Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Indexing Executive Functions with Test Scores, Parent Ratings and ERPs: How Do the Measures Relate in Children versus Adolescents with ADHD?
Authors Häger LA, Øgrim G, Danielsen M, Billstedt E , Gillberg C , Åsberg Johnels J
Received 7 September 2019
Accepted for publication 7 January 2020
Published 17 February 2020 Volume 2020:16 Pages 465—477
DOI https://doi.org/10.2147/NDT.S230163
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Linda Angelica Häger,1,2 Geir Øgrim,1,2 Maria Danielsen,1 Eva Billstedt,2 Christopher Gillberg,2 Jakob Åsberg Johnels2,3
1Neuropsychiatric Team, Åsebråten Clinic, Østfold Hospital Trust, Fredrikstad, Norway; 2Gillberg Neuropsychiatry Centre, Institute of Neuroscience and Physiology, University of Gothenburg, Gothenburg, Sweden; 3Speech and Language Pathology Unit, Institute of Neuroscience and Physiology, University of Gothenburg, Gothenburg, Sweden
Correspondence: Linda Angelica Häger
Neuropsychiatric Team, Åsebråten Clinic, Østfold Hospital Trust, 1740 Grålum, Fredrikstad PB300, Norway
Tel +47 93 28 76 77
Email [email protected]
Objective: Rating scales and neuropsychological tests including continuous performance tests (CPTs) are widely used to assess executive functions (EFs). Event-related potentials (ERPs) are also used to index certain EFs such as action preparation and inhibition. In this descriptive study, we examined the associations between results on an EF rating scale, a CPT and ERP components in ADHD as a function of age.
Methods: Fifty-nine patients with ADHD (and more often than not with comorbid disorders) in two age groups (9– 12 years and 13– 17 years) were assessed using EF ratings, a visual CPT and ERPs (CueP3, P3go and P3no-go).
Results: There were age related changes in the ERPs with the CueP3 amplitude being stronger in children, and the P3no-go amplitude stronger in adolescents. The associations between the EF measures were different in the two age groups. In particular, the P3no-go seemed to reflect different EF-related processes in children versus adolescents.
Conclusion: Age group effects were seen on a selection of ERP amplitudes in this sample of patients with ADHD. Ratings, test scores and EF-related ERPs seem to capture different aspects of EF in ADHD, and the associations differed depending on age group. The results show that different measures of EF are not interchangeable and highlight the importance of age when interpreting ERPs.
Keywords: executive functions, ADHD, electrophysiology, ratings, event related potentials, cognitive control
Introduction
ADHD (attention deficit hyperactivity disorder) is a neurodevelopmental disorder affecting approximately 3–5% of school-aged children.1 The symptoms defining the disorder are attention deficits, impulsivity and/or excess levels of activity. The Diagnostic and Statistical Manual of Mental Disorders, DSM-52 defines three presentations of ADHD – predominantly inattentive (ADHD-I), predominantly hyperactive/impulsive (ADHD-H) and combined (ADHD–C). Across the ADHD population, a very high percentage of comorbid conditions is found.3,4
A large number of studies have described deficits of executive functioning (EF) in ADHD,4–7 although EF deficits are not directly part of the diagnostic criteria. EF is an umbrella term that involves separate, but interrelated, cognitive processes such as initiative, inhibition, working memory, planning, monitoring, focusing and attentional flexibility.8 Cognitive control is often used as a synonym of EF.9,10 Executive processes develop and change over the lifespan.11,12 Because deficits in EF tend to correlate with impaired academic functioning and occupational achievement,13,14 assessment of EF is clinically relevant as a target for possible intervention in ADHD.15–17 Although it has been known for many years that EF is related to the integrity of certain fronto-parietal brain systems,18 many questions remain regarding the precise nature of EF deficits in ADHD, and what the best procedures for assessment are.19,20 Conventionally, EF has been evaluated using neuropsychological performance tests and rating scales.
Continuous performance tests (CPTs) have been used for more than 60 years in clinical populations to assess aspects of attention that require executive control.21 A task analysis suggests that a whole range of executive processes are involved during CPT tasks, including 1) action selection 2) shift from one action to another, 3) action preparation, 4) action execution, 5) working memory, 6) suppression of prepared action, 7) inhibition of ongoing activity, 8) detection of conflict, 9) adjusting future behavior in order to avoid conflicts.9,22 Most CPTs – such as, Conners’ CPT 2,23 the Tova Test24 and the QB test25– apply a go/no-go paradigm, that reports on the number of omission and commission errors, hit reaction time (RT), and RT variability (RTvar). Omission errors and RTvar seem to be most sensitive to ADHD.26,27 A meta-analysis showed that CPT omission errors differed significantly between groups with and without ADHD in 23 of 30 studies.5 Also, CPT results have been significantly, albeit only moderately, effective in discriminating between ADHD and other neurodevelopmental disorders.28
One of the most frequently used questionnaires for rating of EFs in children and adolescents is the Behavior Rating Inventory of Executive Functions (BRIEF),29 a parent and teacher questionnaire for children aged 5–18 years (see Methods for a detailed description). Children with ADHD tend to score in the clinically impaired range on the BRIEF, supporting the validity of BRIEF, as well as the EF deficit theory of ADHD.30–32
Tests and rating measures of EF have both been found to reliably differentiate between ADHD and controls.7,33–35 Compared with neuropsychological tests of EF, rating scales have a number of potential benefits: Data stem from people who know the child well and provide an evaluation of behavior for a longer time period than a single test session. Neuropsychological tests are performed in lab-like settings and therefore may also lack ecological validity. On the other hand, rating scales are potentially sensitive to factors such as negative halo effects36 and temporal instability.37 Moreover, ratings are by necessity subjective, and can often differ significantly across different informants.38
Several studies have examined the relationship between tests and ratings of EF in ADHD. The overlaps appear inconsistent, with correlations sometimes being non-significant or only small-to-moderate.1,39 Such findings may seem surprising. However, Toplak et al39 concluded in a comprehensive treatise of the literature, that EF tests and EF questionnaires to some extent measure different aspects of EF, at different cognitive levels and can be said to have separate clinical utility by reflecting “optimal” (tests) versus “typical” (ratings) EF capacities.
Another information source reflecting EF is event-related brain potentials (ERPs). ERPs are measures of brain responses resulting from a specific sensory, cognitive or motor events, and are increasingly used for studying EF.9,22,38,40–45 More precisely, ERPs are cerebral generated electrical voltages recorded on the scalp in response to specific stimuli or responses.22 Due to the high time resolution of the ERPs, they can potentially give important insights into the temporal neural activity related to behavioral parameters in tasks.40,46,47 The EEG equipment needed to register ERPs is non-invasive, often affordable, and could potentially become a viable clinical tool supplementing tests and ratings.
Cued go/no-go tasks are ERP paradigms often used to study EF.9,22,48–50 As mentioned, a range of cognitive processes are involved. By recording ERP patterns during such tasks, we may potentially get access to underlying operations that occur between stimulus engagement and response execution - data not evident in behavioral test parameters. (See Methods for a detailed description of our go/no-go paradigm).
In cued go/no-go paradigms, ERP components reflecting cognitive control occur in the time interval 300–600 ms. after stimulus presentation.40 A large number of studies in adults and, to a lesser extent, in children and adolescents have explored the neural mechanisms and the functional meanings of ERPs, including the Cue P3, P3go, and P3no-go components as assessed in terms of amplitude and/or latency.9,22 The Cue P3 has a mainly parietal location and is presumed to be associated with the identification of a target in the task. The P3go is a posteriorly positive deflection that peaks after the second stimulus in the go condition, and is thus thought to reflect the association between target and response, ie, response selection.48 The P3no-go is a central positive component linked to activation of alternative responses, inhibition and/or allocation of attention recourses.41,47 It is, however, important to note that ERPs change with age,47 a fact that has sometimes been ignored in clinical ERP research. For instance, the Cue P3 has been found to be stronger in children than in adults, suggesting a stronger preparation process.50 Likewise, the P3go latency has been reported to decrease with age,51 while the P3no-go is often absent in small children and has been shown to increase in amplitude from age nine until adolescence in typically developing populations.50 It has been suggested by several authors52 that ERPs are excellent for studying brain development. Amplitudes and latencies of the ERP components may index development53,54 such as synaptic density, myelination, pruning and increase of pathways (see55). On the other hand, we still do not know enough about how age is related to the functional and clinical meaning of these ERPs.56
ERPs in ADHD have been assessed in a substantial number of studies. In a review,47 several cognitive ERPs related to EF were reported to differ significantly between ADHD and controls, although it was also mentioned that general conclusions were difficult to reach partly due to mixed findings and the lack of standardized procedures in current ERP research. So far ERPs have not been widely used in clinical settings partly because different paradigms and methods of calculation are applied in research studies, but also because more knowledge is needed regarding the functional relevance of ERP signatures in ADHD46 and other disorders. For instance, surprisingly few studies have examined the relationship between EF-related cognitive ERPs and neuropsychological EF test scores38,57 and/or ratings of EF. Moreover, as highlighted by Johnstone et al,47 a greater consideration of possible age/developmental effects is needed.
Current Study
In the current study of 59 children and adolescents with ADHD, we descriptively explore the association between test scores, parent ratings of EF and a selection of EF and ADHD related ERPs, ie Cue P3, P3go and P3no-go amplitudes, as assessed during CPT. In order to better understand the functional meaning and clinical significance of the ERPs, we specifically examined age-related effects by 1) comparing children (age 9–12 years) with adolescents (age 13–17 years) on the ERPs and 2) by conducting separate correlation analyses for children and adolescents between the EF ratings, test scores, and ERPs. Two main questions were asked:
RQ 1: Do children with ADHD differ from adolescents with ADHD differ with regard to EF as indexed in parent ratings, CPT scores and ERPs? RQ 2: How do EF-related parent ratings, test scores and ERPs correlate in children with ADHD and in adolescents with ADHD?
Methods and Materials
Participants
The participants in this study were 59 patients diagnosed with ADHD (ages 9–17), according to the DSM-IV criteria (included before DSM5). Based on description in the review of Johnstone,47 we subdivided the sample into children and adolescents based on cut -off of twelve years, 6 months with a view to examining possible ERP differences related to age in our main analyses.
Participants were recruited from child and adolescent psychiatric outpatient clinics in the county of Østfold, Norway. They had been referred from their GP, due to mental health problems and/or neurodevelopmental concerns. The demographics and characteristics of the sample are shown in Table 1. Common comorbidities such as behavioral and emotional disorders, Tourette syndrome, learning disabilities and autism spectrum disorders, were not excluded since this would have severely impacted the generalizability of the results as an overwhelming majority of ADHD patients show comorbid disorders if these are thoroughly assessed for.3 Exclusion criteria were an IQ score below 70 or diagnosed brain pathology, such as epilepsy. Besides intellectual disability (ie IQ < 70) the comorbidities were not uniformly and consistently assessed in all cases; therefore, we report the diagnoses that are provided for all, but note that there might be additional comorbid cases not fully assessed and refrain from conducting analyses regarding the eventual impact of comorbidity.
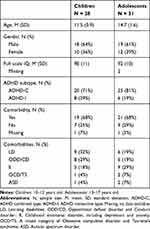 |
Table 1 Sample Characteristics |
There were no significant differences in ADHD subtype or in the proportion of co-morbid conditions between the age groups (Table 1). However, the adolescent group had non-significantly lower FSIQ (t[55] = −1.9, p = 0.063). In order to examine whether this possible difference may have affected the results we performed correlation coefficients between all (CPT, ERP and rating) measures of interest and IQ across the full sample; no significant correlations were obtained (all rho < ± 0.26, all p > 0.05).
Diagnostic Procedures
The participants were diagnosed according to the DSM-IV. All patients were screened medically by a medical doctor, and anamnestic data was collected. Clinical interviews (eg Kiddie Sads), school observations, teacher and parent ratings (Aseba and Conners’ 3) were completed according to standard clinical practice.58–61 Intelligence was tested using the WISC-IV by qualified clinical psychologists.62 Diagnostic decisions were based on all available information in multi-professional teams, comprising at a minimum a child psychiatrist and a psychologist. For the purposes of the research, the patients were referred to the specialist pediatric neuropsychiatric team in Østfold County, Norway, for extended examinations which included BRIEF and registrations of ERPs. Children and parents were informed about the study and gave written consent that clinical data could be used for research purposes. None of the participants were treated with ADHD medication at the time of assessment.
The study was approved by the regional ethics committee (2016/1453).
Brief
The Behavior Rating Inventory of Executive Functions (BRIEF) consists of 86 items organized into subscales31 that form eight sub-scores, two index scores and a summary score, the Global Executive Composite (GEC). To reduce the number of correlations and the risk of type 1 error, we only consider the GEC score here. The BRIEF questionnaires were filled in by the parents, most often the mothers. The ratings were electronically scored and compared with norms. T-scores (Mean = 50, SD = 10) were registered.
Event-Related Potentials and the Cued CPT (Go/No Go) Task
A 19–channel tin electrode cap was used for recording ERP data applying Win EEG software developed by Ponomarev and Kropotov.41 As described in Ogrim and Kropotov,63 input signals were referenced to earlobe electrodes, filtered between 0.5 and 50 Hz, and digitized at a sampling rate of 250 Hz, with impedance kept below 5 khm for all 19 electrodes. Electrodes were placed in accordance with the international 10–20 system. EEG data were re-referenced offline to the common average montage prior to data processing. Eye-blink artefacts were corrected by zeroing the activation curves of individual independent components extracted by independent component analysis (Infomax algorithm) and corresponded to eye-blink topographies. EEG epochs with excessive amplitude (100 μV) and/or excessively fast (35 μV in 20–35 Hz band) and slow (50 μV in 0–1 Hz band) frequency activities were automatically excluded from analysis.
Event-related potentials (ERPs) were recorded using a 20 min cued CPT task consisting of pictures in pairs appearing with inter- stimulus intervals of 1000 ms and inter- trial intervals of 3000 ms. The task was originally designed by J. Kropotov and used in data collection for the Human Brain Institute Database (http://hbimed.com). The task consists of 400 trials. The go condition is a second animal in the pair (animal-animal). The participants were instructed to be equally fast and accurate when responding. Twenty different animals (a), plants (p), and humans (h) are presented randomly in various combinations: a-a, a-p, p-p, p-h; 100 trials in each category. Occasionally, a sound is presented with the human images (p-h). The participants were informed to ignore the sounds; in the current experiment the Cue P3, P3go and P3no-go is in focus. Before starting the task, the participants practiced as long as necessary. They performed the task on a 17- inch computer screen, positioned 1.5 meters in front of them.
In Ogrim and Kropotov,63 amplitudes of the ERP components were measured at the electrodes where the components were observed to be strongest in the grand-average ERP files of the total sample. The time interval for the ERPs were determined by the grand average files. In the present study we use the same time intervals and sites because the samples are partly overlapping and cover the same age range. All component amplitudes were measured manually as the local peak amplitude within the predefined time- intervals. Local peak amplitude refers to the point within the defined time window for the component of interest with the largest amplitude, which is surrounded on both sides by lower voltages, thereby avoiding measuring the offset of preceding or onset of following components. The sites, time intervals and cue stimulus for the ERPs the following: cue-P3 located at Pz (270–370 ms) elicited from all (a) stimuli, P3go located at Pz (260–400 ms) elicited after stimulus 2 in (a-a) stimuli combinations; P3no-go at Cz (300–500 ms) elicited after stimulus 2 in (a-p) stimuli combinations. Following the correction procedures, the ERP waveforms were based on between 40 and 100 trials in all cases. Figure 1 illustrates the cued CPT paradigm.
 |
Figure 1 VCPT: Visual continuous performance test. Notes: The figure shows the four conditions of the VCPT. One hundred trials of each condition are presented randomly. Total test time is 20 min. |
In this study, we focus on amplitudes and not latencies as amplitudes have been shown more reliable in ADHD.64 In addition, this reduced the number of analyses conducted.
Results
Do Children and Adolescents with ADHD Differ with Regard to EF as Indexed in Parent Ratings, CPT Scores and ERPs?
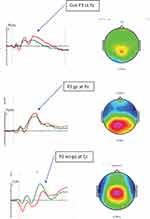
The mean scores for each age group on the BRIEF, neuropsychological measures, and the ERPs are shown in Table 2. The BRIEF GEC data indicates that the groups on average scored in or above the “potentially clinical” range (which traditionally is equal to about 1.5 SD above the normative mean of T = 50 [SD = 10]29). A non-parametric Mann–Whitney U-test revealed no significant age group difference on the BRIEF (p = 0.16). On the CPT test scores, the child group had significantly slower RT (U = 291, z = −2.16, p = 0.031) and larger RTvar (U = 302, z = −2.00, p = 0.045) compared with the adolescent group, whereas no significant differences were seen for the number of omissions or commissions (p >0.12). In terms of ERP amplitudes, the child group had significantly larger Cue p3 amplitude (U = 278, z = −2.37, p = 0.018) but smaller no-go amplitudes (U = 588, z = 2.34, p = 0.019) compared with the adolescent group, whereas no significant difference was seen for the P3go component (p = 0.57). Figure 2 shows the ERP waveforms for each age group as well as topographic maps of the ERP components.
 |
Table 2 Mean (M) and Standard Deviations (SDs) for the BRIEF, the VCPT and ERPs |
How Do EF-Related Parent Ratings, Test Scores and ERPs Correlate in Children and in Adolescents with ADHD?
In order to examine the correlations between measures of EF according to the BRIEF, the CPT test scores, and the ERP data, non-parametric correlations were computed for each age group separately. The correlation matrix for the child group is presented in Table 3 and for the adolescent group in Table 4. In the child group, we found significant and moderately sized correlations between BRIEF ratings and some CPT test scores, ie the number of commissions (rho = 0.390, p < 0.05) and RT (rho = −0.409, p < 0.05). The directions of the correlations show that greater EF difficulties according to parent ratings are associated with a higher number of commissions and faster RTs.
 |
Table 3 Correlations Between BRIEF, Test Scores and ERPs for Age Group 10–12 (N = 28) |
 |
Table 4 Correlations Between BRIEF, Test Scores and ERPs for Age Group 13–17 (N = 31) |
In the adolescent group, however, no significant correlations were seen between BRIEF ratings and CPT test scores (all ps > 0.12).
Turning to the ERPs, in the child group we found significant positive correlations between the P3no-go amplitude and the parent ratings (rho = 0.402, p < 0.05), as well as a negative correlation between P3no-go and reaction time (rho = −0.573, p < 0.01). A significant positive correlation between number of commissions and P3no-go amplitude was also found (rho = 0.497, p < 0.01). The directions of these correlations show that, in children, larger P3no-go amplitudes align with more EF problems according to the BRIEF ratings and with fast and error-prone performance on the CPT.
In the adolescent group, there were significant negative correlations between P3no-go and the number of omissions (rho = −0.377, p < 0.05), reaction time (rho = −0.705, p < 0.01), and RTvar (rho = −0.655, p < 0.01). The direction of these correlations differs from those in the child group, as a large P3no-go amplitude aligned with better CPT test performance in the adolescent group. Unlike in children, there were no significant associations between ERP data and BRIEF ratings in the adolescent group.
Finally, when the CPT and the ERP measures were considered separately, correlations within each type of measure were found, as is reported for descriptive purposes in Tables 3 and 4. The one exception is that in the child group, the P3no-go did not correlate with P3go or Cue p3 amplitudes.
Complementary Analyses in the Full Sample
Although our subdivision in age subgroups was informed by similar distinctions in the existing literature,47 it could be argued that the cut off is rather arbitrary; therefore, we also did complementary analyses using the full sample with collapsed age‐groups for descriptive purposes. When correlating BRIEF ratings, CPT test scores and ERP measures, BRIEF ratings were negatively associated with reaction time (rho = 0.31, p = 0.02), and positively with P3no-go amplitudes (rho = 0.284, p = 0.03). Reaction time variability was also negatively associated with P3go amplitudes (rho = 0.321, p = 0.013). Moreover, chronological age correlated with RT (rho = −0.393, p = 0.002), RTvar (rho = −0.369, p = 0.004), omissions (rho = −0.276, p = 0.034), Cue P3 (rho = −0.379, p = 0.003), P3no-go (rho = −0.350, p = 0.007).
Discussion
Executive function (EF) is an umbrella term for a set of complex functions of cognitive control that are often examined and found impaired in ADHD patients. Traditionally, EFs are assessed applying rating scales and neuropsychological tests. There has been ongoing interest in the clinical utility and the convergence of these EF measures.65 In addition to the conventional measures of EF, cognitive ERPs have been used for several decades in research but are not widely used in clinics. To better understand the relations between these “methods” of EF assessment, and to gain more insight regarding the clinical relevance of ERPs, the aim of this study was to examine how conventional measures of EF (BRIEF ratings and CPT test scores) in ADHD relate to electro-cortical signatures of EF. We also wanted to explore the age effects by comparing children with adolescents and by performing correlations between different measures of EF separately for each age group. In short, results showed that the associations between these indices were subtle and, moreover, that they varied with age.
With regard to research question 1, we noted differences between the two age groups on two of the ERPs. Specifically, the child group had larger Cue p3 amplitude and smaller P3no-go amplitudes compared with the adolescent group, whereas no significant difference was seen for the P3go component. This suggests that the development of ERP amplitudes differs depending on which ERP component is in focus, which is a finding obtained in prior research on non-ADHD samples. Both Jonkman50 and Hämmerer et al66 similarly report that children show increased Cue P3 in children than in adolescents and adults. This might reflect higher alertness in this age group and more generally that the readiness to utilize cues to guide actions is early developing. On the CPT test scores there were differences with regards to the speed and the variability in responding with adolescents performing faster and more stable than children. In the correlation analyses in the full sample, also omissions were fewer with increasing age.
Turning to research question 2, in the adolescent group, no significant correlations between ratings of EF and CPT test data were found. This is in line with many previous findings (see Toplak39). Importantly, this result does not necessarily mean that any (or both) of these measures are invalid indicators of EF. In fact, it has been suggested that these methods should not be used interchangeably as parallel measures of executive function in clinical assessments. Both domains of assessment are potentially useful and valuable, but they provide different types of information in the context of clinical assessment.39 Toplak et al39 further describes how neuropsychological test results indicate how well the individual can perform in a situation with structure and guidance (“optimal performance”), whereas the EF ratings potentially provide a broader picture of how the individual manages to function in a world full of distractions and with a constant need to prioritize.
In the child group, we did find that high problem scores on the BRIEF correlated with more commissions, and a fast reaction time, which collectively indicate problems with impulsivity. This difference in the child vs the adolescent group may help to explain diverging findings obtained in this field, as the age of the child seems to affect how the association between EF test scores and ratings manifest. At this stage, we do not know why associations between ratings and test scores of EF were seen only in the child group. One possibility – inspired by the reasoning of Toplak et al – is that differences between children and adolescents in the amount of independence expected and allowed for in their everyday living will impact the kinds of EF capacities tapped by a parent rating and consequently the strength of the rating’s association with EF test scores. If, as Toplak et al reason, ratings are superior to test scores in indexing the individual’s goal-setting capacities in relation distractions and other environmental factors in everyday life whereas test scores are superior in measuring an individual’s optimal performance during guidance and structure, it might be the case that young children’s everyday life is more similar to a test situation since there is typically adults around for guidance and structure. This hypothesis could potentially help explain why parent ratings and test scores were associated only in the child group. This hypothesis needs to be tested further in future studies.
Regarding ERPs, the analyses revealed some striking differences in the patterns of correlations in the two age groups. In the child group, a significant positive correlation was found between P3no-go and BRIEF scores. The direction of the correlation is somewhat unexpected as is indicates that a strong P3no-go amplitude aligns with high levels of executive difficulties according to parent ratings. In addition, we found in the same subgroup that a high rate of commissions correlated positively with P3no-go. Taken together, the pattern of these correlations indicates that larger P3no-go amplitudes in the 9–12-year-olds with ADHD mirror increased impulsivity. Although the specifics are to be settled, existing accounts of the P3no-go component, informed mainly on research on adults, suggest that a strong component reflects better cognitive control.41 It has further been reported that the component often is small or absent in young children under the age of approx. 9 years.50 In all ERP studies applying paradigms such as ours, P3 no-go amplitude and fast RT are positively correlated. In CPTs impulsivity is characterized by a high number of commission errors and fast RT. This may create a strong P3 no-go component reflecting impulsivity and not cognitive control. If so, a strong amplitude of P3 no-go in a patient with fast RT and many commissions should not be considered an indication of good cognitive control. As mentioned, P3 no-go may be weak or absent in young TD children. This may explain why the “impulsivity P3 no-go” significantly contributed to the P3 no-go amplitude in the child group, but not in the adolescent group. To the best of our knowledge, it has never previously been shown that the amplitude of P3 no-go is associated with increased impulsivity in middle childhood patients with (or without) ADHD, and therefore the current findings are in need of replication. It has, however, been shown previously that ERP components are dynamic phenomena that can be affected by task instructions. For instance, Aasen and Brunner found that asking participants to optimize response speed as compared to instructions to prioritize accurate responding during CPT enhanced the activation of cognitive processes reflected in several of the investigated ERP components, including P3no-go amplitude.67 Even though the instructions were the same for all participants in the current study, it might be that young, more impulsive children with ADHD “chose” to prioritize speed over accuracy to a higher extent and that this could help explain why P3no-go amplitudes seem to mirror impulsivity in the child group.
In the adolescent group, significant negative correlations were found between P3no-go and omissions, reaction time, and RTvar. These findings are in line with a large body of research showing that the amplitude of the P3no-go component is associated with better cognitive control. It is also interesting that the CPT variables that were linked with P3no-go amplitudes – ie omissions and reaction-time variability – are the CPT variables often seen to be particularly impaired in ADHD.26 Collectively, our study provides a combination of new results and corroborations of prior findings, and thus contributes to a refined understanding of brain-behavior relations during executive processes in ADHD. In particular, the results underscore the importance of considering age effects.
Our distinction into age subgroups was motivated by considerations in the existing literature. Indeed, Johnstone et al47 note in their review that prior ERP research have “appropriately used a small age-range, with child participants aged approximately 7–12 years […], or similarly, adolescent participants aged approximately 13–18 years“ (p 645). That said, future research aiming to explore age effects of ERPs in children and adolescents might want to examine how the ERP amplitudes relate to other measures of EF without making a priori decision of where any categorical age distinctions should be placed. Such analyses would likely need larger samples sizes. We finally also did complementary analyses using the full sample with collapsed age-groups for descriptive purposes. Although these linear correlations are silent to the possibility of different functional meaning of the P3 no-go component, they provide descriptive information regarding changes with age.
Clinical Implications
Rating scales, neuropsychological tests (CPTs) and cognitive ERPs have all been proposed as methods for the assessment of EFs. It has been argued that ratings and tests index different levels of executive function, and therefore correlations can be expected to be rather weak. This study is in line with such a proposal. In order to provide best possible support in clinical contexts, the result underscore the need of integrating or comparing different sources of information in order to understand individual’s functioning in different milieus and conditions. In addition, the picture is probably even more complex if we also consider teachers rating and self ratings that were not included in the current study. Hence, it is important to know that these measures can not be considered interchangeable as measures of EF. The pattern of deviances in ERPs may indicate which basic processes that are impaired (preparing, monitoring, inhibition etc.), which in the future, this may guide treatment options (medication response, cognitive training, etc). The current study also suggest that we need more basic research on how the ERPs manifest EF function in children of different ages and how they relate to other measures of EF.
Limitations and Future Directions
A first set of possible limitations has to do with the choice of task paradigm during CPT and ERP assessment. In particular, the associations between ERPs and test data may have been affected by the fact that performance scores and the ERPs were all extracted from the same CPT session. If a separate CPT, not involved in ERP registrations, had been used, we do not know whether the same correlations would have been found. Relatedly, we chose to focus on CPT performance rather than assessing performance on a broader set of neuropsychological capacities such as planning or switching. Finally, we also chose to focus on a selection of ERPs, while not focusing on others ERPs (eg N2, CNV, P1). The ERPs were selected based on a considerations of reliability and that they have been shown to be associated with ADHD and cognitive control/EF in prior research. Moreover, we chose to focus on a rather small number of ERP variables in order not to include too many variables in the analyses. Future research is needed to determine the possible relevance of such methodological choices.
A second possible limitation has to do with the sample. Our findings are based on a group of 59 pediatric ADHD patients, most of whom also had “comorbid” disorders such as learning disorders. The high rate of comorbidity in both age groups reflects clinical reality in the field of pediatric ADHD, but, by implication, we do not know to what degree the results apply specifically for “pure ADHD”, or for other co-occurring neurodevelopmental symptoms or disorders. Therefore, future research should specifically examine if comorbidity patterns affect the manifestation of EF deficits as assessed with ratings, tests and ERPs. A final potential weakness is the lack of a typically developing comparison group of children and adolescents without ADHD as well as “non-ADHD” neurodevelopment disorders cases. While we find it hard to see how the inclusion of such groups could affect the conclusions obtained here, it is important to note that we really do not know whether the pattern of results presented here reflects only children/adolescents with ADHD or if they mirror more general developmental patterns. We believe these are all important topics for future research to address.
Compliance with Ethical Standards
The study was approved by the ethical review board in Norway (project nr 2016/1453), and the procedures conformed to the Helsinki Declaration.
Funding
This research was funded by Østfold Hospital Trust, Fredrikstad, Norway.
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Krieger V, Amador-Campos JA. Assessment of executive function in ADHD adolescents: contribution of performance tests and rating scales. Child Neuropsychol. 2017;24:1–25.
2. American Psychiatric A. Diagnostic and Statistical Manual of Mental Disorders: DSM-5.
3. Kadesjo B, Gillberg C. The comorbidity of ADHD in the general population of Swedish school-age children. J Child Psychol Psychiatry. 2001;42(4):487–492. doi:10.1111/jcpp.2001.42.issue-4
4. Barkley RA. Distinguishing sluggish cognitive tempo from ADHD in children and adolescents: executive functioning, impairment, and comorbidity. J Clin Child Adolescent Psychol. 2013;42(2):161–173. doi:10.1080/15374416.2012.734259
5. Willcutt EG, Doyle AE, Nigg JT, Faraone SV, Pennington BF. Validity of the executive function theory of attention-deficit/hyperactivity disorder: a meta-analytic review. Biol Psychiatry. 2005;57(11):1336–1346. doi:10.1016/j.biopsych.2005.02.006
6. Pliszka S. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894–921. doi:10.1097/chi.0b013e318054e724
7. Nigg JT, Jester JM, Stavro GM, Ip KI, Puttler LI, Zucker RA. Specificity of executive functioning and processing speed problems in common psychopathology. Neuropsychology. 2017;31(4):448–466. doi:10.1037/neu0000343
8. Muscara F, Catroppa C, Anderson V. The impact of injury severity on executive function 7–10 years following pediatric traumatic brain injury. Dev Neuropsychol. 2008;33(5):623–636. doi:10.1080/87565640802171162
9. Kropotov JD. Functional Neuromarkers for Psychiatry: Applications for Diagnosis and Treatment. Academic Press; 2016.
10. Davidson MC, Amso D, Anderson LC, Diamond A. Development of cognitive control and executive functions from 4 to 13 years: evidence from manipulations of memory, inhibition, and task switching. Neuropsychologia. 2006;44(11):2037–2078. doi:10.1016/j.neuropsychologia.2006.02.006
11. Huizinga M, Smidts DP. Age-related changes in executive function: a normative study with the Dutch version of the Behavior Rating Inventory of Executive Function (BRIEF). Child Neuropsychol. 2011;17(1):51–66. doi:10.1080/09297049.2010.509715
12. Williams BR, Ponesse JS, Schachar RJ, Logan GD, Tannock R. Development of inhibitory control across the life span. Dev Psychol. 1999;35(1):205–213. doi:10.1037/0012-1649.35.1.205
13. Andrade BF, Brodeur DA, Waschbusch DA, Stewart SH, McGee R. Selective and sustained attention as predictors of social problems in children with typical and disordered attention abilities. J Atten Disord. 2009;12(4):341–352. doi:10.1177/1087054708320440
14. Clark C, Prior M, Kinsella G. The relationship between executive function abilities, adaptive behaviour, and academic achievement in children with externalising behaviour problems. J Child Psychol Psychiatry. 2002;43(6):785–796. doi:10.1111/jcpp.2002.43.issue-6
15. Biederman J, Monuteaux MC, Doyle AE, et al. Impact of executive function deficits and attention-deficit/hyperactivity disorder (ADHD) on academic outcomes in children. J Consult Clin Psychol. 2004;72(5):757–766. doi:10.1037/0022-006X.72.5.757
16. Biederman J, Petty C, Fried R, et al. Impact of psychometrically defined deficits of executive functioning in adults with attention deficit hyperactivity disorder. Am J Psychiatry. 2006;163(10):1730–1738. doi:10.1176/ajp.2006.163.10.1730
17. Preston AS, Heaton SC, McCann SJ, Watson WD, Selke G. The role of multidimensional attentional abilities in academic skills of children with ADHD. J Learn Disabil. 2009;42(3):240–249. doi:10.1177/0022219408331042
18. Friedman NP, Miyake A. Unity and diversity of executive functions: individual differences as a window on cognitive structure. Cortex. 2017;86:186–204. doi:10.1016/j.cortex.2016.04.023
19. Delis DC, Kramer JH, Kaplan E, Holdnack J. Reliability and validity of the Delis-Kaplan executive function system: an update. J Int Neuropsychol Soc. 2004;10(2):301–303. doi:10.1017/S1355617704102191
20. Kamradt JM, Ullsperger JM, Nikolas MA. Executive function assessment and adult attention-deficit/hyperactivity disorder: tasks versus ratings on the Barkley deficits in executive functioning scale. Psychol Assess. 2014;26(4):1095–1105. doi:10.1037/pas0000006
21. Beck LH, Bransome ED
22. Luck SJ. An Introduction to the Event-Related Potential Technique. MIT press; 2014.
23. Conners CK. Conners Continous Performance Test-II (CPT-II) Computer Program for Windows Technical Guide and Software. Toronto: Multi-Health Systems Inc.; 2000.
24. Greenberg LMM. MN: Author. Minneapolis: Greenberg, L.M. T.O.V.A.™ interpretation manual; 1991.
25. Knagenhjelm P, Ulberstad F. Qbtest-Technical Manual. Stockholm: Qbtech AB; 2010.
26. Egeland J, Kovalik-Gran I. Measuring several aspects of attention in one test: the factor structure of Conners’s continuous performance test. J Atten Disord. 2010;13(4):339–346. doi:10.1177/1087054708323019
27. Ogrim G, Kropotov J, Hestad K. The quantitative EEG theta/beta ratio in attention deficit/hyperactivity disorder and normal controls: sensitivity, specificity, and behavioral correlates. Psychiatry Res. 2012;198(3):482–488. doi:10.1016/j.psychres.2011.12.041
28. Hult N, Kadesjo J, Kadesjo B, Gillberg C, Billstedt E. ADHD and the QbTest: diagnostic Validity of QbTest. J Atten Disord. 2018;22(11):1074–1080. doi:10.1177/1087054715595697
29. Gioia G, Isquith PK, Guy SC, Kenworthy L. Behavior Rating Inventory of Executive Function-Professional Manual. Lutz: Pyychological Assessment Resources, Inc; 2000.
30. Tan A, Delgaty L, Steward K, Bunner M. Performance-based measures and behavioral ratings of executive function in diagnosing attention-deficit/hyperactivity disorder in children. Atten Defic Hyperact Disord. 2018;10(4):309–316. doi:10.1007/s12402-018-0256-y
31. Roth RM, Isquith PK, Gioia GA. Assessment of executive functioning using the Behavior Rating Inventory of Executive Function (BRIEF). In: Goldstein S., Naglieri. J (eds). Handbook of Executive Functioning. Springer; 2014:301–331.
32. Gioia GA, Isquith PK, Kenworthy L, Barton RM. Profiles of everyday executive function in acquired and developmental disorders. Child Neuropsychol. 2002;8(2):121–137. doi:10.1076/chin.8.2.121.8727
33. Scheres A, Oosterlaan J, Geurts H, et al. Executive functioning in boys with ADHD: primarily an inhibition deficit? Arch Clin Neuropsychol. 2004;19(4):569–594. doi:10.1016/j.acn.2003.08.005
34. Mahone EM, Cirino PT, Cutting LE, et al. Validity of the behavior rating inventory of executive function in children with ADHD and/or Tourette syndrome. Arch Clin Neuropsychol. 2002;17(7):643–662. doi:10.1093/arclin/17.7.643
35. Stanovich K, Toplak ME, West RF. The development of rational thought: a taxonomy of heuristics and biases. Adv Child Dev Behav. 2008;36:251–285.
36. Stevens J, Quittner AL, Abikoff H. Factors influencing elementary school teachers’ ratings of ADHD and ODD behaviors. J Clin Child Psychol. 1998;27(4):406–414. doi:10.1207/s15374424jccp2704_4
37. Pelham J, William E, Fabiano GA, Massetti GM. Evidence-based assessment of attention deficit hyperactivity disorder in children and adolescents. J Clin Child Adolescent Psychol. 2005;34(3):449–476. doi:10.1207/s15374424jccp3403_5
38. Ogrim G, Kropotov J, Brunner JF, Candrian G, Sandvik L, Hestad KA. Predicting the clinical outcome of stimulant medication in pediatric attention-deficit/hyperactivity disorder: data from quantitative electroencephalography, event-related potentials, and a go/no-go test. Neuropsychiatr Dis Treat. 2014;10:231–242. doi:10.2147/NDT.S56600
39. Toplak ME, West RF, Stanovich KE. Practitioner review: do performance-based measures and ratings of executive function assess the same construct? J Child Psychol Psychiatry. 2013;54(2):131–143. doi:10.1111/jcpp.12001
40. Brunner JF, Olsen A, Aasen IE, Lohaugen GC, Haberg AK, Kropotov J. Neuropsychological parameters indexing executive processes are associated with independent components of ERPs. Neuropsychologia. 2015;66:144–156. doi:10.1016/j.neuropsychologia.2014.11.019
41. Kropotov JD. Functional Neuromarkers for Psychiatry: Applications for Diagnosis and Treatment. Academic Press; 2016.
42. Yoshimoto S, Araki T, Uemura T, et al. Wireless EEG patch sensor on forehead using on-demand stretchable electrode sheet and electrode-tissue impedance scanner.
43. Bridwell DA, Steele VR, Maurer JM, Kiehl KA, Calhoun VD. The relationship between somatic and cognitive-affective depression symptoms and error-related ERPs. J Affect Disord. 2015;172:89–95. doi:10.1016/j.jad.2014.09.054
44. Grane VA, Brunner JF, Endestad T, et al. Correction: ERP correlates of proactive and reactive cognitive control in treatment-naive adult ADHD. PLoS One. 2016;11(9):e0163404. doi:10.1371/journal.pone.0163404
45. Downes M, Bathelt J, De Haan M. Event-related potential measures of executive functioning from preschool to adolescence. Dev Med Child Neurol. 2017;59(6):581–590. doi:10.1111/dmcn.13395
46. Lenartowicz A, Loo SK. Use of EEG to diagnose ADHD. Curr Psychiatry Rep. 2014;16(11):498. doi:10.1007/s11920-014-0498-0
47. Johnstone SJ, Barry RJ, Clarke AR. Ten years on: a follow-up review of ERP research in attention-deficit/hyperactivity disorder. Clin Neurophysiol. 2013;124(4):644–657. doi:10.1016/j.clinph.2012.09.006
48. Pollak SD, Tolley-Schell SA. Selective attention to facial emotion in physically abused children. J Abnorm Psychol. 2003;112(3):323. doi:10.1037/0021-843X.112.3.323
49. Mueller A, Candrian G, Grane VA, Kropotov JD, Ponomarev VA, Baschera GM. Discriminating between ADHD adults and controls using independent ERP components and a support vector machine: a validation study. Nonlinear Biomed Phys. 2011;5:5. doi:10.1186/1753-4631-5-5
50. Jonkman LM. The development of preparation, conflict monitoring and inhibition from early childhood to young adulthood: a Go/Nogo ERP study. Brain Res. 2006;1097(1):181–193. doi:10.1016/j.brainres.2006.04.064
51. Davis EP, Bruce J, Snyder K, Nelson CA. The X-trials: neural correlates of an inhibitory control task in children and adults. J Cogn Neurosci. 2003;15(3):432–443. doi:10.1162/089892903321593144
52. Coch D, Gullick MM. Event-related potentials and development. In: Kappenman E S., Luck S J (eds). The Oxford Handbook of Event-Related Potential Components. Oxford university press; 2011.
53. De Haan M. Infant EEG and Event-Related Potentials. Psychology Press; 2013.
54. Courchesne E. Neurophysiological correlates of cognitive development: changes in long-latency event-related potentials from childhood to adulthood. Electroencephalogr Clin Neurophysiol. 1978;45(4):468–482. doi:10.1016/0013-4694(78)90291-2
55. Luciana M, Sullivan J, Nelson CA. Associations between phenylalanine-to-tyrosine ratios and performance on tests of neuropsychological function in adolescents treated early and continuously for phenylketonuria. Child Dev. 2001;72(6):1637–1652. doi:10.1111/cdev.2001.72.issue-6
56. Picton TW, Taylor MJ. Electrophysiological evaluation of human brain development. Dev Neuropsychol. 2007;31(3):249–278. doi:10.1080/87565640701228732
57. Larson MJ, Clayson PE. The relationship between cognitive performance and electrophysiological indices of performance monitoring. Cogn Affect Behav Neurosci. 2011;11(2):159–171. doi:10.3758/s13415-010-0018-6
58. Conners CK. Conners’ Rating Scales Revised. Multi-Health Systems, Incorporated; 2001.
59. Achenbach TM, Rescorla LA. Manual for the ASEBA Preschool Forms and Profiles. Vol. 30. Burlington, VT: University of Vermont, Research center for children, youth; 2000.
60. Strauss E, Sherman EM, Spreen O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. American Chemical Society; 2006.
61. Evans AS, Preston A. Conners rating scales. In: Kreutzer JS, DeLuca J, Caplan B, editors. Encyclopedia of Clinical Neuropsychology. New York: Springer New York; 2011:680–681.
62. Wechsler D. Wechsler Preschool and Primary Scale of Intelligence—Fourth Edition. Psychological Corporation San Antonio, TX; 2012.
63. Ogrim G, Kropotov JD. Predicting clinical gains and side effects of stimulant medication in pediatric attention-deficit/hyperactivity disorder by combining measures from qEEG and ERPs in a Cued GO/NOGO task. Clin EEG Neurosci. 2019;50(1):34–43. doi:10.1177/1550059418782328
64. Kompatsiari K, Candrian G, Mueller A. Test-retest reliability of ERP components: a short-term replication of a visual Go/NoGo task in ADHD subjects. Neurosci Lett. 2016;617:166–172. doi:10.1016/j.neulet.2016.02.012
65. Toplak ME, Bucciarelli SM, Jain U, Tannock R. Executive functions: performance-based measures and the behavior rating inventory of executive function (BRIEF) in adolescents with attention deficit/hyperactivity disorder (ADHD). Child Neuropsychol. 2009;15(1):53–72. doi:10.1080/09297040802070929
66. Hammerer D, Li SC, Muller V, Lindenberger U. An electrophysiological study of response conflict processing across the lifespan: assessing the roles of conflict monitoring, cue utilization, response anticipation, and response suppression. Neuropsychologia. 2010;48(11):3305–3316. doi:10.1016/j.neuropsychologia.2010.07.014
67. Aasen IE, Brunner JF. Modulation of ERP components by task instructions in a cued go/no-go task. Psychophysiology. 2016;53(2):171–185. doi:10.1111/psyp.2016.53.issue-2
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.