med. 2016;12(2):725–729. doi:10.3892/etm.2016.3389
46. Mayer EJ, Fox DL, Herdman G, et al. Signal intensity, clinical activity and cross-sectional areas on MRI scans in thyroid eye disease. Eur J Radiol. 2005;56(1):20–24. doi:10.1016/j.ejrad.2005.03.027
47. Yokoyama N, Nagataki S, Uetani M, et al. Role of magnetic resonance imaging in the assessment of disease activity in thyroid-associated ophthalmopathy. Thyroid. 2002;12(3):223–227. doi:10.1089/105072502753600179
48. Feldon SE, Muramatsu S, Weiner JM. Clinical classification of Graves’ ophthalmopathy: identification of risk factors for optic neuropathy. Arch Ophthalmol. 1984;102(10):1469–1472. doi:10.1001/archopht.1984.01040031189015
49. Leung CK-S, Cheung CYL, Weinreb RN, et al. Evaluation of retinal nerve fiber layer progression in glaucoma: a study on optical coherence tomography guided progression analysis. Invest Ophthalmol Vis Sci. 2010;51(1):217–222. doi:10.1167/iovs.09-3468
50. Amini N, Nowroozizadeh S, Cirineo N, et al. Influence of the disc–fovea angle on limits of rnfl variability and glaucoma discrimination. Invest Ophthalmol Vis Sci. 2014;55(11):7332–7342. doi:10.1167/iovs.14-14962
51. Kanamori A, Nakamura M, Escano MF, et al. Evaluation of the glaucomatous damage on retinal nerve fiber layer thickness measured by optical coherence tomography. Am J Ophthalmol. 2003;135(4):513–520. doi:10.1016/S0002-9394(02)02003-2
52. Kanamori A, Nakamura M, Matsui N, et al. Optical coherence tomography detects characteristic retinal nerve fiber layer thickness corresponding to band atrophy of the optic discs. Ophthalmology. 2004;111(12):2278–2283. doi:10.1016/j.ophtha.2004.05.035
53. Park KA, Kim YD, In woo K, et al. Optical coherence tomography measurements in compressive optic neuropathy associated with dysthyroid orbitopathy. Graefes Arch Clin Exp Ophthalmol. 2016;254(8):1617–1624. doi:10.1007/s00417-016-3335-9
54. Park KA, Kim YD, Woo KI. Changes in optical coherence tomography measurements after orbital wall decompression in dysthyroid optic neuropathy. Eye (Lond). 2018;32(6):1123–1129. doi:10.1038/s41433-018-0051-1
55. Cheng S, Yu Y, You Y, et al. Retinal nerve fiber layer thickness measured by optical coherence tomography predicts visual recovery after orbital decompression for dysthyroid optic neuropathy. Int Ophthalmol. 2021;41(9):3121–3133. doi:10.1007/s10792-021-01877-z
56. Gillespie EF, Smith TJ, Douglas RS. Thyroid eye disease: towards an evidence base for treatment in the 21st century. Curr Neurol Neurosci Rep. 2012;12(3):318–324. doi:10.1007/s11910-012-0256-9
57. Zang S, Ponto K, Pitz S, Kahaly G. Dose of intravenous steroids and therapy outcome in Graves’ orbitopathy. J Endocrinol Invest. 2011;34:876–880. doi:10.1007/BF03346732
58. Blandford AD, Zhang D, Chundury RV, Perry JD. Dysthyroid optic neuropathy: update on pathogenesis, diagnosis, and management. Expert Rev Ophthalmol. 2017;12(2):111–121. doi:10.1080/17469899.2017.1276444
59. Bartalena L. Graves’ disease: complications. In: Feingold KR, Anawalt B, Boyce A, editors. Endotext. South Dartmouth (MA): MDText.com, Inc. Copyright © 2000–2021, MDText.com, Inc.; 2000.
60. Perumal B, Meyer DR. Treatment of severe thyroid eye disease: a survey of the American Society of Ophthalmic Plastic and Reconstructive Surgery (ASOPRS). Ophthalmic Plast Reconstr Surg. 2015;31(2):127–131. doi:10.1097/IOP.0000000000000216
61. Ito M, Takahashi Y, Katsuda E, et al. Predictive factors of prognosis after radiation and steroid pulse therapy in thyroid eye disease. Sci Rep. 2019;9(1):2027. doi:10.1038/s41598-019-38640-5
62. Bartalena L, Krassas G, Wiersinga W, et al. Efficacy and safety of three different cumulative doses of intravenous methylprednisolone for moderate to severe and active Graves’ orbitopathy. J Clin Endocrinol Metab. 2012;97:4454–4463. doi:10.1210/jc.2012-2389
63. Mourits M, van Kempen-harteveld M, Garcia M, et al. Radiotherapy for Graves’ orbitopathy: randomised placebo-controlled study. Lancet. 2000;355:1505–1509. doi:10.1016/S0140-6736(00)02165-6
64. Prummel M, Terwee C, Gerding M, et al. A randomized controlled trial of orbital radiotherapy versus sham irradiation in patients with mild Graves’ ophthalmopathy. J Clin Endocrinol Metab. 2004;89:15–20. doi:10.1210/jc.2003-030809
65. Gorman C, Garrity J, Fatourechi V, et al. A prospective, randomized, double-blind, placebo-controlled study of orbital radiotherapy for Graves’. Ophthal Ophthalmol. 2001;108:1523–1534. doi:10.1016/S0161-6420(01)00632-7
66. Rajendram R, Taylor P, Wilson V, et al. Combined immunosuppression and radiotherapy in thyroid eye disease (CIRTED): a multicentre, 2 x 2 factorial, double-blind, randomised controlled trial. Lancet Diabetes Endocrinol. 2018;6:299–309.
67. Prummel M, Mourits M, Blank L, et al. Randomized double-blind trial of prednisone versus radiotherapy in Graves’ ophthalmopathy. Lancet. 1993;342:949–954. doi:10.1016/0140-6736(93)92001-A
68. Marcocci C, Bartalena L, Bogazzi F, et al. Orbital radiotherapy combined with high dose systemic glucocorticoids for Graves’ ophthalmopathy is more effective than radiotherapy alone: results of a prospective randomized study. J Endocrinol Invest. 1991;14:853–860. doi:10.1007/BF03347943
69. Bartalena L, Marcocci C, Chiovato L, et al. Orbital cobalt irradiation combined with systemic corticosteroids for Graves’ ophthalmopathy: comparison with systemic corticosteroids alone. J Clin Endocrinol Metab. 1983;56:1139–1144.
70. McCord CD. Orbital decompression for Graves’ disease. Exposure through lateral canthal and inferior fornix incision. Ophthalmology. 1981;88(6):533–541. doi:10.1016/S0161-6420(81)34995-1
71. Leone CR, Bajandas FJ. Inferior orbital decompression for dysthyroid optic neuropathy. Ophthalmology. 1981;88(6):525–532.
72. Shorr N, Baylis HI, Goldberg RA, Perry JD. Transcaruncular approach to the medial orbit and orbital apex. Ophthalmology. 2000;107(8):1459–1463. doi:10.1016/S0161-6420(00)00241-4
73. Kazim M, Trokel SL, Acaroglu G, Elliott A. Reversal of dysthyroid optic neuropathy following orbital fat decompression. Br J Ophthalmol. 2000;84(6):600–605. doi:10.1136/bjo.84.6.600
74. Jacobs SM, McInnis CP, Kapeles M, Chang S-H. Incidence, risk factors, and management of blindness after orbital surgery. Ophthalmology. 2018;125(7):1100–1108. doi:10.1016/j.ophtha.2018.01.030
75. Kennedy DW, Goodstein ML, Miller NR, Zinreich SJ. Endoscopic transnasal orbital decompression. Arch Otolaryngol Head Neck Surg. 1990;116(3):275–282. doi:10.1001/archotol.1990.01870030039006
76. Chu EA, Miller NR, Lane AP. Selective endoscopic decompression of the orbital apex for dysthyroid optic neuropathy. Laryngoscope. 2009;119(6):1236–1240. doi:10.1002/lary.20240
77. Farling PA. Thyroid disease. Br J Anaesth. 2000;85(1):15–28. doi:10.1093/bja/85.1.15
78. O’Shea JJ, Kontzias A, Yamaoka K, et al. Janus kinase inhibitors in autoimmune diseases. Ann Rheum Dis. 2013;72(suppl2):ii111–ii5. doi:10.1136/annrheumdis-2012-202576
79. Xie W, Huang Y, Xiao S, et al. Impact of Janus kinase inhibitors on risk of cardiovascular events in patients with rheumatoid arthritis: systematic review and meta-analysis of randomised controlled trials. Ann Rheum Dis. 2019;78(8):1048–1054. doi:10.1136/annrheumdis-2018-214846
80. Seavey MM, Dobrzanski P. The many faces of Janus kinase. Biochem Pharmacol. 2012;83(9):1136–1145. doi:10.1016/j.bcp.2011.12.024
81. Stebbing J, Phelan A, Griffin I, et al. COVID-19: combining antiviral and anti-inflammatory treatments. Lancet Infect Dis. 2020;20(4):400–402. doi:10.1016/S1473-3099(20)30132-8
82. Kaur K, Kalra S, Kaushal S. Systematic review of tofacitinib: a new drug for the management of rheumatoid arthritis. Clin Ther. 2014;36(7):1074–1086. doi:10.1016/j.clinthera.2014.06.018
83. Markham A. Baricitinib: first global approval. Drugs. 2017;77(6):697–704. doi:10.1007/s40265-017-0723-3
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.