Back to Journals » International Journal of Nanomedicine » Volume 17
In vivo Detection of Macromolecule Free Radicals in Mouse Sepsis-Associated Encephalopathy Using a New MRI and Immunospin Trapping Strategy
Authors Liu H, Ma C, Xu H, Zhang H, Xu R, Zhang K, Sun R, Li K, Wu Q, Wen L, Zhang L, Guo Y
Received 15 June 2022
Accepted for publication 25 August 2022
Published 1 September 2022 Volume 2022:17 Pages 3809—3820
DOI https://doi.org/10.2147/IJN.S378726
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yan Shen
Hanrui Liu,1 Chengyong Ma,2 Huayan Xu,1 Huan Zhang,3 Rong Xu,1 Kun Zhang,1 Ran Sun,1 Kuan Li,1 Qihong Wu,1 Lingyi Wen,1 Lizhi Zhang,2 Yingkun Guo1
1West China Second University Hospital, Sichuan University, Chengdu, Sichuan, People’s Republic of China; 2West China Hospital, Sichuan University, Chengdu, People’s Republic of China; 3College of Chemistry and Materials Science, Northwest University, Xi’an, People’s Republic of China
Correspondence: Yingkun Guo, Development and Related Diseases of Women and Children Key Laboratory, West China Second University Hospital, Sichuan University, No. 20, Section 3, Renmin South Road, Chengdu, Sichuan, 610041, People’s Republic of China, Tel +86+18180609256, Email [email protected] Lizhi Zhang, Department of Radiology, West China Hospital of Sichuan University, No. 37, Guoxue Road, Chengdu, Sichuan, 610041, People’s Republic of China, Email [email protected]
Introduction: Free radicals in oxidative stress are known to play a pathogenic role in sepsis. A major clinical challenge associated with sepsis is sepsis-associated encephalopathy (SAE). The rapid increase of free radicals in the brain promotes SAE progression. Here, macromolecule free radicals in the mouse brain were uniquely detected by immunospin trapping (IST) and magnetic resonance imaging (MRI).
Methods: The new strategy uses spin trapping agent DEPMPO-biotin to capture macromolecule free radicals in lesions and form biotin-DEPMPO-radical adducts. Then, a targeting MRI probe, avidin-BSA@Gd-ESIO, was used to detect the radical adducts through the highly specific binding of avidin and biotin. The avidin-BSA@Gd-ESIO probe was synthesized and systematically characterized. The detection capability of the new strategy was evaluated in vitro and in vivo using a confocal microscope and a 7T MRI, respectively.
Results: In reactive oxygen species (ROS)–induced microglial cells, the accumulation of the avidin-BSA@Gd-ESIO probe in the DEPMPO-biotin-treated group was significantly higher than that of control groups. In vivo MRI T1 signal intensities were significantly higher within the hippocampus, striatum, and medial cortex of the brain in mice with a mild or severe degree of sepsis compared with the sham control group. Histological analysis validated that the distribution of the avidin-BSA@Gd-ESIO probe in brain tissue slices was consistent with the MRI images. The fluorescence signals of ROS and avidin-BSA@Gd-ESIO probe were overlapped and visualized using immunofluorescent staining. By evaluating the T1 signal changes over time in different areas of the brain, we estimated the optimal MRI detection time to be 30 minutes after the probe administration.
Discussion: This method can be applied specifically to assess the level of macromolecular free radicals in vivo in a simple and stable manner, providing a pathway for a more comprehensive understanding of the role of free radicals in SAE.
Graphical-Abstract:
Keywords: macromolecule radical, ESIO, spin trapping, sepsis-associated encephalopathy
Graphical Abstract:

Introduction
Reactive oxygen species (ROS) are major risk factors that cause oxidative stress and play a pathogenic role in various diseases, including sepsis.1 The increased generation of ROS in sepsis can lead to disseminated oxidative stress and various organ dysfunctions, including brain injury and its cognitive consequences. Sepsis-associated encephalopathy (SAE) is a common complication found in patients with sepsis. Studies have demonstrated that septic encephalopathy and polyneuropathy occur in 70% of sepsis patients, significantly increasing patients’ mortality and influencing sepsis survivors’ quality of life.2 The pathophysiological mechanisms of SAE, however, have been poorly illustrated and often are associated with infection and oxidation stress.3 Therefore, studies on free radicals induced by oxidative imbalances could further clarify the mechanism of SAE and benefit clinical study.
Microglia release a variety of free radicals, such as nitric oxide (NO) and ROS, once activated by sepsis through alterations in the neural pathway, humoral pathway, and blood–brain barrier (BBB).3 The main ROS involved in sepsis pathogenesis include superoxide (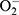 ), hydrogen peroxide (H2O2), and hydroxyl radicals (HO·). These ROS may directly oxidize nucleic acids, proteins, and lipids, causing extracellular and intercellular perturbations in homeostasis. Understanding the extent and timing of free radical–triggered events in an in vivo environment is of importance to our understanding of these major determinants involved in disease evolution and prognosis.
), hydrogen peroxide (H2O2), and hydroxyl radicals (HO·). These ROS may directly oxidize nucleic acids, proteins, and lipids, causing extracellular and intercellular perturbations in homeostasis. Understanding the extent and timing of free radical–triggered events in an in vivo environment is of importance to our understanding of these major determinants involved in disease evolution and prognosis.
Imaging methods have been reported to detect radicals, including techniques such as electron spin resonance (ESR), fluorescence imaging, and magnetic resonance imaging (MRI). MRI potentially has offered a method for detecting the slight content of free radicals generated by specific biological processes. However, spatial resolution and general radical detection are limited by sensitivity requirements. ESR imaging is sensitive, but detection is limited to the injection site of the injected paramagnetic probe and not the general detection of a broad range of radicals. Fluorescence imaging is restricted to excised tissues or isolated cells as a result of limited tissue penetration. Recently, in vivo immune spin trapping (IST)-enhanced MRI showed promise for macromolecule radical detection, including proteins, lipids, and DNA radicals. Several disease models have been studied using this technique, such as tumor-bearing and diabetic mice, in which the appearance or disappearance of the MRI signal indicated a change in macromolecule free radical generation.4–8
IST is a method that uses a spin trapping agent to capture free radicals to form adducts. Then uses the antibody of the spin trapping agent to target the adducts. IST was pioneered by Mason et al as a means to detect protein, lipid, and DNA radical formation for in vitro and in vivo systems using mass spectroscopy, EPR, and immunochemical techniques.8 Towner et al extended this to the detection of biomolecular radicals using an MRI imaging technique.4,6 Typically, bare spin trap DMPO is given to the animal and then an anti-DMPO (biotin-DMPO antibody-albumin-Gd-DTPA) probe is administered. This technique is attractive since it allows for the in-vivo imaging of biomolecular free radicals specifically at the tissue site of their production under oxidative stress conditions. The macromolecule free radicals can be tagged by radical trapping agents to form radical adducts. The antibody of the trapping agent, used to recognize the radical adducts, usually is attached to an MRI probe for imaging. This approach will allow the detection of the correlation between real-time oxidative stress and specific longitudinal pathological conditions associated with specific diseases. The half-life period of the DMPO-radical adduct, however, is only 45 seconds (DMPO-OOH), which is too short and unstable for in vivo imaging. DEPMPO (5-(dihydroxyphosphonyl)-5-methyl-1-pyrroline-n-oxide) is a promising phosphorylated derivative of DMPO, which can form a more stable and easily detectable adducts with radicals. The radical adduct DEPMPO-OOH was reported to last 15 times longer than DMPO-OOH.9 Ke et al also reported that the spin adduct DEPMPO/SO3•− was 7 and 2–4 times more stable than DMPO/SO3•− in vitro and in vivo, respectively.10 Claudine et al verified that the DEPMPO–superoxide spin adduct was significantly more persistent than its DMPO analogue, which allows for the detection of superoxide during the reperfusion of ischemic isolated rat hearts.5 Therefore, compared with DMPO, DEPMPO shows promise for safer and more stable radical detection in vivo by IST-MRI.
In this study, we designed a novel IST strategy for in vivo macromolecule free radical detection. This strategy uses DEPMPO-biotin and a targeting MRI probe avidin-BSA@Gd-ESIO. The DEPMPO-biotin was used to trap macromolecule free radicals in lesions and form radical adducts. Then, we administered the targeting probe avidin-BSA@Gd-ESIO to specifically bond the biotin on the radical adducts. The targeting MRI probe avidin-BSA@Gd-ESIO contains an anti-biotin (avidin), clinical MRI T1 contrast agent Gd-DTPA, and extremely small iron oxide nanoparticles (ESIOs) that have been reported to have superior T1 enhancement property.11 This probe has been designed for the desired T1 contrast morphological image resolution of MRI to detect macromolecule free radicals in vivo (see Figure 1).
This study used a mouse model of SAE for specific investigation of radicals in the mouse brain. The most widely used animal model of sepsis is cecal ligation and puncture (CLP)-induced peritonitis mice.12 We constructed SAE models of different degrees in C57BL/6 mice according to CLP.
Materials and Methods
Synthesis and Characterization of the Avidin-BSA@Gd-ESIO Probe
BSA-Gd-DTPA was prepared using a modified method described in Towner et al.4 In brief, 8 μmol bull serum albumin (BSA, Sigma-Aldrich, St. Louis, MO, USA) was dissolved in a 0.1 M NaHCO3 (Sigma-Aldrich) solution (pH = 8.5). We added 1.4 mmol of Gd-DTPA (Bayer, Leverkusen, Germany) to the solution. Then, the solution was stirred at 4°C for 4 hours. Subsequently, the solution was collected and dialyzed. The BSA-Gd-DTPA product was freeze-dried and stored at −20°C.
ESIO was synthesized as described by Byung H et al.11 Iron-oleate complex (1.8 g, 2 mmol, Sigma), oleic acid (2 mmol; Sigma), and 1.61 g oleyl alcohol (6 mmol; Macklin, Shanghai, China) were dissolved in 10 g diphenyl ether (Macklin) at room temperature. The mixture was heated to 250°C at a constant heating rate of 10°C/min and kept at this temperature for 30 minutes under an inert atmosphere. As the reaction proceeded, the initial brown transparent solution turned black. After the reaction, the mixture containing the nanoparticles was rapidly cooled to room temperature. We added 50 mL of acetone (Macklin) to precipitate the nanoparticles. The nanoparticles were separated by centrifugation (8000 r/min, 15 min) (centrifuge 5840, Eppendorf, Hamburg, German) and dispersed in nonpolar solvents such as n-hexane or chloroform (Macklin). The synthesized ESIO was further modified with dopamine (Sigma), as previously reported.11
ESIO and BSA-Gd-DTPA were conducted through an N-hydroxysuccinimide–N, N-(3-dimethyl aminopropyl)-N’-ethyl-carbodiimide hydrochloride) (NHS-EDC) link between albumin and the NH2 group on the surface of the ESIOs. Avidin was also added to albumin through the NHS-EDC link. Briefly, we dissolved 75 mg BSA-Gd-DTPA in the phosphate buffered saline (PBS; Gibco, Waltham, MA, USA) and added 8.6 mg EDC (Sigma) directly to the solution. The solution was stirred for 30 minutes at room temperature. We added 5.2 mg NHS (Sigma) to the solution and 15 minutes later added 0.5 mL ESIO (1735 μg Fe/mL). The mixed solution was then stirred for another 3 hours. We added 0.5 mg avidin or avidin-FITC (avidin-fluorescein isothiocyanate) (Solarbio, Beijing, China) to the mixture. The reaction was stirred for 3 hours at 4°C. Afterward, the entire solution was dialyzed, freeze-dried, and stored at 4°C.
The physicochemical properties of the avidin-BSA@Gd-ESIO probe were assessed by thermogravimetric analysis (TGA, TGA/DSC2; Mettler Toledo, Nänikon, Switzerland), transmission electron microscopy (TEM, Talos F200X TEM; ThermoFisher, Waltham, MA, USA), and dynamic light scattering (DLS, Zetasizer Nano ZS; Malvern Panalytical, Malvern, UK). The T1 relaxation rate was obtained by measuring the relaxation time of the targeting MRI probe at different concentrations of Fe+Gd under a 7T MRI (MRINOVA 7.0T/16; Time Medical, Shanghai, China). The concentrations of Fe and Gd were measured by inductively coupled plasma-atomic emission spectrometers (ICP-AES, IRIS Advantage; ThermoFisher).
Cell Confocal Experiment
Bv-2 mouse microglial cells (MIC-iCell-n009, iCell, Shanghai, China) were grown to confluency in flasks in complete growth medium (Gibco basal medium; Gibco, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco). Two hours before treatment, the growth medium was replaced with serum-free medium. Before the experiment, the avidin-BSA@Gd-ESIO probe was conjugated with red fluorescent dye rhodamine B through the NHS-EDC link between the NH2 group on the avidin-BSA@Gd-ESIO and the carboxyl group of rhodamine B. Briefly, 0.2 mg rhodamine B (Sigma) were dissolved in PBS and 0.2 mL EDC (1 mg/mL) were added. The mixed solution was stirred for 30 minutes at room temperature. Then, 0.2 mL NHS (1 mg/mL) were added and stirred for another 15 minutes. After that, the reaction solution was added to a 0.5 mL aqueous solution of the avidin-BSA@Gd-ESIO probe (100 mg/mL) and stirred for 5 hours. When finished, the product was washed with water through centrifugation (10,000 rpm/min, 10 minutes) at least three times.
The cells were divided into four groups: (1) mouse microglial cells treated with the avidin-BSA@Gd-ESIO probe; (2) mouse microglial cells treated with DEPMPO-biotin and the avidin-BSA@Gd-ESIO probe; (3) mouse microglial cells treated with ROSup (Solarbio) and the avidin-BSA@Gd-ESIO probe; and (4) mouse microglial cells treated with ROSup and incubated with DEPMPO-biotin and the avidin-BSA@Gd-ESIO probe. Microglial cells were treated with ROSup to stimulate high ROS expression.13 ROSup treatment was conducted according to the manufacturer’s instructions. Briefly, 50 μmol/mL of ROSup was added to the culture dishes for 15 minutes, and then the dishes were washed with PBS three times. A total of 10 µg DEPMPO-biotin (Cayman Chemical, Ann Arbor, MI, USA) were added to the culture dishes and incubated for 15 minutes. To the samples that were treated with the avidin-BSA@Gd-ESIO probe, the probe was added (50 μg [Fe+Gd]/mL), and the cells were incubated for 4 hours. After incubation, the cells were washed with PBS and stained with DAPI for 10 minutes. Any DEPMPO–radical adducts that were not bound to cells or free DEPMPO-biotin were washed away with PBS. The cells were obvious under a confocal microscope (Olympus FV3000, Olympus, Tokyo, Japan).
In vivo Animal Experiments
All animal experiments were conducted according to the National Institutes of Health animal use and welfare guidelines and were approved by the West China Hospital of Sichuan University animal ethics committee. There were two groups of CLP mouse models and a sham control group. The two groups of CLP mouse included the severe-grade CLP group (CLP-severe) and the mild-grade CLP group (CLP-mild). The models were constructed as previously described.14 Briefly, eight-week-old C57BL/6 male mice were anesthetized with isoflurane (2–3%) under aseptic conditions. A laparotomy was performed with a 1.5-cm midline incision through the linea alba. The cecum was exposed and ligated with sterile 3–0 silk below the ileocaecal junction. The severe-grade CLP mice were incised 1.5 cm from the cecum, and the mild-grade CLP mice were incised 1 cm apart. The cecum was punctured once with an 18-gauge needle and returned to the peritoneal cavity. The muscle and skin incisions were closed. After the surgery, the animals were administered 0.5 mL of sterile saline subcutaneously. Mice in the sham group underwent identical procedures, without ligation and punctures of the cecum.
For in vivo MRI, mice were divided into three groups with six mice in each group, namely, the sham, CLP-severe, and CLP-mild groups. DEPMPO-biotin was intraperitoneally administered (25 μL in 100 μL saline) 6 hours after CLP, and then MRI was performed. The avidin-BSA@Gd-ESIO probe was injected intraperitoneally (5 mmol Gd/kg), 15 minutes after DEPMPO-biotin was administrated.10 The MRI (MRINOVA 7.0T/16; Time Medical) was performed for the in vivo study. Signal intensities were obtained through the T1 weighted image (repetition time (TR) 1000 ms, echo time (TE) 20.91 ms, 4.00×4.00 cm2 field of view (FOV), 1-mm slice thickness). MRI experiments were carried out under general anesthesia (1–2% isoflurane, 0.8–1.0 L/min O2). Relative %T1 (percentage difference in T1 weighted image intensity) was measured by comparing the postcontrast (avidin-BSA@Gd-ESIO probe administrated) image to the native image through MRINAVA 7.0T/16 (Time Medical).
Histology and Immunohistochemical Analysis
After the MRI experiment, mice in each group were killed using an anesthetic overdose of 2% pentobarbital sodium. After perfusion with saline, the brain was harvested. Fresh tissues were cut into 4 µm sections for ROS immunohistochemical stain with dihydroethidium (DHE). Paraffin-embedded tissue samples were used for hematoxylin-eosin (HE), Prussian blue and F480 (a marker for macrophages) stain. These slides were observed under both an optical microscope (Nikon Eclipse C1, Tokyo, Japan) and a fluorescence microscope (Nikon Eclipse Ti-SR). The superoxide dismutase (SOD) and malondialdehyde (MDA) contents were tested by enzyme-linked immunosorbent assay (ELISA) kits (A001-1 and A003-1, respectively, Nanjing Jiancheng Bioengineering Institute, Jiangsu, China).
Statistical Analyses
The MRI signal was measured using a 7.0 T MRI software (MRINOVA 7.0T/16, Time Medical), and image subtraction was performed using ImageJ. Statistical differences between the experimental groups and the control groups were analyzed with one-way analysis of variance (ANOVA) and the Student’s t-test using commercially available software (GraphPad Software, San Diego, CA, USA). A p-value less than 0.05 was considered to indicate a statistically significant difference.
Results
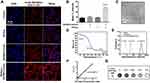
This work verified the detection of macromolecule free radicals in vitro using Bv2 mouse microglial cells. The avidin-BSA@Gd-ESIO probe was conjugated with rhodamine B, showing red fluorescence under the excitation of confocal microscopy. The nuclei were stained with DAPI showing blue fluorescence. The microglial cells in four groups were all incubated with the targeting probe avidin-BSA@Gd-ESIO. Two groups of cells were pretreated with ROSup before the experiment, which stimulates cells to overexpress ROS.13 As shown in Figure 2A, strong fluorescence of the avidin-BSA@Gd-ESIO probes was found to be significantly accumulated in mouse microglial cells that were treated both with DEPMPO-biotin and ROSup, compared with other groups. The ROS upregulated cells incubated without DEPMPO-biotin did not show the same enrichment, and neither did the cells not exposed to ROSup. Mean fluorescence intensity statistics are shown in Figure 2B. The results indicated that the targeting probe avidin-BSA@Gd-ESIO and DEPMPO-biotin could accumulate in ROS overexpressed cells, but did not do so in normal Bv2 cells. This may have been due to the fact that macromolecular free radicals produced with ROS in cells were captured by DEPMPO-biotin and formed radical adducts, which were then targeted by the probe. In cells without large amounts of macromolecular free radicals, excess and free DEPMPO-biotin were washed away by PBS.
Next, the morphology and composition of the targeting probe and its T1 MRI performance were characterized. Figure 2C shows the transmission electron microscope (TEM) image of the synthesized ESIOs. The ESIOs are monodispersed and have a uniform size of approximately 3 nm. Figure 2D shows the thermogravimetric analysis (TGA) curve of the probe. Bound water (the weight dropped before 200°C) accounted for 11.95% and protein, polypeptide, and other organic compounds accounted for 53.12% (200–500°C dropped weight). Pure ESIO core Fe2O3 and Gd elements accounted for only 36.91% (residual weight after 500°C). This indicated that the synthesized targeting probe contained a large number of organic compounds, which should have consisted mainly of BSA and avidin. We further analyzed the dispersion of the targeting probe in the water phase by dynamic light scattering (DLS). Figure 2E shows the hydration particle sizes of ESIO, BSA-Gd-DTPA, and the avidin-BSA@Gd-ESIO probe, which were 6, 45, and 1606 nm, respectively. These results indicated that the synthesized probe may have been formed by cross-linking multiple ESIO particles with multiple BSA-Gd-DTPA molecules. The polydispersity index of the probe was 0.162 (<0.5), indicating a good dispersity. The 1/T1 value of different concentrations of the probe on 7T MRI is shown in Figure 2F. The slope represents the relaxivity (r1) is 2.6 mM-1s-1. T1 MRI images of different concentrations of the probe tended to be brighter with increasing Fe and Gd concentrations (Figure 2G), indicating a good T1 contrast ability. Thus, the targeting probe avidin-BSA@Gd-ESIO was successfully synthesized with monodispersity, biocompatibility, targeting ability, and good MRI performance.
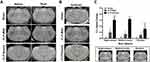
The overall accumulation of the targeting probe avidin-BSA@Gd-ESIO in mice brains is depicted in the MRI images in Figure 3A. And the subtracted image of the post to native is shown in Figure 3B, where the mice with sepsis seemed to accumulate more probes in the brain than sham mice. In Figure 3C, we observed significant increases in T1% signal change in the brains of CLP-severe and CLP-mild mice, compared with sham controls (p <0.001 and p <0.05 for all three regions, respectively). We also observed an increased T1% change in CLP-severe mice compared to CLP-mild mice. This may have been due to the differences in macromolecules radical aggregation.
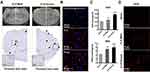
After the MRI experiment, mice in each group were sacrificed for histological and immunohistochemical analysis. The targeting probe avidin-BSA@Gd-ESIO in brain sections was stained with Prussian blue and diaminobenzidine (DAB) enhancement. As shown in Figure 4A, CLP-severe and CLP-mild mouse slices exhibited several brown signals of the avidin-BSA@Gd-ESIO probe. These signals were found in the corresponding region on MRI images in both CLP-severe and CLP-mild mice. These results indicated that the enhanced T1 signal should be attributed to the uptake of the targeting probe. To further verify the distribution of free radicals and the targeting probe, avidin-FITC were used instead of avidin, and ROS in tissue slices were stained with DHE. DHE can enter cells freely and be oxidized by intracellular ROS to form ethidium oxide. Ethidium oxide is incorporated into chromosomal DNA to produce red fluorescence. The excess production of ROS is often accompanied by macromolecular radicals.15–17 Figure 4B shows the ROS (red) and avidin-BSA@Gd-ESIO probe (green) in brain tissue slices of CLP-severe mice. The co-localization of the targeted probe and free radicals indicated the capture of macromolecule free radicals of DEPMPO-biotin. This evidence showed the T1 enhancement in MRI images induced by DEPMPO-biotin and the targeting probe should indicate the macromolecule radicals in septic mice brains.
The SOD and MDA levels in the brain of mice were also tested using ELISA kits. The main function of SOD is to catalyze the disproportionation of superoxide anion radicals into hydrogen peroxide and oxygen. The significant increase in SOD indicates significantly elevated levels of radicals.18,19 When free radicals act on lipids to produce peroxidation, the final oxidation product will be MDA. An increase in MDA also indicates a high content of free radicals. Figure 4C shows the content of SOD and MDA in the brain tissue of CLP-severe, CLP-mild, and sham mice. Compared with sham mice, both SOD and MDA were significantly generated in CLP mice, and the more severe the disease was, the higher the SOD and MDA levels were. This tendency was also consistent with the T1% changes in vivo.
We also stained ROS in mice brains of the control, CLP-mild, and CLP-severe group. As shown in Figure 4D, DHE staining showed abundant ROS in CLP-induced sepsis mouse brains compared with sham mice. CLP-severe mice’s brains also showed more ROS than that in CLP-mild mice. These data demonstrated the viability of this simple and stable methodology that uses DEPMPO-biotin and the designed targeting probe avidin-BSA@Gd-ESIO for macromolecule radical detection in vivo by MRI.
Considering the finite half-life of the DEPMPO-radical adduct, we further investigated the optimal imaging time of macromolecule free radicals in the SAE model detected by the novel IST approach. T1 MRI was performed on CLP-severe and sham mice at different times to measure the changes in T1%, as shown in Figure 5. Figure 5A illustrates the kinetic behavior of the avidin-BSA@Gd-ESIO probe within different brain regions of CLP-severe mice. In most brain regions, we observed sustained T1 signal increases over 30 minutes. The signal then increased in the hippocampus and medial cortex, but decreased in the striatum. This change mainly was due to the accumulation of continuously produced ROS in brain regions before 30 minutes and the fast metabolic clearance in the striatum of the probe after 30 minutes, as there are more blood vessels and circulation. Figure 5B–D show the %T1 signal change of CLP-severe and sham mice in the hippocampus, medial cortex, and striatum respectively before 30 minutes. At 30 minutes, there are significant differences in %T1 signals between CLP-severe and sham mice in all three regions. Therefore, the time of 30 minutes after the targeting probe administration should be the best visualization window for macromolecule free radical detection by this strategy in septic mouse brain.
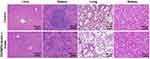
Finally, the safety profiles of the DEPMPO-Biotin and targeting probe avidin-BSA@Gd-ESIO were verified. Healthy mice were injected with DEPMPO-biotin and the probe, while control mice were treated with PBS. The liver, spleen, lung, and kidney were removed 30 minutes after administration of the probe for HE staining. As shown in Figure 6, compared to the control group, we found no difference in the pathology of all organs of mice injected with DEPMPO-biotin and the probe. No obvious tissue damage or inflammatory changes were found in the experimental group. The liver tissue capsule is composed of dense connective tissue with uniform thickness. There is a central vein in the center of the liver lobule, surrounded by liver cells and hepatic sinuses arranged radially. The hepatic plate was arranged regularly and neatly, and there was no obvious expansion or extrusion of hepatic sinuses. In spleen parenchyma, the structure of each layer was clear, and the number and size of each layer did not change significantly. The pulmonary parenchyma consisted of intrapulmonary bronchial branches and a large number of alveoli, with no obvious abnormal bronchial structure. The alveolar structure was clear without obvious inflammatory changes. The renal parenchyma consisted of the superficial cortex and deep medulla. The glomerulus was evenly distributed in the cortex, and the number of cells and matrix in the glomerulus was uniform. In summary, no obvious inflammatory changes were observed.
Discussion
Sepsis and multiple organ failure are associated with many molecular mechanisms of inflammation, including the generation of cytokines, eicosanoids, and ROS/RNS. The underlying mechanism of sepsis involves oxidative damage caused by the generation of free radicals. Oxygen-derived free radicals generated during reperfusion after ischemia or hypoxia or activated neutrophils associated with sepsis are known to be mediators of tissue injury.14 Importantly, oxidative damage is thought to be a major player in the long-lasting cognitive impairment observed in sepsis survivors. Apoptosis and bioenergetic failure from ROS/RNS causing structural membrane damage and mitochondrial dysfunction, respectively, are thought to be the major underlying mechanisms of SAE. In addition, studies by Gavins et al and Gu et al revealed a corresponding increase in BBB permeability during sepsis.3,20,21 We have reported a novel approach for the visualization of macromolecule free radicals in the sepsis mouse brain.
In this study, we combined DEPMPO-biotin and the targeting probe avidin-BSA@Gd-ESIO as a new IST-MRI strategy to detect macromolecule radicals in vivo. The avidin-BSA@Gd-ESIO probe consisted of the excellent T1 contrast agents Gd-DTPA and ESIO, as well as targeting peptide anti-biotin (avidin). Protein, lipid, and DNA radicals resulting from oxidative stress could be trapped by DEPMPO-biotin to form biotin-radical adducts. Then, the biotin-radical adducts were attached by avidin-BSA@Gd-ESIO through the strong avidin-biotin specific link. This strategy should be more stable and sensitive than the previously reported DMPO strategy.6
The confocal images of microglial cells showed that the radicals were distributed both inside the cell and on the cell membrane. The greater the number of radicals, the more probes were enriched in the cell. Note that although most protein, lipid, and DNA free radicals are localized inside the cell, the commonly used DMPO-radical adduct detection based on Gd-DTPA can attach only outside the cell. Reportedly, the Gd-DTPA mainly is attached outside the cell membrane.6,14 The avidin-BSA@Gd-ESIO probe designed in this study also could target macromolecule radicals within cells.
In in vivo experiments, CLP mice showed a significant T1% signal change in the brain, indicating that there were more radicals compared with the sham mice. The avidin-BSA@Gd-ESIO probe was able to cross the BBB, mainly because of the compromised BBB.3,18 In fact, a study by Gavins et al showed that during sepsis, circulating leukocytes decreased, the rolling and adhesion of leukocytes in the brain microvasculature increased, brain myeloperoxidase activity correspondingly increased, and BBB permeability increased.3,11
Figure 3A and B illustrate the diffuse distribution of the avidin-BSA@Gd-ESIO probe in the brains of septic mice compared with sham mice with an intact BBB, which allowed the probe to stay within the major blood vessels. A previous study found similar results and showed that the medical cortex region has more free radicals (greatest T1% change) than other regions. Our data, however, revealed that there were similar amounts of free radicals in all three regions. This difference may be due to the limitation of the previous detection methods and due to the fact that the DMPO-radical adducts have a very short half-life, which may render the detection process unstable. SAE is mainly driven by systemic inflammation. Researchers have found many inflammation and oxidative stress lesions in the hippocampus, cortex, and striatum in SAE models.18,22,23 These findings are consistent with our results.
Interestingly, the T1% increase after 30 minutes of administration in this work reached the same level as the reported study after 2 hours of administration.14 The highest T1% enhancement in this work is 30%, 31%, and 22% in the hippocampus, medial cortex, and striatum, respectively. This T1% enhancement was 1.5- to 2.5-fold higher than the reported work (12%, 20%, and 14% in these three regions, respectively), with a much lower dose of radical trap (25 μL DEPMPO-biotin) than reported (75 µL DMPO).14 This result indicated that the IST method based on DEPMPO-biotin and avidin-BSA@Gd-ESIO probe may be more sensitive and stable than previously reported. Therefore, this novel strategy could be used to efficiently detect macromolecule radicals, such as the C-centered protein, lipid, or DNA radicals, by MRI, noninvasively visualizing the state of oxidative stress and providing reliable technical support for oxidative damage assessment.
Conclusion
In this study, we used a combination of MRI and IST to develop a novel method for detecting macromolecule free radicals in SAE mice with DEPMPO-biotin and the targeting probe avidin-BSA@Gd-ESIO. We demonstrated that this novel approach could target macromolecule radicals with high sensitivity (up to 2.5-fold compared with previous approaches) and a low dose of the trapping agent. We speculated that macromolecule radicals both inside and outside cells could be detected. The new approach simultaneously used the MRI and IST methods, providing in vivo free radical regional targeting imaging and assessment of spatial oxidation differentiation of events in heterogeneous tissue or organ regions. This strategy also provides a method for the detection of free radical–mediated oxidation of cell membrane components. This new method could be applied to evaluate and locate free radical processes in any other pathologic state related to free radicals.
Abbreviations
SAE, sepsis associated encephalopathy; IST, immunospin trapping; MRI, magnetic resonance imaging; ESIO, extremely small iron oxide; ROS, reactive oxygen species; ESR, electron spin resonance; DMPO, 5,5-dimethyl-1-pyrroline n-oxide; DEPMPO, 5-(dihydroxyphosphonyl)-5-methyl-1-pyrroline-n-oxide; CLP, cecal ligation puncture; NHS, N-hydroxysuccinimide; EDC, N, N-(3-dimethyl aminopropyl)-N’-ethyl-carbodiimide hydrochloride; PBS, phosphate buffered saline; TEM, transmission electron microscopy; TGA, thermogravimetric analysis; DLS, dynamic light scattering; DHE, dihydroethidium; HE, hematoxylin-eosin; SOD, superoxide dismutase; MDA, malondialdehyde.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (82120108015, 81971586, 81771897, 82102020, 82071874, 81901712, 81771887), Sichuan Science and Technology Program (2020YFS0050, 2020YJ0029, 2017TD0005, 21ZDYF1967), Fundamental Research Funds for the Central Universities (SCU2020D4132), Clinical Research Finding of Chinese Society of Cardiovascular Disease (CSC) of 2019 (No. HFCSC2019B01), and 1·3·5 projects for disciplines of excellence, West China Hospital, Sichuan University (ZYGD18019).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Zhao Z, Wang Y, Zhou R, et al. A novel role of NLRP3-generated IL-1β in the acute-chronic transition of peripheral lipopolysaccharide-elicited neuroinflammation: implications for sepsis-associated neurodegeneration. J Neuroinflamm. 2020;17(64):1–19. doi:10.1186/s12974-020-1728-5
2. Bolton CF, Bryan GY, Zochodne DW. The neurological complications of sepsis. Ann Neurol. 1993;33(1):94–100. doi:10.1002/ana.410330115
3. Gu M, Mei XL, Zhao YN. Sepsis and cerebral dysfunction: BBB damage, neuroinflammation, oxidative stress, apoptosis and autophagy as key mediators and the potential therapeutic approaches. Neurotox Res. 2021;39(2):489–503. doi:10.1007/s12640-020-00270-5
4. Towner RA, Smith N, Doblas S, et al. In vivo detection of c-Met expression in a rat C6 glioma model. J Cell Mol Med. 2008;12(1):174–186. doi:10.1111/j.1582-4934.2008.00220.x
5. Claudine F, Hakim K, Beatrice T, et al. 5-(Diethoxyphosphoryl)-5-methyl-1-pyrroline N-oxide: a new efficient phosphorylated nitrone for the in vitro and in vivo spin trapping of oxygen-centered radicals. J Med Chem. 1995;38(2):258–265. doi:10.1021/jm00002a007
6. Towner RA, Smith N, Saunders D, et al. In vivo targeted molecular magnetic resonance imaging of free radicals in diabetic cardiomyopathy within mice. Free Radical Res. 2015;49(9):1140–1146. doi:10.3109/10715762.2015.1050587
7. Gomez-Mejiba SE, Zhai Z, Della-Vedova MC, et al. Immuno-spin trapping from biochemistry to medicine: advances, challenges, and pitfalls. Focus on protein-centered radicals. Biochim Biophys Acta. 2014;1840(2):722–729. doi:10.1016/j.bbagen.2013.04.039
8. Mason RP, Douglas G. Immuno-spin trapping of macromolecules free radicals in vitro and in vivo–One stop shopping for free radical detection. Free Radical Bio and Med. 2019;131:318–331. doi:10.1016/j.freeradbiomed.2018.11.009
9. Karoui H, Tordo P, Le Moigne F. Synthesis and spin-trapping properties of a trifluoromethyl analogue of DMPO: 5-Methyl-5-trifluoromethyl-1-pyrroline N -Oxide (5-TFDMPO). Chem-Eur J. 2014;20:4064–4071. doi:10.1002/chem.201303774
10. Liu KJ, Miyake M, Panz T, et al. Evaluation of DEPMPO as a spin trapping agent in biological systems. Free Radical Bio and Med. 1999;26(5–6):714–721. doi:10.1016/S0891-5849(98)00251-2
11. Kim BH, Lee N, Kim H, et al. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution t1 magnetic resonance imaging contrast agents. J Am Chem Soc. 2011;133(32):12624–12631. doi:10.1021/ja203340u
12. Ho J, Yu J, Wong SH, et al. Autophagy in sepsis: degradation into exhaustion? Autophagy. 2016;12(7):1073–1082. doi:10.1080/15548627.2016.1179410
13. Yanyan H, Zhen L, Liu C, et al. Self-assembly of multi-nanozymes to mimic an intracellular antioxidant defense system. Angew Chem Int Edit. 2016;128:6758–6762. doi:10.1002/ange.201600868
14. Towner RA, Garteiser P, Bozza F, et al. In vivo detection of free radicals in mouse septic encephalopathy using molecular mri and immuno-spin-trapping. Free Radical Bio and Med. 2013;65:828–837. doi:10.1016/j.freeradbiomed.2013.08.172
15. Singh B, Kumar A. Radiation formation of functionalized polysaccharide-protein based skin mimicking semi-inter penetrating network for biomedical application. Int J Biol Macromol. 2016;92:1136–1150. doi:10.1016/j.ijbiomac.2016.08.011
16. Khan SR, Baghdasarian A, Nagar PH, et al. Proteomic profile of aminoglutethimide-induced apoptosis in HL-60 cells: role of myeloperoxidase and arylamine free radicals. Chem-Biol Interact. 2015;239:129–138. doi:10.1016/j.cbi.2015.06.020
17. Anirudhan TS, Mohan AM. Novel pH sensitive dual drug loaded-gelatin methacrylate/methacrylic acid hydrogel for the controlled release of antibiotics. Int J Biol Macromol. 2018;110:167–178. doi:10.1016/j.ijbiomac.2018.01.220
18. Semmler A, Okulla T, Sastre M, et al. Systemic inflammation induces apoptosis with variable vulnerability of different brain regions. J Chem Neuroanat. 2005;30(2–3):144–157. doi:10.1016/j.jchemneu.2005.07.003
19. Ighodaro OM, Akinloye OA. First line defense antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): their fundamental role in the entire antioxidant defense grid. Alexandria J Med. 2018;54:287–293. doi:10.1016/j.ajme.2017.09.001
20. Gavins FN, Hughes EL, Buss NA, et al. Leukocyte recruitment in the brain in sepsis: involvement of the annexin 1–FPR2/ALX anti-inflammatory system. FASEB J. 2012;26:4977–4989. doi:10.1096/fj.12-205971
21. Semmler A, Hermann S, Mormann F, et al. Sepsis causes neuroinflammation and concomitant decrease of cerebral metabolism. J Neuroinflamm. 2008;5(1):1–10. doi:10.1186/1742-2094-5-38
22. Ning Q, Liu Z, Wang X, et al. Neurodegenerative changes and neuroapoptosis induced by systemic lipopolysaccharide administration are reversed by dexmedetomidine treatment in mice. Neurol Res. 2017;39(4):357–366. doi:10.1080/01616412.2017.1281197
23. Barichello T, Fortunato J, Vitali AM, et al. Oxidative variables in the rat brain after sepsis induced by cecal ligation and perforation. Crit Care Med. 2006;34:886–889. doi:10.1097/01.CCM.0000201880.50116.12
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.