Back to Journals » Clinical Ophthalmology » Volume 12
In vivo assessment of changes in corneal hysteresis and lamina cribrosa position during acute intraocular pressure elevation in eyes with markedly asymmetrical glaucoma
Authors Gizzi C, Cellini M, Campos EC
Received 13 September 2017
Accepted for publication 30 January 2018
Published 15 March 2018 Volume 2018:12 Pages 481—492
DOI https://doi.org/10.2147/OPTH.S151532
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Corrado Gizzi, Mauro Cellini, Emilio C Campos
Department of Experimental, Diagnostic and Speciality Medicine, Ophthalmology Service, University of Bologna, Bologna, Italy
Purpose: To investigate the biomechanical response of the cornea, lamina cribrosa (LC), and prelaminar tissue (PT) to an acute intraocular pressure (IOP) increase in patients with markedly asymmetrical glaucoma and in healthy controls.
Patients and methods: A total of 24 eyes of 12 patients with markedly asymmetrical primary open-angle glaucoma (POAG) and 12 eyes of 12 healthy patients were examined with spectral-domain optical coherence tomography (SD-OCT) and ocular response analyzer (ORA) at baseline and during acute IOP elevation by means of an ophthalmodynamometer. The displacement of the LC and PT and the change in corneal hysteresis (CH) and corneal resistance factor (CRF) were evaluated.
Results: Following a mean IOP increase of 12.3±2.4 mmHg, eyes with severe glaucoma demonstrated an overall mean anterior displacement of the LC (-6.58±26.09 µm) as opposed to the posterior laminar displacement in eyes with mild glaucoma (29.08±19.28 µm) and in healthy eyes (30.3±10.9; p≤0.001 and p=0.001, respectively). The PT displaced posteriorly during IOP elevation in all eyes. The CH decreased in eyes with severe glaucoma during IOP elevation (from 9.30±3.65 to 6.92±3.04 mmHg; p=0.012), whereas the CRF increased markedly in eyes with mild glaucoma (from 8.61±2.30 to 12.38±3.64; p=0.002) and in eyes with severe glaucoma (from 9.02±1.48 to 15.20±2.06; p=0.002). The increase in CRF correlated with the anterior displacement of the LC in eyes with severe glaucoma.
Conclusion: Eyes with severe glaucoma exhibited a mean overall anterior displacement of the anterior laminar surface, while eyes with mild glaucoma and healthy eyes showed a posterior displacement of the LC during IOP elevation. The CH decreased significantly from baseline only in eyes with severe glaucoma, but the CRF increased significantly in all glaucomatous eyes. The CRF increase correlated with the anterior displacement of the LC in eyes with severe glaucoma.
Keywords: glaucoma, lamina cribrosa, cornea, hysteresis, ophthalmodynamometer, biomechanics, asymmetry
Introduction
Increased intraocular pressure (IOP) is a well-known risk factor for the onset and progression of glaucomatous optic neuropathy (GON).1–3 It is widely accepted that an increase in IOP may lead to strain at the level of the lamina cribrosa (LC) and to a subsequent variable degree of deformation of the laminar tissue. This, in turn, may result in both mechanical and metabolic damage to the optic nerve fibers exiting the intraocular space via the laminar pores.4–6
The biomechanical properties of the LC have been thoroughly investigated,7–14 with a particular focus on the effect of raised IOP on the deformation of the LC. Studies conducted in this respect have employed ex vivo models,15,16 histomorphometric analysis of the LC in animal models,17 and mathematical models based on human eyes.18–20
In recent years, the increased availability of high-resolution imaging devices, such as spectral-domain optical coherence tomography (SD-OCT), allowed in vivo analysis of the LC and prelaminar tissue (PT) in static and dynamic conditions, such as in human eyes with decreased IOP following filtering surgery21 or during an acute increase in IOP.22–24
Central corneal thickness (CCT) is also thought to be involved in glaucoma onset and progression, yet its role as an independent risk factor is still a matter of debate, as it could be just a surrogate for IOP. Nonetheless, other corneal features may prove useful to obtain more information on the overall biomechanical quality of the eye. The corneal properties might be linked to those of the LC and, therefore, provide insight into the individual response to mechanical stress at the level of the LC.25–30 More specifically, corneal features might be a surrogate for extracellular matrix (ECM) quality. In support of this hypothesis, it has been shown that 2 corneal biomechanical parameters, namely, corneal hysteresis (CH) and corneal resistance factor (CRF), are both reduced in Marfan syndrome, which is characterized by a systemic alteration of the ECM.31 Given that ECM components of the cornea, peripapillary scleral ring, and LC are coded by the same gene, it is plausible that they also share a similar biomechanical behavior.32
It is well known that primary open-angle glaucoma (POAG), the most common form of glaucoma worldwide, is often a bilateral disease. However, there are a considerable number of cases where the stage of the GON is markedly asymmetrical or even altogether unilateral. This disparity might reflect the presence of asymmetrical biomechanical properties in the contralateral fellow eye.
The aim of this study was to investigate the behavior of LC and PT following an induced increase in IOP via SD-OCT with enhanced depth imaging (EDI) in patients with markedly asymmetrical glaucoma damage and in control subjects. A protocol similar to the one previously described by Agoumi et al24 was implemented.
Two corneal biomechanical properties (CH and CRF) were also assessed at baseline and during IOP elevation in all the patients enrolled.
Patients and methods
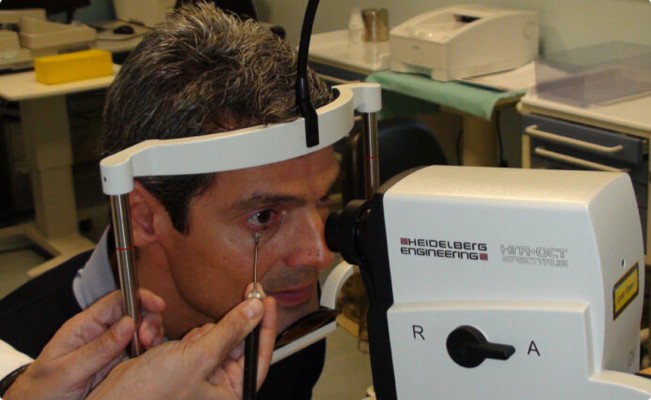
The current study was approved by the local ethics committee of the University of Bologna and adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from all participants before any procedure. Mauro Cellini, MD, received from the person in Figure 1 the informed written consent to have the image published.
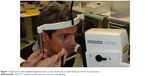  | Figure 1 Application of the ophthalmodynamometer on the infratemporal eyelid during an SD-OCT examination. |
A total of 12 consecutive patients attending the Glaucoma Unit of the Sant’Orsola-Malpighi Hospital (Bologna, Italy) with a diagnosis of POAG and a marked asymmetry in the GON stage between fellow eyes were recruited in the study. A total of 12 control subjects were enrolled among patients attending the eye clinic of the same hospital for a routine review without any specific ocular condition.
Inclusion criteria for glaucoma patients were the following: clinical diagnosis of glaucoma based on visual field (VF) testing, optic disc and retinal nerve fiber layer (RNFL) clinical appearance, open angle at gonioscopy, asymmetrical VF damage with 1 eye presenting a mean deviation (MD) greater than or equal to −6 dB and the fellow eye presenting an MD less than or equal to −12 dB, a satisfactory IOP control (≤21 mmHg) on medications, and a history of IOP >21 mmHg prior to commencement of treatment.
Exclusion criteria for glaucoma patients were the following: threat to fixation, past ocular history of any laser or surgical procedure, non-glaucomatous optic neuropathies, suboptimal IOP control on current medications (IOP >21 mmHg) and/or recent evidence of disease progression, poor fixation due to ocular (eg, low vision and nystagmus) or cognitive conditions (eg, dementia, poor cooperation), or age <18 years. Patients with a connective tissue disease (such as Marfan syndrome) or diabetes mellitus were also excluded from the study.
Inclusion criteria for control subjects were unremarkable ocular condition with a healthy optic disc and RNFL appearance, IOP within 21 mmHg, and normal VF. Exclusion criteria for normal subjects were past ocular history of any laser or surgical procedure, poor fixation, age <18 years, connective tissue diseases, or diabetes mellitus.
All the patients underwent a complete eye examination, including visual acuity assessment, Goldmann applanation tonometry (GAT), gonioscopy, and anterior and posterior segment biomicroscopy. At least 2 previous reliable VF tests (with fixation losses, false positives, and false negatives ≤20%) were required for each glaucoma patient. One additional VF test was performed with the same perimeter and strategy (Humphrey 24-2 SITA Standard; Carl Zeiss Meditec, Dublin, CA, USA) in every partecipant to the study to collect MD and pattern standard deviation (PSD) data.
Based on the perimetric indices, glaucomatous fellow eyes were divided into 2 groups: mild glaucoma when the MD ranged from −2 to −6 dB or severe glaucoma when the MD was equal or lower than −12 dB. The control group had an MD greater than or equal to −2 dB.
Spectral-domain optimal coherence tomography
SD-OCT has greatly enhanced diagnostic capabilities in ophthalmology. The low-coherence interferometer integrated into the device allows the acquisition of in vivo detailed cross-sectional images of the ocular structures. The reference and the reflected beams are simultaneously recorded by a spectrometer to generate A-scans from Fourier-transform time-delay signals.
A Heidelberg Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany) was employed in this study. During the scan, the EDI mode was turned on to obtain enhanced details of deep ocular tissues and, therefore, facilitate the subsequent analysis. A radial scanning pattern (12 high-resolution 15° spaced radial scans each averaged from 30 B-scans with 768 A-scans per B-scan) was centered on the optic nerve of each eye included in the study. Glaucoma patients had both eyes imaged, whereas a randomly selected eye was scanned in each control subject.
The eye tracker function available in the Spectralis OCT was capable of obtaining a baseline image for each eye and to repeat the scan at the same position during the application of an ophthalmodynamometer on the temporal side of the inferior eyelid. Images with insufficient details were discarded, and the scans were repeated to obtain a quality level appropriate for further analysis.
Ocular response analyzer (ORA)
Corneal biomechanics data were collected with the ORA (Reichert Ophthalmic Instruments Inc, Depew, NY, USA). The device, which is similar to a noncontact tonometer, detects the change of shape of the corneal surface, while an air jet is delivered by the instrument. During the examination, the cornea is first deflected inward by the air puff, and then it regains its prolate configuration when the air stimulus terminates. An integrated infrared beam allows the recording of the forces required to applanate the cornea during the inward and outward movements of the corneal surface. The data collected on the force-in and force-out applanations are then used to calculate 2 biomechanical parameters, CH and CRF. The CH value provides a better characterization of the corneal contribution to IOP measurement than CCT alone. The CRF is a measure of corneal resistance, which is relatively independent of IOP.33
An ORA examination was performed at baseline and during IOP elevation by means of an ophthalmodynamometer applied to the temporal side of the lower eyelid. Four measurements were collected for each eye, and the one with the highest quality score was selected for analysis.
Each patient underwent the ORA examination following the SD-OCT scan, with an interval of at least 1 hour between the 2 examinations. This was considered a sufficient time to allow each eye to recover from the previous ophthalmodynamometer compression.
IOP elevation
An ophthalmodynamometer (Inami, Tokyo, Japan) was used to increase the IOP during the SD-OCT and ORA examinations. The procedure was similar to the one employed by Agoumi et al.24
The device was applied at the temporal side of the lower eyelid and pressure was exerted to reach a reading of ~30 Pa (Figure 1). An IOP check was performed with a Tono-Pen tonometer (Innova Medical Ophthalmics, Toronto, ON, Canada) at baseline and during the application of the ophthalmodynamometer. The device was held in place during collection of the second set of OCT scans and the second set of ORA measurements. Both the tests took ~<1 min each. No patient experienced pain during the application of the ophthalmodynamometer; only a sensation of pressure on the globe was reported. No patient experienced changes in the quality or comfort of vision after the pressure elevation.
SD-OCT scan processing and analysis
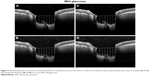
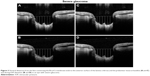
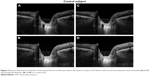
Among the 12 radial scans obtained during each examination, 2 corresponding sets of 4 images that provided the best anatomical details at baseline and during IOP elevation were selected for each eye.
The images were processed with open-source vector graphics software (Inkscape, http://www.inkscape.org). A reference line connecting both sides of the termination of the retinal pigment epithelium–Bruch’s membrane interface was drawn. A set of equidistant lines perpendicular to the reference line were traced to intersect the anterior edge of the PT, and the image was saved. Then the same lines were drawn further down to intersect the anterior edge of the LC, which is seen as a highly reflective tissue beneath the PT, and a second image was saved. The same process was repeated for all the set of images acquired at baseline and during IOP elevation (Figures 2–4).
The vertical distances from the reference line to both the anterior limit of the PT and the LC were measured with ImageJ (National Institutes of Health, Bethesda, MD, USA), after calibrating the software with the scale bar included in each image.
The prelaminar and laminar tissue displacements were calculated as the mean difference between the corresponding vertical distances from the reference line in the baseline and elevated IOP conditions. Positive values were suggestive of an overall posterior movement of the tissues, whereas negative values were interpreted as an overall anterior displacement of the tissues examined. All the images were analyzed by the same trained examiner (CG) who was masked on the source of the images.
Statistical analysis
The Mann–Whitney U-test, Wilcoxon signed-rank test, and Friedman test were employed as appropriate for statistical analysis. Correlations were evaluated using Spearman correlation coefficients. The statistical analysis was performed with R (R software; provided in the public domain by the R Foundation for Statistical Computing, Vienna, Austria; available at www.r-project.org), and a p-value <0.05 was considered statistically significant.
Results
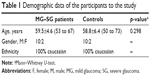
A total of 24 eyes of 12 patients with POAG and a significant degree of asymmetry in the stage of the disease between fellow eyes and 12 eyes of 12 healthy subjects were recruited for the study. The demographic data of the 3 groups are represented in Table 1.
  | Table 1 Demographic data of the participants to the study |
Baseline ocular data (VF, CCT, IOP, CH and CRF) are shown in Table 2. As expected, at baseline there was a significant difference in the VF indices among the groups. Moreover the b-CRF (baseline CRF) was significantly lower in eyes with mild glaucoma (8.61±2.3 mmHg) and severe glaucoma (9.02±1.48 mmHg) compared with controls (p=0.039 and p=0.034, respectively).
Table 3 provides the details on IOP measured with GAT, Tonopen, and ORA.
The increase in IOP and the forces exerted following the application of the ophthalmodynamometer during both the OCT scans and the ORA measurements were not statistically different among the 3 groups (Table 4). The ophthalmodynamometer-induced CH (i-CH) was significantly lower in eyes with mild glaucoma (7.23±2.29 mmHg; p=0.011) and in eyes with severe glaucoma (6.92±3.04 mmHg; p=0.003) compared to controls (10.59±1.78 mmHg; Table 5). The i-CH was also significantly lower than baseline CH (b-CH) in eyes with severe glaucoma (6.92±3.04 vs 9.30±3.65 mmHg; p=0.012), whereas no significant changes were detected in the other groups (Table 6).
The ophthalmodynamometer-induced CRF (i-CRF) was significantly higher in eyes with severe glaucoma (15.2±2.06 mmHg) than in eyes with mild glaucoma and in controls (12.38±3.65 and 11.27±1.46 mmHg; p=0.027 and p=0.019, respectively). The i-CRF was also significantly higher than the b-CRF in both eyes with mild and severe glaucoma (12.38±3.64 vs 8.61±2.30 mmHg and 15.20±2.06 vs 9.02±1.48 mmHg; p=0.002 and p=0.002, respectively), whereas no significant change was observed in the control group (11.27±1.46 vs 10.64±1.28, p=0.275).
In terms of the anterior laminar surface response to the ophthalmodynamometer compression, we found that the laminar displacement (LD) was not statistically different between eyes with mild glaucoma and control eyes (29.08±19.28 and 30.3±10.9 μm, respectively; p=0.833). On the other hand, eyes with severe glaucoma exhibited an overall mean anterior displacement of the LC (−6.58±26.09 μm), which was significant when compared with both the control and mild glaucoma groups (p=0.001 and p<0.001, respectively).
It was also observed that the PT displacement (PTD) was reduced in eyes with severe glaucoma (13.08±17.06 μm) when compared with the control group (36.0±17.9 μm; p=0.001). On the other hand, the PTD in eyes with mild glaucoma (22.33±33.14 μm) was not significantly different to the PTD in eyes with severe glaucoma and in controls (p=0.518 and p=0.131, respectively). The data are summarized in Table 3.
Finally, we found a correlation (r=−658; p=0.019) between the anterior LD and an increase in CRF in the severe glaucoma group (Figure 5).
  | Figure 5 Scatterplot illustrating the inverse correlation by Spearman’s test between CRF and LD in eyes with severe glaucoma. |
Discussion
The biomechanical properties of the LC have attracted the interest of many researchers in recent years as they might lead to a better understanding of glaucoma pathophysiology.34 An increase in the IOP is thought to induce a strain on the laminar tissue that may put the integrity of the optic nerve fibers at risk. The damage induced on the axons might be the result of a mechanical stress,35 a vascular impairment,36 an increase in the translaminar pressure gradient,12,37–40 or a combination of all these mechanisms.
The importance of IOP on the dynamics of the optic nerve structures has been highlighted by the observation of an anterior displacement of the optic nerve surface after IOP reduction both surgically and with medical treatment.41–43 On the other hand, it has been demonstrated that an increase in the IOP may result in a posterior displacement of the optic nerve head (ONH).22,23 Similar outcomes have been achieved in experimental studies on animals.44
The availability in recent years of SD-OCT with EDI allowed the study of the LC and PT with unprecedented accuracy. This advanced technology provides considerable details even when the globe is compressed and deformed by the application of an external force via an ophthalmodynamometer.24
We observed that an average IOP increase of 12.3±2.4 mmHg determined a mean posterior displacement of the LC both in healthy controls and in eyes with mild glaucoma. However, in eyes with advanced glaucoma, we detected a mean anterior displacement of the LC.
All the groups demonstrated a mean posterior displacement of the PT, although it was significantly lower in the advanced glaucoma group compared with the control group.
Anterior laminar surface response to mechanical strain has been investigated in different study cohorts and with different protocols. This, together with the high degree of biomechanical complexity of the LC and scleral canal, might explain the dissimilar and sometimes contradictory results obtained.
Agoumi et al24 did not find any significant displacement of the LC with SD-OCT following an induced IOP increase with an ophthalmodynamometer in both glaucoma and control subjects. Differences in the baseline characteristics of the patients investigated, including the glaucoma stage (the average MD was −2.8 dB in the study by Agoumi et al), might at least partially explain the inconsistency of our results.
Fazio et al45 found that a biphasic behavior of the LC was dependent on the IOP level in healthy subjects of European descent. The LC displaced posteriorly until the IOP elevation from baseline was within 12 mmHg; after that value, the LC displaced anteriorly.
Previous studies on healthy enucleated human eyes15,16 and primate eyes with 3D histomorphometric analysis46 demonstrated a variable response of the lamina to an increased IOP, ranging from a posterior displacement to a nonsignificant displacement.
On the basis of mathematical models, some authors described the behavior of the laminar tissue as the result of the biomechanical and anatomical properties not only of the LC itself but also of the adjacent scleral tissue.19,20 Recent studies supported the hypothesis of a high degree of complexity in the interaction between the laminar tissue and the peripapillary scleral tissue, which may cause a variable response to the mechanical strain exerted by IOP.47–54
We found that eyes with advanced glaucoma had an overall mean anterior displacement of the LC during acute IOP elevation, which is contrary to the posterior displacement observed in mild glaucoma and healthy eyes. We speculate that the different behavior reflected progressive changes at the level of the laminar and peripapillary scleral tissue. An increasing stiffening of the laminar tissue relative to the surrounding sclera might explain the trend toward an anterior bowing of the anterior laminar surface in eyes with advanced glaucoma during IOP elevation as opposed to that noted in eyes with mild glaucoma and in healthy eyes. The opposite direction of displacement observed in fellow eyes might reflect the progressive anatomical and biomechanical changes occurring within the ocular tissues as the glaucoma stage progresses.
The CCT was previously found to be correlated with optic disc features and the likelihood of VF progression in glaucoma.25–29 However, CCT is not considered a reliable reflection of the rigiditiy of the ocular structures and may not provide a significant description of the biomechanics of the eye.55 Hence, CCT values, which were found to be not significantly different among the groups, have probably a marginal role in the interpretation of our results. Corneal biomechanical parameters, such as CH and CRF, are much more closely related to ocular biomechanics and have been correlated to glaucoma onset and progression.30 While CH is a measure of the viscoelastic dampening features of the cornea, CRF indicates the whole viscoelastic resistance. These indices might also be linked to the overall quality of the ECM.31
We performed an ORA examination at baseline and during an ophthalmodynamometer-induced increase in IOP. We found that b-CH was not significantly different among the groups. On the other hand, the CRF was significantly lower in eyes with glaucoma than in control eyes, but no significant difference was found between the eyes with mild and severe glaucoma.
During the ophthalmodynamometer-induced increase in the IOP, mean CH was significantly lower in eyes with mild and advanced glaucoma than in the controls. However, compared with baseline, only eyes with advanced glaucoma exhibited a significant decrease in CH.
The CRF during IOP elevation was significantly higher in eyes with severe glaucoma compared with the other groups. The mean CRF value was also significantly higher than baseline in eyes with mild and severe glaucoma. We also observed a correlation between the increase in CRF during IOP increase and the anterior displacement of the LC in the severe glaucoma group.
The corneal behavior observed in our experiment is in line with a previously described viscoelastic model of the cornea, where a stiffening of the corneal structure (CRF increase) is accompanied by the parallel reduction of the viscous component (CH reduction).56 A CH reduction and a CRF increase, such as the one observed in eyes with glaucoma during IOP elevation, might be linked to an LC with an impaired capacity to dampen IOP changes. This might increase the susceptibility to glaucoma damage.
A reduction in the CH was previously noted in glaucomatous subjects57–59 and was thought to be related to deterioration in VF.28,60–64 The potential role of CH in glaucoma as a surrogate for ONH biomechanics is also suggested by the observation that in glaucomatous eyes, CH is correlated to optic nerve surface deformation.23,65 Corneal biomechanical properties might therefore give indirect information on the structural features and vulnerability of the ONH. However, other authors reported a lack of correlation between ORA parameters (both CH and CRF), MD, and the severity of glaucoma.66
Although we observed a lower CH in the glaucoma groups compared with the control group, the difference among the groups failed to reach statistical significance.
In summary, we found that in patients with markedly asymmetrical glaucoma, eyes with advanced glaucoma responded to an acute IOP elevation with an overall mean anterior displacement of the LC as opposed to the posterior displacement observed in eyes with mild glaucoma and healthy eyes.
The CH baseline value was lower (but not significantly) in eyes with glaucoma compared with the controls. During acute IOP elevation, the CH decreased markedly in both the advanced and mild glaucoma eyes to a mean value significantly lower than the one found in healthy eyes.
The CRF value at baseline was lower in glaucoma eyes compared with healthy eyes. However, CRF increased significantly during acute IOP elevation in all the groups, and the increase was significantly higher in eyes with advanced glaucoma.
We also found that in patients with markedly asymmetrical glaucoma damage, the eye with advanced glaucoma shows a positive correlation between the CRF increase and the anterior displacement of the LC during IOP elevation.
In light of our findings, we may formulate a hypothesis to interpret our results despite the complexity of the matter. Raised IOP in POAG patients causes progressive anatomical and biomechanical changes at the level of the LC and adjacent scleral tissue. These alterations might be reflected by the biomechanical response of the cornea and LC to an induced increase in the IOP, which might, in turn, correlate to the stage of glaucoma.
There are several limitations in this study. The groups were small, and the laminar features were manually analyzed instead of being automatically detected and measured by the software. The measurements obtained on the laminar and PTs were limited to 4 scans per eye, which might be insufficient to estimate the actual response of these tissues to biomechanical strain. However, despite these limitations, the inclusion of patients with markedly asymmetrical disease between fellow eyes offered the chance to investigate differences in biomechanical response in different stages of the disease. Further studies with larger groups are necessary to validate our preliminary results.
Acknowledgment
The authors thank mechanical engineer Marco Cellini for his contribution to the understanding of corneal biomechanics after an acute increase in intraocular pressure.
Disclosure
The authors report no conflicts of interest in this work.
References
Gordon MO, Beiser JA, Brandt JD, et al. The Ocular Hypertension Treatment Study: baseline factors that predict the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):714–720. | ||
Leske MC, Heijl A, Hussein M, et al; Early Manifest Glaucoma Trial Group. Factors for glaucoma progression and the effect of treatment: the early manifest glaucoma trial. Arch Ophthalmol. 2003;121(1):48–56. | ||
Leske MC, Heijl A, Hyman L, Bengtsson B, Dong L, Yang Z; EMGT Group. Predictors of long-term progression in the early manifest glaucoma trial. Ophthalmology. 2007;114(11):1965–1972. | ||
Varma R, Minckler DS. Anatomy and pathophysiology of the retina and optic nerve. In: Ritch R, Shields MB, Krupin T, editors. The Glaucomas. 2nd ed. vol. 1. St. Louis, MO: Mosby; 1996:139–175. | ||
Sigal IA, Flanagan JG, Tertinegg I, Ethier CR. Predicted extension, compression and shearing of optic nerve head tissues. Exp Eye Res. 2007;85(3):312–322. | ||
Sigal IA, Flanagan JG, Tertinegg I, Ethier CR. Modeling individual-specific human optic nerve head biomechanics. Part I: IOP-induced deformations and influence of geometry. Biomech Model Mechanobiol. 2009;8(2):85–98. | ||
Downs JC, Roberts MD, Burgoyne CF. Mechanical environment of the optic nerve head in glaucoma. Optom Vis Sci. 2008;85(6):425–435. | ||
Burgoyne CF, Downs JC, Bellezza AJ, Suh JK, Hart RT. The optic nerve head as a biomechanical structure: a new paradigm for understanding the role of IOP-related stress and strain in the patho-physiology of glaucomatous optic nerve head damage. Prog Retin Eye Res. 2005;24(1):39–73. | ||
Sigal IA, Flanagan JG, Ethier CR. Factors influencing optic nerve head biomechanics. Invest Ophthalmol Vis Sci. 2005;46(11):4189–4199. | ||
Bellezza AJ, Hart RT, Burgoyne CF. The optic nerve head as a biomechanical structure: initial finite element modeling. Invest Ophthalmol Vis Sci. 2000;41(10):2991–3000. | ||
Burgoyne CF, Downs JC, Bellezza AJ, Hart RT. Three-dimensional reconstruction of normal and early glaucoma monkey optic nerve head connective tissues. Invest Ophthalmol Vis Sci. 2004;45(12):4388–4399. | ||
Downs JC, Blidner RA, Bellezza AJ, Thompson HW, Hart RT, Burgoyne CF. Peripapillary scleral thickness in perfusion-fixed normal monkey eyes. Invest Ophthalmol Vis Sci. 2002;43(7):2229–2235. | ||
Ethier CR, Johnson M, Ruberti J. Ocular biomechanics and biotransport. Annu Rev Biomed Eng. 2004;6:249–273. | ||
Edwards ME, Good TA. Use of a mathematical model to estimate stress and strain during elevated pressure induced lamina cribrosa deformation. Curr Eye Res. 2001;23(3):215–225. | ||
Levy NS, Crapps EE. Displacement of optic nerve head in response to short-term intraocular pressure elevation in human eyes. Arch Ophthalmol. 1984;102(5):782–786. | ||
Yan DB, Coloma FM, Metheetrairut A, Trope GE, Heathcote JG, Ethier CR. Deformation of the lamina cribrosa by elevated intraocular pressure. Br J Ophthalmol. 1994;78(8):643–648. | ||
Bellezza AJ, Rintalan CJ, Thompson HW, Downs JC, Hart RT, Burgoyne CF. Deformation of the lamina cribrosa and anterior scleral canal wall in early experimental glaucoma. Invest Ophthalmol Vis Sci. 2003;44(2):623–637. | ||
Sigal IA, Flanagan JG, Tertinegg I, Ethier CR. Modeling individual-specific human optic nerve head biomechanics. Part I: IOP-induced deformations and influence of geometry. Biomech Model Mechanobiol. 2009;8(2):85–98. | ||
Sigal IA, Flanagan JG, Ethier CR. Factors influencing optic nerve head biomechanics. Invest Ophthalmol Vis Sci. 2005;46(11):4189–4199. | ||
Sigal IA, Flanagan JG, Tertinegg I, Ethier CR. Modeling individual-specific human optic nerve head biomechanics. Part II: influence of material properties. Biomech Model Mechanobiol. 2009;8(2):99–109. | ||
Yuen D, Buys YM. Disc photography and Heidelberg retinal tomography documentation of reversal of cupping following trabeculectomy. Graefes Arch Clin Exp Ophthalmol. 2010;248(11):1671–1673. | ||
Azuara-Blanco A, Harris A, Cantor LB, Abreu MM, Weinland M. Effects of short term increase of intraocular pressure on optic disc cupping. Br J Ophthalmol. 1998;82(8):880–883. | ||
Wells AP, Garway-Heath DF, Poostchi A, Wong T, Chan KC, Sachdev N. Corneal hysteresis but not corneal thickness correlates with optic nerve surface compliance in glaucoma patients. Invest Ophthalmol Vis Sci. 2008;49(8):3262–3268. | ||
Agoumi Y, Sharpe GP, Hutchison DM, Nicolela MT, Artes PH, Chauhan BC. Laminar and prelaminar tissue displacement during intraocular pressure elevation in glaucoma patients and healthy controls. Ophthalmology. 2011;118(1):52–59. | ||
Pakravan M, Parsa A, Sanagou M, Parsa CF. Central corneal thickness and correlation to optic disc size: a potential link for susceptibility to glaucoma. Br J Ophthalmol. 2007;91(1):26–28. | ||
Hong S, Kim CY, Seong GJ, Hong YJ. Central corneal thickness and visual field progression in patients with chronic primary angle closure glaucoma with low intraocular pressure. Am J Ophthalmol. 2007;143(2):362–363. | ||
Rogers DL, Cantor RN, Catoira Y, Cantor LB, WuDunn D. Central corneal thickness and visual field loss in fellow eyes of patients with open-angle glaucoma. Am J Ophthalmol. 2007;143(1):159–161. | ||
Medeiros FA, Sample PA, Zangwill LM, Bowd C, Aihara M, Weinreb RN. Corneal thickness as a risk factor for visual field loss in patients with preperimetric glaucomatous optic neuropathy. Am J Ophthalmol. 2003;136(5):805–813. | ||
Jonas JB, Stroux A, Velten I, Juenemann A, Martus P, Budde WM. Central corneal thickness correlated with glaucoma damage and rate of progression. Invest Ophthalmol Vis Sci. 2005;46(4):1269–1274. | ||
Congdon NG, Broman AT, Bandeen-Roche K, Grover D, Quigley HA. Central corneal thickness and corneal hysteresis associated with glaucoma damage. Am J Ophthalmol. 2006;141(5):868–875. | ||
Beene LC, Traboulsi EI, Seven I, et al. Corneal deformation response and ocular geometry: a noninvasive diagnostic strategy in marfan syndrome. Am J Ophthalmol. 2016;161:56.e1–64.e1. | ||
Karali A, Russell P, Stefani FH, Tamm ER. Localization of myocilin/trabecular meshwork-inducible glucocorticoid response protein in the human eye. Invest Ophthalmol Vis Sci. 2000;41(3):729–740. | ||
Luce DA. Determining in vivo biomechanical properties of the cornea with an ocular response analyzer. J Cataract Refract Surg. 2005;31(1):156–162. | ||
Burgoyne FC, Downs JC. Premise and prediction-how optic nerve head biomechanics underlines the susceptibility and clinical behavior of the aged optic nerve head. J Glaucoma. 2008;17(4):318–328. | ||
Gaasterland D, Tanishima T, Kuwabara T. Axoplasmic flow during chronic experimental glaucoma. 1. Light and electron microscopic studies of the monkey optic nerve head during development of glaucomatous cupping. Invest Ophthalmol Vis Sci. 1978;17(9):838–846. | ||
Hiraoka M, Inoue K, Ninomiya T, Takada M. Ischaemia in the Zinn-Haller circle and glaucomatous optic neuropathy in macaque monkeys. Br J Ophthalmol. 2012;96(4):597–603. | ||
Maumenee AE. Causes of optic nerve damage in glaucoma. Robert N. Shaffer lecture. Ophthalmology. 1983;90(7):741–752. | ||
Berdahl JP, Allingham RR, Johnson DH. Cerebrospinal fluid pressure is decreased in primary open-angle glaucoma. Ophthalmology. 2008;115(5):763–768. | ||
Jonas JB, Berenshtein E, Holbach L. Anatomic relationship between lamina cribrosa, intraocular space, and cerebrospinal fluid space. Invest Ophthalmol Vis Sci. 2003;44(12):5189–5195. | ||
Morgan WH, Yu DY, Balaratnasingam C. The role of cerebrospinal fluid pressure in glaucoma pathophysiology: the dark side of the optic disc. J Glaucoma. 2008;17(5):408–413. | ||
Irak I, Zangwill L, Garden V, Shakiba S, Weinreb RN. Change in optic disk topography after trabeculectomy. Am J Ophthalmol. 1996;122(5):690–695. | ||
Lesk MR, Spaeth GL, Azuara-Blanco A, et al. Reversal of optic disc cupping after glaucoma surgery analyzed with a scanning laser tomograph. Ophthalmology. 1999;106(5):1013–1018. | ||
Bowd C, Weinreb RN, Lee B, Emdadi A, Zangwill LM. Optic disk topography after medical treatment to reduce intraocular pressure. Am J Ophthalmol. 2000;130(3):280–286. | ||
Morgan WH, Chauhan BC, Yu DY, Cringle SJ, Alder VA, House PH. Optic disc movement with variations in intraocular and cerebrospinal fluid pressure. Invest Ophthalmol Vis Sci. 2002;43(10):3236–3242. | ||
Fazio MA, Johnstone JK, Smith B, Wang L, Girkin CA. Displacement of the lamina cribrosa in response to acute intraocular pressure elevation in normal individuals of African and European descent. Invest Ophthalmol Vis Sci. 2016;57(7):3331–3339. | ||
Yang H, Downs JC, Sigal IA, Roberts MD, Thompson H, Burgoyne CF. Deformation of the normal monkey optic nerve head connective tissue after acute IOP elevation within 3-D histomorphometric reconstructions. Invest Ophthalmol Vis Sci. 2009;50(12):5785–5799. | ||
Sigal IA, Ethier CR. Biomechanics of the optic nerve head. Exp Eye Res. 2009;88:799–807. | ||
Girard MJ, Suh JK, Bottlang M, Burgoyne CF, Downs JC. Biomechanical changes in the sclera of monkey eyes exposed to chronic IOP elevations. Invest Ophthalmol Vis Sci. 2011;52(8):5656–5669. | ||
Grytz R, Meschke G, Jonas JB. The collagen fibril architecture in the lamina cribrosa and peripapillary sclera predicted by a computational remodeling approach. Biomech Model Mechanobiol. 2011;10(3):371–382. | ||
Roberts MD, Liang Y, Sigal IA, et al. Correlation between local stress and strain and lamina cribrosa connective tissue volume fraction in normal monkey eyes. Invest Ophthalmol Vis Sci. 2010;51(1):295–307. | ||
Roberts MD, Sigal IA, Liang Y, Burgoyne CF, Downs JC. Changes in the biomechanical response of the optic nerve head in early experimental glaucoma. Invest Ophthalmol Vis Sci. 2010;51(11):5675–5684. | ||
Downs JC, Suh JK, Thomas KA, Bellezza AJ, Hart RT, Burgoyne CF. Viscoelastic material properties of the peripapillary sclera in normal and early-glaucoma monkey eyes. Invest Ophthalmol Vis Sci. 2005;46(2):540–546. | ||
Girard MJ, Downs JC, Bottlang M, Burgoyne CF, Suh JK. Peripapillary and posterior scleral mechanics – part II: experimental and inverse finite element characterization. J Biomech Eng. 2009;131(5):051012. | ||
Yan D, McPheeters S, Johnson G, Utzinger U, Vande Geest JP. Microstructural differences in the human posterior sclera as a function of age and race. Invest Ophthalmol Vis Sci. 2011;52(2):821–829. | ||
Pallikaris IG, Kymionis GD, Ginis HS, Kounis GA, Tsilimbaris MK. Ocular rigidity in living human eyes. Invest Ophthalmol Vis Sci. 2005;46(2):409–414. | ||
Glass DH, Roberts CJ, Litsky AS, Weber PA. A viscoelastic biomechanical model of the cornea describing the effect of viscosity and elasticity on hysteresis. Invest Ophthalmol Vis Sci. 2008;49(9):3919–3926. | ||
Shah S, Laiquzzaman M, Mantry S, Cunliffe I. Ocular response analyser to assess hysteresis and corneal resistance factor in low tension, open angle glaucoma and ocular hypertension. Clin Experiment Ophthalmol. 2008;36(6):508–513. | ||
Narayanaswamy A, Su DH, Baskaran M, et al. Comparison of ocular response analyzer parameters in chinese subjects with primary angle-closure and primary open-angle glaucoma. Arch Ophthalmol. 2011;129(4):429–434. | ||
Grise-Dulac A, Saad A, Abitbol O, et al. Assessment of corneal biomechanical properties in normal tension glaucoma and comparison with open-angle glaucoma, ocular hypertension and normal eyes. J Glaucoma. 2012;21(7):486–489. | ||
Anand A, De Moraes CG, Teng CC, Tello C, Liebmann JM, Ritch R. Corneal hysteresis and visual field asymmetry in open angle glaucoma. Invest Ophthalmol Vis Sci. 2010;51(12):6514–6518. | ||
Medeiros FA, Meira-Freitas D, Lisboa R, Kuang TM, Zangwill LM, Weinreb RN. Corneal hysteresis as a risk factor for glaucoma progression: a prospective longitudinal study. Ophthalmology. 2013;120(8):1533–1540. | ||
De Moraes CV, Hill V, Tello C, Liebmann JM, Ritch R. Lower corneal hysteresis is associated with more rapid glaucomatous visual field progression. J Glaucoma. 2012;21(4):209–213. | ||
Mansouri K, Leite MT, Weinreb RN, Tafreshi A, Zangwill LM, Medeiros FA. Association between corneal biomechanical properties and glaucoma severity. Am J Ophthalmol. 2012;153(3):419.e1–427.e1. | ||
Vu DM, Silva FQ, Haseltine SJ, Ehrlich JR, Radcliffe NM. Relationship between corneal hysteresis and optic nerve parameters measured with spectral domain optical coherence tomography. Graefes Arch Clin Exp Ophthalmol. 2013;251(7):1777–1783. | ||
Prata TS, Lima VC, de Moraes CG, et al. Factors associated with topographic changes of the optic nerve head induced by acute intraocular pressure reduction in glaucoma patients. Eye (Lond). 2011;25(2):201–207. | ||
Nongpiur ME, Png O, Chiew JW, et al. Lack of association between corneal hysteresis and corneal resistance factor with glaucoma severity in primary angle closure glaucoma. Invest Ophthalmol Vis Sci. 2015;56(11):6879–6885. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.