Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Influence of Emphysema and Air Trapping Heterogeneity on Pulmonary Function in Patients with COPD
Authors Li K, Gao Y, Pan Z, Jia X, Yan Y , Min X , Huang K, Jiang T
Received 3 July 2019
Accepted for publication 23 November 2019
Published 6 December 2019 Volume 2019:14 Pages 2863—2872
DOI https://doi.org/10.2147/COPD.S221684
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chunxue Bai
Kun Li,1,* Yanli Gao,1,* Zhenyu Pan,1 Xiuqin Jia,1 Yuchang Yan,1 Xiaohong Min,1 Kewu Huang,2 Tao Jiang1
1Department of Radiology, Beijing Chao-Yang Hospital, Capital Medical University, Beijing, Republic of China; 2Department of Pulmonary and Critical Care Medicine, Beijing Chao-Yang Hospital, Capital Medical University, Beijing Institute of Respiratory Medicine, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Tao Jiang
Department of Radiology, Beijing Chao-Yang Hospital, Capital Medical University, Beijing, People’s Republic of China
Tel +86 13718875778
Fax +86 1065935237
Email [email protected]
Purpose: To explore the influence of emphysema and air trapping heterogeneity on pulmonary function changes in patients with stable chronic obstructive pulmonary disease (COPD).
Patients and methods: One hundred and seventy-nine patients with stable COPD were enrolled in this prospective study. All patients underwent low-dose inspiratory and expiratory CT scanning and pulmonary-function tests. CT quantitative data for the emphysema index (EI) on full-inspiration and air trapping (AT) on full-expiration were measured for the whole lung, the right and left lungs, and the cranial-caudal lung zones. The heterogeneity index (HI) values for emphysema and air trapping were determined as the ratio of the difference to the sum of the respective indexes. The cranial-caudal HI and left–right lung HI were compared between mild-to-moderate (GOLD stage I and II) and severe (GOLD stage III and IV) disease groups. The associations between HI and pulmonary-function measurements adjusted for age, sex, height, smoking history, EI and AT of the total lung were assessed using multiple linear regression analysis.
Results: The absolute values for cranial-caudal HI (AT_CC_HI) and left–right lung HI (AT_LR_HI) on full-expiration were significantly larger in the mild-to-moderate group, while no significant intergroup differences were observed on full-inspiration. COPD patients with lower-zone and/or left-lung predominance showed significantly lower pulmonary function than those with upper-zone and/or right-lung predominance on full-expiration, whereas no significant differences were observed on full-inspiration. The absolute values of AT_CC_HI and AT_LR_HI significantly correlated with pulmonary-function measurements. Higher AT_CC_HI and lower AT_LR_HI absolute values indicated better pulmonary function, after adjusting for age, sex, height, smoking history, EI and AT of the total lung.
Conclusion: Subjects with more heterogeneous distribution and/or upper-zone predominant and/or right-lung predominant patterns on full-expiration tend to have better pulmonary function. Thus, in comparison with emphysema heterogeneity, AT heterogeneity better reflects the pulmonary function changes in COPD patients.
Keywords: pulmonary disease, chronic obstructive, small airway disease, heterogeneous distribution, tomography, X-Ray computed
Introduction
Chronic obstructive pulmonary disease (COPD) is defined by the presence of airflow obstruction that is “not fully reversible”.1 Pulmonary emphysema and airway lesions are the main pathological changes in patients with COPD. Emphysema in COPD patients shows considerable heterogeneity in its extent and spatial distribution,2 and the heterogeneity in the distribution of emphysema might be associated with the clinical severity of the disease.3–6 As a result, the importance of emphysema distribution in COPD assessment has been widely recognized. Accurate phenotyping of emphysema distribution might be useful in predicting disease progression and prognosis and designing effective personalized therapeutic or management protocols for preventing disease progression.7
To date, spirometry remains the primary method for diagnosis and monitoring of COPD. However, traditional pulmonary-function tests (PFTs) have a low sensitivity for diagnosing early-stage COPD, since 30% of the patients may suffer from emphysema before exhibiting any detectable decline in pulmonary function.8 Furthermore, abnormalities in regional function and morphology cannot be evaluated by PFTs. As an alternative to PFTs, CT examinations offer substantial advantages in the assessment of regional destruction of pulmonary parenchyma and airway remodeling. Advances in CT scanning and image-processing software have allowed precise measurement of the extent and heterogeneity of emphysema and air trapping (AT), which may correlate with functional changes.
Several previous studies have focussed on the effect of the cranial-caudal emphysema heterogeneity on pulmonary function on full-inspiration, but their results were not completely consistent.7,9–11 The heterogeneity in the distribution in the left and right lungs in patients with COPD was seldom reported. Furthermore, the effect of AT heterogeneity on pulmonary function on full-expiration has not been fully discussed.
Small airway lesions are known to be the main factors leading to irreversible airflow limitation in COPD patients. Expiratory flow limitation is the pathophysiologic hallmark of these patients. Small airway lesions can be indirectly assessed by measurement of AT on expiration.12 Our previous study has shown that AT on full-expiration shows better correlation with pulmonary function in COPD patients.13 Therefore, we speculated that AT heterogeneity may be more advantageous in reflecting pulmonary function. To test this hypothesis, our study aimed to evaluate the influence of the heterogeneity distribution on pulmonary-function parameters by using inspiratory-to-expiratory CT scans in COPD patients.
Materials and Methods
Subjects
The protocol for this prospective clinical study was approved by the Human Research Ethics Board of Beijing Chao-Yang Hospital (study 2016-KE-183-1) and conducted in accordance with the Declaration of Helsinki. A signed written informed consent document was obtained from each patient prior to participation.
The study population was randomly selected from the Department of Pulmonary and Critical Care Medicine outpatients at our hospital between June 2016 and June 2018 by using the following inclusion criteria: (1) age 40–75 years; (2) a history of chronic respiratory symptoms, including wheeze, cough, expectoration, and dyspnea; and (3) pre-bronchodilator (pre-BD) forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio <70%. Subjects were excluded if they met any of the following criteria: (1) post-bronchodilator (post-BD) FEV1/FVC ratio ≥70%; (2) unable to cooperate with respiration; (3) presence of congenital heart disease or congestive heart failure; or (4) presence of severe pulmonary lesions or dysplasia. A total of 179 patients were eventually selected on the basis of these criteria and divided into mild-to-moderate and severe groups (GOLD stage I and II vs GOLD stage III and IV). Both CT scanning and PFTs were performed post-BD (salbutamol) administration.
CT Protocols
All patients were scanned in the supine position with a 64-MDCT system (Light Speed VCT, GE Healthcare, Milwaukee, WI, USA) that was calibrated routinely. The patients received thorough pre-scan instructions and were carefully followed up to guarantee full-inspiration and full-expiration levels for the CT examinations. The scans were obtained with the following parameters: tube voltage, 120 kV; tube current, 50 mAs; pitch, 1.0; and field of view, 280 to 350 mm. The images were acquired from the lung apices to the bases without the use of intravenous contrast and were reconstructed with a 1.25-mm reconstruction slice thickness and 1.25-mm interval using a standard algorithm. After post-BD administration of salbutamol (Ventolin Aerosol; 400 μg, GlaxoSmithKline), PFT was performed within 30 min and CT scanning was performed within 90 min.
CT Densitometry
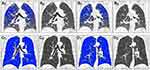
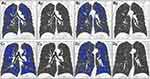
All CT images were analyzed using the GE ADW 4.5 densitometry workstation (Thoracic-VCAR; GE Healthcare), which automatically extracts lung segments. The trachea and the main bronchi were also eliminated. Manual modification was performed if necessary to correct extraction errors for lung regions. Emphysema index (EI) was quantified as the percentage of lung voxels with attenuation lower than –950 HU on full-inspiration. AT was defined as the percentage of lung voxels with a CT attenuation value below −856 HU on full-expiration. EI and AT for the right lung, left lung, and whole lung were measured. The right and left lungs were then divided into the upper zone, middle zone, and lower zone, based on the level of the carina and the inferior right pulmonary vein. The EI and AT of each zone were also measured.
The absolute value of the cranial-caudal heterogeneity index on full-inspiration (EI_CC_HI) or on full-expiration (AT_CC_HI) between the upper and lower zones was computed Eq. (1,2). Similarly, the absolute value of the heterogeneity index (HI) between the left–right lungs on full-inspiration (EI_LR_HI) or on full-expiration (AT_LR_HI) was computed Eq. (3,4). EIupper (ATupper) was the percentage of emphysema (AT) voxels as the sum of that in left upper, right upper, left middle, and right middle zones, while EIlower (ATlower) was the percentage of emphysema (AT) voxels as the sum of that in left lower and right lower zones. A higher absolute value of the HI represented a more heterogeneous distribution of emphysema or AT, and a lower absolute value of the HI represented a homogeneous distribution.
(1) (2) (3) (4)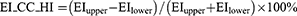
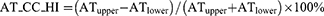
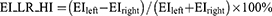
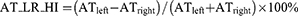
PFT Technique
Pulmonary function was assessed via spirometry and plethysmography (Jaeger MasterScreen; Viasys Healthcare, Höchberg, Germany) according to European Respiratory Society (ERS)/American Thoracic Society (ATS) Guidelines.14 For each subject, the following functional parameters were assessed post-BD: FEV1, FVC, the FEV1/FVC ratio, the FEV1% predicted (FEV1(%pred)), and the inspiratory capacity (IC)/total lung capacity (TLC). The PFT parameters of COPD patients are summarized in Table 1. The modified British medical research council (mMRC) score, COPD assessment test (CAT) score, and number of COPD acute exacerbations in the past year (N1AE) were also recorded (Table 1).
 |
Table 1 Patient Characteristics |
Statistical Analyses
All statistical analyses were performed with the SPSS statistical software package, version 21.0 (IBM Corporation, Armonk, NY, USA). K-means clustering data-driven analysis was performed on the HI to divide subjects into two groups of subtypes: upper-zone-predominant and lower-zone-predominant emphysema (AT) subtypes as well as left-lung-predominant and right-lung-predominant emphysema (AT) subtypes. The intergroup differences were evaluated with Student’s t test or the Mann–Whitney U-test when the data did not pass the normality test (Shapiro–Wilk test). Spearman rank correlation analysis was used to determine the association between pairs of variables for non-parametric data. Associations between the absolute value of the HI and the lung function measures, including FEV1, FEV1(%pred), FVC, FEV1/FVC, and IC/TLC, were assessed by using multiple linear regression analysis after adjusting for age, sex, height, smoking history (pack-years), and EI and AT of the total lung. P values below 0.05 (two-tailed) were considered statistically significant.
Results
The demographic, imaging, clinical, and PFT parameters in participants with COPD are shown in Table 1. On full-expiration, the absolute values of AT_CC_HI and AT_LR_HI showed significant intergroup differences. Subjects with a more homogeneous distribution of AT had poorer lung function than those showing a heterogeneous distribution. On full-inspiration, the absolute values of EI_CC_HI and EI_LR_HI showed no significant intergroup differences (Table 2, Figure 1A–D, Figure 2A–D).
 |
Table 2 Comparison of the Absolute Values of HI Between Patients with Mild-to-Moderate and Severe Disease (GOLD Stage I and II vs GOLD Stage III and IV) |
On full-expiration, the COPD patients with lower-zone and/or left-lung-predominant AT showed significantly lower pulmonary function than the patients with upper-zone and/or right-lung-predominant AT. There were no statistically significant differences in pulmonary function between lower-zone and upper-zone predominance and between left-lung and right-lung predominance on full-inspiration (Tables 3 and 4).
 |
Table 3 A Comparison of PFT Parameters Between Patients with Upper-Zone and Lower-Zone Predominance |
 |
Table 4 A Comparison of PFT Parameters Between Patients with Left-Lung and Right-Lung Predominance |
Correlation between the absolute values of EI_CC_HI, AT_CC_HI, EI_LR_HI or AT_LR_HI and the PFT results is analysed. The absolute value of AT_CC_HI was significantly positively associated with FEV1 (r = 0.374, p < 0.001), FEV1(%pred) (r = 0.333, p < 0.001), FVC (r = 0.268, p < 0.001), FEV1/FVC (r = 0.346, p < 0.001), and IC/TLC (r = 0.269, p < 0.01) (Figure 3A–E). The absolute value of AT_LR_HI was positively associated with FEV1 (r = 0.149, p < 0.05), FEV1(%pred) (r = 0.167, p < 0.05), and IC/TLC (r = 0.224, p < 0.05), whereas, no correlation was observed between the absolute value of AT_LR_HI and FVC or FEV1/FVC. There was no correlation between the absolute values of EI_CC_HI and EI_LR_HI and the PFT results. Multivariate linear regression analysis showed that a higher absolute value of AT_CC_HI and lower absolute value of AT_LR_HI indicated better pulmonary function. In contrast, the absolute values of EI_CC_HI and EI_LR_HI were not included in the model, after adjusting for age, sex, height, smoking history (pack-years), and EI and AT of the total lung (Table 5).
 |
Table 5 Multivariate Linear Regression Analysis Showing the Relationship Between CT Parameters and Pulmonary Function Parameters |
Discussion
We comprehensively studied cranial-caudal and left–right lung heterogeneity in COPD patients by using inspiratory-to-expiratory CT. The findings of this study showed that the heterogeneity in the distribution of AT correlated with pulmonary function changes, which may be helpful in understanding the development of airflow limitation in COPD patients.
In our study, subjects with a more homogeneous distribution of air trapping had worse lung function than those showing a heterogeneous distribution on full-expiration CT, whereas the absolute value of EI_CC_HI showed no significant intergroup difference on full-inspiration. This finding is consistent with a previous study in which there were no statistical differences in emphysema distributions for different clinical or physiologic parameters.9 Some studies10,11,15,16 have shown that patients with more homogeneous emphysema distribution have worse lung function on full-inspiration. This may be related to the different proportions of patients under GOLD classifications in the study group. Our results suggest that AT heterogeneity on full-expiration better reflects pulmonary function than emphysema heterogeneity on full-inspiration, which is rarely reported in the literature. A more heterogeneous distribution of emphysema and/or AT corresponds to relatively more residual normal lung parenchyma, better synchronization of respiratory movements that can be quantitatively assessed by strain measurement using dynamic-ventilation CT, and better pulmonary function.17,18 In the early stage of small airway lesions, expiratory dysfunction manifests exclusively, while with the progression of disease, both inspiratory and expiratory functions show impairment. The structural changes in expiratory CT are consistent with the pulmonary functional changes.
Our study also showed that the absolute value of AT_LR_HI was significantly different between the two groups on full-expiration, whereas the absolute values of EI_LR_HI showed no significant intergroup differences on full-inspiration, which was consistent with the literature.10 This result also confirmed our hypothesis that AT heterogeneity on full-expiration may be more valuable in reflecting pulmonary function.
The cranial-caudal heterogeneity of emphysema and/or AT distribution is an important predictor of the response to pulmonary function. However, there is little consensus regarding the definition of upper-zone-predominant and lower-zone-predominant emphysema and/or AT subtypes. Therefore, on the basis of HI, K-means clustering data-driven analysis was performed to divide the patients into upper-zone-predominant and lower-zone-predominant emphysema or AT subtypes with respect to cranial-caudal heterogeneity; and left-lung predominant and right-lung predominant subtypes between the left and right lungs. Previous studies3,7,9,19–21 have shown that subjects with upper-zone predominance had better functional status on full-inspiration. Although the subjects showed no obvious differences in PFT parameters in the upper- versus lower-lung predominance on full-inspiration in our research, our study found the same results on full-expiration. Our study also found that subjects with right-lung-predominant AT had significantly better functional status than those with left-lung-predominant AT on full-expiration, whereas there were no statistically significant differences in pulmonary function on full-inspiration, which is consistent with the previous study.10 These results further confirm our hypothesis that AT heterogeneity on full-expiration may be more advantageous in reflecting pulmonary function. It is not easy to identify the exact factors underlying the variety in heterogeneous distribution among patients with COPD. On the basis of previous investigations, specific genes19,20 (e.g., MMP-9, EPHX1, GSTP1) were found to be associated with distinct groups of subjects with upper lobe-predominant emphysema. Different etiologies may cause differences in the heterogeneous distribution. Early emphysema and AT may be located in the upper lung in patients with COPD caused by smoking, while early emphysema and AT caused by chronic bronchitis may begin in the lower lung. The COPD patients showing upper-zone dominant and/or right-lung-predominant distribution of AT had better pulmonary function, which may be related to the greater potential activity of the diaphragm. Lung hyperinflation has a major impact on the shape and position of the diaphragm due to the changes in the size of the lungs and chest,21,22 leading to diaphragmatic dysfunction, especially in lower-zone dominant and/or left-lung-predominant distribution. Our results may indicate that the airflow limitation in COPD patients develops from certain lung zones and/or one lung to the both lungs with disease progression. Furthermore, AT heterogeneity may provide a reference for estimating pulmonary function in COPD patients who are unable to cooperate with pulmonary function tests.
This study had several limitations. First, 179 subjects were enrolled with various degrees of disease severity, and patients classified as GOLD stage I (15.6%) and IV (6.7%) were limited. Second, as there are important sex-related differences in emphysema severity and distribution, the small proportion of participating women is a limitation of the present study. Additionally, the correlation between CT heterogeneity and clinical manifestations was not fully discussed in this study. Thus, future studies with larger sample sizes may yield more information about the heterogeneity of emphysema or AT via quantitative CT, as well as its correlation with clinical and functional changes.
Conclusion
In summary, subjects with more heterogeneous distribution and/or upper-zone-predominant and/or right-lung-predominant patterns on full-expiration tend to have better pulmonary function. Thus, in comparison with emphysema heterogeneity, AT heterogeneity better reflects the pulmonary function changes in COPD patients.
Author Contributions
All authors had full access to the study data, conducted the analysis, and take responsibility for the integrity of the data and the accuracy of the analysis. All authors contributed to drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Pauwels RA, Buist AS, Ma P, et al. GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: National Heart, Lung, and Blood Institute and World Health Organization Global Initiative for Chronic Obstructive Lung Disease (GOLD): executive summary. Respir Care. 2001;46:798–825.
2. Han MK, Bartholmai B, Liu LX, et al. Clinical significance of radiologic characterizations in COPD. COPD. 2009;6:459–467. doi:10.3109/15412550903341513
3. Haraguchi M, Shimura S, Hida W, et al. Pulmonary function and regional distribution of emphysema as determined by high-resolution computed tomography. Respiration. 1998;65:125–129.
4. Mair G, Miller JJ, McAllister D, et al. Computed tomographic emphysema distribution: relationship to clinical features in a cohort of smokers. Eur Respir J. 2009;33:536–542. doi:10.1183/09031936.00111808
5. Chae EJ, Seo JB, Song JW, et al. Slope of emphysema index: an objective descriptor of regional heterogeneity of emphysema and an independent determinant of pulmonary function. AJR Am J Roentgenol. 2010;194:W248–255. doi:10.2214/AJR.09.2672
6. Nakano Y, Sakai H, Muro S, et al. Comparison of low attenuation areas on computed tomographic scans between inner and outer segments of the lung in patients with chronic obstructive pulmonary disease: incidence and contribution to lung function. Thorax. 1999;54:384–389. doi:10.1136/thx.54.5.384
7. Tanabe N, Muro S, Tanaka S, et al. Emphysema distribution and annual changes in pulmonary function in male patients with chronic obstructive pulmonary disease. Respir Res. 2012;13:31. doi:10.1186/1465-9921-13-31
8. Gurney JW, Jones KK, Robbins RA, et al. Regional distribution of emphysema: correlation of high-resolution CT with pulmonary function tests in unselected smokers. Radiology. 1992;183:457–463. doi:10.1148/radiology.183.2.1561350
9. De Torres JP, Bastarrika G, Zagaceta J, et al. Emphysema presence, severity, and distribution has little impact on the clinical presentation of a cohort of patients with mild to moderate COPD. Chest. 2011;139:36–42. doi:10.1378/chest.10-0984
10. Ju J, Li R, Gu S, et al. Impact of emphysema heterogeneity on pulmonary function. PLoS ONE. 2014;9:e113320. doi:10.1371/journal.pone.0113320
11. Bastos HN, Neves I, Redondo M, et al. Influence of emphysema distribution on pulmonary function parameters in COPD patients. J Bras Pneumol. 2015;41:489–495. doi:10.1590/s1806-37562015000000136
12. Lee SM, Seo JB, Lee SM, et al. Optimal threshold of subtraction method for quantification of air-trapping on coregistered CT in COPD patients. Eur Radiol. 2016;26:2184–2192. doi:10.1007/s00330-015-4070-z
13. Yanli Gao, Xiaoli Zhai, Kun Li, et al. Asthma COPD overlap syndrome on CT densitometry: a distinct phenotype from COPD. COPD. 2016;13:471–476. doi:10.3109/15412555.2015.1102874
14. Miller MR, Crapo R, Hankinson J, et al. General considerations for lung function testing. Eur Respir J. 2005;26:153–161. doi:10.1183/09031936.05.00034505
15. Boueiz A, Chang Y, Cho MH, et al. Lobar emphysema distribution is associated with 5-year radiological disease progression. Chest. 2018;153:65–76. doi:10.1016/j.chest.2017.09.022
16. Saitoh T, Koba H, Shijubo N, et al. Lobar distribution of emphysema in computed tomographic densitometric analysis. Invest Radiol. 2000;35:235–243. doi:10.1097/00004424-200004000-00004
17. Yamashiro T, Moriya H, Matsuoka S, et al. Asynchrony in respiratory movements between the pulmonary lobes in patients with chronic obstructive pulmonary disease: continuous measurement of lung density by 4-dimensional dynamic-ventilation CT. Int J Chron Obstruct Pulmon Dis. 2017;12:2101–2109. doi:10.2147/COPD.S140247
18. Xu Y, Yamashiro T, Moriya H, et al. Strain measurement on 4-dimensional dynamic-ventilation CT: quantitative analysis of abnormal respiratory deformation of the lung in COPD. Int J Chron Obstruct Pulmon Dis. 2019;14:65–72. doi:10.2147/COPD.S183740
19. Ito I, Nagai S, Handa T, et al. Matrix metalloproteinase-9 promoter polymorphism associated with upper lung dominant emphysema. Am J Respir Crit Care Med. 2005;172:1378–1382. doi:10.1164/rccm.200506-953OC
20. DeMeo DL, Hersh CP, Hoffman EA, et al. Genetic determinants of emphysema distribution in the national emphysema treatment trial. Am J Respir Crit Care Med. 2007;176:42–48. doi:10.1164/rccm.200612-1797OC
21. Hellebrandová L, Chlumský J, Vostatek P, et al. Airflow limitation is accompanied by diaphragm dysfunction. Physiol Res. 2016;65:469–479.
22. Scheibe N, Sosnowski N, Pinkhasik A, et al, Sonographic evaluation of diaphragmatic dysfunction in COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;10:1925–1930.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.