Back to Journals » Clinical Ophthalmology » Volume 8
Incidence rate and occurrence of visually significant cataract formation and corneal decompensation after implantation of Verisyse/Artisan phakic intraocular lens
Authors Moshirfar M , Imbornoni L, Ostler E, Muthappan V
Received 30 December 2013
Accepted for publication 4 February 2014
Published 8 April 2014 Volume 2014:8 Pages 711—716
DOI https://doi.org/10.2147/OPTH.S59878
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Majid Moshirfar,1 Lauren M Imbornoni,2 Erik M Ostler,1 Valliammai Muthappan1
1Moran Eye Center, Department of Ophthalmology and Visual Sciences, University of Utah, Salt Lake City, UT, USA; 2University of Arizona College of Medicine, Tucson, AZ, USA
Purpose: To evaluate the incidence rate and indications for explantation of Verisyse phakic intraocular lenses (pIOLs) over a 13.6 year period.
Subjects and methods: Case series describing six cases of explantation of Verisyse pIOLs, out of 213 eyes with Verisyse pIOLs, implanted over a 13.6 year period by one surgeon at one institution, with mean follow-up of 5.6 years per eye.
Results: Four pIOLs were removed to facilitate extraction of a visually significant cataract, one was removed due to both cataract formation and development of corneal decompensation, and another was removed due to development of localized peripheral corneal decompensation. The incidence rate of pIOL removal was five per 1,000 patient-years with pIOL. Mean time from insertion of pIOL to cataract removal was 9.3 years (range 4.0–12.6 years). Mean time from insertion of pIOL to development of corneal decompensation was 10 years (range 6.9–13 years). There was no increase in the rate of cataract extraction in the pIOL population compared to the general population.
Conclusion: Cataracts may develop in patients with pIOLs, necessitating removal of the pIOL. Corneal decompensation is a serious complication that appears to be directly related to pIOLs. Corneal decompensation can occur up to 13 years following implantation of pIOLs, and long-term follow-up is important to monitor for this complication.
Keywords: Verisyse lens, Artisan lens, phakic IOL, corneal decompensation, cataract
Introduction
The Verisyse® phakic intraocular lens (pIOL), manufactured by Abbott Medical Optics (Santa Ana, CA, USA), is reported to provide excellent refractive outcomes, predictability, and stability, with few complications.1–4 However, despite the advantages and benefits of this procedure, serious complications have been reported, including corneal decompensation,5–7 elevated intraocular pressure,1,4 endophthalmitis,1 and retinal detachment.2,4 Cataract formation has also been reported, and is one of the most common indications for removal of pIOLs.
The Verisyse pIOL was approved by the US Food and Drug Administration (FDA) for correction of moderate-to-high myopia in 2004. Our institution has been using these lenses since 1999 as part of the FDA-supervised Clinical Investigation of the Artisan Myopia Lens undertaken by Ophtec USA (Boca Raton, FL, USA).8 We followed 213 eyes of 121 patients who have received Verisyse pIOLs over a 13.6 year period, with a mean follow-up of 5.6 years per eye. Of the 121 patients, 70.2% have continued to be followed up as recently as within the last year, and 83.3% have been followed up within the last 2 years. We report all (six) cases of pIOL explantation encountered during this 13.6 year period; reasons for explantation include visually significant cataracts or corneal decompensation. The patient was considered to have a visually significant cataract if there was a loss of three or more lines of corrected distance visual acuity (CDVA), which necessitated surgical removal of the cataract. Corneal decompensation was defined as corneal swelling that caused visual disturbances and loss of best-corrected visual acuity.
Case reports
Case 1
A 45-year-old woman presented to the Moran Eye Center in 1999, interested in refractive surgery for severe myopia. She had a spherical equivalent (SE) of −6.875 in the right eye and −11.875 in the left eye. At the time of presentation, the Verisyse pIOL was being evaluated by an FDA Phase III clinical trial, and the patient received a pIOL in the left eye through this study. Postoperative CDVA was 20/20 with an SE of −1.75. The patient elected for laser-assisted in situ keratomileusis (LASIK) in the right eye approximately a year later. She subsequently developed a visually significant posterior subcapsular cataract (PSC) in the right eye 5 years after LASIK, and underwent cataract extraction and intraocular lens implantation. Ten years after left pIOL implantation, the patient began to develop a nuclear sclerotic cataract (NSC) in the left eye. Three years later (13 years postimplantation), the left CDVA had decreased to 20/50+2, and it was decided that cataract surgery was necessary. The pIOL was removed, and phacoemulsification with posterior chamber intraocular lens (PCIOL) placement was performed on the left eye. At the most recent follow-up 5 months after removal of the pIOL, CDVA in the left eye was 20/20 and SE was plano.
Case 2
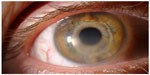
A 35-year-old man with a history of right retinal detachment, repaired with scleral buckle 20 years ago, presented to the Moran Eye Center in 2000 desiring correction for severe myopia. A Verisyse pIOL was implanted in the left eye as part of the Verisyse FDA clinical trial. Preoperative endothelial cell density in the left eye was 2,146 cells/mm2. Three years later, a Verisyse pIOL was implanted in the right eye for myopia, with preoperative right SE of −15.125 and preoperative endothelial cell density of 2,053 cells/mm2. Postoperative right uncorrected distance visual acuity (UDVA) was 20/20. Ten years later, the patient developed cloudy vision in the right eye with CDVA of 20/30−. Slit-lamp examination identified corneal edema in the right eye. Right endothelial cell density was 1,364 cells/mm2. The patient was medically managed with prednisolone acetate 1% eyedrops, Muro® 128 5% eyedrops (Bausch and Lomb, Rochester, NY, USA) and ointment for 6 months, with improvement of vision. However, after 2 years, the patient’s vision worsened to counting fingers, correctable only to 20/100. Slit-lamp examination identified increasing corneal edema (Figure 1) and early nuclear sclerosis. The patient underwent explantation of the pIOL, cataract extraction with placement of PCIOL, and Descemet’s stripping automated endothelial keratoplasty (DSAEK). At the most recent examination, 4 weeks after surgery, the cornea was clear centrally, in the area of the DSAEK graft, with remaining peripheral corneal edema. The right eye CDVA was 20/25, and SE was +1.125.
Case 3
A 45-year-old woman presented to the Moran Eye Center in 2002 for correction of high myopia, with SE of −11.75 in the right eye and −7.75 in the left eye. Verisyse pIOLs were implanted in both eyes, 4 months apart, in the FDA Verisyse clinical trial. Postoperative CDVA was 20/20 in the right eye and 20/25+2 in the left. Ten years later, on routine follow-up, the patient reported decreased vision in the right eye, with a CDVA of 20/60. Slit-lamp examination identified nuclear sclerosis in the right eye. The patient subsequently underwent pIOL explantation and cataract extraction, with PCIOL placement in the right eye. At the last examination, 1 year after pIOL removal, CDVA was 20/20+, and SE was −0.75 in the right eye. The patient has reported no problems post-pIOL in the left eye.
Case 4
A 53-year-old woman was referred in 2003 for correction of high myopia, with SE of −15.75 and −14.375 in the right and left eyes, respectively. The patient underwent uncomplicated pIOL implantation in both eyes, 9 months apart, as part of the Verisyse FDA clinical trial. At 2-year follow-up, the CDVA was 20/20 with SE of 0.125 in the right eye, and 20/20–2 with SE of 0.625 in the left eye. Seven years after implantation of the right pIOL, the patient was found on annual exam to have developed trace nuclear sclerosis in the right eye. Two years later, right CDVA was 20/50–2 and left CDVA was 20/25–2. Slit-lamp examination identified 2+ nuclear sclerosis and PSC in the right eye, and a 1−2+ NSC in the left eye. There were no known comorbidities in this patient. The patient subsequently underwent right pIOL explantation and cataract extraction, with PCIOL placement. At the most recent follow-up, 2 weeks after surgery, the patient’s CDVA was 20/20 in the right eye, and SE was −0.75.
Case 5
A 23-year-old man with microspherophakia presented in 2006 for correction of high myopia, with SE of −14.375 and −14.25 in the right and left eye, respectively. Preoperative anterior chamber depth (ACD), measured from the epithelium, was 2.87 mm in the right eye and 2.80 mm in the left eye. Preoperative mean endothelial cell density was 3,077 cells/mm2 in the right eye and 2,825 cells/mm2 in the left eye. The patient subsequently underwent pIOL implantation, in the right and left eyes, 2 months apart. At 4-year follow-up, UDVA was 20/25 in both eyes. ACD was 2.46 mm in the right eye and 2.30 mm in the left eye. Mean endothelial cell density was 2,890 cells/mm2 in the right eye and 2,785 cells/mm2 in the left eye.
In 2012, on routine follow-up, the patient reported worsening visual acuity in the right eye, associated with irritation and dryness. The patient admitted to rubbing the right eye secondary to localized irritation. UDVA in the right eye was 20/100, correctable to 20/80. Slit-lamp examination showed localized peripheral edema and bullous changes in the superonasal region of the cornea. The nasal enclavation site appeared closer to the corneal endothelium and less securely attached than the temporal site. The patient was treated with prednisolone acetate 1%, Muro 128 5% and Combigan® eyedrops (Allergan, Inc., Irvine, CA, USA) in the right eye for elevated IOP – 24 mmHg. The left eye did not require these treatments. Two weeks later, the corneal edema had only slightly improved, and it was decided to remove the right pIOL. At the most recent follow-up, 10 weeks post-pIOL removal, CDVA was 20/80, with SE of −12.50. The corneal edema was significantly improved. The lower-than-expected visual acuity was attributed to a combination of residual corneal edema, irregular astigmatism, and a secondary glaucoma associated with microspherophakia. The patient has been referred for further glaucoma workup and a fitting of rigid gas-permeable contact lens to see if this will improve his vision.
Case 6
A 51-year-old woman presented in 2008 for correction of high myopia, with SE of −7.125 in the right eye and −8.5 in the left. The patient underwent pIOL implantation in both eyes, 1 month apart. Postoperative CDVA at 2 months was 20/20–2 with SE of −0.125 in the right eye, and 20/25–2 with SE of −0.25 in the left eye. A year later, on routine follow-up, trace nuclear sclerosis and PSCs were identified in both eyes, worse in the right than the left. The patient was followed at 6-month intervals for 2 years, until it was decided to remove the cataract in the right eye. Preoperative right CDVA was 20/30−. The patient underwent uncomplicated pIOL removal and phacoemulsification with PCIOL insertion. Postoperative right CDVA at 6 weeks was 20/20, with SE of −0.75.
Results
Over a period of 13.6 years, our institution has implanted Verisyse pIOLs in 213 eyes (69 eyes from men and 144 eyes from women) of 121 patients (42 men and 69 women), with a mean follow-up of 5.6 years per eye. The average age of implantation was 40.89 years. The occurrence of cataract formation in this patient population was 2.3% (five of 213), and the occurrence of corneal decompensation requiring lens removal was 0.9% (two of 213). The incidence rate was calculated using total patient-years with the pIOL. The incidence rate of cataract formation was 4.2 per 1,000 patient-years, and the incidence rate of corneal decompensation was 1.7 per 1,000 patient-years. The overall incidence of pIOL removal was 5.0 per 1,000 patient-years. Clinical findings in patients who underwent pIOL removal are summarized in Table 1.
In the five patients who developed visually significant cataracts, the mean duration from pIOL insertion to pIOL removal was 9.3 years (range 4.0–12.6 years), and the mean age at visually significant cataract requiring surgical removal was 55 years (range 46–62 years). Three patients (cases 1, 2, and 3) developed NSCs, and two (cases 4 and 6) developed both NSCs and PSCs. The surgical approach for the patients who developed visually significant cataracts started with an initial temporal incision to disenclavate the haptics of the lens. A large superior wound was then created to explant the pIOL, with the added advantage of reducing with-the-rule astigmatism in these eyes. Phacoemulsification and PCIOL insertion were subsequently performed using the initial temporal incision. In the four patients (cases 1, 3, 4, and 6) who developed a cataract without corneal decompensation, CDVA following pIOL removal was 20/20 or better. There were no instances of refractive surprise.
Discussion
Visually significant cataract formation has been cited as a complication of Verisyse/Artisan pIOL implantation.2,4 Our occurrence rate of 2.3% is consistent with prior reports in the literature. A 3-year FDA clinical trial found development of cataracts in 1.5% (two of 137) of eyes with Artisan pIOLs.4 A 5-year follow-up of 399 Artisan pIOLs identified cataract formation in two patients (0.5%),2 and a 3.5-year study of 160 patients identified cataract development in 3.42% of patients with pIOLs.9 A more recent study identified visually-significant cataract formation requiring surgery in four eyes (7.5%) over 6 years.10 Studies have suggested a causal relationship between pIOLs and cataract formation, theorizing that the close approximation of the pIOL to the crystalline lens may cause cataract formation,7 or that the lens may become cloudy due to surgically induced trauma during pIOL insertion.11
Although cataract formation with the use of pIOLs has been documented, the relationship between pIOL implantation and cataract development has not been clearly shown. We are unconvinced that pIOLs contribute to cataract formation. The NSCs and PSCs in our four pIOL patients who developed cataracts only are more likely age-related than pIOL-related. The reported 10-year incidence of cataract surgery in adults at least 49 years of age is 17.8%, and in adults ages 55–64 is 9.1%.12 The mean age of our total patient population was 47.7 years. However, our four post-pIOL patients with visually significant cataract and without corneal decompensation were between 55 and 62 years of age. Of our total population, 59 patients were between the ages of 55 and 64 years, for an occurrence of cataract surgery in this age-group of four of 59 or 6.8%. We have found that in our patients, pIOLs did not increase the occurrence of cataract surgery above what is expected for age in the general population.
The patients with pIOLs who developed cataracts may have had additional risk factors for cataract development, such as high myopia or corticosteroid use. The patient discussed in case 1 received a pIOL in the left eye only, but subsequently developed visually significant cataracts in both eyes. The youngest patient requiring cataract extraction in our series was 46 years old (case 2); however, he was treated extensively with steroids for corneal edema before the NSC developed. While prednisolone acetate eyedrops typically cause PSC, the chronic ophthalmic steroid use may have contributed to the development of nuclear sclerosis. Additionally, patients requiring pIOLs for correction of high myopia may be inherently more likely to develop cataracts. There is evidence of a significant association between high myopia and development of both nuclear cataract and PSCs.13 Cataract development was found to be four times more frequent in patients with high myopia than in the general population.9 Additionally, the majority of pIOL-associated cataracts are anterior subcapsular cataracts, and the strongest correlation is between posterior chamber pIOLs and anterior subcapsular cataracts.11 None of our patients developed anterior subcapsular cataracts, supporting our theory that pIOLs did not cause cataracts in our patients.
The risk of corneal decompensation following pIOL implantation is well established.2,4–7,14 Possible mechanisms for endothelial cell loss and decompensation include surgical trauma, early postoperative elevated intraocular pressure, chronic anterior chamber inflammation secondary to the implant, contact between the cornea and the lens haptics, or transient contact between the lens optic and the corneal endothelium.5 Two of our 213 patients required pIOL removal due to corneal decompensation. One of these patients (case 2) had a history of retinal detachment and scleral buckle in the eye that developed corneal decompensation. There were no problems with the pIOL in the patient’s other eye. The preoperative endothelial cell density in the eye with corneal decompensation was 2,053 cells/mm2, lower than in the other eye. The low endothelial cell density may be secondary to prior ocular surgery. Endothelial cell loss is known to occur at a faster rate in those with pIOLs. A 3-year study reported a mean loss of 1.8% per year;4 however, a more recent study reported a mean endothelial cell loss of 6.9% after 10 years.10 Low preoperative endothelial cell density is a contraindication for pIOL use. This patient was treated with pIOL removal and DSAEK, and has recovered nicely, with a most recent CDVA of 20/25.
The second patient with corneal decompensation (case 5) received a pIOL at age 23 years for correction of microspherophakia. At the time of insertion of the pIOL, the patient’s ACD was 2.87 mm, which met the manufacturer’s recommendation of a minimum depth of 2.6 mm at the time of the surgery in 2006.15 Smaller ACD is correlated with increased endothelial cell loss,16 and the current recommendation for minimum ACD is 3.2 mm. Four years after implantation, the right ACD was 2.46 mm, and right endothelial cell density was 2,890 cells/mm2. There was a larger decrease in endothelial cell density in the right eye compared to the left eye, and the distance between the corneal endothelium to the site of pIOL enclavation was smaller in the right eye compared to the left. At this time, there was no evidence of corneal decompensation. The small ACD may have contributed to the corneal edema in this patient. On examination, 6 years after implantation, the nasal enclavation site in the right eye appeared to be less securely attached than the temporal site. It is possible that this instability led to the localized edema in the superonasal region of the cornea. We also considered the possibility of Brown–McLean syndrome in this patient as the cause of peripheral edema, although this patient lacked the characteristic endothelial pigmentation. Brown–McLean syndrome is most often associated with intracapsular lens extraction,17 although it has been reported in eyes with phakic anterior chamber intraocular lens.18 In our patient, removal of the pIOL resulted in improvement in visual acuity and corneal edema, though his uncorrected vision may be limited due to corneal astigmatism and glaucoma.
Our institution has been using the Verisyse pIOL for 13.6 years, with a mean follow-up of 5.6 years. To our knowledge, there has not been a similar study reported with a longer follow-up time. We have been using these lenses since 1999 as part of the FDA clinical trial, 5 years before they gained FDA approval. Long-term follow-up is important for pIOL patients, as our two patients with corneal decompensation did not develop corneal edema until 6 and 13 years after implantation. We believe that it is important to report that serious complications can occur even after a decade of use with no problems. Additionally, our study contributes prognostic data following pIOL removal. The four patients who developed visually significant cataracts all attained CDVA of 20/20 following pIOL removal and PCIOL insertion. In the patients with corneal decompensation, the first attained CDVA of 20/25 following pIOL removal. The second patient’s CDVA was 20/80 following pIOL removal; however, we believe this may be partially attributed to comorbidities secondary to the patient’s microspherophakia.
In conclusion, our institution has seen a relatively low rate of pIOL removal over the last 14 years. However, the cases we report suggest that pIOLs are not without risk. Cataracts may develop in patients with pIOLs, necessitating removal of the pIOL. The role of pIOLs in cataract formation is unclear; however, the individual risk of cataract development should be considered prior to pIOL implantation. Corneal decompensation continues to be a serious complication that appears to be directly related to the pIOL. It is necessary to monitor patients for early signs of corneal decompensation. Corneal decompensation usually requires removal of the pIOL. In general, Verisyse lenses are shown to be effective and predictable, and remain a refractive surgical option for patients with high myopia. Good long-term follow-up should be ensured to monitor these patients for complications necessitating removal of the implant.
Disclosure
The authors report no conflicts of interest in this work.
References
Gonnermann J, Klamann MK, Maier AK, et al. Visual outcome and complications after posterior iris-claw aphakic intraocular lens implantation. J Cataract Refract Surg. 2012;38(12):2139–2143. | |
Güell JL, Morral M, Gris O, Gaytan J, Sisquella M, Manero F. Five-year follow-up of 399 phakic Artisan-Verisyse implantation for myopia, hyperopia and/or astigmatism. Ophthalmology. 2008;115(6):1002–1012. | |
Moshirfar M, Holz HA, Davis DK. Two-year follow-up of the Artisan/Verisyse iris-supported phakic intraocular lens for the correction of high myopia. J Cataract Refract Surg. 2007;33(8):1392–1397. | |
Stulting RD, John ME, Maloney RK, Assil KK, Arrowsmith PN, Thompson VM. Three-year results of Artisan/Verisyse phakic intraocular lens implantation. Ophthalmology. 2008;115(3):464–472. | |
Coullet J, Mahieu L, Malecaze F, et al. Severe endothelial cell loss following uneventful angle-supported phakic intraocular lens implantation for high myopia. J Cataract Refract Surg. 2007;33(8):1477–1481. | |
Kim M, Kim JK, Lee HK. Corneal endothelial decompensation after iris-claw phakic intraocular lens implantation. J Cataract Refract Surg. 2008;34(3):517–519. | |
Patel SR, Chu DS, Ayres BD, Hersh PS. Corneal edema and penetrating keratoplasty after anterior chamber phakic intraocular lens implantation. J Cataract Refract Surg. 2005;31(11):2212–2215. | |
Alexander L, John M, Cobb L, Noblitt R, Barowsky RT. US clinical investigation of the Artisan myopia lens for the correction of high myopia in phakic eyes. Report of the results of phases 1 and 2, and interim phase 3. Optometry. 2000;71(10):630–642. | |
Alió JL, de la Hoz F, Ruiz-Moreno JM, Salem TF. Cataract surgery in highly myopic eyes corrected by phakic anterior chamber angle-supported lenses. J Cataract Refract Surg. 2000;26(9):1303–1311. | |
Torun N, Bertelmann E, Klamann M, Maier AK, Liekfeld A, Gonnermann J. Posterior chamber phakic intraocular lens to correct myopia: long-term follow-up. J Cataract Refract Surg. 2013;39(7):1023–1028. | |
Moshirfar M, Mifflin M, Wong G, Chang JC. Cataract surgery following phakic intraocular lens implantation. Curr Opin Ophthalmol. 2010;21(1):39–44. | |
Kanthan GL, Wang JJ, Rochtchina E, et al. Ten-year incidence of age-related cataract and cataract surgery in an older Australian population. Ophthalmology. 2008;115(5):808–814. | |
Younan C, Mitchell P, Cumming RG, Rochtchina E, Wang JJ. Myopia and incident cataract and cataract surgery: the Blue Mountains eye study. Invest Ophthalmol Vis Sci. 2002;43(12):3625–3632. | |
van Eijden R, de Vries NE, Cruysberg LP, Webers CA, Berenschot T, Nuijts RM. Case of late-onset corneal decompensation after iris-fixated phakic intraocular lens implantation. J Cataract Refract Surg. 2009;35(4):774–777. | |
Moshirfar M, Meyer JJ, Schliesser JA, Espandar L, Chang JC. Iris-fixated phakic intraocular lens implantation for correction of high myopia in microspherophakia. J Cataract Refract Surg. 2010;36(4):682–685. | |
Saxena R, Boekhoorn SS, Mulder PGH, Noordxij B, van Rij G, Luyten GP. Long-term follow-up of endothelial cell change after Artisan phakic intraocular lens implantation. Opthlamology. 2008;115(4):608–613. | |
Pareja-Esteban J, Montes MA, Peréz-Rico C, Jiménez-Parras R, Bolívar G. [Brown-McLean syndrome after insertion of an anterior chamber intraocular lens: description of one case]. Arch Soc Esp Oftalmol. 2007;82(5):315–318. Spanish. | |
Díaz-Llopis M, Garcia-Delpech S, Salom D, Udaondo P. [Brown-McLean syndrome and refractive phakic anterior chamber intraocular lenses]. Arch Soc Esp Oftalmol. 2007;82(12):737–740. Spanish. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.