Back to Journals » Clinical Interventions in Aging » Volume 15
Impact of Hearing Loss on Geriatric Assessment
Authors Völter C, Götze L , Dazert S, Wirth R , Thomas JP
Received 14 September 2020
Accepted for publication 22 November 2020
Published 30 December 2020 Volume 2020:15 Pages 2453—2467
DOI https://doi.org/10.2147/CIA.S281627
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Christiane Völter,1 Lisa Götze,1 Stefan Dazert,1 Rainer Wirth,2 Jan Peter Thomas1
1Department of Otorhinolaryngology, Head and Neck Surgery, Ruhr-University Bochum, St. Elisabeth-Hospital, Bochum 44787, Germany; 2Department of Geriatric Medicine, Ruhr-University Bochum, Marien Hospital Herne, Herne 44625, Germany
Correspondence: Christiane Völter
Department of Otorhinolaryngology, Head and Neck Surgery, Ruhr-University Bochum, St. Elisabeth-Hospital, Bleichstraße 15, Bochum 44787, Germany
Tel +49 234 509 8390
Email [email protected]
Background: Due to the aging society, the incidence of age-related hearing loss (ARHL) is strongly increasing. Hearing loss has a high impact on various aspects of life and may lead to social isolation, depression, loss of gain control, frailty and even mental decline. Comorbidity of cognitive and sensory impairment is not rare. This might have an impact on diagnostics and treatment in the geriatric setting.
Objective: The aim of the study was to evaluate the impact of hearing impairment on geriatric assessment and cognitive testing routinely done in geriatrics.
Material and Methods: This review is based on publications retrieved by a selective search in Medline, including individual studies, meta-analyses, guidelines, Cochrane reviews, and other reviews from 1960 until August 2020.
Results: Awareness of sensory impairment is low among patients and health professionals working with elderly people. The evaluation of the hearing status is not always part of the geriatric assessment and not yet routinely done in psychiatric settings. However, neurocognitive testing as an important part can be strongly influenced by auditory deprivation. Misunderstanding of verbal instructions, cognitive changes, and delayed central processes may lead to a false diagnosis in up to 16% of subjects with hearing loss. To minimize this bias, several neurocognitive assessments were transformed into non-auditory versions recently, eg the most commonly used Hearing-Impaired Montreal Cognitive Assessment (HI-MoCA). However, most of them still lack normative data for elderly people with hearing loss.
Conclusion: Hearing loss should be taken into consideration when performing geriatric assessment and cognitive testing in elderly subjects. Test batteries suitable for ARLH should be applied.
Keywords: age-related hearing loss, cognitive screening, dementia, geriatric assessment
Introduction
Prevalence of Hearing Loss
According to the WHO, normal hearing is defined as an averaged hearing threshold of 25dB or less in the frequencies 0.5, 1, 2 and 4kHz in the better hearing ear. Currently, 466 million people worldwide are affected by disabling hearing loss; by 2050 this number will even double.1 Common causes are ototoxic medication, head injuries, a sudden loss or an inflammation such as meningitis. But also age itself accelerates the decline of hearing loss. After the age of 60, hearing thresholds decline each year by 1dB even in subjects without otological problems (ISO/CD 7029:2014).2 30% of men and 20% of women have a disabling hearing loss of at least 40dB in the better hearing ear, and this number rises up to 55 in sexagenarians and 45% in octogenarians.3 Hearing loss is the third most common disabling chronic disease.4
Despite its high prevalence, hearing loss is frequently undetected and untreated in elderly subjects. A large Canadian study on 3964 subjects aged 40 to 79 years revealed that 54% of the study population had at least a mild hearing loss, but only 6% noticed symptoms.5 More than 25% of people over 50 years have never informed themselves about hearing loss as an online questionnaire study showed; only 20% of people aged 65 years or older with moderate to profound hearing loss identify themselves as being hearing-impaired.6 This might be due to the slow progression of hearing loss the people affected get used to.7
Consequences of Hearing Loss
Besides speech understanding and communication pattern, hearing loss also impacts on a variety of physical and psychosocial aspects. People with hearing loss are more likely to be affected by depression or anxiety,8–11 women even more than men.11 However, the degree of hearing loss is not always linked to the severity of depression but in case of dual sensory impairment, the rate of depression and total mortality increases.12,13
Hearing loss is also associated with frailty.14,15 People with hearing loss are more likely to get affected by a frail condition as shown by Liljas in 2017 in 2836 elderly subjects aged 60 and older even after adjustment for confounders.14 But also the acceleration rate in the development from the pre-frail to the frail condition is increased in the hearing impaired as reported by Gordon 2020 in 656 adults aged 40–75 (mean 59.9).15 The incidence of falls rises by 1.4 for every 10dB the hearing thresholds decline16 and the risk of incident hospitalization in patients aged 70 to 79 is 16% greater in those with mild and 21% in those with moderate or greater hearing loss.17
Besides, hearing loss has an impact on cognitive performance as already mentioned by Clark in 1960.18 Later Uhlmann supported these findings in 1989 in 71 Alzheimer patients.19 However, it took more than 20 years until a longitudinal study done by Lin showing that the risk for dementia increases by 1.89 for mild hearing impairment and even by almost 5 for severe hearing loss raised awareness of the impact of hearing loss on cognition in the elderly.20
In the meantime, a huge number of studies on this cohort have been undertaken. Although the studies vary a lot in quality, a systematic review and meta-analysis published by Loughrey in 2018 comprising 36 studies on 20,264 participants clearly demonstrated a minor but statistically significant association between an objective measured hearing loss and cognitive functions in all cognitive domains including global cognition, executive functions, episodic and semantic memory, visuospatial ability and processing speed.21 This has recently been confirmed by Alattar in 1164 subjects aged 73.5. Subjects with a severe hearing loss are twice as likely to experience cognitive decline by the MMSE than elderly with only a mild hearing impairment over a period of 24 years.22
Nowadays, hearing loss is considered to be one of twelve modifiable risk factors in the development of dementia.23 Because of the high prevalence of hearing impairment in the elderly subjects, its treatment in the middle age has a large impact on the prevention or delay of dementia in up to 8% of the population.24
Association Between Cognition and Hearing
Cognition and hearing are closely related and influence each other. On the one hand, neurocognitive functions have an impact on speech understanding especially in challenging acoustic situations as summarized by Rönnberg in the Ease of Language Understanding (ELU) model.25 In optimal listening conditions, understanding is easy and does not require any effort as the incoming signal perfectly matches with the phonological information stored in the long-term memory. In challenging acoustic surroundings, however, top-down mechanisms have to be used to overcome the mismatch between the distorted incoming signal and the information stored in the long-term memory.25
As working memory which is known to be an important neurocognitive domain regarding speech understanding is limited and declines with age, linguistic abilities which largely remain stable and contextual information help to compensate age-related sensory and cognitive decline.26 Clinically, increased listening effort and cognitive fatigue have been reported by impaired subjects, especially in the elderly population.27,28
On the other hand, hearing loss also influences cognitive performance. On the neuronal level, peripheral auditory processing disorders directly or indirectly impact on the morphology of brain structures and their functions.29 Besides a reduction of the volume of the primary auditory cortex and a diminished integrity of relevant subcortical pathways, a broader reactivation and redistribution of the cognitive resources for the processing of the auditory stimuli and an increased full-brain connectivity have been described.30 Reduced neuronal plasticity associated with neurodegenerative diseases like Alzheimer may also impair neuronal adaptation processes that are relevant to speech processing.
Despite the large body of publications describing the association between cognitive functions and hearing loss one has to question whether the results may be partially related to the oral presentation of the tests applied. Notably, the most popular cognitive test was the auditory-based MMSE (Mini-Mental Status Examination) and researchers included subjects with a wide range of different levels of hearing loss, including slight to severe hearing impairment.
The underlying mechanisms of the close association between hearing status and cognition are not yet fully understood.31–35 The common cause hypothesis postulates that both hearing loss and cognitive decline are related to a pathology that impairs multiple organs, such as vascular or metabolic diseases.23,36 Another approach that contributes to the deprivation hypothesis is that social isolation due to sensory impairment also may cause a decline in cognitive performance.37,38 In contrast, higher cognitive demand for speech understanding is supposed to cause cognitive overload which withdraws cognitive capacity for other mentally challenging tasks.39,40 Cognitive decline in the course of hearing loss progression follows a cascade and might be pushed by brain alterations due to different pathogenic reasons (dual shot hypothesis).
The aim of the study was to evaluate the impact of hearing impairment on geriatric assessment including cognitive testing in elderly subjects in the geriatric setting.
Materials and Methods
Computer-based search was performed from 1960 to 08.2020 according to the guidelines of PRISMA (preferred reporting items for systematic reviews and meta-analysis) on PubMed, Ebsco, Scopus and web of science. Data management and deduplication were performed using Citavi 6, after deduplication screening of titles and abstracts was done according to the inclusion and exclusion criteria by two independent researchers (L.G. and C.V.). In case of incongruence, a third researcher (J.P.T.) provided consent. Afterwards studies were checked for quality and (test/retest, detection, performance and selection) bias.
First, a search was done using MeSH based on the keywords “geriatric assessment” AND “hearing impairment.” Inclusion criteria were (1) application on elderly, (2) assessment tool and performance tests routinely applied in geriatric medicine, (3) hearing impairment either self-assessed or by 4-PTA.
Furthermore, another search was performed in Pubmed, Ebsco and Scopus using the keywords “hearing impairment” AND “cognitive assessment” AND “elderly subjects.” Only studies (1) dealing with adult patients aged 18 or older, (2) using objective assessments, (3) studies which compared participants with hearing loss or a simulated hearing loss and NH subjects, (4) full-text availability in English or German were included. Studies on the impact of hearing aid use or cochlear implantation on cognition or those that focus on central auditory processing or on the relation between speech perception (in noise) and cognitive functions or studies that evaluate the relationship between linguistic abilities and speech perception have been excluded.
For statistical analysis studies on the impact of hearing loss on the performance in cognitive assessments were selected. In 3 cases the MoCA and in 1 case the MMSE was used. All other studies had to be excluded due to heterogeneity or missing data. Means and standard deviations for the mean difference in the MoCA and the MMSE score for each study for the hearing impaired (HI) and normal hearing (NH) subjects were calculated and Forest-plot analysis was performed.
Results
Impact of Hearing Loss on Geriatric Assessments in General
Only a few studies analyzed the influence of impaired hearing on geriatric performance tests. 118 studies were found of which 37 were excluded after screening the titles and another 50 after screening the abstracts. 31 were eligible for full-text analysis and 3 were included in the data analysis (see Figure 1A, Table 1).
 |
Table 1 Publications Dealing with Impact of Hearing Loss on Geriatric Assessment (See Flow Chart Figure 1A) |
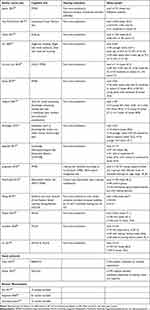 |
Table 2 Publications Dealing with Impact of Hearing Loss on Cognitive Testing (See Flow Chart Figure 1B) |
 |
Figure 1 (A) Flow chart of literature search on impact of hearing loss on geriatric assessment. (B) Flow chart of literature search on impact of hearing loss on cognitive testing. |
Ibrahim described in 2084 elderly aged 68.7 that a self-reported vision and/or hearing loss was associated with longer duration of 0.6 seconds assessed in the Time up and Go Test (TUG).41
Besides walking also gait speed and mobility may be hampered depending on the severity of the hearing loss as shown by Chen in the Health, Aging and Body Composition study.42 In 2190 subjects the SPPB (Short Physical Performance Battery) score was significantly lower in subjects with mild and moderate hearing impairment than in NH subjects (10.04 versus 10.36, respectively). Furthermore, gait speed dropped from 1.22 to 0.88 m/s in NH in contrast to 1.18 to 0.80 m/s in HI during a follow-up period of 11 years. Interestingly, gender differences were detected. Women with moderate or severe hearing impairment had an increased risk of incident disability in 31%.42
Furthermore, HI need significantly more help to perform the activities of daily life (ADL) as shown by Gopinath in 1952 subjects aged >60, 686 with an hearing impairment aged 70.4 and 886 without one aged 77.2.43 Subjects with moderate to severe hearing loss had a 2.9-fold increased likelihood of reporting difficulties in basic and instrumental ADL compared to NH although HI were significantly younger. Subjects aged <75 years with hearing loss had 2-fold higher odds of impaired ADL compared to NH.43
So far, geriatric assessment does not frequently include an evaluation of hearing status such as the popular ISAR (Identification Seniors at Risk). Some rely only on self-assessment by questionnaires as it is the case in the manageable geriatric assessment (MAGIC) which asks the question whether it is difficult to follow conversations.44 Only a few include a whispering test such as in the German Lachs Geriatric screening test battery and the Brief assessment tool by the family physician (BAF) where patients are asked to repeat three numbers that have been whispered before.45 However, outcome in whispering tests might vary due to an inter-rater difference in the loudness of whispering.
Impact of Hearing Loss on Cognitive Testing
In total, 958 PubMed, 33 Scopus, 144 web of science and 1890 Ebsco articles matched to the search items. The remaining 912 articles after removal of duplicates were screened for titles and abstracts (see Figure 1B). 687 articles were removed after title screening, 225 articles were eligible for abstract screening. Studies with vision loss (n=4), studies including only a cognitive self-assessment (n=2), studies in other languages apart from the above mentioned (n=3), studies on central auditory processing (n=4) and studies with a different focus (n=109) were excluded. Out of the remaining 103 articles, the majority (n=47) analyzed the interaction between cognitive functions and hearing status. 6 articles focused on central auditory function, 9 on speech perception abilities in noise, and 20 had another focus of interest. After detailed full-text analysis, 21 articles dealing with the impact of hearing loss on cognitive testing were identified. One article was not available in full-text version,46 two publications were reviews,47,48 one was a review and a meta-analysis49 and two were study protocols.50,51
15 studies including participants of different age groups and hearing abilities were analyzed in detail (see Table 2). Hearing impairment was either simulated in subjects with normal hearing (NH) or mild, medium, or severe to profound. Testing was also partially done with a hearing aid or a cochlear implant. Both cognitively healthy patients and subjects with cognitive impairment were enrolled. The test batteries used were the MMSE, the MoCA, the ALAcog, the abbreviated mental test (AMT) and the Continuous Visual Memory Test.
4 studies, 3 of them dealing with the MoCA and 1 with the MMSE, presented mean values and were selected for a Forest-plot. All other studies had to be excluded due to heterogeneity or missing data.
In total, HI subjects performed 2.94 points (SD 0.47, range from 2.01 to 3.86) lower than NH subjects in these 4 studies covering 425 subjects (197 HI and 228 NH). In total, this difference was highly statistically significant (p<0.0005).
Cognitive Testing in Subjects with Hearing Loss
Assessment of cognitive functioning in people with sensory impairment is challenging and even healthcare professionals claim lack of appropriate assessment tools and the need for clear guidelines.52,53
The majority of currently used neurocognitive test batteries are based on oral instructions and include subtests relying on auditory functions which rise the bias of false positive results in multiple ways, especially in subjects with a lower educational background.54–57 In the most frequently used neurocognitive screening batteries, the MMSE and the MoCA (Montreal Cognitive Assessment), 7 versus 10 out of 30 points depend on auditory presented stimuli, respectively.47,48,58,59 This is also true for the DemTect, which is highly sensitive to (mild cognitive impairment) MCI detection and quite popular in Germany.60 It consists of 5 subtests covering attention, (delayed) recall, transcoding of numbers, and language abilities and it has been validated in 363 patients with age-dependent cut-off scores. However, half of the subtests of the DemTect rely on verbal instructions as well (see Table 3).
 |
Table 3 Cognitive Screening Test Batteries. Some Subtests (Indicated by *) Might Be Influenced by Hearing Impairment |
Therefore, overdiagnosis of cognitive decline can happen in people with hearing loss. This has already been described by Jorgensen in 125 healthy NH young subjects with a simulated hearing loss.54 Whereas mild hearing loss did not have an influence on cognitive performance, 16% of the subjects with at least moderate hearing loss were misdiagnosed with dementia. In line with that Gaeta found that the performance in the MMSE was comparable in 30 elderly hearing impaired with a mean age of 69.4 and 30 younger adults aged 24.2 when a hearing loss was simulated.56 Also, Verhaegen undermined these findings in a verbal recall task.61 Augmentation by hearing aid use can reverse the bad performance in hearing-impaired subjects as shown by Jorgensen, Gaeta, Wong and MacDonald.54,56,62,63
The cognitive domain which seems to be mostly influenced by hearing loss is working memory and associative learning. Jayakody demonstrated in a study on 119 subjects, 47 NH, 51 with a mild to moderate HI and 21 with a severe to profound hearing loss that for every dB improvement in the hearing threshold the working memory task assessed by the CANTAB test battery improved by 2.85 points and the learning task by 11.79 points.64 For the tasks that were only visually presented no difference was found between 210 NH and 87 HI subjects.65
Cognitive Assessment Tools for Subjects with Sensory Impairment
As the awareness of the influence of sensory impairment on cognitive tasks has recently risen, several attempts have been made to adapt the existing screening tools to the needs of people with sensory impairment either by introducing a new cut-off score for people with hearing loss, removing of auditory stimuli, or providing non-auditory presentation of the stimuli.66,67
The first one to pay attention to this topic was Uhlmann in 1989 who presented the instructions of the MMSE in a written version in addition to the original one in Alzheimer patients with and without hearing loss.19 Silva et al tested 55 NH subjects and 27 subjects with moderate-to-severe hearing loss with a mean age of 81.2 using both a written and the original version of the MMSE. 75% of the subjects with hearing loss preferred the written version to the auditory presentation whereas only 58% of the NH subjects preferred the written version.66 This may further be the case because HI rely more on visual cues as shown in 48 normal and HI subjects.68 Another approach was made by Dupuis when introducing different cut-offs for the MoCA.69 In a sample of 165 NH and 135 subjects with hearing loss, the NH subjects still outperformed the subjects with hearing loss even if the auditory presented parts of the tests were scored differently for the subjects with hearing loss. However, the number of subjects with hearing loss that passed the test increased by 21% by applying different cut-off scores.
Others adapted the cognitive testing by removing the subtests relying on auditory functions. However, this strategy may decrease the validity of the test. In a study on 277 elderly patients without cognitive decline, with MCI or mild Alzheimer’s disease (AD), the deletion of the delayed recall items of the MoCA decreased the sensitivity to detect MCI from 90% in the original version to 53% and even to 43% when all auditory-based subtests were removed. In contrast, sensitivity to detect AD was still between 87% and 100% in comparison to 100% in the original version.70
Assessment tools that are fully independent of verbal instructions or responses such as the Raven`s Progressive Matrices, which is commonly used for the evaluation of the general intelligence71 may be another choice. It`s usability as a non-verbal and language-independent test instrument has been proven. Unfortunately, this test is worse in the differentiation between MCI and AD as shown by Ambra 2016 in 30elderly subjects with MCI or AD and 31 healthy controls. Whereas AD patients scored significantly poorer than the healthy controls, the number of errors was similar between the healthy controls and the MCI subjects and differed only in 2 out of 4 subcategories between the MCI and AD subjects.72
To overcome the bias, computer-based assessments have been developed. These test batteries are time-saving, require less resources, and are highly objective in the provision of stimuli and assessment of test results. These digital tests are less dependent on auditory thresholds as shown by Gallacher 2012 who followed 1057 men for 17 years.39 Beside 4-PTA, an interview-administered test battery for cognitive decline and computer tests were applied. Auditory thresholds were associated with incident dementia and cognitive decline (OR of 1.42). Interestingly, the association was lower in the computer tests than in the interview-based tests. This might be due to the fact that cognitive overload may be diminished by using non-auditory computer-based batteries, which do not depend on auditory functions and allow the participant to re-read a question.
The tests that are currently available are mostly designed for a defined target group and focus on one certain feature. As shown in different studies, computer-based tests are also useful for testing elderly people.73,74 However, it is recommended to do a pretest run in advance to adapt subjects who are less experienced with computers to digital media.75
A large variety of computer-based testing tools, screening tools, and comprehensive test batteries are available now.76 Visual stimuli have been developed for the application in subjects with hearing loss, such as in the Repeatable Battery for the Assessment of Neuropsychological Status for Hearing-Impaired Individuals (RBANS-H), the Cambridge Neuropsychological Test Automated Battery (CANTAB), the ALAcog and the Cogstate battery which have mainly been used in the past to study the benefit of cochlear implantation for cognitive functions in older subjects.50,77–80 However by using visual stimuli instead of auditory stimuli different ways of processing might be assessed and a combined audiovisual presentation of stimuli might even alter the performance.
In 2017, Lin introduced a visually adapted version of the MoCA (HI-MoCA) by using PowerPoint slides to present the written test instructions to 103 cognitively healthy NH elderly subjects with a mean age of 68.4 and 49 age-matched subjects with severe hearing loss with a mean age of 70.2. Both groups achieved similar results on the adapted test version. When comparing the two test versions in the total sample of 152 people, the two versions slightly differed in the recall task and in the language ability task.81 In line with that, Parada tested 21 cochlear-implanted subjects with a mean age of 68.9 with the MoCA and the HI-MoCA. Especially in the recall task, patients obtained significantly better scores in the HI-MoCA than in the original version.
Assessments specifically tailored to subjects with hearing loss have been developed (see Table 4). The ReaCT Kyoto, a Japanese test instrument, was introduced by Okano in 2020.82 It includes measures on registration, repletion, delayed recall, visuospatial recognition, executive functions as measured by verbal fluency and orientation; it has already been applied in 115 healthy subjects and subjects with cognitive impairment with a sensitivity of 90%. To date, this test instrument is only available in Japanese.82
A new approach was introduced by Bruhn in 2018. A Tactile Test Battery (TTB) comprising established standard tests transformed for tactile use was applied to 60 subjects, 20 of them with dementia, 20 with dual sensory impairment, and 20 controls aged 63–92. The different subtests covering learning, memory, naming, spatial perception, and processing speed clearly differed cognitively healthy subjects from subjects with cognitive impairment.83
Discussion
So far sensory impairment has often insufficiently been considered by patients themselves but also by health professionals.6,53 Screening for hearing loss in the elderly population is not regularly done as this is the case for other chronic diseases associated with age, such as screening programs to check for breast or prostate cancer which are both well established.84 Pure tone audiometry, which is the gold standard or auditory steady-state response (ASSR) evaluation as a valid method for hearing assessment in people with cognitive impairment, is rather time-consuming and not viable in an outpatient setting.85
Efforts have been made to develop inventories to screen for sensory loss. The most commonly used for hearing impaired are the Hearing Handicap Inventory for the Elderly (HHIE), the Measure of Severity of Hearing Loss, and the Hyperacusis Questionnaire.86 Moreover, Osterloo recently published promising results that one single multiple-choice-question detects and differentiates between mild and moderate hearing loss in older adults.87 Löhler also published a short questionnaire; the sensitivity for people aged 60 years and older, however, is lower than in the younger age group.88
Several Apps are available which allow people to screen for hearing loss on their own.89 Some of them, such as the Digit- Triplet Test (Hear ContrOL, Hörtech Oldenburg) which is based on a combination of 3 digits presented in background noise, has already shown to be suitable and sensitive for automatic self-screening.90,91
Lycke proposed in 2015 to include a screening tool (App uHear) which allows to determine air conduction thresholds in each ear separately within the comprehensive geriatric assessment.92
But as shown by different studies hearing loss has an impact on cognitive screening tests, although not all subtests are affected in the same way.69,93 In particular, outcomes on recall and delayed recall tasks tend to be affected by hearing loss as reported by Wong in 2019 who analyzed 82 cognitively healthy NH older aged adults between 55 and 85 years using the Hopkins Verbal Learning Testing-Revised (HVLT-R). NH were able to remember almost twice as many words in the recall and in the delayed recall as subjects with a mean hearing loss of 49.2dB. In a cross condition with a simulated hearing loss for the NH subjects and an increased audibility for the subjects with hearing loss, the results were vice versa.62
Moreover, the number of words that have to be remembered and recalled influences the outcome. Lim and Loo investigated the performance on the MoCA and the MMSE in 111 older participants with varying degrees of hearing loss. Regression analysis demonstrated that for every 10dB of hearing loss MMSE scores decreased by 2.8% and MoCA scores by 3.5%.93 This may be related to the number of words that have to be remembered, three words for the MMSE and five for the MoCA.93 Shen investigated a small sample size of 24 elderly with the MoCA and the “Word Auditory Recognition and recall Measure” (WARRM) which includes 100 monosyllabic words to be recalled either in 2, 3, 4, 5, or 6 items. Testing was performed in an audio design, visually, and with amplification. Overall, the MoCA score was not significantly influenced by the test modality but with the WARRM, better scores were obtained when the sound pressure level (SPL) was amplified.67
In line with that MacDonald reported that amplification by hearing aid use in 192 subjects aged 82.4 was related to significantly better test results in the MMSE, but not in the AMT (Abbreviated Mental Test) in which only one out of 10 points relies on auditory stimuli.63
So far, there are only few investigations on the potential impact of different degrees of hearing loss on cognitive testing. According to a study done by Jorgensen, speech recognition of at least 40% in the NU-6 speech recognition test is necessary to ensure understanding of test instructions.94 Jupiter underlined this finding analyzing 101 Alzheimer's disease patients between 68 and 108. Subjects with hearing thresholds better than 40dB performed better in the MMSE.95 This is in line with Saunders who tested 42 participants with a mild hearing loss in the MoCA once without and once with hearing aids. Amplification did not significantly improve the performance.96 Therefore, cognitive testing might be hampered only in severe to profound hearing-impaired subjects.
Moreover, the examiner needs to clearly scrutinize the test setting. Background noise, for example, in an emergency unit or at the ward, which ranges between approximately 50 and 70dB needs to be considered as cognitive screening tests are usually performed at the bedside.97,98 Dupuis analyzed this aspect in 60 subjects who underwent cognitive testing with the MoCA either in a low background noise setting (+20dB Signal Noise Ratio) or in a high background noise setting (−12dB Signal Noise Ratio).57 Subjects were further divided into 3 groups: older NH subjects with a mean age of 71.4, older subjects with hearing loss with a mean age of 73.7, and younger NH subjects with a mean age of 18.8. All groups performed on average 3 points poorer in the high background noise setting. In the low background noise setting, however, NH elderly participants were able to compensate this obstacle whereas the elderly with hearing loss could not. In the louder setting, both groups achieved equal results; they even scored below the cut-off of 25 points which may indicate a mild cognitive impairment. Thus, geriatric assessment should be performed in a quiet setting.
But hearing impairment does not only have a consequence on the sensory perceptual level, but also a negative downstream effect on processing resources in cognitive testing. The constant cognitive effort which hearing loss imposes in order to maintain successful understanding of speech leads to mental fatigue in hearing-impaired subjects.25,39 This extra burden was demonstrated by Tun in 2009 in 24 younger and 24 older adults with different degrees of hearing acuity.99 HI needed a greater effort to perform a secondary task while recalling words even though the stimuli were presented to the HI at a sound intensity which was adapted to the hearing thresholds in order to allow them to understand.99 Similar results were obtained by McCoy who found that the recall of the first and the second-to-last word significantly differed between NH and HI aged 66 to 81 years especially if the context was low.100
Conclusion
Hearing loss has a considerable impact on different parts of well-being, such as cognition, mobility and quality of life; but also on cognitive or geriatric performance tests. This aspect has often been neglected so far. Recently, the demand for appropriate assessments has risen due to population aging and the growing number of elderly people with dementia.52,101 Different approaches have been made to minimize the effect of audibility on cognitive performance by removing subtests or developing appropriate test batteries. However, normative data are still missing and further research is necessary.51,57,70,74
Acknowledgments
We are very thankful to Ursula Lehner-Mayrhofer, Med-EL, for helpful proofreading on a version of the manuscript. We further appreciate the support by the DFG Open Access Publication Funds of the Ruhr-University Bochum.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The Department of Otorhinolaryngology, Head and Neck Surgery at the Katholisches Klinikum in Bochum, Ruhr-University of Bochum, received unrelated third-party funds from MED-EL. Christiane Völter, Jan Peter Thomas, Stefan Dazert received travel expense support from MED-EL. The authors report no other conflicts of interest in this work.
References
1. World Health Organization Regional Office for South-East Asia. Deafness and hearing impairment survey—report of the consultative meeting of principal investigators SEARO, New Delhi, 7–9 May 2001. WHO, New Delhi.
2. Fischer N, Weber B, Riechelmann H. Presbyakusis - Altersschwerhorigkeit. Laryngorhinootologie. 2016;95:497–510.
3. Arvin B, Prepageran N, Raman R. “High frequency presbycusis”-is there an earlier onset? Indian J Otolaryngol Head Neck Surg. 2013;65:480–484.
4. Blackwell DL, Lucas JW, Clarke TC Summary health statistics for US adults: National health interview survey, 2012. Vital and health statistics. Series 10, Data from the National Health Survey. 2014:1.
5. Ramage-Morin PL, Banks R, Pineault D, Atrach M. Unperceived hearing loss among Canadians aged 40 to 79. Health Rep. 2019;30:11–20.
6. D’Haese PSC, de Bodt M, van Rompaey V, van de Heyning P. Awareness of hearing loss in older adults: results of a survey conducted in 500 subjects across 5 european countries as a basis for an online awareness Campaign. Inquiry. 2018;55:46958018759421.
7. Carson AJ. “What brings you here today?” The role of self-assessment in help-seeking for age-related hearing loss. J Aging Stud. 2005;19:185–200.
8. Cosh S, Carriere I, Daien V, et al. The relationship between hearing loss in older adults and depression over 12 years: findings from the Three-City prospective cohort study. Int J Geriatr Psychiatry. 2018;33:1654–1661.
9. Ciorba A, Bianchini C, Pelucchi S, Pastore A. The impact of hearing loss on the quality of life of elderly adults. Clin Interv Aging. 2012;7:159–163.
10. Kim SY, Min C, Lee CH, Park B, Choi HG. Bidirectional relation between depression and sudden sensorineural hearing loss: two longitudinal follow-up studies using a national sample cohort. Sci Rep. 2020;10:1482.
11. Choudhury M. Hearing loss a triggering factor to depression: a systematic review. SJO. 2019;2.
12. Cosh S, von Hanno T, Helmer Cet al. The association amongst visual, hearing, and dual sensory loss with depression and anxiety over 6 years: the Tromsø Study. Int J Geriatr Psychiatry. 2018;33:598–605.
13. Gopinath B, Schneider J, McMahon CM, Burlutsky G, Leeder SR, Mitchell P. Dual sensory impairment in older adults increases the risk of mortality: a population-based study. PLoS one. 2013;8(3), e55054.
14. Liljas AEM, Carvalho LA, Papachristou E, et al. Self-reported hearing impairment and incident frailty in english community-dwelling older adults: a 4-year follow-up study. J Am Geriatr Soc. 2017;65:958–965.
15. Gordon SJ, Baker N, Kidd M, Maeder A, Grimmer KA. Pre-frailty factors in community-dwelling 40-75 year olds: opportunities for successful ageing. BMC Geriatr. 2020;20:96.
16. Lin FR, Ferrucci L. Hearing loss and falls among older adults in the United States. Arch Intern Med. 2012;172:369–371.
17. Genther DJ, Betz J, Pratt S, et al. Association Between Hearing Impairment and Risk of Hospitalization in Older Adults. J Am Geriatr Soc. 2015;63(6):1146-1152. doi:10.1111/jgs.13456
18. Clark JW. The aging dimension: a factorial analysis of individual differences with age on psychological and physiological measurements. J Gerontol. 1960.
19. Uhlmann RF, Larson EB, Rees TS, et al. Relationship of hearing impairment to dementia and cognitive dysfunction in older adults. JAMA. 1989;261:1916–1919.
20. Lin FR, Metter EJ, O’Brien RJ, Resnick SM, Zonderman AB, Ferrucci L. Hearing loss and incident dementia. Arch Neurol. 2011;68:214–220.
21. Loughrey DG, Kelly ME, Kelley GA, Brennan S, Lawlor BA. Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia: a systematic review and meta-. JAMA Otolaryngol Head Neck Surg. 2018;144:115–126.
22. Alattar AA, Bergstrom J, Laughlin GA, et al. Hearing impairment and cognitive decline in older, community-dwelling adults. J Gerontol a Biol Sci Med Sci. 2020;75:567–573.
23. Livingston G, Sommerlad A, Orgeta V, et al. Dementia prevention, intervention, and care. The Lancet. 2017;390:2673–2734.
24. Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396:413–446.
25. Rönnberg J, Lunner T, Zekveld A, et al. The Ease of Language Understanding (ELU) model: theoretical, empirical, and clinical advances. Front Syst Neurosci. 2013;7:31.
26. Wingfield A. Evolution of models of working memory and cognitive resources. Ear Hear. 2016;37:35–43.
27. Holman JA, Drummond A, Naylor G. The effect of hearing loss and hearing device fitting on fatigue in adults: a systematic review. Ear Hear. 2020.
28. Pichora-Fuller KM, Kramer SE, Eckert MA, Edwards B. Hearing impairment and cognitive energy: the Framework for Understanding Effortful Listening (FUEL). Ear Hear. 2016;37 Suppl 1:5S–27S.
29. Ren F, Ma W, Li M, et al. Gray matter atrophy is associated with cognitive impairment in patients with presbycusis: a comprehensive morphometric study. Front Neurosci. 2018;12:744.
30. Bidelman GM, Mahmud MS, Yeasin M, Shen D, Arnott SR, Alain C. Age-related hearing loss increases full-brain connectivity while reversing directed signaling within the dorsal-ventral pathway for speech. Brain Struct Funct. 2019;224:2661–2676.
31. Heywood R, Gao Q, Nyunt MSZ, et al. Hearing loss and risk of mild cognitive impairment and dementia: findings from the singapore longitudinal ageing study. Dement Geriatr Cogn Disord. 2017;43:259–268.
32. Lin FR, Ferrucci L, Metter EJ, An Y, Zonderman AB, Resnick SM. Hearing loss and cognition in the Baltimore Longitudinal Study of Aging. Neuropsychology. 2011;25:763–770.
33. Lin FR, Albert M. Hearing loss and dementia - who is listening? Aging Ment Health. 2014;18:671–673.
34. Fortunato S, Forli F, Guglielmi V, et al. A review of new insights on the association between hearing loss and cognitive decline in ageing. Acta otorhinolaryngologica Italica. 2016;36:155–166.
35. Uchida Y, Sugiura S, Nishita Y, Saji N, Sone M, Ueda H. Age-related hearing loss and cognitive decline - The potential mechanisms linking the two. Auris Nasus Larynx. 2019;46:1–9.
36. Oh I-H, Lee JH, Park DC, et al. Hearing loss as a function of aging and diabetes mellitus: A cross sectional study. PLoS One. 2014;9:e116161.
37. Rutherford BR, Brewster K, Golub JS, Kim AH, Roose SP. Sensation and psychiatry: linking age-related hearing loss to late-life depression and cognitive decline. Am J Psychiatry. 2018;175:215–224.
38. Dawes P, Emsley R, Cruickshanks KJ, et al. Hearing loss and cognition: the role of hearing AIDS, social isolation and depression. PLoS One. 2015;10:e0119616.
39. Gallacher J, Ilubaera V, Ben-Shlomo Y, et al. Auditory threshold, phonologic demand, and incident dementia. Neurology. 2012;79:1583–1590.
40. Tun PA, Williams VA, Small BJ, Hafter ER. The effects of aging on auditory processing and cognition. Am J Audiol. 2012;21:344–350.
41. Ibrahim A, Singh DKA, Shahar S. ‘Timed Up and Go’ test: age, gender and cognitive impairment stratified normative values of older adults. PLoS One. 2017;12:e0185641.
42. Chen DS, Betz J, Yaffe K, et al. Association of hearing impairment with declines in physical functioning and the risk of disability in older adults. J Gerontol Ser A. 2015;70:654–661.
43. Gopinath B, Schneider J, McMahon CM, Teber E, Leeder SR, Mitchell P. Severity of age-related hearing loss is associated with impaired activities of daily living. Age Ageing. 2012;41:195–200.
44. Barkhausen T, Junius-Walker U, Hummers-Pradier E, Mueller CA, Theile G. “It’s MAGIC”–development of a manageable geriatric assessment for general practice use. BMC Fam Pract. 2015;16:4.
45. Mueller YK, Monod S, Locatelli I, Büla C, Cornuz J, Senn N. Performance of a brief geriatric evaluation compared to a comprehensive geriatric assessment for detection of geriatric syndromes in family medicine: a prospective diagnostic study. BMC Geriatr. 2018;18:72.
46. Nixon G, Sarant JZ, Tomlin D, Dowell R. The relationship between peripheral hearing loss and higher order listening function on cognition in older Australians. Int J Audiol. 2019;58:933–944.
47. Pye A, Charalambous AP, Leroi I, Thodi C, Dawes P. Screening tools for the identification of dementia for adults with age-related acquired hearing or vision impairment: A scoping review. Int Psychogeriatr. 2017;29:1771–1784.
48. Raymond M, Barrett D, Lee DJ, Peterson S, Raol N, Vivas EX. Cognitive screening of adults with postlingual hearing loss: a systematic review. Otolaryngol Head Neck Surg. 2020;0194599820933255.
49. Utoomprurkporn N, Woodall K, Stott J, Costafreda SG, Bamiou DE. Hearing-impaired population performance and the effect of hearing interventions on Montreal Cognitive Assessment (MoCA): systematic review and meta-analysis. Int J Geriatr Psychiatry. 2020.
50. Claes AJ, van de Heyning P, Gilles A, Hofkens-Van den Brandt A, van Rompaey V, Mertens G. Impaired Cognitive Functioning in Cochlear Implant Recipients Over the Age of 55 Years: A Cross-Sectional Study Using the Repeatable Battery for the Assessment of Neuropsychological Status for Hearing-Impaired Individuals (RBANS-H). Front Neurosci. 2018;12:580.
51. Dawes P, Pye A, Reeves D, et al. Protocol for the development of versions of the Montreal Cognitive Assessment (MoCA) for people with hearing or vision impairment. BMJ Open. 2019;9:e026246.
52. Leroi I, Himmelsbach I, Wolski L, et al. Assessing and managing concurrent hearing, vision and cognitive impairments in older people: an international perspective from healthcare professionals. Age Ageing. 2019;48:580–587.
53. Raymond MJ, Lee AC, Schader LM, Moore RH, Raol NR, Vivas EX. Practices and perceptions of cognitive assessment for adults with age-related hearing loss. Laryngoscope Invest Otolaryngol. 2020;5:137–144.
54. Jorgensen LE, Palmer CV, Pratt S, Erickson KI, Moncrieff D. The effect of decreased audibility on MMSE performance: a measure commonly used for diagnosing dementia. J Am Acad Audiol. 2016;27:311–323.
55. Füllgrabe C. On the Possible overestimation of cognitive decline: the impact of age-related hearing loss on cognitive-test performance. Front Neurosci. 2020;14:454.
56. Gaeta L, Azzarello J, Baldwin J, et al. Effect of reduced audibility on mini-mental state examination scores. J Am Acad Audiol. 2019;30:845–855.
57. Dupuis K, Marchuk V, Pichora-Fuller MK. Noise affects performance on the montreal cognitive assessment. Can J Aging. 2016;35:298–307.
58. Nasreddine ZS, Phillips NA, Bedirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–699.
59. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198.
60. Kalbe E, Kessler J, Calabrese P, et al. DemTect: a new, sensitive cognitive screening test to support the diagnosis of mild cognitive impairment and early dementia. Int J Geriatr Psychiatry. 2004;19:136–143.
61. Verhaegen C, Collette F, Majerus S. The impact of aging and hearing status on verbal short-term memory. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2014;21:464–482.
62. Wong CG, Rapport LJ, Billings BA, Ramachandran V, Stach BA. Hearing loss and verbal memory assessment among older adults. Neuropsychology. 2019;33:47–59.
63. MacDonald AA, Joyson A, Lee R, Seymour DG, Soiza RL. The effect of hearing augmentation on cognitive assessment scales at admission to hospital. Am J Geriatr Psychiatry. 2012;20:355–361.
64. Jayakody DMP, Friedland PL, Eikelboom RH, Martins RN, Sohrabi HR. A novel study on association between untreated hearing loss and cognitive functions of older adults: baseline non-verbal cognitive assessment results. Clin Otolaryngol. 2018;43:182–191.
65. Hay-McCutcheon MJ, Hyams A, Yang X, et al. An exploration of the associations among hearing loss, physical health, and visual memory in adults from west central Alabama. J Speech Lang Hear Res. 2017;60:2346–2359.
66. de Silva ML, Mclaughlin MT, Rodrigues EJ, Broadbent JC, Gray AR, Hammond-Tooke GD. A mini-mental status examination for the hearing impaired. Age Ageing. 2008;37:589–593.
67. Shen J, Sherman M, Souza PE. Test administration methods and cognitive test scores in older adults with hearing loss. Gerontology. 2020;66:24–32.
68. Hällgren M, Larsby B, Lyxell B, Arlinger S. Evaluation of a cognitive test battery in young and elderly normal-hearing and hearing-impaired persons. J Am Acad Audiol. 2001;12:357–370.
69. Dupuis K, Pichora-Fuller MK, Chasteen AL, Marchuk V, Singh G, Smith SL. Effects of hearing and vision impairments on the Montreal Cognitive Assessment. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2015;22:413–437.
70. Al‐Yawer F, Pichora‐Fuller MK, Phillips NA. The Montreal Cognitive Assessment after omission of hearing‐dependent subtests: psychometrics and clinical recommendations. J Am Geriatr Soc. 2019;67:1689–1694.
71. Raven JC, Court JH. Raven’s Progressive Matrices: Western Psychological Services. Los Angeles, CA; 1938.
72. Ambra FI, Iavarone A, Ronga B, et al. Qualitative patterns at Raven’s colored progressive matrices in mild cognitive impairment and Alzheimer’s disease. Aging Clin Exp Res. 2016;28:561–565.
73. Collerton J, Collerton D, Arai Y, et al. A comparison of computerized and pencil-and-paper tasks in assessing cognitive function in community-dwelling older people in the Newcastle 85+ Pilot Study. J Am Geriatr Soc. 2007;55:1630–1635.
74. Völter C, Götze L, Falkenstein M, Dazert S, Thomas JP. Application of a computer-based neurocognitive assessment battery in the elderly with and without hearing loss. Clin Interv Aging. 2017121681–1690.
75. Fazeli PL, Ross LA, Vance DE, Ball K. The relationship between computer experience and computerized cognitive test performance among older adults. J Gerontol B Psychol Sci Soc Sci. 2013;68:337–346.
76. Zygouris S, Tsolaki M. Computerized cognitive testing for older adults: a review. Am J Alzheimers Dis Other Demen. 2015;30:13–28.
77. Völter C, Götze L, Dazert S, Falkenstein M, Thomas JP. Can cochlear implantation improve neurocognition in the aging population? Clin Interv Aging. 2018;13:701–712.
78. Moberly AC, Doerfer K, Harris MS. Does cochlear implantation improve cognitive function? 2019;2208–2209.
79. Jayakody DMP, Friedland PL, Nel E, Martins RN, Atlas MD, Sohrabi HR. Impact of cochlear implantation on cognitive functions of older adults: pilot test results. Otol Neurotol. 2017;38:e289–e295.
80. Sarant J, Harris D, Busby P, et al. The effect of cochlear implants on cognitive function in older adults: initial baseline and 18-month follow up results for a prospective international longitudinal study. Front Neurosci. 2019;13:789.
81. Lin VYW, Chung J, Callahan BL, et al. Development of cognitive screening test for the severely hearing impaired: hearing-impaired MoCA. Laryngoscope. 2017;127(Suppl 1)):4–11.
82. Okano T, Yamamoto Y, Kuzuya A, et al. Development of the Reading Cognitive Test Kyoto (ReaCT Kyoto) for early detection of cognitive decline in patients with hearing loss. J Alzheimers Dis. 2020;73:981–990.
83. Bruhn P, Dammeyer J. Assessment of dementia in individuals with dual sensory loss: application of a tactile test battery. Dement Geriatr Cogn Dis Extra. 2018;8:12–22.
84. Siu AL. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279–296.
85. Villeneuve A, Hommet C, Aussedat C, Lescanne E, Reffet K, Bakhos D. Audiometric evaluation in patients with Alzheimer’s disease. Eur Arch Otorhinolaryngol. 2017;274:151–157.
86. Wittich W, Jarry J, Höbler F, McGilton KS. Agreement on the use of sensory screening techniques by nurses for older adults with cognitive impairment in long-term care: a mixed-methods consensus approach. BMJ Open. 2019;9:e027803.
87. Oosterloo BC, Homans NC, Baatenburg de Jong RJ, Ikram MA, Nagtegaal AP, Goedegebure A. . Assessing hearing loss in older adults with a single question and person characteristics; Comparison with pure tone audiometry in the Rotterdam Study. PLoS One. 2020;15:e0228349.
88. Löhler J, Walther L, Schlattmann P. The Mini-Audio-Test (MAT). Screening inventory for hearing loss starting at the age of 50. Laryngo-rhino-otologie. 2013;92:815.
89. Bexelius C, Honeth L, Ekman A, et al. Evaluation of an internet-based hearing test—comparison with established methods for detection of hearing loss. J Med Internet Res. 2008;10:e32.
90. Cullington HE, Aidi T. Is the digit triplet test an effective and acceptable way to assess speech recognition in adults using cochlear implants in a home environment? Cochlear Implants Int. 2017;18:97–105.
91. Vercammen C, Goossens T, Wouters J, van Wieringen A. Digit triplet test hearing screening with broadband and low-pass filtered noise in a middle-aged population. Ear Hear. 2018;39:825–828.
92. Lycke M, Debruyne PR, Lefebvre T, et al. The use of uHear™ to screen for hearing loss in older patients with cancer as part of a comprehensive geriatric assessment. Acta Clin Belg. 2018;73:132–138.
93. Lim MYL, Loo JHY. Screening an elderly hearing impaired population for mild cognitive impairment using Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA). Int J Geriatr Psychiatry. 2018;33:972–979.
94. Jorgensen LE, Palmer CV, Fischer G. Evaluation of hearing status at the time of dementia diagnosis. Audiol Today. 2014;26:38–45.
95. Jupiter T. Cognition and screening for hearing loss in nursing home residents. J Am Med Dir Assoc. 2012;13:744–747.
96. Saunders GH, Odgear I, Cosgrove A, Frederick MT. Impact of hearing loss and amplification on performance on a cognitive screening test. J Am Acad Audiol. 2018;29:648–655.
97. Filus W, ABMd L, Albizu E. Ambient noise in emergency rooms and its health hazards. Int Arch Otorhinolaryngol. 2015;19:205–209.
98. Tijunelis MA, Fitzsullivan E, Henderson SO. Noise in the ED. Am J Emerg Med. 2005;23:332–335.
99. Tun PA, McCoy S, Wingfield A. Aging, hearing acuity, and the attentional costs of effortful listening. Psychol Aging. 2009;24:761–766.
100. McCoy SL, Tun PA, Cox LC, Colangelo M, Stewart RA, Wingfield A. Hearing loss and perceptual effort: downstream effects on older adults’ memory for speech. Quart J Exp Psychol Sect A. 2005;58:22–33.
101. Wolski L, Leroi I, Regan J, et al. The need for improved cognitive, hearing and vision assessments for older people with cognitive impairment: a qualitative study. BMC Geriatr. 2019;19:328.
102. Randolph C, Tierney MC, Mohr E, Chase TN. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): preliminary clinical validity. J Clin Exp Neuropsychol. 1998;20:310–319.
103. Olaithe M, Weinborn M, Lowndes T, et al. Repeatable battery for the assessment of neuropsychological status (RBANS): normative data for older adults. Arch Clin Neuropsychol. 2019.
104. Falkenstein M, Hoormann J, Hohnsbein J. ERP components in Go/Nogo tasks and their relation to inhibition. Acta Psychol (Amst). 1999;101:267–291.
105. Wild-Wall N, Falkenstein M, Gajewski PD. Age-related differences in working memory performance in a 2-back task. Front Psychol. 2011;2:186.
106. Maruff P, Thomas E, Cysique L, et al. Validity of the CogState brief battery: relationship to standardized tests and sensitivity to cognitive impairment in mild traumatic brain injury, schizophrenia, and AIDS dementia complex. Arch Clin Neuropsychol. 2009;24:165–178.
107. Mielke MM, Machulda MM, Hagen CE, et al. Performance of the CogState computerized battery in the Mayo Clinic Study on Aging. Alzheimers Dement. 2015;11:1367–1376.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

