Back to Journals » Clinical and Experimental Gastroenterology » Volume 12
Iatrogenic bile duct injury: impact and management challenges
Authors Pesce A , Palmucci S , La Greca G , Puleo S
Received 23 November 2018
Accepted for publication 23 January 2019
Published 6 March 2019 Volume 2019:12 Pages 121—128
DOI https://doi.org/10.2147/CEG.S169492
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Everson L.A. Artifon
Antonio Pesce, Stefano Palmucci, Gaetano La Greca, Stefano Puleo
Department of Medical, Surgical Sciences and Advanced Technologies “G.F. Ingrassia”, University of Catania, Catania, Italy
Abstract: Iatrogenic bile duct injuries (BDIs) after laparoscopic cholecystectomy, being one of the most common performed surgical procedures, remain a substantial problem in gastrointestinal surgery with a significant impact on patient’s quality of life. The primary aim of this review was to discuss the classification of BDIs, the proposed methods to prevent biliary lesions, the associated risk factors, and the management challenges depending on the timing of recognition of the injury, its extension, the patient’s clinical condition, and the availability of experienced hepatobiliary surgeons. Early recognition of BDI is of paramount importance and limiting the diagnosis delay is crucial for an optimal postoperative outcome. The therapeutic management depends on the type and gravity of the biliary lesion, and includes endoscopic, radiologic, and surgical approaches.
Keywords: bile duct injury, laparoscopic cholecystectomy, biliary anatomy, early recognition, management challenge, endoscopic treatment, surgical repair
Introduction
Laparoscopic cholecystectomy (LC) is one of the most frequently performed procedures worldwide in general surgery.1 Annually, more than 750,000 procedures are performed in the US.2 Bile duct injury (BDI) represents the most serious complication of LC, with an incidence of 0.3%–0.7%, resulting in a significant impact on quality of life (QoL), overall survival, and frequent medico-legal liabilities.1–4 The incidence of iatrogenic BDI has significantly gone up since LC became the “gold standard” in the treatment of symptomatic cholelithiasis. It has been demonstrated that the primary cause of BDI is the misinterpretation of biliary anatomy in 71%–97% of all cases.1 Over the years, various classifications of biliary injuries have been proposed and different methods have been described to prevent iatrogenic biliary tract lesions. Early recognition of BDI is of paramount importance. The optimal treatment is influenced by the timing of recognition of the injury, the extent of BDI, the patient’s clinical condition, and the availability of experienced hepatobiliary surgeons. This article aims to discuss the classification and management challenges of iatrogenic BDIs after cholecystectomy.
Classification of BDIs
Before the advent of LC, the most frequent lesion to biliary tract was represented by bile duct stricture, and biliary injuries were classified using the “Bismuth classification”5 according to the level of biliary injury. This classification includes five types of bile duct lesions according to the distance from the hepatic hilus, the level of injury, the involvement of bile duct bifurcation, and individual right sectoral duct. Few years later, Strasberg et al6 proposed a new classification, similar to the Bismuth, but incorporating additional biliary injuries seen more commonly in the laparoscopic era, in particular biliary leaks. In the “Strasberg classification”, biliary injuries were stratified form type A to type E, as summarized in Table 1. McMahon et al7 proposed another classification by distinguishing major and minor biliary injuries: lacerations under 25% of the common bile duct (CBD) diameter or cystic duct-common hepatic duct (CD-CHD) junction were considered as minor injury, whereas transection or laceration over 25% of CBD diameter and postoperative bile duct stricture were classified as major injury. The “Stewart-Way classification”8 involved four classes based on the mechanism and anatomy of biliary injury, as shown in Figure 1. This classification arose from the analysis of operative reports, providing the human mistakes and cognitive processes involved in the mechanisms of BDIs. Class I injury occurs in two ways: 1) either the CBD is mistaken for the cystic duct, but the error is recognized before CBD is divided in 72% of cases, or 2) an incision made in the cystic duct for the cholangiogram catheter is unintentionally extended into the CBD (28% of cases). Class II injuries consist of lateral damage to the CHD with resultant stricture and/or leak. This type of injury occurs when clips or electro-cautery are used too close to the CBD, mainly in cases where visibility is limited due to severe inflammation or excessive bleeding. Class III injuries are the most common (about 60% of cases) and involve complete transection of the main bile duct, which always includes the CD-CHD junction. Class IV injuries include transection or leak of the right hepatic duct (RHD) or posterolateral sectoral duct, often combined with injury to the right hepatic artery.
  | Table 1 Bismuth and Strasberg classification Abbreviations: –, not applicable; RHD, right hepatic duct; CBD, common bile duct; CHD, common hepatic duct. |
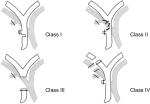  | Figure 1 Stewart-Way classification of bile duct injuries. Notes: Adapted with permission from Wolters Kluwer Health, Inc.: Way LW, Stewart L, Gantert W, et al. Causes and prevention of laparoscopic bile duct injuries: analysis of 252 cases from a human factors and cognitive psychology perspective. Ann Surg. 2003;237(4):460–469.8 Copyright © 2003 Lippincott Williams & Wilkins, Inc. Available from: https://journals.lww.com/annalsofsurgery/Abstract/2003/04000/Causes_and_Prevention_of_Laparoscopic_Bile_Duct.4.aspx. The Creative Commons license does not apply to this content. Use of the material in any format is prohibited without written permission from the publisher, Wolters Kluwer Health, Inc. Please contact [email protected] for further information. |
The “Amsterdam classification” proposed by Bergman et al in 19969 is also very commonly used in many articles, especially among endoscopists. This classification includes four types of ductal injury: type A with leakage from cystic ducts or peripheral hepatic radicles, type B presenting as major bile duct leakage, type C showing an isolated ductal stricture, and type D with complete transection of the bile duct.
For an easy reading, we have chosen the Stewart-Way classification system in this manuscript, as it better describes the mechanisms of biliary lesion during LC, including also concomitant vascular injuries.
Prevention of iatrogenic BDI
Over the years, various methods have been proposed and described in the scientific literature to prevent iatrogenic biliary tract lesions. They are summarized in Table 2. The “critical view of safety (CVS)” technique was introduced by Strasberg in 199510 and it is considered the gold standard to perform a safe cholecystectomy with identification of biliary structures during dissection. Three criteria are required to achieve the CVS: 1) the hepatocystic triangle must be cleared of adipose and fibrotic tissues; the CBD and CHD must not be exposed; 2) the lower third of the gallbladder must be separated from the liver bed to expose the cystic plate; and 3) two and only two structures should be seen entering the gallbladder. CVS was conceived not as a way to do LC but as a way to avoid biliary injury.
  | Table 2 Summary of proposed methods to prevent bile duct injuries Note: B-SAFE: B, bile duct; S, sulcus of Rouvière; A, hepatic artery; F, umbilical fissure; E, enteric/duodenum. |
In such cases, some surgeons prefer performing the infundibular method in order to work very close to gallbladder infundibulum, reducing the risk of biliary injuries, but care should be taken with “hidden cystic duct” syndrome which presents a deceptive appearance of a false infundibulum that misleads the surgeon into identifying the CBD as the cystic duct.11 The “antegrade dissection or fundus first/dome-down technique” represents a way of dissection from the gallbladder fundus up to the infundibulum away from Calot’s triangle.12 In this way, the gallbladder is left pedunculated by the cystic artery and cystic duct, which can be clipped and divided in turn, reducing the risk of biliary lesions. Another easy, safe, and definitive operation for the “difficult gallbladder” is described and has been termed “subtotal or partial cholecystectomy”,13 consisting in leaving the posterior wall of the gallbladder attached to the liver and securing the cystic duct at its origin from within the gallbladder.
Different anatomic landmarks have been described: among them, in 1924, Henri Rouvière, a French surgeon, described a 2–5 cm sulcus running to the right of liver hilum, anterior to the caudate lobe, and usually containing the right portal triad or its branches.14 It can be considered a useful landmark site to start dissection of the hepatocystic triangle during LC.15 The “Rouvière’s sulcus” is present in the majority of patients, ranging from 68% to 90%,16 and is clearly visible during laparoscopy, being visible in one form or the other (a sulcus, a slit, or a scar). Another anatomic landmark to guide gallbladder dissection is represented by the “cystic lymph node” or Mascagni’s node, which always lies lateral to the biliary tree and should form the medial end point of dissection.17
Some authors proposed a mnemonic “B-SAFE method”, by using five anatomic landmarks (B, bile duct; S, sulcus of Rouvière; A, hepatic artery; F, umbilical fissure; E, enteric/duodenum) to correctly place their cognitive map during dissection.18
The routine use of “intra-operative cholangiography (IOC)” has been proposed for the better declaration of biliary anatomy, detection of silent CBD stones, and reduction of incidence of BDIs, but it is burdened by morbidity and mortality and hence should be performed in facilities where the necessary equipment and experience are available.1,19,20 The opinions about the “routine” or “selective” use of IOC still represent a matter of debate.1,19
“Laparoscopic ultrasound (LUS)” was shown to provide highly sensitive mapping of the extra-hepatic biliary anatomy but the difficult learning curve and the lack of randomized controlled trials have reduced its use in clinical practice.21,22
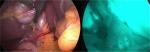
One of the latest innovations in minimally invasive technology is fluorescence image-guided surgery; “near-infrared fluorescent cholangiography (NIRF-C)” represents a novel intra-operative imaging technique that allows a real-time enhanced visualization of the extrahepatic biliary tree by fluorescence.23–25 NIRF-C well represents a useful method to identify the CVS with the aid of real-time fluorescent vision with a perfect combination between surgical anatomy and technical innovation, as shown in Figure 2.26
In some difficult situations, the opinion of a surgical colleague in vicinity during operation may also represent a valid alternative, the so-called “colleaguography”, in place of IOC or other methods, as proposed by some authors.27
Despite the plethora of publications and debates, there is still no consensus as to which method is the best to prevent BDIs during LC. Recently, highest consensus was achieved on the importance of the CVS and NIRF-C, as well as antegrade dissection or fundus first/dome-down technique and partial cholecystectomy as alternative techniques.28,29
A general recommendation is that in all cases of complicated cholecystectomies, the surgeon must not hesitate when considering conversion to open approach because there is no substitute for experience and caution in biliary surgery.
Risk factors
There are several risk factors which can contribute to the iatrogenic injury of the biliary tract: anatomical factors; patient-related factors; and factors related to the gallbladder disease, the surgical technique, and the surgeon. Among the anatomical factors, the numerous anatomical variants of the biliary tract represent a possible cause of iatrogenic lesion, for example, the different variants of the cystic duct, such as short cystic duct, cystic duct running parallel to the CBD, anomalies of the CD-CHD junction, presence of the hepatocystic duct, accessory cystic duct, the existence of aberrant bile ducts (eg, Luschka duct).30 Among the patient-related factors, severe obesity, previous surgery on the biliary tract, and underlying liver disease seem to be predisposing factors for peri-operative complications.
However, acute cholecystitis causes a series of modifications of the local anatomy (adhesions, thickening of the tissues, inflammation, bleeding) that are associated with an increased risk of iatrogenic lesion.31,32 Among the factors related to the technique, the laparoscopic approach itself represents a risk factor, hence the importance of a correct “learning curve” for young surgeons.33,34 The human factors also play a crucial role in the pathophysiology of the iatrogenic bile lesion: the excessive safety of the surgeon, the rush to finish the intervention, the fatigue and personal concerns of the surgeon, the performance anxiety, the superficiality of the surgical act, and the lack of humility in converting to open surgery in doubtful cases may determine a relevant damage to the biliary tree.8–35
LC can be seen as a trivial intervention but it could hide many difficulties with dramatic consequences. For these reasons, caution and experience are essential in biliary surgery.
Clinical presentation
Clinical presentation depends essentially on the extent and type of iatrogenic lesion, if this is not discovered intra-operatively. The presence of a possible biliary leak can start with non-specific symptoms, such as nausea, vomiting, bloating, widespread abdominal pain, general discomfort, and anorexia. Rarely, the patient presents the clinical picture of biliary peritonitis, despite large amounts of bile in the abdomen. The evolution of biliary leak is very subtle, because of the presence of these vague symptoms. Patients with stricture at level of the CBD, in cases where the CBD is mistaken for the cystic duct, will present the classic Charcot’s triad with pain in the right hypochondrium, fever, and jaundice. A general recommendation is that any alteration in the normal postoperative course after LC must suggest a possible damage to the biliary tract. For these reasons, a careful clinical evaluation of patients is essential. A delay in the diagnosis is crucial for the postoperative outcome.
Timing of recognition
Early recognition of BDI is of paramount importance. Intra-operative recognition should typically prompt immediate surgical repair as this confers the best outcomes; however, <25%–30% of BDIs are recognized during operation and not all surgeons perform an intra-operative cholangiography.36,37 If the surgeon is inexperienced, an intra-abdominal drain should be placed, and the patient should be immediately transferred to a high-volume hepatobiliary center. There are several data suggesting that these injuries are best managed by a surgeon with expertise in biliary reconstruction with better outcomes.37–39 There are several factors that lead to a recognition of intra-operative lesion, but the most important is the surgeon’s awareness to suspect and/or evaluate for a BDI. Diagnosis often occurs in the immediate postoperative period (within 6 weeks post-intervention) or, in some cases, later (over 6 weeks). A careful clinical evaluation helps for a correct diagnosis. The appearance of persistent or abnormal abdominal pain in the right hypochondrium, the leakage of bile from drainage (if placed during surgery), a picture of biliary peritonitis, and the appearance of fever and jaundice with increased liver function tests are all elements supporting a diagnosis of any iatrogenic lesion of the biliary tree. Radiologic investigations should be obtained for the correct identification of the damage, its extension, and gravity and to plan therapeutic strategies. Abdominal ultrasound represents the first diagnostic tool aimed at finding any liver collections, dilatation of the CBD, and any associated vascular lesions.40 The abdominal computed tomography (CT) scan is the principal investigation that allows to define the possible presence of focal intra- or peri-hepatic fluid collections, ascites, biliary obstruction with upstream dilatation, or long-term sequelae of a long-standing bile stricture, such as lobar hepatic atrophy or signs of secondary biliary cirrhosis. The CT scan also allows to identify any associated vascular lesions, such as to the right hepatic artery.
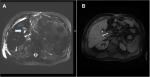
Magnetic resonance cholangio-pancreatography (MRCP) represents the “gold standard” for the complete morphological evaluation of the biliary tree as it offers detailed information about the integrity of the biliary tract.41,42 MRCP can also be done with a contrast agent (a gadolinium-based contrast agent, the gadoxetic acid and its salt, gadoxetate disodium), and it is much more informative in bile leaks than plain MRCP.42 The use of a contrast agent during MRCP allows detection of active bile leakage by direct visualization of contrast material extravasation into fluid collections in addition to demonstrating the anatomical site of the leakage and the type of BDI. For example, in Figure 3 cystic duct leak was demonstrated at MRCP after gadoxetic acid injection. In the same way, hepatobiliary scintigraphy may confirm the presence of an active biliary leak with the progressive accumulation of the radiotracer inside the peritoneal cavity, but this examination is much less reliable. However, if fluid collection is found in ultrasound, CT, or MRCP, an abdominal drain should be placed right away under radiological guidance in order to improve patient’s clinical condition (peritonitis, sepsis, tissue repairing). If there is a delay of more than a week in operative treatment of Stewart-Way Class III or IV injuries, there should be a timeout for 2–3 months before operation. In this time frame, the patient needs to be stabilized and optimized for surgery. An optimal control of any intra-abdominal fluid collection, inflammation, and infection is essential, and is best achieved with percutaneous drainage.37
In the postoperative period, re-laparoscopy can identify the biliary injury, drainage of any intra-abdominal collection, and exclusion of injury to other surrounding organs, which can lead to the decision between conservative management and early or late definitive surgical repair.43
There is no role for explorative laparotomy, as this is associated with increased morbidity and mortality.
Therapeutic management
The endoscopic retrograde cholangio-pancreatography (ERCP) examination has an important therapeutic role in cases where a conservative therapeutic approach is preferred based on the type and extent of the biliary lesion (cystic duct leaks, Stewart-Way Class I injuries, and most Class II injuries).
Cystic duct leaks are well managed conservatively; the endoscopic procedure allows to perform a sphincterotomy with the aim of reducing the pressure in the biliary tree and to place a plastic stent or a naso-biliary tube to limit or exclude the fistula (depending on the location of the biliary leak). After the right patient selection, the success rate of endoscopic treatment can approach 100% for cystic duct or peripheral radical leaks. Endoscopic sphincterotomy is an effective and cost-effective single procedure with success rate similar to endoscopic sphincterotomy and biliary stenting, as proposed by Rainio et al.44 Self-expanding biodegradable biliary stents have only recently become available in order to avoid second endoscopy with good results.45 The limits of the endoscopic treatment concern the lack of visualization of aberrant or sectioned bile ducts which do not communicate with the CBD (eg, an aberrant RHD), the cases of complete transection of the bile duct, and the difficulty in visualization of possible intra-hepatic proximal leaks. In this specific case, the percutaneous transhepatic cholangiography (PTC) is superior in the definition of the lesion and in the biliary drainage.
Stewart-Way Class I injuries, which are by definition recognized intra-operatively, can be immediately repaired by using fine monofilament absorbable suture. Insertion of a T-tube catheter is rather contraindicated, as extension of the laceration to facilitate T-tube insertion results in worsening and progression of the injury and an increased likelihood of stricture.37,46
Stewart-Way Class II injuries consist of lateral damage to the CHD with resultant stricture and/or leak. The management of biliary leak requires a multidisciplinary approach with endoscopy and radiological-guided drainage as the first therapeutic options. Stewart-Way Class II injuries with stricture can be treated with multiple plastic stents and self-expanding covered metallic or biodegradable stents, depending on the gravity of biliary lesion.45
Stewart-Way Class III and IV treatment is mostly operative, but even in such conditions Stewart-Way Class III lesions can also be treated mini-invasively by endoscopic and radiologic extra-anatomical rendezvous reconstruction.47,48 Fiocca et al48 proposed a combined endoscopic–radiologic rendezvous technique to treat the complete transection of the main bile duct, as it is possible to avoid difficult surgical reintervention that presents some morbidity and mortality.
Class IV injuries that involve a sectoral bile duct without transection of the duct can often be managed non-operatively, with drainage and stenting via either ERCP or PTC, with good outcomes in many cases.49–53 Class IV injuries involving transection of the bile ducts require reconstruction of the duct (either the RHD or a right sectoral duct) into a defunctionalized Roux limb.
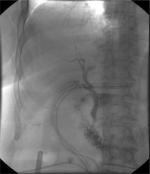
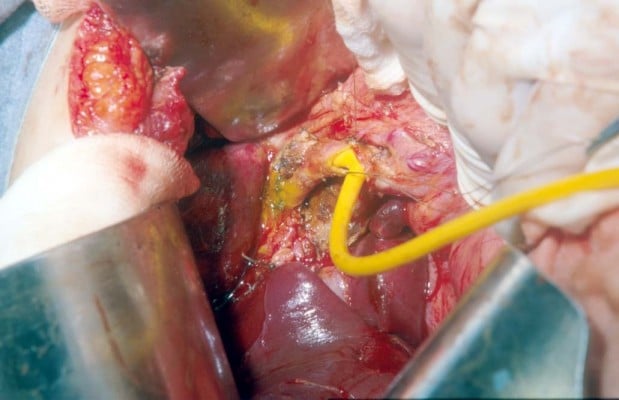
With regard to reparative surgery, there are some basic rules, and the difficulty in repairing damage derives from both the size and caliber of the bile duct and from local inflammatory and sclerotic changes. The timing of the operation is an extremely important factor in the repair of the lesion; therefore, an early identification of the lesion plays a key role. In case of partial or complete transection of the CBD, it is possible to perform a direct end-to-end suture if the distal choledochus is free and without tension. It is also preferable to protect the suture by inserting a Kehr T-tube, as shown in Figure 4. There is also a second type of T-tube available for biliary drainage, the so-called internal Y-drainage,54 which involves the insertion of the two short branches into both the right and left hepatic ducts, splinting of the anastomosis, and conducting of its long branch into the duodenum by the papilla of Vater. This tube can be removed endoscopically after healing of the end-to-end ductal anastomosis. Moreover, it was suggested as the drainage of choice in end-to-end ductal anastomosis.55–58 In case of impossibility to perform a direct suture or in cases of severe biliary strictures, a biliodigestive anastomosis is recommended, a Roux-en-Y hepaticojejunostomy in most cases, as demonstrated in Figure 5. If a hepaticojejunostomy cannot be performed due to the presence of dense adhesions with infected and friable tissues, some authors have proposed a combination of endoscopic biliary stenting and pedicled omental patch repair of the bile duct to control bile leak and sepsis as a bridging procedure to definitive hepaticojejunostomy.59 In case of strictures involving the bifurcation or left or RHD, bilateral hepaticojejunostomies may be necessary. For many years, biliary repair has been the prerogative of open surgery. Nowadays, the improvement in surgical technology has allowed to perform reconstruction of iatrogenic biliary lesions even by laparoscopic or robotic approaches with good results.60–62 In a recent paper, Cuendis-Velázquez et al61 have demonstrated that the robotic approach is similar to the laparoscopic technique regarding safety and efficacy in attaining primary patency of hepaticojejunostomy for bile duct repair.
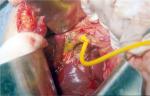  | Figure 5 A hepaticojejunostomy was performed for a stricture at biliary confluence after the positioning of T-tube. |
Liver resection is very rarely indicated in cases of extensive hepatic necrosis. In a small subset of patients with iatrogenic BDIs, failure of surgical or non-surgical management might lead to acute or chronic liver failure necessitating liver transplantation.63
Bismuth and Strasberg classifications do not include concomitant vascular injuries during LC. In the Stewart-Way classification system, the Class IV biliary injuries are frequently associated with concurrent right hepatic artery lesions. The right hepatic artery injuries may present with hepatic abscess, hemobilia, and right hepatic lobe ischemia.64 Many of these patients require angioembolization for treatment of hemobilia and drainage of hepatic abscesses, and some require hepatectomy.65–68
Furthermore, some authors have also analyzed the consequences of biliary injuries in terms of patient’s QoL. Flores-Rangel et al69 reported that the QoL of the patients undergoing a bilioenteric derivation due to a biliary injury, independently by surgeon’s experience, is reduced significantly when compared to patients undergoing uncomplicated cholecystectomy. Moreover, Booij et al70 have analyzed the long-term impact of BDIs on QoL and found that BDIs are associated with a significant decrease in patient’s QoL and work-related limitations.
Conclusion
Iatrogenic BDIs represent a serious complication which can be brought on by cholecystectomy. The errors leading to laparoscopic bile duct lesions stem principally from misperception of the biliary anatomy. Any effort toward the reduction of the risk profile of everyday cholecystectomy is appreciated. The key points to successful treatment are characterized by early recognition, control of any intra-abdominal fluid collection and infection, nutritional balance, multidisciplinary approach, and surgical repair by an experienced surgeon in biliary reconstruction.
Disclosure
The authors report no conflicts of interest in this work.
References
Pesce A, Portale TR, Minutolo V, Scilletta R, Li Destri G, Puleo S. Bile duct injury during laparoscopic cholecystectomy without intraoperative cholangiography: a retrospective study on 1,100 selected patients. Dig Surg. 2012;29(4):310–314. | ||
Archer SB, Brown DW, Smith CD, Branum GD, Hunter JG. Bile duct injury during laparoscopic cholecystectomy: results of a national survey. Ann Surg. 2001;234(4):549–558. | ||
Woods MS. Estimated costs of biliary tract complications in laparoscopic cholecystectomy based upon Medicare cost/charge ratios. A case-control study. Surg Endosc. 1996;10(10):1004–1007. | ||
Carroll BJ, Birth M, Phillips EH. Common bile duct injuries during laparoscopic cholecystectomy that result in litigation. Surg Endosc. 1998;12(4):310–314. | ||
Bismuth H, Majno PE. Biliary strictures: classification based on the principles of surgical treatment. World J Surg. 2001;25(10):1241–1244. | ||
Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg. 1995;180(1):101–125. | ||
McMahon AJ, Fullarton G, Baxter JN, O’Dwyer PJ. Bile duct injury and bile leakage in laparoscopic cholecystectomy. Br J Surg. 1995;82(3):307–313. | ||
Way LW, Stewart L, Gantert W, et al. Causes and prevention of laparoscopic bile duct injuries: analysis of 252 cases from a human factors and cognitive psychology perspective. Ann Surg. 2003;237(4):460–469. | ||
Bergman JJ, van den Brink GR, Rauws EA, et al. Treatment of bile duct lesions after laparoscopic cholecystectomy. Gut. 1996;38(1):141–147. | ||
Strasberg SM, Brunt LM. Rationale and use of the critical view of safety in laparoscopic cholecystectomy. J Am Coll Surg. 2010;211(1):132–138. | ||
Daly SC, Deziel DJ, Li X, et al. Current practices in biliary surgery: do we practice what we teach? Surg Endosc. 2016;30(8):3345–3350. | ||
Neri V, Ambrosi A, Fersini A, Tartaglia N, Valentino TP. Antegrade dissection in laparoscopic cholecystectomy. JSLS. 2007;11(2):225–228. | ||
Bornman PC, Terblanche J. Subtotal cholecystectomy: for the difficult gallbladder in portal hypertension and cholecystitis. Surgery. 1985;98(1):1–6. | ||
Rouvière H. On the configuration and the significance of the groove of the caudate process. Newsletter and Memoirs of The Anatomical Society of Paris. 1924;94:355–358. | ||
Hugh TB, Kelly MD, Mekisic A. Rouvière’s sulcus: a useful landmark in laparoscopic cholecystectomy. Br J Surg. 1997;84(9):1253–1254. | ||
Singh M, Prasad N. The anatomy of Rouviere’s sulcus as seen during laparoscopic cholecystectomy: a proposed classification. J Minim Access Surg. 2017;13(2):89–95. | ||
Ferzli G, Timoney M, Nazir S, Swedler D, Fingerhut A. Importance of the node of Calot in gallbladder neck dissection: an important landmark in the standardized approach to the laparoscopic cholecystectomy. J Laparoendosc Adv Surg Tech A. 2015;25(1):28–32. | ||
Sutherland F, Dixon E. The importance of cognitive MAP placement in bile duct injuries. Can J Surg. 2017;60(6):424–425. | ||
Flum DR, Dellinger EP, Cheadle A, Chan L, Koepsell T. Intraoperative cholangiography and risk of common bile duct injury during cholecystectomy. JAMA. 2003;289(13):1639–1644. | ||
Nickkholgh A, Soltaniyekta S, Kalbasi H. Routine versus selective intraoperative cholangiography during laparoscopic cholecystectomy: a survey of 2,130 patients undergoing laparoscopic cholecystectomy. Surg Endosc. 2006;20(6):868–874. | ||
Dili A, Bertrand C. Laparoscopic ultrasonography as an alternative to intraoperative cholangiography during laparoscopic cholecystectomy. World J Gastroenterol. 2017;23(29):5438–5450. | ||
Pesce A, Portale TR, di Stefano B, Costa S, Cammisuli F. Clinical value of intra-operative ultrasonography during laparoscopic cholecystectomy. Laparosc Surg. 2018;2:7. | ||
Ishizawa T, Bandai Y, Kokudo N. Fluorescent cholangiography using indocyanine green for laparoscopic cholecystectomy: an initial experience. Arch Surg. 2009;144(4):381–382. | ||
Pesce A, Piccolo G, La Greca G, Puleo S. Utility of fluorescent cholangiography during laparoscopic cholecystectomy: a systematic review. World J Gastroenterol. 2015;21(25):7877–7883. | ||
Pesce A, Latteri S, Barchitta M, et al. Near-infrared fluorescent cholangiography – real-time visualization of the biliary tree during elective laparoscopic cholecystectomy. HPB (Oxford). 2018;20(6):538–545. | ||
Pesce A, Diana M. Critical view of safety during laparoscopic cholecystectomy: from the surgeon’s eye to fluorescent vision. Surg Innov. 2018;25(3):197–198. | ||
Kapoor VK. “Colleaguography” in place of cholangiography, to prevent bile duct injury during laparoscopic cholecystectomy. J Minim Access Surg. Epub 2018 Aug 7. | ||
Conrad C, Wakabayashi G, Asbun HJ, et al. IRCAD recommendation on safe laparoscopic cholecystectomy. J Hepatobiliary Pancreat Sci. 2017;24(11):603–615. | ||
van de Graaf FW, Zaïmi I, Stassen LPS, Lange JF. Safe laparoscopic cholecystectomy: a systematic review of bile duct injury prevention. Int J Surg. 2018;60:164–172. | ||
Nuzzo G, Giuliante F, Giovannini I, et al. Bile duct injury during laparoscopic cholecystectomy: results of an Italian national survey on 56 591 cholecystectomies. Arch Surg. 2005;140(10):986–992. | ||
Kum CK, Eypasch E, Lefering R, Paul A, Neugebauer E, Troidl H. Laparoscopic cholecystectomy for acute cholecystitis: is it really safe? World J Surg. 1996;20(1):43–49; discussion 48-9. | ||
Georgiades CP, Mavromatis TN, Kourlaba GC, et al. Is inflammation a significant predictor of bile duct injury during laparoscopic cholecystectomy? Surg Endosc. 2008;22(9):1959–1964. | ||
Gigot J, Etienne J, Aerts R, et al. The dramatic reality of biliary tract injury during laparoscopic cholecystectomy. An anonimous multicenter Belgian survey of 65 patients. Surg Endosc. 1997;11:1171–1178. | ||
Moore MJ, Bennett CL. The learning curve for laparoscopic cholecystectomy. The Southern Surgeons Club. Am J Surg. 1995;170:55–59. | ||
Hugh TB. New strategies to prevent laparoscopic bile duct injury – surgeons can learn from pilots. Surgery. 2002;132(5):826–835. | ||
Seeras K, Kalani AD. Bile Duct Repair. Treasure Island, FL: StatPearls Publishing LLC; 2018. Available at: https://www.ncbi.nlm.nih.gov/books/NBK525989/. | ||
Stewart L. Iatrogenic biliary injuries: identification, classification, and management. Surg Clin North Am. 2014;94(2):297–310. | ||
Stewart L, Way LW. Bile duct injuries during laparoscopic cholecystectomy. Factors that influence the results of treatment. Arch Surg. 1995;130(10):1123–1128; discussion: 1129. | ||
de Reuver PR, Grossmann I, Busch OR, Obertop H, van Gulik TM, Gouma DJ. Referral pattern and timing of repair are risk factors for complications after reconstructive surgery for bile duct injury. Ann Surg. 2007;245(5):763–770. | ||
Latteri S, Malaguarnera G, Mannino M, et al. Ultrasound as point of care in management of polytrauma and its complication. J Ultrasound. 2017;20(2):171–177. | ||
Palmucci S, Mauro LA, Coppolino M, et al. Evaluation of the biliary and pancreatic system with 2D SSFSE, breathhold 3D FRFSE and respiratory-triggered 3D FRFSE sequences. Radiol Med. 2010;115(3):467–482. | ||
Palmucci S, Mauro LA, La Scola S, et al. Magnetic resonance cholangiopancreatography and contrast-enhanced magnetic resonance cholangiopancreatography versus endoscopic ultrasonography in the diagnosis of extrahepatic biliary pathology. Radiol Med. 2010;115(5):732–746. | ||
Gupta V, Jayaraman S. Role for laparoscopy in the management of bile duct injuries. Can J Surg. 2017;60(5):300–304. | ||
Rainio M, Lindström O, Udd M, Haapamäki C, Nordin A, Kylänpää L. Endoscopic therapy of biliary injury after cholecystectomy. Dig Dis Sci. 2018;63(2):474–480. | ||
Siiki A, Vaalavuo Y, Antila A, et al. Biodegradable biliary stents preferable to plastic stent therapy in post-cholecystectomy bile leak and avoid second endoscopy. Scand J Gastroenterol. 2018;5:1–5. | ||
Podda M, Polignano FM, Luhmann A, Wilson MS, Kulli C, Tait IS. Systematic review with meta-analysis of studies comparing primary duct closure and T-tube drainage after laparoscopic common bile duct exploration for choledocholithiasis. Surg Endosc. 2016;30(3):845–861. | ||
Donatelli G, Vergeau BM, Derhy S, et al. Combined endoscopic and radiologic approach for complex bile duct injuries (with video). Gastrointest Endosc. 2014;79(5):855–864. | ||
Fiocca F, Salvatori FM, Fanelli F, et al. Complete transection of the main bile duct: minimally invasive treatment with an endoscopic-radiologic rendezvous. Gastrointest Endosc. 2011;74(6):1393–1398. | ||
Schizas D, Papaconstantinou D, Moris D, et al. Management of segmental bile duct injuries after cholecystectomy: a systematic review. J Gastrointest Surg. Epub 2018 Nov 6. | ||
Li J, Frilling A, Nadalin S, et al. Surgical management of segmental and sectoral bile duct injury after laparoscopic cholecystectomy: a challenging situation. J Gastrointest Surg. 2010;14(2):344–351. | ||
Mazer LM, Tapper EB, Sarmiento JM. Non-operative management of right posterior sectoral duct injury following laparoscopic cholecystectomy. J Gastrointest Surg. 2011;15(7):1237–1242. | ||
Perera MTPR, Monaco A, Silva MA, et al. Laparoscopic posterior sectoral bile duct injury: the emerging role of nonoperative management with improved long-term results after delayed diagnosis. Surg Endosc. 2011;25(8):2684–2691. | ||
Perini RF, Uflacker R, Cunningham JT, Selby JB, Adams D. Isolated right segmental hepatic duct injury following laparoscopic cholecystectomy. Cardiovasc Intervent Radiol. 2005;28(2):185–195. | ||
Jabłonska B. End-to-end ductal anastomosis in biliary reconstruction: indications and limitations. Can J Surg. 2014;57(4):271–277. | ||
Kapoor BS, Mauri G, Lorenz JM. Management of biliary strictures: state-of-the-art review. Radiology. 2018;289(3):590–603. | ||
Renz BW, Bösch F, Angele MK. Bile duct injury after cholecystectomy: surgical therapy. Visc Med. 2017;33(3):184–190. | ||
Jabłońska B, Lampe P, Olakowski M, et al. Hepaticojejunostomy vs end-to-end biliary reconstructions in the treatment of iatrogenic bile duct injuries. J Gastrointest Surg. 2009;13(6):1084–1093. | ||
Gazzaniga GM, Filauro M, Mori L. Surgical treatment of iatrogenic lesions of the proximal common bile duct. World J Surg. 2001;25(10):1254–1259. | ||
Ng JJ, Kow AWC. Pedicled omental patch as a bridging procedure for iatrogenic bile duct injury. World J Gastroenterol. 2017;23(36):6741–6746. | ||
Cuendis-Velázquez A, Morales-Chávez C, Aguirre-Olmedo I, et al. Laparoscopic hepaticojejunostomy after bile duct injury. Surg Endosc. 2016;30(3):876–882. | ||
Cuendis-Velázquez A, Trejo-Ávila M, Bada-Yllán O, et al. A new era of bile duct repair: robotic-assisted versus laparoscopic hepaticojejunostomy. J Gastrointest Surg. Epub 2018 Nov 6. | ||
Giulianotti PC, Quadri P, Durgam S, Bianco FM. Reconstruction/repair of iatrogenic biliary injuries: is the robot offering a new option? short clinical report. Ann Surg. 2018;267(1):e7–e9. | ||
Parrilla P, Robles R, Varo E, et al. Liver transplantation for bile duct injury after open and laparoscopic cholecystectomy. Br J Surg. 2014;101(2):63–68. | ||
Halasz NA. Cholecystectomy and hepatic artery injuries. Arch Surg. 1991;126(2):137–138. | ||
Buell JF, Cronin DC, Funaki B, et al. Devastating and fatal complications associated with combined vascular and bile duct injuries during cholecystectomy. Arch Surg. 2002;137(6):703–710. | ||
Tzovaras G, Dervenis C. Vascular injuries in laparoscopic cholecystectomy: an underestimated problem. Dig Surg. 2006;23(5–6):370–374. | ||
Schmidt SC, Settmacher U, Langrehr JM, Neuhaus P. Management and outcome of patients with combined bile duct and hepatic arterial injuries after laparoscopic cholecystectomy. Surgery. 2004;135(6):613–618. | ||
Li J, Frilling A, Nadalin S, et al. Management of concomitant hepatic artery injury in patients with iatrogenic major bile duct injury after laparoscopic cholecystectomy. Br J Surg. 2008;95(4):460–465. | ||
Flores-Rangel GA, Chapa-Azuela O, Rosales AJ, Roca-Vasquez C, Böhm-González ST. Quality of life in patients with background of iatrogenic bile duct injury. World J Surg. 2018;42(9):2987–2991. | ||
Booij KAC, de Reuver PR, van Dieren S, et al. Long-term impact of bile duct injury on morbidity, mortality, quality of life, and work related limitations. Ann Surg. 2018;268(1):143–150. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.