Back to Journals » Clinical Ophthalmology » Volume 13
In vitro demodicidal activity of commercial lid hygiene products
Authors Kabat AG
Received 18 March 2019
Accepted for publication 9 July 2019
Published 6 August 2019 Volume 2019:13 Pages 1493—1497
DOI https://doi.org/10.2147/OPTH.S209067
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Alan G Kabat
Pennsylvania College of Optometry, Salus University, Elkins Park, PA 19027, USA
Purpose: Demodex folliculorum is a ubiquitous mite that can infest the eyelash follicles. Two commercial lid hygiene products have asserted their effectiveness in killing Demodex mites, yet there has never been a comparative trial between these two products. This study evaluated the demodicidal activity of 0.01% hypochlorous acid (HOCl) solution (Avenova; NovaBay Pharmaceuticals; Emeryville, CA) and 4% terpinen-4-ol (T4O) solution (Cliradex, Bio-Tissue; Miami, FL) in comparison to mineral oil (MO), a negative control.
Methods: Live Demodex mites were obtained from volunteers. Samples were immersed in 1–2 drops of test solution: 0.01% HOCl, 4% T4O, or 100% MO. Samples were observed under the microscope every 10 mins for up to 90 mins. Kill time was defined as the elapsed time between the addition of test solution and all cessation of movement of the body, legs, mouth and pedipalps for a minimum of 60 seconds.
Results: T4O demonstrated a mean kill time of 40±0.0 mins. HOCl had a mean kill time of 87.86±4.23 mins, with 79% of samples surviving the full 90 mins. In the MO group, all samples survived through the 90 min mark. Kill time was statistically significant in favor of T4O as compared to HOCl (p=0.0005). There was no statistically significant difference in kill time between HOCl and MO (p=0.25).
Conclusion: 4% T4O effectively killed all adult mite samples within 40 mins of exposure. In contrast, the demodicidal activity of 0.01% HOCl was minimal, and comparatively similar to 100% MO.
Keywords: Demodex, blepharitis, lid hygiene, tea tree oil, terpinen-4-ol, hypochlorous acid
Introduction
Demodex is an obligatory parasite of the skin surface, which in large populations can cause a pathological response.1 In humans, only two species are known to inhabit the skin of the face and eyelids: Demodex folliculorum and Demodex brevis. Of these, D. folliculorum is more prevalent on the eyelids and eyelashes, while D. brevis resides deeper in the pilosebaceous and meibomian glands.2,3
Demodex infestation of the eyelids and eyelashes can result in a condition known as Demodex-associated blepharitis (DAB). The hallmark sign of DAB is the presence of “cylindrical dandruff” (CD) at the base of the lashes.3 Treatment options for DAB vary, but often include one or more of the following: mechanical debridement of the lid margins with a surfactant cleanser (microblepharoexfoliation); in-office treatment with highly concentrated tea tree oil, a known demodicidal agent; home therapy with lower concentrations of tea tree oil or derivatives thereof; and oral therapy with ivermectin (Stromectol, Merck & Company, Inc.; Kenilworth, NJ) over 2–4 weeks.4–6
Recently, promotional claims have been made regarding a lid hygiene product that contains 0.01% HOCl in non-preserved saline (Avenova, NovaBay Pharmaceuticals; Emeryville, CA) suggesting that it has acaricidal activity against Demodex mites. Statements made by notable thought leaders in ophthalmology and optometry include the following: “Laboratory studies show that [Avenova] effectively and rapidly kills the nymph form of the demodex mites … ” and “Avenova, a product from NovaBay Pharmaceuticals, safely kills the mites … ”.7,8 If true, the studies that validate these claims remain unpublished in the peer-reviewed literature.
The goal of this study was to determine the ability of 0.01% HOCl solution to kill Demodex folliculorum in vitro as compared to 4% T4O solution (Cliradex, Bio-Tissue; Miami, FL), and also 100% mineral oil, which is known to be innocuous.
Materials and methods
Materials
Test solutions consisted of 0.01% HOCl, 4% T4O and 100% mineral oil. The HOCl solution was purchased directly from NovaBay Pharmaceuticals (Emeryville, CA, USA). Likewise, the T4O solution was obtained directly from Bio-Tissue (Doral, FL, USA). While this product is only commercially available as a saturated facial wipe, the company was able to provide 20 mL of solution upon request. The mineral oil was obtained from Wal-Mart Stores, Inc. (Bentonville, AR, USA).
Patients
This study followed the tenets of The Helsinki Declaration and was approved by the Institutional Review Board at Southern College of Optometry (Memphis, TN). Subjects ≥18 years of age were recruited from a large optometric facility; those with CD and symptoms consistent with DAB were screened and provided written informed consent. These symptoms are delineated in Table 1. Individuals using any form of lid hygiene for blepharitis or any known demodicidal agents were excluded.
 |
Table 1 Common symptoms of Demodex-associated blepharitis |
Collection and counting of Demodex mites
Patients were positioned at the slit lamp. Lashes with CD were isolated and, using a rotational technique, extracted with forceps, being careful to preserve the lash follicle. Up to 4 lashes were extracted from each subject. The samples were transferred to glass slides and immersed in 1–2 drops of test solution (ie HOCl, T4O or MO) before applying a cover slip. The slides were examined under a microscope (at magnifications ranging from 40–100X) every 10 mins for up to 90 mins. The kill time was defined as the elapsed time between the addition of test solution and all cessation of movement of the body, legs, mouth and pedipalps for a minimum of 60 seconds. Additionally, maximum survival time was calculated for two of the samples in which mites remained viable beyond the 90 min mark (one from the HOCl group and one from the MO group). These samples were observed at intervals of 1–14 hrs after the conclusion of the original 90 min protocol. The maximum survival time was defined as the final time point at which movement of the mites’ body parts was observed.
Because Demodex at an earlier stage of life may be more vulnerable, only adult Demodex folliculorum with four pairs of well-developed legs were counted. Also, because Demodex mites buried in CD might delay or preclude the killing effect, only organisms that were fully exposed to the test solution were considered. The total number of live, adult, exposed mites per slide was calculated and recorded.
Results
In vitro killing effect
In total, 35 viable adult Demodex mites were recovered from 3 human subjects with blepharitis and CD (mean/sample =3.89) (Figures 1 and 2). The observation was performed 3 times for each of the 3 test solutions, on different days (total trials =9). The results of these series are shown in Table 2.
 |
Table 2 In vitro kill time for Demodex in test solution |
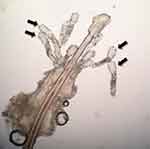 |
Figure 1 Demodex folliculorum mites on a lash follicle (black arrows). |
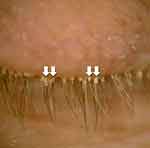 |
Figure 2 Demodex-associated blepharitis with classic cylindrical dandruff (white arrows). |
Mean kill time for Demodex exposed to T4O was 40±0.0 mins. In contrast, HOCl had a mean kill time of 87.86±4.23 mins, with 79% of the exposed mites surviving the full 90 mins of observation. As expected, MO was found to have no effect on Demodex, with all mites surviving through 90 mins. Kill time was statistically significant in favor of T4O as compared to HOCl (p=0.0005). There was no statistically significant difference in kill time between HOCl and MO (p=0.25).
Maximum survival time
In five of the slide samples, Demodex mites survived through the 90 min mark. Two of these slide samples were randomly selected for continued observation over time, including one from the HOCl group and one from the MO group. The maximum survival time noted for mites exposed to HOCl (n=9) was 210 mins (3.5 hrs). The maximum survival time noted for mites exposed to mineral oil (n=3) was 1,470 mins (24.5 hrs). This information is displayed graphically in Figure 3.
 |
Figure 3 Maximum survival time for Demodex folliculorum mites by direct observation in test solutions. |
Discussion
The present study confirms that 4% T4O solution has the capacity to neutralize adult Demodex mites in vitro. No tested samples survived longer than 40 mins in T4O. In contrast, the majority of adult Demodex mites (79%) exposed to HOCl survived for the entire study duration of 90 mins. Additionally, a sub-group of mites that was observed for a longer period of time was found to be viable for up to 3.5 hrs. Statistically, 0.01% HOCl was no more effective against Demodex mites in vitro than 100% MO, which is known to be innocuous to these organisms.4
In 2005, Gao and associates demonstrated that tea tree oil was effective at killing Demodex mites in a concentration-dependent fashion.4 100% tea tree oil was found to have the most rapid kill time when compared to all other treatments, including 100% caraway oil, 100% alcohol, 10% povidone-iodine and 4% pilocarpine.4 In 2013, Tighe and associates concluded that T4O is the most active chemical component of tea tree oil to kill Demodex mites, demonstrating even more rapid kill times than tea tree oil at similar concentrations.5 Even a dilution of 1% T4O was able to kill Demodex mites in <90 mins, whereas 10% tea tree oil could not achieve this outcome.5 Presently in the United States, Cliradex is the only commercially available product specifically designed for ophthalmic use (ie lid hygiene) that contains T4O.
HOCl has been noted to kill a range of bacteria as well as some fungal pathogens, including Staphylococcus aureus, Staphylococcus capitis, Staphylococcus epidermidis, Pseudomonas aeruginosa, Escherichia coli, Enterococcus faecalis and Candida albicans. The antimicrobial efficacy of HOCl appears to be pH-dependent, with more acidic formulations yielding faster rates of eradication.9–11 Despite this evidence, a recent in vitro study that assessed the bactericidal activity of 0.01% HOCl showed persistent growth of S. aureus, Streptococcus pneumoniae and Haemophilus influenza at serial dilutions of 1:20, 1:400 and 1:1600.12 With regard to the demodicidal properties of HOCl, there has been no published research to date.
One potential shortcoming of this study was found to be the stability of 0.01% HOCl. As saline-based solution, this product demonstrated a much more rapid rate of evaporation from the test slides as compared to 4% T4O or 100% mineral oil, which are both lipid-based. In the one HOCl test slide that showed a kill time of 80 mins, it was observed that the solution had completely evaporated beneath the cover slip. Hence, it was impossible to determine if the Demodex mites had ceased movement due to lethal effects of the solution or simply due to dehydration.
Along these same lines, the addition of test solution prior to mounting the cover slip may have resulted in displacement of some nonadherent Demodex mites, resulting in undercounting of the organisms. This limitation may be corrected in future investigations by developing a method to add test solutions to the slide after the cover slip has been secured in place.
Finally, it must be acknowledged that the sample size for Demodex mites in this study was relatively small. A larger number of mites would likely have produced more stable results. This should be addressed in future investigations.
Despite these issues, the clinical relevance of this study is substantial. Demodex has been implicated in a number of chronic disorders of the eyelids and ocular surface.13–22 Practitioners need to recognize the presence of Demodex in patients with ocular disorders and employ an effective therapy that both limits their numbers and controls their pathogenicity. Tea tree oil and derivatives thereof, such as T4O, have consistently demonstrated in vitro efficacy in killing Demodex mites. As new commercial lid hygiene products enter the market, their efficacy against Demodex must be ascertained, and claims must be substantiated by scientific evidence.
Acknowledgments
The author wishes to thank Hosam Sheha, MD, PhD for his insights and assistance with the statistical analysis of this study. Financial support for this study was provided by TissueTech, Inc. This data set and its conclusions were originally presented as a poster at the Association for Research in Vision and Ophthalmology (ARVO) Annual Meeting 2018. The poster’s abstract was published in Investigative Ophthalmology and Visual Science, July 2018, Vol. 59, 905, https://iovs.arvojournals.org/article.aspx?articleid=2693620.
Disclosure
Dr Alan G Kabat is a consultant for Bio-Tissue, Inc. Dr Alan G Kabat reports non-financial support from TissueTech, Inc., during the conduct of the study; personal fees from Avellino Labs, Bio-Tissue, Inc., Bruder Healthcare Company, Inc., Lacrivera, OCuSOFT, Inc., Shire, Sight Sciences, Sun Pharmaceuticals, Vmax Vision, Kala Pharmaceuticals, Inc., National Vision Administrators, LLC, Quidel, TearScience and EyeGate Pharma, outside the submitted work. The author reports no other conflicts of interest in this work.
References
1. Czepita D, Kuźna-Grygiel W, Kosik-Bogacka D. Investigations on the occurrence as well as the role of Demodex folliculorum and Demodex brevis in the pathogenesis of blepharitis. Klin Oczna. 2005;107(1–3):80–82.
2. Liu J, Sheha H, Tseng SCG. Pathogenic role of Demodex mites in blepharitis. Curr Opin Allergy Clin Immunol. 2010;10(5):505–510. doi:10.1097/ACI.0b013e32833df9f4
3. Gao YY, Di Pascuale MA, Li W, et al. High prevalence of Demodex in eyelashes with cylindrical dandruff. Invest Ophthalmol Vis Sci. 2005;46(9):3089–3094. doi:10.1167/iovs.05-0275
4. Gao YY, Di Pascuale MA, Li W, et al. In vitro and in vivo killing of ocular Demodex by tea tree oil. Br J Ophthalmol. 2005;89(11):1468–1473. doi:10.1136/bjo.2005.072363
5. Tighe S, Gao YY, Tseng SC. Terpinen-4-ol is the most active ingredient of tea tree oil to kill demodex mites. Transl Vis Sci Technol. 2013;2(7):2. doi:10.1167/tvst.2.7.2
6. Holzchuh FG, Hida RY, Moscovici BK, et al. Clinical treatment of ocular Demodex folliculorum by systemic ivermectin. Am J Ophthalmol. 2011;151(6):1030–1034.e1. doi:10.1016/j.ajo.2010.11.024
7. Donnenfeld E. Fighting the mites that live on eyelids. Summer. 2015. Available from: http://avenova.com/wp-content/uploads/2016/06/2-fighting-the-mites-that-live-on-eyelids-eric-donnenfeld1.pdf. Accessed May 2, 2017.
8. Hauser W Fighting the mites that live on your eyelids; April 25, 2017. Available from: https://www.linkedin.com/pulse/fighting-mites-live-your-eyelids-whitney-hauser.
9. Ono T, Yamashita K, Murayama T, Sato T. Microbicidal effect of weak acid hypochlorous solution on various microorganisms. Biocontrol Sci. 2012;17(3):129–133.
10. Romanowski EG, Stella NA, Yates KA, et al. In vitro evaluation of a hypochlorous acid hygiene solution on established biofilms. Eye Contact Lens. 2018. [Epub ahead of print]. doi:10.1097/ICL.0000000000000456
11. Wang L, Bassiri M, Najafi R, et al. Hypochlorous acid as a potential wound care agent: part I. Stabilized hypochlorous acid: a component of the inorganic armamentarium of innate immunity. J Burns Wounds. 2007;11(6):e5.
12. Grob SR, Shumate K, Booth DG, MacQuaid EP. Bactericidal effect of hypochlorous acid in blepharitis. Am Acad Ophthalmol 2017.2017;Abstract PO051. New Orleans, LA.
13. de Venecia AB
14. Liang L, Ding X, SC T. High prevalence of demodex brevis infestation in chalazia. Am J Ophthalmol. 2014;157(2):342–348.e1. doi:10.1016/j.ajo.2013.09.031
15. Tarkowski W, Moneta-Wielgoś J, Młocicki D. Demodex sp. as a potential cause of the abandonment of soft contact lenses by their existing users. Biomed Res Int. 2015;2015:259109. doi:10.1155/2015/259109
16. Liu J, Sheha H, Tseng SC. Pathogenic role of Demodex mites in blepharitis. Curr Opin Allergy Clin Immunol. 2010;10(5):505–510. doi:10.1097/ACI.0b013e32833df9f4
17. Kheirkhah A, Casas V, Li W, et al. Corneal manifestations of ocular demodex infestation. Am J Ophthalmol. 2007;143(5):743–749. doi:10.1016/j.ajo.2007.01.054
18. Tarkowski W, Owczyńska M, Błaszczyk-Tyszka A, Młocicki D. Demodex mites as potential etiological factor in chalazion - a study in Poland. Acta Parasitol. 2015;60(4):777–783. doi:10.1515/ap-2015-0110
19. Li Y, Kim GE, Yoon KC, Choi W. First report of palpebral conjunctival inflammatory nodule associated with Demodex species. Indian J Ophthalmol. 2018;66(9):1365–1367. doi:10.4103/ijo.IJO_375_18
20. Tarkowski W, Moneta-Wielgoś J, Młocicki D. Do Demodex mites play a role in pterygium development? Med Hypotheses. 2017;98:6–10. doi:10.1016/j.mehy.2016.09.003
21. Zhang XB, Ding YH, He W. The association between demodex infestation and ocular surface manifestations in meibomian gland dysfunction. Int J Ophthalmol. 2018;11(4):589–592. doi:10.18240/ijo.2018.04.08
22. Luo X, Li J, Chen C, Tseng S, Liang L. Ocular demodicosis as a potential cause of ocular surface inflammation. Cornea. 2017;36(Suppl 1):S9–S14. doi:10.1097/ICO.0000000000001361
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
