Back to Journals » Patient Preference and Adherence » Volume 15
Influence of Patient-Reported Treatment Satisfaction on Psychological Health and Quality of Life Among Patients Receiving Divya-Swasari-Coronil-Kit Against COVID-19: Findings from a Cross-Sectional “SATISFACTION COVID” Survey
Authors Balkrishna A, Raj P, Singh P, Varshney A ![]()
Received 20 January 2021
Accepted for publication 25 March 2021
Published 29 April 2021 Volume 2021:15 Pages 899—909
DOI https://doi.org/10.2147/PPA.S302957
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Johnny Chen
Video abstract of "Divya-Swasari-Coronil-Kit against COVID-19" [ID 302957].
Views: 237
Acharya Balkrishna,1,2 Preeti Raj,3 Pratima Singh,3 Anurag Varshney1– 3
1Patanjali Research Foundation Trust, Patanjali Yogpeeth, Haridwar, Uttarakhand, 249 405, India; 2Department of Allied and Applied Sciences, University of Patanjali, Haridwar, Uttarakhand, 249 405, India; 3Clinical Research Division, Patanjali Research Institute, Haridwar, Uttarakhand, 249 405, India
Correspondence: Anurag Varshney
Clinical Research Division, Patanjali Research Institute, NH-58, Haridwar, Uttarakhand, 249 405, India
Email [email protected]
Introduction: The correlation among treatment satisfaction with demographic characteristics, health symptoms or psychological health, and quality of life with the prophylactic regime against COVID-19 is rather unexplored. This real-world exploratory study was conducted to determine patient-perspectives regarding their treatment satisfaction receiving Divya-Swasari-Coronil-Kit with correlative impacts on psychological health (PH) and Quality of life (QoL) based on four hypotheses each relating to PH, QoL, Demographic characteristics, and Treatment satisfaction.
Methods: This cross-sectional, web-based survey collected data on demographic characteristics and psychological health with DASS-21; QoL with 5-level 5-dimension EuroQol instrument; and treatment satisfaction using Treatment Satisfaction Questionnaire for Medication (TSQM) V9. Pearson correlation coefficient analysis was used to examine the relation between TSQM and PH and the demographic variables. Factor analysis was used for multi-collinearity tests, and multiple linear regression analysis was used to explore demographic variables and TSQM.
Results: Out of 421 initial screenings, 367 patient-participants were included in the analysis. The mean age of included participants was 33.61 ± 9.47 years. Marital status and socio-economic class positively correlated with TSQM. Physical symptoms in patients are positively correlated with depression, anxiety, and stress; and in contrast, negatively with QoL. Global satisfaction with Divya-Swasari-Coronil-Kit medication negatively correlated with depression, anxiety, stress, effectiveness, convenience; whereas global satisfaction correlated positively with QoL.
Conclusion: Present study (SATISFACTION COVID) indicates that treatment satisfaction due to avaliablity and treatment of Divya-Swasari-Coronil-Kit has constructive and beneficial implications on psychological health, Quality of life and demographic factors. In addition, web-based patient-reported perspectives may well be a feasible way to provide better insights into treatment satisfaction, in relation to psychological health and Quality of life.
Keywords: Traditional Indian Medicine, mental health well-being, health-related quality of life, medication satisfaction, Divya-Swasari-Coronil-Kit, SATISFACTION COVID
Introduction
Originated as a cluster of unexplained cases of pneumonia in Wuhan, China; COVID-19, caused by Severe Acute Respiratory Syndrome Corona Virus 2 (SARS-CoV-2) has profoundly affected every aspect of day-to-day life around the world. Clinically, the disease is heterogeneous in nature, with a wide range of symptoms and rather uncertain prognosis.
With incalculable socio-economic effects and the imposition of unfamiliar public health measures that infringe on personal freedoms, the COVID-19 pandemic has emerged as a threat to the world community. Providing exact information on the number of infected individuals and infection fatality ratio is intricate since the health crisis is not yet under control and the death toll is on its rise.1 In India, COVID-19 pandemic began on 3 February 2020, and till 18 January 2021 the number of infected individuals was more than 10.5 million; of those 152,456 lost lives, as reported by the Indian Council of Medical Research (ICMR), New Delhi, India and World Health Organization (WHO).2,3
As a highly contagious viral disease beyond threatening physical health and life, researchers and clinicians have voiced concern over its impact on mental health.4,5 This biological disaster has prevailed fear and uncertainty, due to lack of end-point which is precipitating psychopathology such as depressive and anxiety disorders in individuals.4 In particular, susceptibility towards the risk of exposure, concerns about infecting their loved ones or having to isolate away from them, and being associated with ethically difficult decisions around resource-allocation may cause a profound psychological burden.5–7 At present, with no cure identified yet, therapeutic strategies to deal with the infection are only supportive, and prevention aimed at reducing transmission in the community as the best option.8 With the established integrative healthcare system combining both traditional medicines and modern medicine, India has achieved good clinical success in the prevention and control of the pandemic. In India, the practice of the indigenous system of medicine such as Ayurveda has existed for ages with extensive use in wide sections of the population.9 The classical textbook of Ayurveda “Charaka Samhita” mentions epidemic management with a broader emphasis on building the strength of body and mind to cope with various stressors with arresting the progression for maintaining the homeostasis10,11 Divya-Swasari-Coronil-Kit is a polyherbal ayurvedic medicine kit prepared in India, developed specifically as a remedy for COVID-19.
The Divya-Swasari-Coronil-Kit contains Swasari Vati and Coronil tablet with Anu Taila as a combined treatment regime. These formulations have been shown to suppress inflammatory storm, modulate immune function and reduce lung injury caused by SARS-CoV-2 S-protein under in-vitro and in-vivo experimental conditions.12–15 The fundamental research and clinical study with its constituents showed that Divya-Swasari-Coronil-Kit promising bidirectional regulation in the symptoms associated with SARS-CoV-2 infection. Results from a double-blind, randomized, controlled trial showed expedite virological clearance and concomitantly reduce the risk of viral dissemination. Reduced inflammation markers suggested less severity of SARS-CoV-2 infection in the treatment group.16 Overall, the ayurvedic treatment was associated with faster recovery from SARS-CoV-2 infection in asymptomatic and mildly symptomatic cases (CTRI No. CTRI/2020/05/025273). Available data highlights the role of traditional and complementary medicine with enormous therapeutic and prophylactic potential that can be proven effective against COVID-19.17
Considering the therapeutic effect of Divya-Swasari-Coronil-Kit, the ministry of AYUSH, Government of India has approved this formulation as the supportive measure in management of COVID-19.18 This traditional formulation has been employed as a pre-emptive remedy and an adjunct treatment option on a precisely wide level with repurposing its effectiveness against the COVID-19 pandemic amid the conundrum of other drug development which would take time as human trials are underway.
Divya-Swasari-Coronil-Kit has been consumed as a prophylactic regimen against COVID−19 on an extensive scale by the Indian population. From a scientific viewpoint, treatment satisfaction is a valuable domain in clinical practice guidelines with an apparent link and pivotal role in psychological well-being and Health-related Quality of life (QoL) for treatment satisfaction.19 Little is known about patient experience and perspectives with Divya-Swasari-Coronil-Kit in real-world settings as treatment satisfaction is not assessed in randomized control trials (RCT). Moreover, treatment satisfaction proposes insight on patient-related decision making.
Based on this perspective, in this exploratory study (SATISFACTION COVID), we sought to determine the feasibility of garnering and understanding real-world patient accounts on their treatment satisfaction with correlative impact on Psychological Health (PH) and Quality of life (QoL). SATISFACTION COVID study is based on the following four hypotheses (Figure 1) relating to PH and QoL, demographic characteristics, and treatment satisfaction:
Study Hypothesis 1 (H1): Demographic variables significantly relates treatment satisfaction.
Study Hypothesis 2 (H2): Treatment satisfaction significantly modulates psychological health.
Study Hypothesis 3 (H3): Treatment satisfaction significantly modulates health-related Quality of Life.
Study Hypothesis 4 (H4): PH and QoL correlate with demographic variables.
Materials and Methods
Study Design and Participants
A prospective, cross-sectional web-based survey was conducted to assess treatment satisfaction and Quality of Life concerning psychosomatic impact among patients receiving Divya-Swasari-Coronil-Kit as a prophylactic ayurvedic regimen. Electronic medical record systems of patients prescribed with Divya-Swasari-Coronil-Kit were extracted from Patanjali Electronic Medical Record (PEMR) system. Patients' inclusion criteria were: age ≥18 years, access to the internet, willingness to participate in the online survey voluntarily, administered with Divya-Swasari-Coronil-Kit.
Procedure
A call for the invitation to participate in SATISFACTION COVID survey was sent to registered patients through SMS and emails in both Hindi and English languages with a unique identification code to ensure that the eligible candidates complete the survey, only once. Data were collected from September 2020 until October 2020. No personal identifiers were used with survey responses. In India, a non-interventional questionnaire without any linked identifiers and disclosures is exempted from approval by the institutional ethics committee. This exemption is consistent with the Ethical Review Procedures of National Ethical Guidelines for Biomedical and Health Research Involving Human Participants, as specified by 2017 guidelines of Indian Council of Medical Research.20 The study was conducted following the checklist for reporting of Internet E-surveys (CHERRIES) guidelines.21
Measures
The survey questionnaire was developed under the guidelines of the ayurvedic practitioner in practice (first author) and patient taking Divya-Swasari-Coronil-Kit (second author). Their real-world familiarity provided us better insight into the psychological impact of COVID-19 and satisfaction with the availability of Divya-Swasari-Coronil-Kit which resonates better with other patients.
As the study candidates click on the shared link, they were given a brief description on survey study design, required procedures and were explicitly asked if they wish to continue with the study enrollment process. If the study candidates continued further via the web link, it was considered as the informed consent, and subsequent click on the process button proceeded to the next section consisting brief questions on study eligibility.
Background Characteristics
The eligibility questions of the SATISFACTION COVID study questionnaire included (socio-demographic details: age, gender, marital status, education, occupation, and socio-economic status (calculated based on Modified Kuppuswamy socio-economic scale).22 Information regarding the presence of physical symptoms like the presence of cough, cold, headache, breathing difficulties, fever, and fatigue, and specific purpose for use of Divya-Swasari-Coronil-Kit like, infected with COVID-19, direct contact with COVID-19 positive person, relative infection with COVID-19, residing in containment zone, travel history, for which all responses are dichotomous (No =0 and Yes =1).
Psychological Health (Depression, Anxiety and Stress)
The patient-reported psychological impact was measured using survey instrument DASS-21, which consists of a twenty-one-item instrument divided into three subscales that are depression, anxiety, and stress with each subscale allocated for seven items.23,24 Each response was rated on a 4-point scale, where 0 indicates “Did not apply to me” and 3 indicated “Applied to me most of the time”. Depression was assessed from questions 3, 5, 10, 13, 16, 17, and 21. The total score depression subscale score was subdivided into normal (0–9), mild (10−12), moderate (13−20), severe (21–27), and extremely severe depression (28–42). Anxiety was assessed from questions 2, 4, 7, 9, 15, 19, and 20. The stress subscale was assessed from questions 1, 6, 8, 11, 12, 14, and 18. The total score of stress subscale was subdivided into normal (0−10), mild (11–18), moderate (19–26), severe (27–34), and extremely severe stress (35–42). The DASS-21 has been demonstrated as valid and reliable for assessing mental health in previous COVID-19 related researches in the Indian population.25,26
Abbreviated Treatment Satisfaction Questionnaire for Medication (TSQM)
The TSQM version 9 is a generic and multilingual validated questionnaire that is a conceptual framework to assess treatment satisfaction and patients’ satisfaction with their medication.27 The 9-item questionnaire is a psychometrically reliable and valid instrument to assess most common dimensions on TS on three scales which includes; the effectiveness scale (questions 1 to 3), the convenience scale (questions 4 to 8), and the global satisfaction scale (questions 9 to 11).27,28 The results for each scale are presented from 0 to 100, where higher scores represent higher satisfaction in that domain.
Heath-Related Quality of Life
The EQ-5D-5L is a generic patient-reported health-related Quality of Life (HR-QoL) questionnaire composed of 5 questions (Descriptive system) on mobility, self-care, usual activities, pain or discomfort, and anxiety or depression and a visual analog scale (EQ-VAS).29 The latter records patient’s health on a 20-cm vertical visual analog scale with endpoints labeled as “best health you can imagine” and “the worst health you can imagine”. The descriptive system questions have replies of five levels corresponding to “no problem”, “some problem”, and “extreme problem” no problems, slight problems, moderate problems, severe problems.
Statistical Analysis
Descriptive statistics were conducted for demographic variables and clinical parameters (like physical health and purpose of medication) using SPSS 22.0 (IBM SPSS Statistics, New York, USA). Pearson correlation coefficient analysis was used to examine the relation between TSQM and PH and the demographic variables. The method of factor analysis was used for multi-collinearity tests, and multiple linear regression analysis was used to explore demographic variables and TSQM. All the tests were two-tailed, with differences considered to be statistically significant at p<0.05 levels.
Results
According to the guidelines by Comfrey,30 a sample size of 100 is too small, although 200 is adequate, 300 is good, 500 is very good and 1000 is excellent.31 For a general analysis of 45 item factors, in most cases, 250 samples are considered sufficient.32 The sample size of our study was 367, which is in-line with the expected criteria for meaningful research data analysis and its interpretations.
Patient Demographics and Clinical Characteristics
Out of the 680 invitations sent for participation in the SATISFACTION COVID survey, 259 patients were excluded (invitations returned undeliverable or subjects who did not meet the eligibility criteria). Of the remaining 421 subjects, 367 subjects who responded and verified eligible on inclusion criteria with informed consent and completed the entire survey were included in the study. Patient demographics and clinical characteristics are shown in Table 1. The mean (SD) age of included participants was 33.61 (9.47) years. Most respondents were female (70.8%), between 29 and 38 years (40.9%). Majority of participants were married (75.2%); with a professional degree (52.9%), professional occupation (60.5%), and monthly income in Indian currency ₹15,754 - ₹31,506 (33.8%) respectively. The upper socio-economic class (31.9%) in socio-economic status was in preponderance. Cold/cough (29.7%) and relative being infected with COVID-19 (28.06%) were major patient-reported purpose of taking Divya-Swasari-Coronil-Kit (Figure 2A and B).
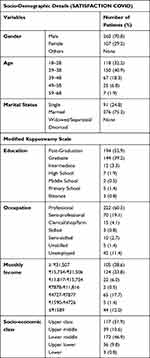 |
Table 1 Demographic Characteristics and Frequencies at the Baseline for the Study Sample (N=367) |
Correlation Matrix Between Treatment Satisfaction, Quality of Life, Psychological Health, and Demographic Characteristics
Table 2 gives descriptive statistics of psychological health (depression, anxiety, stress), Quality of Life, treatment satisfaction, and demographic variables, and also shows correlation variables. These observed correlation are shown in Table 2, and bold data represent statistically significant correlations (p< 0.05).
 |
Table 2 Correlation Matrix for the TSQM, HR-QoL, PH, and Demographic Variables |
Effectiveness, convenience, and global satisfaction are positively correlated with Quality of Life, and with all domains of treatment satisfaction from medication. Regression correlation coefficient (r) was observed at optimal level (r<0.6, p<0.05). These significant correlations are highlighted in Table 2. Generally, there could be a multicollinearity problem when the correlation coefficient (r) is above 0.9,33,34 so correlation coefficient of less than 0.6 would be an acceptable baseline for validated analysis. Therefore, Pearson analysis results support study hypothesis 3 (H3) showing treatment satisfaction for medication positively affects the Quality of Life.
Global satisfaction is correlated with depression, anxiety, and stress. These results positively support our study hypothesis 2 (H2), showing treatment satisfaction negatively affects psychological health. The correlation of anxiety with depression exceeds 0.8, at a value of 0.81. We conducted a multi-collinearity test on all study data. Often variance of inflation is directly proportional to multi-collinearity. More concretely, multicollinearity is not a problem if the tolerance value is greater than 0.10 or the variance of inflation (VIF) is less than 10.35,36 In our study, the lowest tolerance value is 0.436 and, the highest variance of inflation is 2.345. Subsequently, multi-collinearity does not seem to be a significant concern in our data-sheet.
Physical symptoms are positively correlated with depression, anxiety, and stress and in contrast negatively with Quality of Life which validates our hypothesis 4 (H4) that exhibits demographic variables are positively correlated with psychological health.
The results in multiple linear regression analysis in Table 3 to extent support hypothesis 1 (H1). Marital status and socioeconomic class are significantly associated with all the domains of treatment satisfaction of medication. Age is significantly correlated with effectiveness whereas physical symptoms are significantly correlated with global satisfaction and effectiveness.
 |
Table 3 Multiple Regression Analysis with Treatment Satisfaction from Medication as the Dependent Variable |
Discussion
In this study, we demonstrated the feasibility of collecting measurable patient-reported experiences treatment satisfaction concerning psychological well-being and HR-QoL among patients on the prophylactic ayurvedic regimen during the COVID-19 outbreak in India through a cross-sectional survey.
Inconsistent with the hypothesis, the first important finding of the study was the marital status and socio-economic class correlates with TSQM. With no study conducted so far on treatment satisfaction in a similar context, we could not relate our findings with other studies in the literature. Although some studies have reported treatment satisfaction consistent with age.37,38 Our study did not reveal treatment satisfaction with all the domains except effectiveness. However, Fox and Storms39 emphasized the lack of consistency of the effect of age in satisfaction studies. Because satisfaction studies are concentrated on variety of deterministic concepts, such as satisfaction with health services, satisfaction with hospital management, and satisfaction with medication or treatment. The deficit of age-based relation to TSQM reveals that our study population was too homogenous in consideration of age.
Second, high global satisfaction from the medication was associated with low psychological health. Our findings are congruent with other studies conducted on patients with diabetes mellitus and coronary artery disease.40,41 Although, it is unclear from the present finding whether poor treatment satisfaction leads to greater distress or whether greater distress negatively impacts the patient’s view of his or her treatment satisfaction. Further trials aimed at inspecting the impact of psychological health on treatment satisfaction will need to be performed to explore the possible interpretation and the potential point of intervention.
Third, higher vitality scores, as assessed by the EQ-5D-5L, were associated with higher treatment satisfaction from medication. The association in our study between high Quality of Life scores and higher treatment satisfaction, as assessed by TSQM is in line with similar findings previously reported by Revicki and co-workers.42 The association between variables of treatment satisfaction and Quality of Life is a stronger one. Cleary and McNeil43 reported positive correlations between health status and satisfaction. However, further research is needed for better understanding determinants of satisfaction in correlation with health status before the intervention or with health status after the intervention.
Fourth, we found a significant correlation between physical symptoms with psychological health and HR-QoL. The more decrease in physical symptoms reported better Psychological well-being and HR-QoL status. Physical symptoms concerning COVID P−19 can precipitate the perceived risk of psychological distress and negatively impact the Quality of Life. These findings suggest that the meaning-focused resources (ie, availability of treatment to help control the risk perception) can paradoxically act as a coping strategy in an environment full of panic and uneasiness in the state of emergency brought up by the pandemic. To our knowledge, the COVID-19 patients had uncommon symptoms, including headache, cold/cough, and chest pain, reported during mild to moderate cases.44 In addition to physical symptoms, other factors such as fear of illness, and extreme uncertainty during the COVID-19, leads to low vitality scores. Albeit, the confounding variables are evenly distributed across the treatment groups, their influence on psychological health and QoL has been reported in previous studies. Health and symptom status are known to influence QoL depending upon their frequency, severity and duration. Similarly, the patient perception is also affected by emotional impact of the disease.45,46 Improvement in physical symptoms, in particular, plays a critical role in mitigating health-related Quality of Life.47
Our current study highlights patient-reported experiences with treatment experience among patients on prophylactic ayurvedic regimen against COVID-19 in an Indian real-world setting. The current results may be unique, as with survey-based studies, patient perspectives may be subject to the patients' bias and experience. There are few shortcomings and limitations of this study. Firstly, comprehensive diagnosis and treatment history are not available in this cross-sectional study. Additionally, QoL and psychological well-being before initiation of Divya-Swasari-Coronil-Kit calls for open re-call bias. Because survey participation was offered online, only respondents with the access of internet were included. Our sample size may adequately represent complete analysis on patient satisfaction as our recruitment method did not allow participation of patients with discontinued treatment. However, our goal was not to assess treatment satisfaction of those who continued versus those who discontinued. Rather, our goal was to evaluate treatment satisfaction from medication in relation to psychological well-being and health-related Quality of Life. Although evaluation of treatment satisfaction is dynamically complex as no COVID-19 specific generic instrument are available. Future researches are planned to examine symptomatic improvement and evaluation of patient experience in comparison with other therapeutic agents appears more practical and imperative in the present scenario.
Conclusion
Using a web-based survey, our results showed that all domains of treatment satisfaction which comprises effectiveness, convenience, and global satisfaction positively correlates with Quality of Life. Additionally, global satisfaction from the treatment regime negatively correlates with psychological health. From all the categories of socio-demographic details, marital status and socio-economic class correlate with treatment satisfaction from medication. The more decrease in physical symptoms reported better psychological well-being and Health-Related Quality of Life status. This pilot endeavor indicates that treatment satisfaction from Divya-Swasari-Coronil-Kit has its implication on psychological health, Quality of Life, and demographic factors indicated in our study and is feasible and applicable in future studies.
Data Sharing Statement
The data generated and/or analyzed in the SATISFACTION COVID study are for academic purposes and are available on appropriate requests.
Ethical Approval
The study was conducted in accordance with the Principles of the Declaration of Helsinki (1975). An institutional ethical committee exemption was obtained from IEC-Patanjali Bhartiya Ayurvigyan Evam Anusandhan Sansthan, Haridwar, India, for the study and data collection, as per National Ethical Guidelines for Biomedical and Health Research Involving Human Participants, ICMR (2017).
Informed Consent
Informed consent was obtained electronically from the eligible patients volunteered in participating in the SATISFACTION COVID survey.
Acknowledgments
The authors are thankful to the volunteers who agreed to participate in the study and to Mr Deepak Rawat for assistance in patient recruitment. We extend our gratitude to Ms. Priyanka Kandpal, Mr. Tarun Rajput, Mr. Gagan Kumar, and Mr. Lalit Mohan for their swift administrative supports.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
Divya-Swasari-Coronil-Kit has been manufactured by Divya Pharmacy, Haridwar India. Divya Pharmacy was not involved in any aspect of research reported in this study. Acharya Balkrishna is an honorary trustee in Divya Yog Mandir Trust; in addition, he holds an honorary managerial position in Patanjali Ayurved Ltd., Haridwar, India; he reports no other potential conflicts of interest for this work. All other authors declare no conflicts of interest for this work.
References
1. Roda WC, Varughese MB, Han D, Li MY. Why is it difficult to accurately predict the COVID-19 epidemic? Infect Dis Model. 2020;5:271–281. doi:10.1016/j.idm.2020.03.001
2. World Health Organization. WHO Coronavirus (COVID-19) dashboard; 2020. Available from: https://covid19.who.int/.
3. Indian Council of Medical Research (ICMR). COVID-19 Toll Update. Available from: https://www.icmr.gov.in.
4. Pedersen A, Zachariae R, Bovbjerg DH. Influence of psychological stress on upper respiratory infection – a meta-analysis of prospective studies. Psychosom Med. 2010;72(8):823–832. doi:10.1097/PSY.0b013e3181f1d003
5. Tsamakis K, Rizos E, Manolis A, et al. COVID-19 pandemic and its impact on mental health of healthcare professionals. Exp Ther Med. 2020;19(6):3451–3453. doi:10.3892/etm.2020.8646
6. Pfefferbaum B, North CS. Mental health and the Covid-19 pandemic. N Engl J Med. 2020;383(6):510–512. doi:10.1056/nejmp2008017
7. Kang L, Li Y, Hu S, et al. The mental health of medical workers in Wuhan, China dealing with the 2019 novel coronavirus. Lancet Psychiatry. 2020;7(3). doi:10.1016/S2215-0366(20)30047-X
8. Cascella M, Rajnik M, Cuomo A, Dulebohn SC, Di Napoli R. Features, Evaluation and Treatment Coronavirus (COVID-19). In: StatPearls [Internet] 2021. Available from: https://www.ncbi.nlm.nih.gov/books/NBK554776.
9. Shankar D, Patwardhan B. AYUSH for New India: vision and strategy. J Ayurveda Integr Med. 2017;8(3):137. doi:10.1016/j.jaim.2017.09.001
10. Bahadur S, Mukherjee P, Milan Ahmmed S, Kar A, Harwansh R, Pandit S. Metabolism-mediated interaction potential of standardized extract of Tinospora cordifolia through rat and human liver microsomes. Indian J Pharmacol. 2016;48(5):576. doi:10.4103/0253-7613.190758
11. Samhita C, Hridaya A. History of Ayurveda. Bull Indian Inst Hist Med Hyderabad. 2000;30(1):1–14..
12. Balkrishna A, Solleti SK, Verma S, Varshney A. Application of humanized zebrafish model in the suppression of SARS-CoV-2 spike protein induced pathology by tri-herbal medicine coronil via cytokine modulation. Molecules. 2020;25(21):5091. doi:10.3390/molecules25215091
13. Balkrishna A, Solleti SK, Singh H, Tomer M, Sharma N, Varshney A. Calcio-herbal formulation, Divya-Swasari-Ras, alleviates chronic inflammation and suppresses airway remodelling in mouse model of allergic asthma by modulating pro-inflammatory cytokine response. Biomed Pharmacother. 2020;126:110063. doi:10.1016/j.biopha.2020.110063
14. Balkrishna A, Solleti SK, Singh H, et al. Herbal decoction Divya-Swasari-Kwath attenuates airway inflammation and remodeling through Nrf-2 mediated antioxidant lung defence in mouse model of allergic asthma. Phytomedicine. 2020;78:153295. doi:10.1016/j.phymed.2020.153295
15. Balkrishna A, Pokhrel S, Singh H, et al. Withanone from Withania somnifera attenuates SARS-CoV-2 RBD and host ACE2 interactions to rescue spike protein induced pathologies in humanized zebrafish model. Drug Des Dev Ther. 2021;Volume 15:1111–1133. doi:10.2147/DDDT.S292805
16. Devpura G, Tomar BS, Nathiya D, et al. Randomized placebo-controlled pilot clinical trial on the efficacy of ayurvedic treatment regime on COVID-19 positive patients. Phytomedicine. 2021;84:153494. doi:10.1016/j.phymed.2021.153494
17. Xu J, Zhang Y. Traditional Chinese Medicine treatment of COVID-19. Complement Ther Clin Pract. 2020;39:101165. doi:10.1016/j.ctcp.2020.101165
18. Business Standard. Patanjali’s Coronil, 1st “proof-based” drug to fight Covid, gets govt nod. Business Standard; 2021. Available from: https://www.business-standard.com/article/current-affairs/ramdev-releases-paper-on-patanjali-s-1st-proof-based-covid-drug-coronil-coronavirus-treatment.
19. Larsson B, Dragioti E, Gerdle B, Björk J. Positive psychological well-being predicts lower severe pain in the general population: a 2-year follow-up study of the SwePain cohort. Ann Gen Psychiatry. 2019;18(1):1. doi:10.1186/s12991-019-0231-9
20. Mathur R, Swaminathan S. National ethical guidelines for biomedical and health research involving human participants. Indian Council Med Res. 2017;36–37.
21. Eysenbach G. Improving the quality of web surveys: the checklist for reporting results of internet E-Surveys (CHERRIES). J Med Internet Res. 2004;6(3):e34. doi:10.2196/jmir.6.3.e34
22. Mohd Saleem S. Modified Kuppuswamy socioeconomic scale updated for the year 2019. Indian J Forensic Community Med. 2020;7(1):1–3. doi:10.18231/2394-6776.2019.0001
23. Lovibond PF, Lovibond SH. The structure of negative emotional states: comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behav Res Ther. 1995;33(3):335–343. doi:10.1016/0005-7967(94)00075-u
24. Henry JD, Crawford JR. The short-form version of the depression anxiety stress scales (DASS-21): construct validity and normative data in a large non-clinical sample. Br J Clin Psychol. 2005;44(2):227–239. doi:10.1348/014466505X29657
25. Verma S, Mishra A. Depression, anxiety, and stress and socio-demographic correlates among general Indian public during COVID-19. Int J Soc Psychiatry. 2020;66(8):756–762. doi:10.1177/0020764020934508
26. Nathiya D, Suman S, Singh P, Raj P, Tomar BS. Mental health outcome and professional quality of life among healthcare workers during COVID-19 pandemic: a frontline COVID survey. Hamdan Med J . 2020;13(4):196. doi:10.4103/HMJ.
27. Atkinson MJ, Sinha A, Hass SL, et al. Validation of a general measure of treatment satisfaction, the Treatment Satisfaction Questionnaire for Medication (TSQM), using a national panel study of chronic disease. Health Qual Life Outcomes. 2004;2(1):1–3. doi:10.1186/1477-7525-2-12
28. Atkinson MJ, Kumar R, Cappelleri JC, Mass SL. Hierarchical construct validity of the treatment satisfaction questionnaire for medication (TSQM Version II) among outpatient pharmacy consumers. Value Heal. 2005. doi:10.1111/j.1524-4733.2005.00066.x
29. Wu P, Fang Y, Guan Z, et al. The psychological impact of the SARS epidemic on hospital employees in China: exposure, risk perception, and altruistic acceptance of risk. Can J Psychiatry. 2009;54(5):302–311. doi:10.1177/070674370905400504
30. Pearson RH, Mundfrom DJ. Recommended sample size for conducting exploratory factor analysis on dichotomous data. J Mod Appl Stat Methods. 2010;9(2):5. doi:10.22237/jmasm/1288584240
31. Preacher KJ, Hayes AF. Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav Res Methods. 2008;40(3):879–891. doi:10.3758/BRM.40.3.879
32. Kline RB. Principles and Practice of Structural Equation Modelling.
33. Liu H, Ke W, Wei KK, Gu J, Chen H. The role of institutional pressures and organizational culture in the firm’s intention to adopt internet-enabled supply chain management systems. J Oper Manag. 2010;28(5):372–384. doi:10.1016/j.jom.2009.11.010
34. Sun JJ. Analysing the influence factors of single task pricing based on public packet system: an empirical study in China. J Phys Conf Ser. 2020. doi:10.1088/1742-6596/1437/1/012100
35. Dowell AC, Hamilton S, McLeod DK. Job satisfaction, psychological morbidity and job stress among New Zealand general practitioners. N Z Med J. 2000;113(1113):269.
36. de Carvalho JF, Draper NR, Smith H. Applied regression analysis. J Am Stat Assoc. 1981. doi:10.2307/2287608
37. Blanchard CG, Labrecque MS, Ruckdeschel JC, Blanchard EB. Physician behaviors, patient perceptions, and patient characteristics as predictors of satisfaction of hospitalized adult cancer patients. Cancer. 1990;65(1):186–192. doi:10.1002/1097-0142(19900101)65:1<186::AID-CNCR2820650136>3.0.CO;2-4
38. Zahr LK, William G, Ayam EH. Patient satisfaction with nursing care in Alexandria, Egypt. Int J Nurs Stud. 1991;28(4):337–342. doi:10.1016/0020-7489(91)90060-G
39. Fox JG, Storms DM. A different approach to sociodemographic predictors of satisfaction with health care. Soc Sci Med Part a Med Psychol Med. 1981;15(5):557–564. doi:10.1016/0271-7123(81)90079-1
40. Spertus JA, McDonell M, Woodman CL, Fihn SD. Association between depression and worse disease-specific functional status in outpatients with coronary artery disease. Am Heart J. 2000;140(1):105–110. doi:10.1067/mhj.2000.106600
41. Musselman DL, Betan E, Larsen H, Phillips LS. Relationship of depression to diabetes types 1 and 2: epidemiology, biology, and treatment. Biol Psychiatry. 2003;54(3):317–329. doi:10.1016/S0006-3223(03)00569-9
42. Revicki DA, Zodet MW, Joshua-Gotlib S, Levine D, Crawley JA. Health-related quality of life improves with treatment-related GERD symptom resolution after adjusting for baseline severity. Health Qual Life Outcomes. 2003;1(1):1–9. doi:10.1186/1477-7525-1-73
43. Cleary PD, McNeil BJ. Patient satisfaction as an indicator of quality care. Inquiry. 1988;25(1):25–36.
44. Li K, Wu J, Wu F, et al. The clinical and chest CT features associated with severe and critical COVID-19 pneumonia. Invest Radiol. 2020. doi:10.1097/RLI.0000000000000672
45. Hall JA, Dornan MC. Patient sociodemographic characteristics as predictors of satisfaction with medical care: a meta-analysis. Soc Sci Med. 1990;30(7):811. doi:10.1016/0277-9536(90)90205-7
46. Ross CK, Steward CA, Sinacore JM. A comparative study of seven measures of patient satisfaction. Med Care. 1995. doi:10.1097/00005650-199504000-00006
47. Brophy S, Davies H, Dennis MS, et al. Fatigue in ankylosing spondylitis: treatment should focus on pain management. Semin Arthritis Rheum. 2013;42:361. doi:10.1016/j.semarthrit.2012.06.002
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


