Back to Journals » Clinical Interventions in Aging » Volume 15
Impact of Different Geriatric Conditions on Choice of Therapy and In-Hospital Outcomes in Elderly Patients with Acute Coronary Syndrome
Authors Rittger H, Stadelmaier C , Kieschnick T, Büber D, Rank K, Vitali-Serdoz L, Bastian D , Waliszewski M
Received 10 February 2020
Accepted for publication 9 April 2020
Published 25 May 2020 Volume 2020:15 Pages 723—731
DOI https://doi.org/10.2147/CIA.S249017
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Walker
Harald Rittger,1,* Christoph Stadelmaier,1,* Thomas Kieschnick,1 Duygu Büber,1 Kristina Rank,1 Laura Vitali-Serdoz,1 Dirk Bastian,1 Matthias Waliszewski2,3
1Medizinische Klinik 1, Klinikum Fürth, Fürth, Germany; 2Medical Scientific Affairs, B. Braun Melsungen AG, Berlin, Germany; 3Department of Internal Medicine and Cardiology, Charité – Universitätsmedizin Berlin, Campus Virchow, Berlin, Germany
*These authors contributed equally to this work
Correspondence: Harald Rittger
Klinikum Fuerth, Medizinische Klinik 1, Jakob-Henle-Strasse 1, Fuerth 90766, Germany
Tel +49-911-7580-1101
Fax +49-911-7580-1104
Email [email protected]
Introduction: The clinical benefit of invasive therapy in elderly patients with acute coronary syndrome (ACS) remains unclear. Furthermore, the decision-making process to treat this growing patient group is also debatable. The purpose of this study was to assess the association between elderly ACS patients, the treatment choice and their in-hospital outcomes after non-ST-elevation (NSTE)-ACS in a consecutive series of patients > 75 years of age.
Methods and Results: Consecutive patients > 75 years presenting with NSTE-ACS in our hospital between July 2017 and July 2018 were included during the first 2 days of hospital admission. Demographic data, prior medical history and present medical condition were documented. During day 0 and day 2, geriatric assessments (Clinical Frailty Scale [CFS], Barthel index, Charlson comorbidity index, “timed up and go” test [TUG], Mini-Mental Status Test [MMS], Geriatric Depression Scale [GDS], SF-36 for quality of life, instrumental activities of daily living [IADL], Killip-score, Grace-score and Euro-score) were conducted. After 6 months, patients were re-evaluated. In 106 patients (mean age 81.9± 5.3 years, 57% male gender), 68 patients (64%) were treated interventionally, and 38 patients had conservative treatment (36%). Patients treated with intervention were significantly younger (80.9± 4.7 years vs 83.5± 6.0 years, p=0.015), had a lower rate of prior cerebral events (17.6% vs 26.3%; p=ns) and suffered more often from chronic obstructive pulmonary disease (17.6% vs 34.2%; p=0.050). All other demographic variables were comparable between both groups. The composite clinical endpoint (death, re-infarction, bleeding) was reached in 7 patients (10.3%) of the invasive and in 2 patients (5.3%) of the conservative group. They were not significantly different between both groups. A frailty index, consisting of commonly used parameters of functional impairment in elderly patients, namely, MMS ≤ 2 at baseline, IADL ≤ 7, CFS ≥ 7 and age ≥ 85 years, significantly predicted conservative treatment.
Conclusion: Effective revascularization techniques are still underused in patients of older age in the case of ACS. For decision-making, geriatric tests alone may not predict treatment in those patients, but the combination of different tests may better predict treatment and perhaps the clinical outcomes in those patients. Furthermore, frail patients are at higher risk for not receiving guideline recommended therapy.
Keywords: acute coronary syndromes, elderly patients, frailty, interventional treatment
Introduction
The rapidly aging population in the Western world leads to higher incidence rates of coronary artery disease (CAD) and patients with acute coronary syndrome (ACS). There is a substantial part of elderly patients suffering from geriatric preconditions like frailty and comorbidity, an increasing number of these patients are also treated invasively.1 Guidelines recommend an invasive therapy even in elderly patients with Non-ST-elevation myocardial infarction-ACS (NSTEMI-ACS).2 A recently published randomized trial, specifically designed to study elderly patients, revealed that this patient group benefits from invasive therapy.2 Unfortunately, this study included highly selected patients in relatively good health but neglected specific geriatric preconditions, like frailty and comorbidity, which are very common in these patients. There is strong evidence that prognosis is worse for common outcome measures in these patients.4–8 Not only frailty is associated with worse outcomes, pre-existing comorbid conditions as well as dementia have an unfavorable prognosis in the presence of ACS.9–11 Frailty is a condition characterized by loss of biological reserves, which leads to failure of homeostatic mechanisms following stressor events.12 An acute myocardial infarction is an example of a stressor event from which an older person with frailty may be at greater risk for adverse events, compared to a physically active older person. Consequently, frailty is independently associated with increased mortality following ACS. Possibly as a consequence, older patients are less likely to receive guideline indicated ACS care.13 It remains unclear if invasive therapy is beneficial, when these preconditions are evident in patients with NSTEMI-ACS. Likewise, the decision-making process of the treating physician is not well elucidated. The purpose of this study was to assess the association between these factors, the choice of treatment and in-hospital outcomes in consecutively recruited NSTE-ACS patients >75 years.
Methods
Patient Population and Study Protocol
The present study is a prospective observational study including consecutive patients admitted to our center for the treatment of NSTEMI-ACS.
NSTEMI-ACS was defined as acute chest pain based on the following criteria:
- Elevated troponin T (hsTNT) levels at admission or during the course of the hospital stay without other reasons for TNT elevation like congestive heart failure, myocarditis or atrial fibrillation (see below).
- ECG-changes susceptible for myocardial ischemia
To avoid possible confounding influences of other reasons for elevation of cardiac markers or ST-segment-changes like congestive heart failure, trauma, left ventricular aneurysm or persistent tachyarrhythmia due to atrial fibrillation, patients were not included if there were elevated TNT-levels detected without angiographic confirmation of CAD. Patients originally admitted with unstable angina, who developed myocardial infarction (MI) during the hospital course were classified as having ACS.
The decision to perform angiography and percutaneous coronary intervention (PCI) was left to the decision of the treating physician taking also into account the patients´ personal preferences or the preferences of the patients´ relatives.
Coronary angiography, PCI and stent implantation were performed according to institutional standards using a transfemoral or transradial approach in all patients. The decision for PCI as well as the decision to implant a stent after balloon dilatation or to conduct direct stent implantation without prior balloon dilatation was left to the discretion of the treating physician. In the case of stent implantation, drug-eluting stents (DES) were used in all patients. Heparin was given intravenously to achieve an activated clotting time of >250 s during the intervention and all patients without specific contraindications received standard antithrombotic therapy. Patients without antithrombotic therapy prior to admission received a loading dose of 500 mg aspirin intravenously and 300–600 mg clopidogrel or 180 mg of ticagrelor orally. During their hospital stay all patients received a standard dose of 100 mg aspirin and 75 mg clopidogrel or 2x90 mg ticagrelor daily. In patients with an indication for oral anticoagulation, aspirin was given as “triple therapy” during the hospital stay. Patients were discharged with dual therapy consisting of clopidogrel and reduced dose of an oral anticoagulant.
Data Collection and Geriatric Assessment
Patients were consecutively included, exclusion criteria were (1) patients refused to participate in the survey and (2) patients not suitable to obtain geriatric tests due to physical impairment and (3) patients not able to provide informed consent or to perform the various tests due to cognitive impairment. If the patient was not able to provide signed informed consent in case of care assistance, it was not substituted by a representative or the caregiver. Data were prospectively collected during admission within the first 48 hours.
Baseline clinical and laboratory data, past medical history, cardiac and non-cardiac concomitant diseases were analyzed together with established cardiovascular risk factors i.e. diabetes, hypertension, and dyslipidemia.
Patients were stratified according to their cardiac risk based on the Euro-score,14 Killip-class15 and the Grace risk score (Global Registry of Acute Coronary Events).16
We evaluated any serious adverse events during the in-hospital stay such as death, MI, and complications during his/her hospital stay due to bleeding, reinfarction, need for mechanical ventilation, respiratory tract infection, acute renal failure and resuscitation.
Follow-up after 6 months was done by clinical appointment either in the hospital, at the patients´ home, or in the nursery home. During this visit, all above-mentioned tests were repeated for comparison with baseline values.
Geriatric assessment was conducted within the first 48 hours of admission, assessing comorbidity with the Charlson comorbidity index,17 cognitive function with the Mini-Mental State Examination (MMS),18 Clinical Frailty Scale (CFS) for the extent of frailty,19 Barthel index,20 “timed up and go” test (TUG) for gait function21 and the instrumental activities of daily living (IADL).22 The extent of depression was measured with the Geriatric Depression Scale.23 The institutional board of the University of Erlangen approved the protocol prior to recruitment. This study was conducted according to the Declaration of Helsinki in its latest form.
Statistical Analysis
For dichotomous variables, either the two-sided Fisher’s exact test or the Chi2 statistic was used whenever applicable. In the case of continuous variables, the unpaired t-test was used. In case the Shapiro–Wilk test revealed a strong deviation from a normal distribution, the Mann–Whitney U-test was preferred for non-Gaussian data. Logistic regressions were done with the dependent variable of angiography done (yes/no) while independent variables also included patient demographics and the used scales and scores. The significance level α of 0.05 was used for all tests. For all descriptive statistical analyses, SPSS version 24.0 (IBM, Munich, Germany) was used.
Results
A total of 106 consecutive patients >75 years admitted for ACS between January 2017 and March 2018 were included. Of these, 60 (56.6%) were male, 46 (43.4%) were female. Of all patients 38 patients (35.9%, mean age 83.5±6.0 years) were treated conservatively, 68 (64.1%) were transferred to angiography. In these 65 patients (95.6%) PCI was performed. Mean systolic blood pressure at admission was 147±23 mmHg, diastolic blood pressure 81±16 mmHg, mean heart rate was 88±25 beats/minute. Ejection fraction, measured angiographically or with echocardiography was >60% in 53 (56.4%) patients.
Baseline demographic characteristics were very similar in the two groups (Table 1). Patients, who had angiography, were significantly younger as compared to patients who were treated conservatively. Male gender was predominant in the overall cohort, ie, more male patients received angiography as compared to female patients. All other demographic data were not significantly different.
 |
Table 1 Baseline Demographic Data |
Angiographic Data
Coronary angiography was conducted in 68 (64.2%) of all patients. Of those, in 54 patients (79.4%) angiography was performed within the first 72 hours. PCI was done in 51 patients (75%), in 38 patients (55.9%) one vessel, in 12 patients (17.6%) two vessels, and in 1 patient (1.5%) 3 vessels were treated during the index procedure. In all patients, procedures were successful with the implantation of at least one stent. None of the patients were treated with balloon angioplasty alone.
Geriatric Assessment at Baseline
Euro-score, Killip-class and Grace-score were assessed in all patients. There were no significant differences found between both groups. Mini-Mental state test for dementia was 25.2±5.8 for all patients, showing mild cognitive impairment for all patients, but it was not different between patients managed conservatively or invasively. Geriatric Depression Scale was 4.4±2.8, indicating no prevalent depression in our patients. Clinical Frailty Scale (CFS) was 4.0±2.0 in patients not forwarded to angiography and 3.4±1.6 in patients managed invasively [non-frail (CFS 1-4), mild-to-moderately frail (CFS 5-6), and severely frail (CFS 7-8)] and therefore in both groups in average patients were not frail. IADL were less present in patients managed conservatively (5.4±2.7 vs 6.3±2.5, p=0.087). Comorbidity as measured with the Charlson comorbidity index was 3.2±2.5 vs 3.0±1.9 (p=0.691) (Table 2, Figure 1)
 |
Table 2 Geriatric Scores According to the Type of Diagnosis and Treatment |
 |
Figure 1 Overview of the Geriatric Scores According to the Type of DIagnosis and Treatment. |
Predictors of Choice of Treatment
The purpose of this analysis was, to identify factors, which possibly could impact the decision to treat the patients either interventionally or conservatively. To identify these potential confounders, a logistic regression analysis was performed on the basis of the available demographic data and the results of the specific geriatric tests. Age was identified as a significant single predictor for conservative treatment. Of all other items, individual tests were not able to predict treatment. A frailty index, consisting of commonly used parameters of functional impairment in elderly patients, namely, MMS at baseline ≤2, IADL ≤ 7, Clinical Frailty Scale ≥7 and age ≥85 years significantly predicted conservative treatment (Table 3).
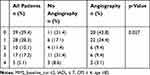 |
Table 3 Predictors of Angiography According to Level of Dementia, Activities of Daily Living, Clinical Frailty Scale and Age |
In-Hospital Mortality Rates and Complications
A total of 4 deaths (3.8%) occurred during hospitalization, the overall mortality was higher in patients who had angiography, but this finding was not significant (p=0.127). Bleeding was similar in both groups (2 patients [5.3%] vs 3 patients [4.4%], p=0.841), the incidence of acute renal failure was higher in the conservative group (2 patients [5.3%] vs 1 patient [1.5%], p=0.259). There was no in-hospital MI in either groups (Table 4).
 |
Table 4 In-Hospital Outcomes |
Discussion
The purpose of this study was to evaluate the process of physicians’ decision-making when treating elderly ACS patients, since there is a paucity of clinical evidence for the optimal management of frail and comorbid elderly patients. One of the main results of our study is that age is still a risk factor leading to less referrals to interventional treatment in ACS patients. Secondly, individual geriatric tests may not predict treatment in these patients. However, the combination of different tests may better predict treatment and, possible outcomes in this patient group. In the present study, a MMS ≤ 2, IADL ≤ 7 and a CFS of higher than 5 independently predicted conservative treatment of ACS. In addition, our study shows, that frail patients are at higher risk for not receiving guideline-recommended therapy. It is currently being discussed, if ACS patients with a high frailty index benefit from invasive treatment.24 This study revealed that dementia was part of risk evaluation in elderly ACS patients and that after the inclusion of the MMS test, our score was able to predict the type of treatment. Therefore, the addition of the dementia severity in risk evaluation ought to be part of the risk stratification, even for outcome measurements.
Evidence of how to deal with this patient group is still limited but has been growing in recent years. There are only a few randomized trials to compare the different treatment strategies in elderly ACS patients. The significant heterogeneity in this patient group allows only a minority of possible candidates to participate in randomised trials. This is underlined by high exclusion rates in some trials (48.5% of screened patients included in the Italian Elderly ACS trial25 and 10.9% in the After Eighty trial3). Furthermore, this pronounced diversity in patients might confine selection criteria for different tests. Therefore, even in this study, included patients differed to some extent substantially from real-life ACS patients who were treated at community hospitals. As a result, the applicability of a specific test in different clinical and individual scenarios for this heterogeneous patient group greatly limits the applicability for each test or score. Therefore, even the applicability of a screening score consisting of several tests will only have limited value.
There is growing evidence to treat elderly ACS patients. In the After-Eighty Study,3 patients >80 years presenting with ACS were randomized to either conservative or interventional treatment in 16 Norwegian hospitals. The primary outcome (composite of MI, revascularization, stroke and death) was significantly in favour of interventionally treated patients (457 patients included, primary endpoint 40.6% vs 61.4%). Prevalence of comorbidities and frailty in patients was low. This study had important in- and exclusion criteria, eg, ongoing angina was an exclusion criterion. The Italian ACS study randomized 313 patients >75 years presenting with NSTEMI-ACS to interventional or initially conservative treatment.25 The primary endpoint was a composite of death, MI, disabling stroke and hospitalization or severe bleeding and occurred in 27.9% of the invasive and 34.6% of the conservative group (p=0.260). In contrary to these two studies, with a high prevalence of relatively healthy participants, the MOSCA trial included 106 patients >60 years with a higher proportion of patients with comorbidities.8 After 2.5 years, the authors did not find a significant difference between groups regarding the combined endpoint consisting of all-cause mortality, reinfarction and readmission.
Alonso Salinas et al5 found in 234 ACS patients aged >75 years, that frailty was an independent predictor of the combination of death or non-fatal myocardial infarction. Of 234 patients, 40.2% fulfilled the frailty criteria and those patients had a higher risk profile according to age and worse outcomes regardless of treatment type.
The LONGEVO-SCA registry included NSTE-ACS patients >80 years while a geriatric assessment during the first 72 hours of hospitalization26 was conducted. Of 531 patients, 27.3% were frail, 76.6% of all patients underwent invasive assessment. Even in this registry, patients who underwent the invasive strategy were younger and a lower proportion of those were frail. Conservative management in non-frail patients was strongly associated with a higher incidence of the primary endpoint (sHR 3.85, 95% CI 2.13–6.95, p=0.001). In contrast, no significant association was observed in patients with established frailty criteria (sHR1.40, 95% CI 0.72–2.75, p=0.325). Interestingly, this association remained significant in the overall cohort and in non-frail patients but not in frail persons. The interaction invasive strategy-frailty was significant (p=0.032). This registry was the first to show that the success of the interventional treatment might be extenuated with the extent of frailty. The authors concluded that with an increasing degree of frailty, the benefit of an invasive therapy decreases. Moreover, they recommended larger studies and randomized trials to clarify the role of intervention in these patients are mandatory.
Campo et al showed in 402 patients >70 years with ACS that the assessment of the physical performance with SPPB scale before hospital discharge in addition to the GRACE- and TIMI risk scores increased the ability to predict adverse events in older ACS patients. They concluded that this might be useful in the clinical decision-making process.27
Even non-cardiovascular comorbidities (NCC) like diabetes, renal disease or anemia can provoke worse outcomes. Ofori-Asenso et al showed in 1488 older adults hospitalized for NSTE-ACS that having ≥2 NCC was associated with a significantly increased likelihood of longer hospital stays and in-hospital death rates (HR 1.79, 95% CI: 1.06–3.03; p=0.029), respectively.28
The patient number in the present study was too low to show significant differences in in-hospital outcomes, but interestingly the presence of dementia was a predictor for the choice of therapy. Again this reflects the problem in decision-making when elderly patients are treated. The pivotal question of which test is suitable for which individual patient is highly pertinent.
Current guidelines emphasize individualized treatment in elderly patients, respecting the overall condition and the comorbidity burden, but decision-making in the face of frail patients is complex. Our data underline the fact that this decision-making is highly individual respecting the patient, patient wishes and even the wishes of patients’ relatives.
Analogously, the large multinational observational global registry of acute coronary events (GRACE) has been used to derive regression models to predict death in hospital and death after discharge in patients with ACS,29,30 but since “numerical” age is a major risk factor in this model and part of the risk score itself, a special risk score for decision-making in elderly patients should be developed. This stratification should aim to identify elderly patients with ACS, who are suitable for reperfusion therapy with the largest survival benefit. But not only survival in this patient group is of paramount interest. The improvement of functional outcomes should also be integrated into risk estimation.
Limitations
This is a non-randomized, single-centre, observational study with 106 patients and relatively low event rates and low sample sizes in each group. Since this was an observational study, we are not able to rule out any selection bias. Due to the heterogeneity of the elderly population, recruitment of patients was slow, and total number of included patients remained low due to several circumstances like physical or cognitive impairment and resulting inability to perform the array of tests. According to the design of this observational study, there is a considerable selection bias regarding treatment, which remains a limitation of this study.
Because of the explorative nature of this study, more statistical tools could have been used to adjust for potential confounders, which was not done due to the small sample size.
Conclusion
Effective revascularization techniques are still underused in patients with high “numerical” age in the case of ACS. Furthermore, frail patients are at higher risk for not receiving guideline-recommended therapy. With the absence of randomized data, which guide decision-making regarding useful therapies in the case of NSTEMI-ACS in elderly frail patients, our results show, that the combination of different geriatric tests better predicted the chosen treatment. A randomized outcome trial, specifically designed for elderly frail patients with NSTEMI-ACS, to guide decision-making in this patient group is inevitably warranted.
Abbreviations
ACS, acute coronary syndrome; CAD, coronary artery disease; CFS, Clinical Frailty Scale; COPD, chronic obstructive pulmonary disease; GDS, Geriatric Depression Scale; ECG, electro cardiogram; DES, drug-eluting stent; hsTNT, high sensitivity Troponin T; IADL, instrumental activities of daily living; NSTEMI-ACS, non-ST-elevation myocardial infarction-ACS; MI, myocardial infarction; MMS, Mini-Mental Status Test; PCI, percutaneous coronary intervention; TUG, timed up and go test.
Disclosure
Thomas Kieschnick is employed by Klinikum Fürth Kardiologie and received monthly payment for works at the hospital ward. Matthias Waliszewski is affiliated with B. Braun. The authors report no other conflicts of interest in this work.
References
1. Ofori-Asenso R, Chin KL, Mazidi M, et al. Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(8):e198398. doi:10.1001/jamanetworkopen.2019.8398
2. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40:87–165.
3. Tegn N, Abdelnoor M, Aaberge L, et al.; After Eghty study investigators. Invasive versus conservative strategy in patients aged 80 years or older with non-ST-Elevation myocardial infarction or unstable angina pectoris (After Eighty study): an open-label randomized controlled trial. Lancet. 387;2016:1057–1065. doi:10.1016/S0140-6736(15)01166-6
4. Khandelwal D, Goel A, Kumar U, Gulati V, Narang R, Dey AB. Frailty is associated with longer hospital stay and increased mortality in hospitalized older patients. J Nutr Health Aging. 2012;16:732–735. doi:10.1007/s12603-012-0369-5
5. Alonso Salinas GL, Sanmartin Fernandez M, Pascual Izco M, et al. Frailty is a short-term prognostic marker in acute coronary syndrome in elderly patients. Eur Heart J Acute Cardiovasc Care. 2016;5:434–440. doi:10.1177/2048872616644909
6. Nunez J, Ruiz V, Bonanad C, et al. Percutaneous coronary intervention and recurrent hospitalizations in elderly patients with non ST-segment acute coronary syndrome: the role of frailty. Int J Cardiol. 2017;228:456–458. doi:10.1016/j.ijcard.2016.11.151
7. Graham MM, Galbraith PD, O´Neill D, Rolfson DB, Dando C, Norris CM. Frailty and outcome in elderly patients with acute coronary syndrome. Can J Cardiol. 2013;29:1610–1615. doi:10.1016/j.cjca.2013.08.016
8. Ekerstad N, Swahn E, Janzon M, et al. Frailty is independently associated with short.term outcomes for elderly patients with non-ST-segment elevation myocardial infarction. Circulation. 2011;124:2397–2404. doi:10.1161/CIRCULATIONAHA.111.025452
9. Sanchis J, Nunez E, Barrabés E, et al. Randomized comparison between the invasive and conservative strategies in comorbid elderly patients with non-St elevation myocardial infarction. Eur J Intern Med. 2016;35:89–94. doi:10.1016/j.ejim.2016.07.003
10. Sánchez E, Vidán MT, Serra JA, et al. Prevalence of geriatric syndromes and impact on clinical and functional outcomes in older patients with acute cardiac diseases. Heart. 2011;97:1602–1606. doi:10.1136/hrt.2011.227504
11. Noriega FJ, Vidán MT, Sánchez E, et al. Incidence and impact of delirium on clinical and functional outcomes in older patients hospitalized for acute cardiac diseases. Am Heart J. 2015;170:938–944. doi:10.1016/j.ahj.2015.08.007
12. Afilalo J, Alexander KP, Mack MJ, et al. Frailty assessment in the cardiovascular care of older adults. J Am Coll Cardiol. 2014;68:747–762. doi:10.1016/j.jacc.2013.09.070
13. Alonso Salinas GL, Sanmartin M, Pascual Izco M, et al. Frailty is an independent prognostic marker in elderly patients with myocardial infarction. Clin Cardiol. 2017;40:925–931. doi:10.1002/clc.22749
14. Nashef SAM, Roques F, Sharples LD, et al. EuroSCORE II. Eur J Cardiothorac Surg. 2012;41:734–745. doi:10.1093/ejcts/ezs043
15. Killip T, Kimball JT. Treatment of myocardial infarction in a coronary care unit: a two year experience of 250 patients. Am J Cardiol. 1967;20:457–464. doi:10.1016/0002-9149(67)90023-9
16. Granger CB, Goldberg RJ, Dabbous O, et al.; Global Registry of Acute Coronary Events Investigators. Predictors of hospital mortality in the global registry of acute coronary events. Arch Intern Med. 2003;163:2345–2353. doi:10.1001/archinte.163.19.2345
17. Charlson M, Pompei P, Ales K, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383. doi:10.1016/0021-9681(87)90171-8
18. Folstein MF, Folstein SE, McHugh PR. Mini-mental state: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. doi:10.1016/0022-3956(75)90026-6
19. Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol. 2007;62A:722–727. doi:10.1093/gerona/62.7.722
20. Mahoney FI, Barthel DW. Functional evaluation: the Barthel index. Md State Med J. 1965;14:61–65.
21. Podsiadlo D, The Timed RS. “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–148. doi:10.1111/j.1532-5415.1991.tb01616.x
22. Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179–186. doi:10.1093/geront/9.3_Part_1.179
23. Yesavage JA, Brink TL, Rose TL, et al. Development and validation of a geriatric depression screening scale: a preliminary report. J Psychiatr Res. 1982;17:37–49. doi:10.1016/0022-3956(82)90033-4
24. Alegre O, Ariza-Solé A, Vidán MT, et al. Impact of Frailty and Other Geriatric Syndromes on Clinical Management and Outcomes in Elderly Patients With Non-ST-Segment Elevation Acute Coronary Syndromes: rationale and Design of the LONGEVO-SCA Registry. Clin Cardiol. 2016;397:373–377. doi:10.1002/clc.22550
25. Savonitto S, Cavallini C, Petronio AS, et al.; Italian Elderly ACS Trial Investigators. Early aggressive versus initially conservative treatment in elderly patients with non-ST-segment elevation acute coronary syndrome: a randomized controlled trial. JACC Cardiovasc Interv. 5;2012:906–16 elderly trial. doi:10.1016/j.jcin.2012.06.008
26. Llao I, Ariza-Solé A, Sanchis J, et al. Invasive strategy and frailty in very elderly patients with acute coronary syndromes. Eurointervention. 2018;14:336–342. doi:10.4244/EIJ-D-18-00099
27. Campo G, Maietti E, Tonet E, et al. The assessment of scales of frailty and physical performance improves prediction of major adverse cardiac events in older adults with acute coronary syndrome. J Gerontol a Biol Sci Med Sci. 2019. doi:10.1093/gerona/glz123
28. Ofori-Asenso R, Zomer E, Chin KL, et al. Prevalence and impact of non-cardiovascular comorbidities among older adults hospitalized for non-ST segment elevation acute coronary syndrome. Cardiovasc Diagn Ther. 2019;3:250–261. doi:10.21037/cdt.2019.04.06
29. Avezum A, Makdisse M, Spencer F, et al. Impact of age on management and outcome of ACS: observations from the GRACE registry. Am Heart J. 2005;149:67–73. doi:10.1016/j.ahj.2004.06.003
30. Devlin G, Gore JM, Elliott J, et al.; GRACE Investigators. Management and 6-month outcomes in elderly and very elderly patients with high-risk non-ST-elevation acute coronary syndromes: the Global Registry of Acute Coronary Events. Eur Heart J. 2008;10:1275–1282.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
