Back to Journals » Journal of Asthma and Allergy » Volume 14
In vitro Culture with Cytokines Provides a Tool to Assess the Effector Functions of ILC2s in Peripheral Blood in Asthma
Authors Drake LY, Bartemes KR, Bachman KA, Hagan JB, Kita H
Received 14 October 2020
Accepted for publication 10 December 2020
Published 11 January 2021 Volume 2021:14 Pages 13—22
DOI https://doi.org/10.2147/JAA.S286695
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Amrita Dosanjh
Li Y Drake,1,2 Kathleen R Bartemes,1,3 Kay A Bachman,1 John B Hagan,1 Hirohito Kita1,4
1Division of Allergic Diseases and Department of Medicine, Mayo Clinic, Rochester, MN, USA; 2Department of Anesthesiology and Perioperative Medicine, Mayo Clinic, Rochester, MN, USA; 3Department of Otorhinolaryngology, Mayo Clinic, Rochester, MN, USA; 4Division of Allergy, Asthma, and Clinical Immunology and Department of Medicine, Mayo Clinic Arizona, Scottsdale, AZ, USA
Correspondence: Hirohito Kita
Division of Allergy, Asthma, and Clinical Immunology and Department of Medicine, Mayo Clinic Arizona, Scottsdale, AZ 85259, USA
Tel +1 480 301 9616
Fax +1 480 301 7017
Email [email protected]
Background: Group 2 innate lymphoid cells (ILC2s) play crucial roles in type 2 immunity and asthma development. While ILC2s are resident in mucosal tissues, they also circulate in peripheral blood. It remains controversial whether ILC2s are increased in the peripheral blood of patients with asthma.
Purpose: The goal of this project was to study the effector functions of ILC2s in peripheral blood samples by in vitro culture with cytokines.
Patients and Methods: Peripheral blood mononuclear cells (PBMCs) were collected from 11 adult patients with mild asthma and 12 healthy control subjects. The number of peripheral blood ILC2s in PBMCs was analyzed by flow cytometry. PBMCs were cultured with IL-33 and IL-25 without any antigens, and the amounts of type 2 cytokines in cell-free supernatants were analyzed by ELISA. In selected experiments, production of cytokines by ILC2s was analyzed by intracellular cytokine staining and flow cytometry.
Results: In response to either IL-33 or IL-25 stimulation, PBMCs from patients with mild asthma produced larger amounts of IL-5 and IL-13 than PBMCs from healthy control subjects. However, ILC2 numbers or proportions were not significantly different between these two groups. Flow cytometric analysis confirmed production of IL-5 by ILCs when stimulated with IL-33.
Conclusion: In vitro culture of PBMCs with a cocktail of cytokines, such as either IL-33 or IL-25 plus IL-2, may provide a valuable tool to assess the effector functions of ILC2s and may serve as a biomarker for human asthma.
Keywords: IL-33, IL-25, IL-5, IL-13
Introduction
Group 2 innate lymphoid cells (ILC2s) play important roles in type 2 immune responses and allergic airway diseases.1 In response to epithelium-derived cytokines, such as interleukin (IL)-33 and IL-25, ILC2s produce type 2 cytokines, including IL-5 and IL-13, and other mediators involved in inflammation and tissue remodeling.1 ILC2s are mostly mucosal tissue-resident cells with a small number of cells in circulation.1 Human ILC2s have been identified in tonsils, lungs, nasal tissues, bronchoalveolar lavage (BAL), induced sputum, and peripheral blood.2–5 Studies show that ILC2 numbers in BAL fluid and induced sputum are increased in asthma patients.3–5 However, the results of studies that examined ILC2 numbers in the peripheral blood of asthma patients have been inconsistent. Some studies, including our own, found increased numbers of circulating ILC2s in asthma patients,5–8 whereas other studies revealed no significant differences in circulating ILC2s between asthmatic and non-asthmatic control subjects.4,9,10
Peripheral blood would provide a versatile non-invasive source for the assessment of disease phenotypes and endotypes. The complexity of ILC2 phenotypes that overlap with those of other ILC groups makes it challenging to enumerate them precisely.8 Therefore, the goal of this project was to characterize the effector functions of ILC2s in peripheral blood samples by straightforward in vitro culture with cytokines. Using this approach, we found that although peripheral blood ILC2 numbers were not significantly different between mild asthma patients and healthy control subjects, peripheral blood mononuclear cells (PBMCs) from mild asthma patients produced stronger type 2 cytokine responses in vitro as compared to PBMCs from healthy control subjects.
Patients and Methods
Study Subjects
Peripheral blood was obtained from normal subjects or subjects with mild allergic asthma. Inclusion criteria for asthma patients were as follows: 1) a pre-bronchodilator forced expiratory volume in one second (FEV1) >60% of predicted normal value with a post-bronchodilator increase in flow >12% and/or a reduction in FEV1 >20% following 1–5 breaths of 25 mg/dl inhaled methacholine, 2) physician-diagnosed asthma, 3) allergic sensitization to aeroallergens confirmed by new or existing skin testing or IgE serum immunoassays, and 4) the absence of nasal polyps. Exclusion criteria for study subjects were as follows: 1) systemic glucocorticoid therapy during the past 3 months, 2) previous (>10 pack year smoking history) or current smokers, 3) a lower or upper airway infection (eg, “cold”) within the last 4 weeks, 4) diagnosed with immunodeficiency, cystic fibrosis, or any other respiratory or general medical problem which the investigator felt would preclude inclusion into the study, eg, infection, malignancy, 5) immunotherapy with any allergen, 6) pregnancy, and 7) immunosuppressive medication. The Mayo Clinic Institutional Review Board approved this study, and informed consent was obtained from all participants. This study complies with the Declaration of Helsinki.
Reagents
FITC-labeled antibodies to CD3 (SK7), CD14 (MϕP9), CD19 (4G7), CD34 (581), and CD56 (NCAM16.2) were purchased from BD Biosciences. FITC-labeled antibodies to CD11c (3.9), CD94 (DX22), CD123 (6H6), and FcεRIα (AER-37) were purchased from eBioscience. PE-labeled antibody to CD127 (hIL7R-m21) was purchased from BD Biosciences. PE-labeled antibody to IL-5 (TRFK5) was purchased from BioLegend. AF647-labeled antibody to CRTH2 (BM16) and BV510-labeled antibody to CD45 (HI30) were purchased from Biolegend. Recombinant IL-33 and IL-12 were purchased from R&D Systems. Recombinant IL-25 was purchased from Biolegend. Recombinant human IL-2 was purchased as Proleukin (aldesleukin) from Novartis Pharmaceuticals Corporation.
PBMC Isolation and Stimulation for Cytokine Production
Heparinized peripheral blood was layered over an equal volume of Histopaque 1077 (Sigma-Aldrich) and centrifuged according to the manufacturer’s instructions. Mononuclear cells were collected from the interface between the Histopaque and the plasma. PBMCs were washed and resuspended in RPMI 1640 media (Gibco/Life Technologies) containing 10% heat-inactivated human AB serum (Sigma-Aldrich), penicillin (100 U/mL), and streptomycin (100 µg/mL; Gibco/Life Technologies). Cells were cultured in 96-well round-bottom plates (5 × 105 cells/well) at 37°C with 5% CO2 for 7 days with IL-2 (20 U/mL), IL-33 or IL-25 at the indicated concentrations (0.5–50 ng/mL), and IL-12 (5 ng/mL). IL-5, IL-13, and IFN-γ concentrations in the cell-free supernatants were determined using ELISA kits as recommended by the manufacturer (Thermo Fisher Scientific).
ILC2 Analysis by Flow Cytometry
PBMCs were stained with antibodies to lineage markers (CD3, CD11c, CD14, CD19, CD34, CD56, CD94, CD123, and FcεRIα) and antibodies to ILC2 surface markers CD45, CD127, and CRTH2. After staining, cells were washed and fixed with 1% paraformaldehyde and analyzed using flow cytometry. Conventional ILC2s were defined as the Lineage−CD45+CD127+CRTH2+ population. Alternatively, nonconventional ILC2s were identified as the Lineage−CD45+CD127+CRTH2− population. The absolute numbers of ILC2s were calculated by multiplying the total number of PBMCs per milliliter of blood by the ILC2 percentage of a given sample. For IL-5 intracellular staining experiments, PBMCs were cultured in 6-well plates (3 x107/well) for 4 days with IL-2 (20 U/mL) and IL-33 (50ng/mL). Brefeldin A (GolgiPlug, BD Biosciences) was added for the final 3 hours. Cells were then stained with fixable viability dye eFluor 780, FITC-labeled antibodies to lineage markers, BV421-labeled antibody to CD127; fixed and permeabilized with a Cytofix/Cytoperm kit (BD Biosciences), and stained intracellularly with a PE-labeled antibody to IL-5.
Statistical Analyses
Data are presented as the mean ± SEM for the numbers of subjects as indicated. Differences were evaluated using paired Student’s t-tests, and p < 0.05 was considered statistically significant.
Results
Blood ILC2 Numbers are Similar in Mild Asthma Patients and Healthy Controls
Previously, we reported that the number of ILC2s and IL-33 or IL-25-induced type 2 cytokine production were increased in PBMCs from patients with allergic asthma.6 The caveats of that study were that the numbers and functions of ILC2s were analyzed in different subject populations. Furthermore, asthma patients with various degrees of disease severity were included in the study, making it difficult to rule out the potential influence of the medications, such as systemic glucocorticoids, especially in patients with severe asthma. To overcome these potential problems from the previous study, we designed the current study to simultaneously assess the quantity and function of peripheral blood ILC2s from patients with mild asthma. PBMCs were obtained from 11 adult patients with mild asthma and 12 healthy control subjects (Table 1). The definition of mild asthma followed the National Asthma Education and Prevention Program Expert Panel Report 3 guideline.11 To assess the numbers of circulating blood ILC2s, we stained PBMCs with antibodies to CD45, CRTH2, CD127 and lineage markers including CD3, CD14, CD16, CD19, CD56, FcεRIα, CD11c, CD34, CD94, and CD123 (Figure 1A). The conventional ILC2s were identified as Lin−CD45+CRTH2+CD127+. The nonconventional ILCs were identified as Lin−CD45+CRTH2−CD127+. When analyzed by flow cytometry, both control subjects and asthma patients showed considerable heterogeneity in ILC2 numbers within each study population (Figure 1B and C). There was no significant difference in the proportions of conventional ILC2s within the PBMC preparations or absolute numbers of conventional ILC2s in peripheral blood between control subjects and asthma patients (Figure 1C and D). No difference was observed in absolute numbers of the Lin−CD45+CD127+CRTH2− ILC population (Figure 1E).
 |
Table 1 Study Subjects |
PBMCs from Mild Asthma Patients Produce More IL-5 After IL-33 and IL-25 Stimulation
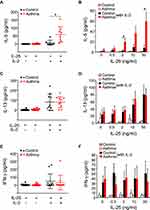
Next, we analyzed the effector functions of blood ILC2s by culturing freshly isolated PBMCs with IL-33 and IL-25 as described previously.6 When cultured with IL-33 or IL-25 alone, PBMCs from asthma patients or control subjects produced minimal amounts of IL-5, IL-13, and interferon (IFN)-γ (Figures 2 and 3). In control subjects, the addition of IL-2 to IL-33 or IL-25 modestly enhanced the ILC2 response, and the levels of IL-5 became detectable in some subjects (Figures 2A and B and 3A and B); greater amounts of IL-13 were detected in the majority of these control subject samples (Figures 2C and D and 3C and D). In asthma patients, this added IL-2 also enhanced the ILC2 response to IL-33 or IL-25 (Figures 2A–D and 3A–D). Importantly, significantly greater amounts of IL-5 were produced by asthma patient samples compared to control subject samples following stimulation with IL-33 plus IL-2 (Figure 2A and B, p < 0.05). Robust differences were observed upon stimulation with higher concentrations of IL-33. Similarly, greater amounts of IL-5 were produced in asthma patient samples than in control subject samples following stimulation of the PBMCs with IL-25 plus IL-2 (Figure 3A and B, p< 0.05). Although IL-13 levels in asthma patient samples trended higher, they were not significantly different from the IL-13 levels in healthy control samples when PBMCs were stimulated with IL-33 plus IL-2 or with IL-25 plus IL-2 (Figures 2C and D and 3C and D). Production of IFN-γ was minimally affected by the addition of IL-2 to IL-33 or IL-25, and no differences were observed between the subject groups (Figures 2E and F and 3E and F). Together these data indicate that PBMCs from patients with mild asthma produce more IL-5 in response to IL-33 or IL-25 stimulation in vitro.
IL-12 Inhibits IL-33-Induced Type 2 Cytokine Production by PBMCs
Recent studies in mice suggest that the effector functions of ILC2s are suppressed by several cytokines, including IFN-γ and IL-27.12 IL-12 is produced by innate immune cells and plays a major role in linking innate and adaptive type 1 immunities.13 IL-12 has been shown to inhibit IL-2/IL-33/TSLP-induced type 2 immune response by transdifferentiating healthy blood ILC2s into IFN-γ-producing ILC1s.14,15 We examined whether IL-12 has inhibitory effects on IL-33-induced type 2 cytokine responses in asthmatic PBMCs. In the presence of IL-2 and IL-33, IL-12 stimulated similar levels of IFN-γ production by PBMCs from both control subjects and asthma patients (Figure 4A). Addition of IL-12 nearly abolished IL-5 production by PBMCs from asthma patients when PBMCs were stimulated with IL-33 plus IL-2 (Figure 4B). These data suggest that the innate type 2 immune response in asthma patients may be suppressed by IL-12.
ILC2s are a Major Source for IL-5 Production in PBMCs
Besides ILC2s, the IL-33 receptor is also expressed by many other immune cells, including T lymphocytes, natural killer cells, and monocytes.16 These cells are present in PBMCs and potentially can produce type 2 cytokines upon IL-33 stimulation. Previously, we showed that a FACS-sorted Lin−CD127+CRTH2+ cell population but not a Lin+ population in human peripheral blood produced and released large amounts of type 2 cytokines upon stimulation with IL-33 plus IL-2 in vitro.6 To further verify that ILC2s are likely a major source of IL-5 when PBMCs are stimulated with IL-33 plus IL-2 in vitro, we performed IL-5 intracellular staining on IL-33-stimulated whole asthmatic PBMCs and analyzed IL-5 expression by flow cytometry. The fixation and permeabilization in the intracellular staining procedures diminished CRTH2 antibody staining in our hands. Thus, we analyzed IL-5 expression gating on Lin+ cells and Lin−CD127+ cells that likely included both conventional and nonconventional ILC2s. We found very few IL-5+ cells in the Lin+ population (less than 0.05%) and more than 30% IL-5+ cells in the Lin−CD127+ population, suggesting that Lin−CD127+ ILCs are likely a major source of IL-5 production when asthmatic PBMCs are stimulated with IL-33 plus IL-2 in vitro (Figure 5).
Discussion
Several studies have explored the possibility of using blood ILC2 number as a biomarker for asthma.4–10 So far, the results have not been consistent. Whereas some studies showed that blood ILC2 numbers were increased in asthmatic patients,5–8 other studies revealed no changes in blood ILC2 numbers.4,9,10 This discrepancy may be caused by differences in patient population selection criteria, including the severity of asthma and usage of medication, and differences in flow cytometry gating strategies. Our group previously showed that asthmatic patients had increased numbers of blood ILC2s.6 The current study shows that the blood ILC2 numbers are similar in asthma patients and healthy control subjects when only mild asthma patients are included and a more stringent lineage gating strategy is applied. In contrast to the variability of ILC2 numbers, we consistently observed in this study that IL-33- or IL-25-induced type 2 cytokine production by PBMCs (likely circulating ILC2s) is upregulated in asthma patients, even in those with mild asthma. These data suggest that a functional assay may better reflect a clinical phenotype. Notably, in both our previous study and in the current study, IL-5 levels, but not IL-13 levels, were significantly upregulated in asthmatic PBMCs,6 suggesting that IL-33- or IL-25-induced IL-5 production is a more sensitive biomarker as compared to IL-13 production. Similar findings have been reported by another study.5 These data also indicate that IL-5 and IL-13 signaling pathways maybe differentially regulated in asthmatic PBMCs.
There are several limitations to our study that must be considered. First, this study is comparatively preliminary due to its relatively small sample size with 11 asthma patients and 12 control subjects. Second, both our previous study and this current study showed that ILC2s were the main cellular source for IL-33-induced IL-5 production in PBMC cultures.6 However, we cannot completely exclude the possibility that other cell types within the PBMC specimens may influence cytokine production by ILC2s. We recognize that cytokine intracellular staining would provide more convincing evidence that the increased IL-5 levels in asthmatic PBMCs are derived from blood ILC2s. However, ELISA assays are more streamlined and applicable for future large clinical studies. Third, because of its rather small sample size, this study was not powered sufficiently to detect potential sex difference and influences of asthma medications, variables which should be addressed in future larger studies. Nonetheless, our study provides good evidence that the straightforward in vitro culture of PBMCs with a cocktail of cytokines, such as either IL-33 or IL-25 plus IL-2, may be a valuable tool for assessing the effector functions of ILC2s and may serve as a biomarker for human asthma.
Conclusion
We have confirmed our previous finding that the innate type 2 cytokine response is increased in the peripheral blood of asthma patients. In a rigorously defined population of subjects with mild asthma, we found that the numbers of circulating ILC2s are not significantly different from healthy controls. Overall, our results suggest that the function, but not the number, of peripheral blood ILC2s may serve as a useful biomarker for human asthma.
Acknowledgments
This work was supported by a grant from the National Institutes of Health (R01 HL117823) and the Mayo Foundation.
Disclosure
Dr. Drake has nothing to disclose. Dr. Bartemes reports grants from the NIH during the conduct of the study. Ms. Bachman has nothing to disclose. Dr. Hagan serves as the local PI on a multicenter Astra-Zeneca clinical nasal polyp trial. Dr. Kita reports grant support from NIH, NHLBI and NIAID during the conduct of the study. The authors report no other potential conflicts of interest in this work.
References
1. Vivier E, Artis D, Colona M, et al. Innate lymphoid cells: 10 years on. Cell. 2018;174:1054–1066.
2. Mjosberg JM, Trifari S, Crellin NK, et al. Human IL-25- and IL-33-responsive type 2 innate lymphoid cells are defined by expression of CRTH2 and CD161. Nat Immunol. 2011;12:1055–1062.
3. Christianson CA, Goplen NP, Zafar I, et al. Persistence of asthma requires multiple feedback circuits involving type 2 innate lymphoid cells and IL-33. J Allergy Clin Immunol. 2015;136:59–68.
4. Nagakumar P. Type 2 innate lymphoid cells in induced sputum from children with severe asthma. J Allergy Clin Immunol. 2016;137:624–626.
5. Smith SG, Chen R, Kjarsgaard M, et al. Increased numbers of activated group 2 innate lymphoid cells in the airways of patients with severe asthma and persistent airway eosinophilia. J Allergy Clin Immunol. 2016;137:75–86.
6. Bartemes KR, Kephart G, Fox SJ, Kita H. Enhanced innate type 2 immune response in peripheral blood from patients with asthma. J Allergy Clin Immunol. 2014;134:671–678.
7. Miao Q, Wang Y, Liu YG, et al. Seasonal variation in circulating group 2 innate lymphoid cells in mugwort-allergic asthmatics during and outside pollen season. Allergy Asthma Clin Immunol. 2018;14:6.
8. Liu S, Sirohi K, Verma M, et al. Optimal identification of human conventional and nonconventional (CRTH2(-) IL7Ralpha (-)) ILC2s using additional surface markers. J Allergy Clin Immunol. 2020;146:390–405.
9. Barnig C, Cernadas M, Dutile S, et al. Lipoxin A4 regulates natural killer cell and type 2 innate lymphoid cell activation in asthma. Sci Transl Med. 2013;5:174ra26.
10. Maggi L. Human circulating group 2 innate lymphoid cells can express CD154 and promote IgE production. J Allergy Clin Immunol. 2017;139:964–976.
11. Guideline for the Diagnosis and Management of Asthma. U.S. Department of health and human serviced. National Institute Health, National Heart, Lung, Blood Institue. 2007. NIH Publication Number 08-5846.
12. Kabata H, Moro K, Koyasu S. The group 2 innate lymphoid cell (ILC2) regulatory network and its underlying mechanisms. Immunol Rev. 2018;286:37–52.
13. Vignali DA, Kuchroo VK. IL-12 family cytokines: immunological playmakers. Nat Immunol. 2012;13:722–728.
14. Bal SM, Bernink JH, Nagaswa M, et al. IL-1beta, IL-4 and IL-12 control the fate of group 2 innate lymphoid cells in human airway inflammation in the lungs. Nat Immunol. 2016;17:636–645.
15. Ohne Y, Silver J, Thompson-Snipes L, et al. IL-1 is a critical regulator of group 2 innate lymphoid cell function and plasticity. Nat Immunol. 2016;17:646–655.
16. Drake LY, Kita H. IL-33: biological properties, functions, and roles in airway disease. Immunol Rev. 2017;278:173–184.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.