Back to Journals » International Journal of Women's Health » Volume 9
Hysterosalpingo-foam sonography: patient selection and perspectives
Authors Rajesh H, Lim SL, Yu SL
Received 2 October 2016
Accepted for publication 24 November 2016
Published 28 December 2016 Volume 2017:9 Pages 23—32
DOI https://doi.org/10.2147/IJWH.S123710
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Elie Al-Chaer
Hemashree Rajesh, Serene Liqing Lim, Su Ling Yu
Centre of Assisted Reproduction, Department of Obstetrics and Gynecology, Singapore General Hospital, Singapore
Abstract: We present an overview of the common types of tubal patency tests, with a focus on hysterosalpingo-foam sonography (HyFoSy). Current evidence suggests that HyFoSy is an accurate alternative to X-ray hysterosalpingography (HSG) for outpatient tubal evaluation in women who are at low risk for tubal disease. It may be superior to saline hysterosalpingo-contrast sonography (HyCoSy) in excluding tubal occlusion. A hyperechogenic medium may enhance contrast visualization and enable clearer delineation of tubal anatomy. This may enhance confidence in the diagnosis of tubal patency, reduce false occlusion results, and improve the diagnostic yield of the test. It would be reasonable to deduce that HyFoSy would have similar performance characteristics as HyCoSy with other positive contrast agents. The available evidence supports the accuracy of HyFoSy compared to other forms of tubal investigation. We suggest a decision-making pathway based on the most current professional recommendations and available evidence. However, in this article, we do not provide a definitive exposition of the methods used for investigating tubal patency. Rather, we explore the contexts in which the various investigations are most and least suitable, and identify their strengths and limitations. Finally, we also discuss challenges encountered when performing tubal contrast sonography in clinical practice, including the problem of false occlusion results.
Keywords: hysterosalpingo-contrast sonography, hysterosalpingo-foam sonography, tubal patency testing, infertility, tubal occlusion
Introduction
Tubal disease is an important cause of infertility, accounting for 25%–35%1 of female infertility, and tubal patency testing is an important part of the infertility investigation. Tubal disease encompasses a range of pathologies including obstruction, stenosis, dilatation, and impaired peristaltic function.2 Pelvic inflammatory disease is by far the most common cause of tubal disease. Other causes of tubal infertility include endometriosis, previous pelvic surgery, fibroids, and pelvic tuberculosis.3
Treatment of tubal infertility is largely dependent on the site and extent of the disease. Accurate evaluation of the fallopian tubes is crucial because an incorrect diagnosis may result in unnecessary tubal reconstructive surgery or in vitro fertilization (IVF).3 Recommendations by the American Society of Reproductive Medicine (ASRM) state that all available methods for evaluation of tubal factors have technical limitations that must be considered when any one technique yields abnormal results. Further evaluation with a second, complementary method is prudent whenever the specific diagnosis or best treatment strategy is uncertain.4 The most commonly employed methods for investigating tubal patency include laparoscopy and dye chromopertubation (lap-and-dye), X-ray hysterosalpingography (HSG), and hysterosalpingo-contrast sonography (HyCoSy).
We aim to review the literature on ExEm® foam (GynaecologIQ, Delft, the Netherlands) and evaluate its role in tubal contrast sonography (Table 1). First, we provide a brief overview of the common types of tubal patency tests and discuss their strengths and limitations. Second, the background and technical procedure for tubal contrast sonography using ExEm® foam are described. Third, we discuss challenges encountered when performing tubal contrast sonography in clinical practice and evaluate the impact of the type of contrast medium on the diagnostic accuracy of HyCoSy, with a focus on ExEm® foam. Fourth, we assess the safety aspect of ExEm® foam. Finally, we propose a decision-making pathway for selecting the appropriate type of tubal patency investigation based on the most current professional recommendations and available evidence.
Common types of tubal patency testing
Lap-and-dye is considered the gold standard investigation for tubal assessment. It allows for direct inspection of the fimbrial ends and assessment of pelvic pathology such as endometriosis and adhesions. It also enables hysteroscopy and therapeutic procedures such as tubal surgery, hysterosopic tubal cannulation, ablation of endometriosis, ovarian cystectomy, and adhesiolysis to be undertaken concurrently. However while lap-and-dye enables direct visualization of external tubal morphology, assessment of the internal architecture of the tube is not permitted and it is not always possible to identify the site of tubal occlusion. Lap-and-dye is also more invasive and costly than other forms of tubal assessment and carries a risk of surgical morbidity and mortality.
HSG is the traditional method of assessing tubal patency and can be performed in the outpatient setting. To some extent, it can be considered complementary to lap-and-dye because it allows assessment of luminal abnormalities and identification of the site of tubal occlusion. It also enables concurrent assessment of the uterine cavity. However, its use is limited in assessing pelvic pathology, and it is unable to detect abnormalities in the ovaries and myometrium, such as endometriotic cysts and adenomyosis, which can lead to infertility. It also involves exposure to ionizing radiation and is associated with a small risk of iodine allergy.
HyCoSy was introduced as an alternative to HSG for outpatient tubal assessment, thereby avoiding exposure to the risks of iodine allergy or ionizing radiation. It allows for real-time ultrasound assessment of the pelvic organs including the ovaries and myometrium and is reported to be superior to HSG at detecting abnormalities in the uterine cavity. The patient does not need to be referred to another team for tubal assessment as HyCoSy is usually performed within the fertility unit. The test shows good concordance with lap-and-dye (80.4%–92.5%) and HSG (83.8%–90.5%).5 The main disadvantage of HyCoSy is that it is more operator-dependent compared to HSG and has a high false occlusion rate.5 Positive contrast media, color Doppler, and 3D technology have been incorporated into HyCoSy assessment in an attempt to improve its diagnostic performance.
Using foam as a contrast medium (HyFoSy)
Initially saline6,7 or a mixture of saline and air8,9 were used as contrast media in HyCoSy. Subsequently more hyperechogenic media such as Echovist® (Schering AG, Berlin, Germany), Infoson® (MBI, San Diego, CA, USA), and SonoVue® (Bracco, Milan, Italy) were developed. These contrast agents are either no longer commercially available or not licensed for intrafallopian use.
In 2007, a non-embryo-toxic gel known as ExEm® gel (GynaecologIQ) containing hydroxyethylcellulose and glycerol was introduced.10 The gel is diluted in water, and foam is mechanically created when this solution is pushed through narrow openings in syringes and tubes. The resultant turbulence causes a pressure drop that forces air to dissolve in the solution in the form of echogenic micro-air bubbles. The diluted gel is fluid enough to pass through patent tubes but not so viscous such that the air bubbles remain suspended for a longer duration in it than water. This results in a more stable foam mixture that can maintain its echogenicity for about 7 minutes, allowing it to be used as a contrast medium for hysterosalpingo-foam sonography (HyFoSy).11
Procedure for HyFoSy
The European Society of Human Reproduction and Embryology (ESHRE) guidelines advocate semen analysis and ovulation assessment before a test of tubal patency is performed.12
Our unit’s procedure for HyFoSy was previously described in a randomized controlled, crossover trial comparing efficacy and patient comfort between saline vs ExEm® foam as the contrast medium.13 The procedure is usually performed by a trained clinician and ultrasonographer. Prior to the examination, a transvaginal ultrasound is done to assess the pelvic anatomy, in particular noting the position of the ovaries and presence of any hydrosalpinges. We also routinely perform a 3D volume scan of the uterus and note the presence of any congenital uterine malformations and intracavitary abnormalities.
We use the GIS catheter (GynaecologIQ),11 which is balloon-less and has a tapered soft tip for our HyFoSy procedures. The cervix is visualized with a Cussco speculum, and the catheter is introduced into the cervical os under aseptic conditions using a tenaculum if needed. The speculum is then removed, taking care not to dislodge the catheter, and the ultrasound transducer is introduced vaginally. We first perform uterine cavity assessment by instilling sterile saline to distend and visualize the uterine cavity in the longitudinal plane. The saline is aspirated before ExEm® foam is instilled. In our experience, uterine cavity assessment is best performed prior to instillation of ExEm® foam and tubal evaluation as the echogenicity of the foam could mask intracavitary abnormalities. This also allows the examiner to confirm correct placement of the cervical catheter and exclude leakage before introducing ExEm®foam.13
Alternatively, undiluted ExEm® gel can be used to assess intracavitary abnormalities with less leakage and more optimal and stable distention of the uterine cavity compared to saline.14 There is some evidence that gel infusion sonohysterography may be associated with lower pain scores15,16 and fewer technical failures17 compared to saline infusion sonohysterography with comparable accuracy.
Van Schoubroeck et al18 recommended using a pediatric Foley balloon catheter with stylet for infusion of ExEm® foam, with the balloon placed in the lower uterine cavity to prevent backflow. We occasionally use a Foley balloon catheter for our HyFoSy procedures if there is difficulty inserting the GIS catheter because of a stenotic os or if there are multiple extrusions of the GIS catheter or excessive backflow from a patulous os.13
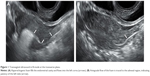
The foam contrast is reconstituted by mechanically mixing 10 mL ExEm® gel and 10 mL of sterile water.19 The foam is slowly injected into the endometrial cavity through the GIS catheter in repeated small (0.5–1 mL) boluses while observing for antegrade flow through the uterine cornua at the transverse plane using B-mode. Distal flow of contrast is followed through each tube until peritoneal spill is visualized (Figure 1). It is easier to visualize spill of the foam into the peritoneal cavity after locating the ovaries.19 The foam usually maintains its echogenicity long enough to allow acquisition of 3D volume images.
In the event of apparent cornual block, it has been suggested that this may be secondary to tubal spasm. Van Schoubroeck et al20 reported that temporarily withholding further foam injection then trying again after a certain period may correct tubal spasm.
Diagnostic accuracy of HyCoSy and the impact of the type of contrast medium
Both false patency and false occlusion results can occur with HyCoSy, irrespective of the type of contrast medium used. One meta-analysis5,21 on HyCoSy with Echovist® (Schering AG) reported a false occlusion rate of 10.3% and false patency rate of 6.7%. False patency results from HyCoSy can arise in the following situations. First, bilateral cornual block can result in back pressure of Echovist® within the uterine cavity with intravazation into the extrauterine vessels and mimic tubal filling. Second, Echovist® can track along the pouch of Douglas from a patent tube to the other side, giving the impression of bilateral tubal patency.22 These problems should be minimized with careful ultrasound technique and can theoretically also occur with HyFoSy. The pattern of contrast flow should be noted in relation to its injection. It is also essential to carefully follow the flow of ExEm® foam from the cornual region distally and ensure that flow is in an antegrade direction.22
On the other hand, false occlusion results are more difficult to correct and may be unavoidable. It is easier to diagnose tubal patency than it is tubal occlusion by HyCoSy. With good sonographic technique, the clinician would be fairly confident that the tube is truly patent if contrast fill is traced in an antegrade manner throughout the length of the tube with associated peritoneal spill. However, if tubal occlusion is suspected, it is usually not possible to differentiate between true and false occlusion.
There are many factors that can give rise to a false occlusion result. First, the tubal lumen may be temporarily occluded by a mucous plug, blood clot, myometrial spasm, or mucosal edema.5,23–27 The proximal segment being the narrowest part of the fallopian tube is especially prone to this resulting in a diagnosis of a cornual block. Second, technical difficulty can arise from multiple reasons. Contrast leakage or cervical stenosis can occur resulting in failure to achieve adequate pressure of hydrotubation.22 Tubal convolution or distorted anatomy from pelvic adhesions can lead to difficulty in tracing the course of the tubes. The multiplanar course of the tubes aggravates the problem of uncertain results.5,23–27 If a low viscosity medium such as saline is used, rapid contrast dissipation limits observation time.
While tracing the flow of contrast through the entire tubal length increases confidence in the diagnosis of proximal and distal patency, this is more technically demanding. Sometimes only proximal patency can be demonstrated by visualizing paracornual flow of contrast. It is thought that more hyperechogenic media28 may enhance contrast visualization and enable clearer delineation of tubal anatomy. This may enhance confidence in the diagnosis of tubal patency, reduce false occlusion results, and improve the diagnostic yield of the test. However, few studies have directly compared the use of commercially available hyperechogenic contrast agents with saline solution. One study26 reported that SonoVue® (Bracco) was more accurate than saline and air medium, especially in the reduction of false occlusion results. Another study,27 which compared Infoson® (MBI) with saline, found that Infoson®-enhanced HyCoSy provided a significantly larger number of correct diagnoses than saline HyCoSy with an accuracy comparable to that of HSG. However, a recent systematic review29 found no benefit of commercially available hyperechogenic contrast agents over saline solution in diagnosing tubal patency. As the viscosity of ExEm® foam is even lower than Echovist®,30 we could expect that ExEm® foam would perform at least as well as Echovist in terms of demonstrating tubal patency. Hamilton et al28 evaluated the performance of HyCoSy with Echovist® in 500 women and reported a concordance of 85.8% with lap-and-dye.
HyFoSy in comparison with saline HyCoSy
We randomized 40 patients to HyFoSy and HyCoSy with saline with the aim of comparing the diagnostic yield of both investigations.13 Patients first underwent tubal evaluation with their assigned medium. Only those with at least one possibly occluded or unexaminable tube on the initial test underwent crossover testing with the other medium. As both groups were appropriately randomized and found to be homogenous in terms of baseline characteristics and risk factors for tubal occlusion, it was assumed that there should be no significant difference in the proportion of truly occluded tubes between the two groups. Therefore, it is reasonable to assume that any differences in detection of patent tubes can be attributed to the performance of the test medium rather than differences in the proportion of patent tubes between the two groups.
Complete (ie, proximal and distal) tubal patency was defined as flow over the whole length of the tube, fimbrial outflow, or peritoneal spillage of contrast. This was demonstrated in a higher proportion of tubes in the HyFoSy group (60.0% vs 35.0%, P=0.04) (Figure 2). A higher proportion of patients in the HyCoSy group were found to have at least one possibly occluded or unexaminable tube and underwent crossover testing (80.0% [16/20] vs 45.0% [9/20], P=0.02). On crossover testing, 41.7% (10/24) of possibly occluded or unexaminable tubes in the HyCoSy group were reclassified as patent when examined with ExEm® foam, compared to 8.3% (1/12) of possibly occluded or unexaminable tubes in the HyFoSy group (P=0.03). Our results suggest that ExEm® foam was better than saline at detecting patent tubes and demonstrating complete tubal patency. We concluded that tubal patency assessment with HyFoSy might improve the diagnostic yield and efficacy over HyCoSy with saline.
  | Figure 2 Results of a randomized, controlled, crossover trial comparing efficacy and patient comfort between saline versus ExEm® foam as the contrast medium. |
HyFoSy in comparison with HSG and lap-and-dye
There are not many studies specifically addressing the accuracy of HyFoSy compared to HSG and lap-and-dye. It would be reasonable to deduce that HyFoSy would have similar performance characteristics as HyCoSy with other positive contrast agents with respect to HSG and lap-and-dye. False occlusion results can occur with HSG. This problem also applies to lap-and-dye, which is not a perfect gold standard and is susceptible to false occlusion results.2,28,31
The available evidence supports the accuracy of HyFoSy compared to other forms of tubal investigation. A nonrandomized observational study of 20 women who underwent lap and dye immediately following HyFoSy found 100% agreement between both forms of tubal assessment.18
In an observational study, Emanuel et al19 evaluated 73 women with HyFoSy. 21.9% (16/73 patients) subsequently underwent HSG because of inconclusive results on HyFoSy, either due to technical failure or suspected tubal blockage. The results for HSG and HyFoSy were discordant with regards to evaluation of cervical occlusion or tubal patency in 9.6% (7/73 patients).
A prospective study by Dreyer et al32 evaluated the accuracy of HyFoSy compared to HSG for assessing tubal blockage after Essure® (Conceptus, Inc., Mountain View, CA, USA) placement for 38 hydrosalpinges in patients who were planned for IVF. The concordance between HyFoSy and HSG was 97.4% (95% confidence interval [CI]: 92.34%–100.0%). HyFoSy could correctly identify 33 of 34 occluded hydrosalpinges and all four hydrosalpinges that remained patent after treatment. The authors concluded that HyFoSy is an accurate alternative to HSG for confirming proximal tubal occlusion after placement of Essure® to treat hydrosalpinges. As there was one tube that was classified as falsely patent, the authors advised that if tubes are found to be patent on HyFoSy, this should be confirmed with HSG.
Safety aspects and pain associated with HyFoSy
A literature search conducted 6 years after the introduction of ExEm® gel did not reveal any concerns, and there have been no reports of allergic reactions or teratogenicity. The individual components of ExEm® gel have been tested for safety.14
There is a paucity of data regarding complications related to the HyFoSy procedure. Given that the procedure for HyFoSy is similar in nature to that of HyCoSy and HSG, it would be expected that the adverse effect profile of HyFoSy would be the same. Dessole33 performed saline HyCoSy on 1,153 women and reported an overall complication rate of 8.8%. The incidence of vasovagal symptoms was 3.5%. In Emanual’s study, five patients (5/73; 6.8%) had vasovagal discomfort during or after the HyFoSy procedure that resolved spontaneously in time.19
The risk of pelvic inflammatory disease following HSG is reported to be 0.3%–3.1%.34 Guidelines from the American Congress of Obstetricians and Gynecologists35 recommend that antibiotic prophylaxis with doxycycline be given to patients with a history of pelvic infection or tubal abnormalities noted at the time of the test.
In contrast, the pelvic infection risk associated with HyCoSy is less certain. The decision for prophylactic antibiotics is left largely up to the provider’s discretion as there are no guidelines addressing this issue.35 Most providers assume that the postprocedural infection risk following HyCoSy is similar to that of HSG.35 However, there is some evidence to suggest that the risk may be even lower30 than commonly assumed. In Dessole’s series,33 the postprocedural infection rate after saline HyCoSy was 0.095%. No infections associated with HyFoSy have been reported in the literature,13,14,18–20,32,36 but this could be due to small sample size of the individual studies performed and insufficient power to detect uncommon complications.
Studies that evaluated pain associated with HyFoSy found that the procedure is generally well tolerated. A randomized controlled trial by Dreyer et al36 found that HyFoSy was less painful than HSG with a lower median visual analog scale pain score (1.7 vs 3.7, P<0.01). Another prospective study by the same authors32 evaluated pain in patients who underwent HyFoSy to check for proximal occlusion after Essure® placement. They also found lower pain scores with HyFoSy compared to HSG in this group of women. Schoubroeck et al20 performed HyFoSy on 216 consecutive patients. 92.1% thought that the procedure was at worst painful but tolerable; only 7.9% reported that it was really painful.
Patient selection and proposed decision-making pathway for choice of tubal patency investigation
We suggest a decision-making pathway (Figure 3) based on professional recommendations and available evidence. The National Institute for Health and Clinical Excellence guidelines37 recommend that women who are likely to have comorbidities such as pelvic inflammatory disease, previous ectopic pregnancy, and endometriosis should be offered lap-and-dye so that other pelvic pathology can be assessed at the same time. Similarly, the 2008 ESHRE position paper12 states that women thought to have comorbidities should be offered laparoscopy so that any tubal and other pelvic pathology can be investigated and treated at the same time.
  | Figure 3 Decision-making pathway for choice of tubal patency investigation. |
For women at low risk of tubal disease, the choice lies between HSG and HyCoSy. The National Institute for Health and Clinical Excellence opinion37 is that HSG should be offered in these women to screen for tubal occlusion. Alternatively, HyCoSy can be considered because it is an effective alternative to HSG in women who are not known to have comorbidities. The ASRM4 recommends HSG as the traditional and standard method for evaluating tubal patency. It suggests that HyCoSy performed by observing for the appearance of fluid in the pouch of Douglas would not be useful in differentiating between unilateral and bilateral patency. However, advances in HyCoSy technique and ultrasound technology have now made it possible to identify the side of tubal blockage with comparable accuracy to that of HSG.23,29,38,39
While HSG and HyCoSy are good for confirming tubal patency, both investigations are not as reliable for diagnosing tubal occlusion and are susceptible to false occlusion results.5,23–27 The ASRM4 recommends that findings suggestive of proximal tubal occlusion require further evaluation with selective tubal cannulation to exclude false occlusion resulting from tubal spasm or catheter position. This is more commonly performed either through the hysteroscopic or fluoroscopic approach.3,4 Therefore, an advantage of performing a HSG over HyCoSy is that it allows for a “see-and-treat” approach in cases of proximal tubal obstruction. In contrast, ultrasound-guided tubal cannulation is more technically difficult and requires expertise that is rarely available.40–42
In women at low risk of tubal disease, HSG should be offered as the standard method of tubal evaluation that offers the advantage of reducing false occlusion results for apparent proximal occlusion by fluoroscopic tubal cannulation if available. HyCoSy may be considered in women who have an iodine allergy or require sonographic assessment of their pelvic organs. It is the investigation of choice in women who wish to avoid radiation. It may be associated with a lower risk of postprocedural infection compared to HSG.30,33,35 We suggest that a positive contrast medium be used for performing HyCoSy whenever possible because it offers better visualization which can potentially reduce false occlusion results and minimize need for second-line investigations.26–28 If HyCoSy is abnormal, verification should be carried out with diagnostic laparoscopy because intrauterine insemination can be offered if the tubes are patent. Alternatively HSG can be offered in patients who wish to avoid an invasive procedure, although it is not the gold standard. If proximal tubal occlusion is suspected, selective tubal cannulation either through the hysteroscopic or fluoroscopic approach should be considered during the second-line tubal evaluation.2,4
Contraindications to performing HyCoSy should apply for HyFoSy. HyFoSy should not be done if there is active pelvic infection or pregnancy.30,34 As such it is best to arrange for the procedure during the preovulatory phase of the menstrual cycle.30,34
Current controversies and future directions
Greater research efforts should be directed toward establishing the accuracy and value of HyFoSy for first-line tubal patency testing. There is limited information on pregnancy outcomes after HyFoSy. It is commonly believed that tubal flushing can restore patency and fertility.34 Only one study by Emanuel et al19 reported that the conception rate was 19.2% within a median of 3 months after HyFoSy. On the other hand, a 2015 Cochrane review reported that HSG performed with oil-based contrast significantly increased the likelihood of pregnancy compared to no intervention (odds ratio: 3.59, 95% CI: 2.06–6.26). However this benefit was not seen when water-soluble contrast media was used.43
The complication rate associated with HyFoSy can be better defined. This would require the performance of large-scale studies in order to detect uncommon complications such as procedural-related infection, which is an independent cause of tubal infertility.
It would also be worthwhile exploring the value of 3D technology in improving the diagnostic performance of HyFoSy. A recent systematic review39 reported that 3D-HyCoSy shows high diagnostic accuracy with a pooled sensitivity of 98% and pooled specificity of 90%. 3D-HyCoSy offers several advantages over 2D-HyCoSy. 3D volume acquisition and reconstruction of the multiplanar tubal anatomy allows easy visualization of the contrast medium’s flow throughout the entire tubal length. This reduces false patency and uncertain results due to difficulty tracing the course of convoluted tubes with 2D-HyCoSy. As static images of the tube are captured in an automated process, the procedure is less operator dependent, with no need for difficult probe movements and the captured images can be stored for later review. Since image reconstruction can be performed offline, the procedure time is shorter and less contrast is required, which may help to reduce pain. Nevertheless, 3D-HyCoSy should be regarded as complementary and cannot fully replace 2D-HyCoSy. The main limitations of 3D technology are that of cost, availability, and inability to visualize the real-time passage of contrast medium through the fallopian tubes.23–25,39–40
Conclusion
HSG is considered the traditional standard for outpatient tubal patency testing in patients who are at low risk for tubal disease. However HyCoSy is a viable alternative that offers advantages over HSG with proven accuracy. A common limitation of conventional tubal patency evaluation is that of false occlusion results. In the case of HyCoSy, advances in ultrasound technology and introduction of positive contrast media have helped to reduce false occlusion results and improve its accuracy. HyFoSy is a modification of tubal contrast sonography that uses foam created from diluted ExEm® gel as a hyperechogenic contrast agent.
A review of the available literature suggests that HyFoSy has comparable accuracy to that of HSG with the advantages of less pain and shorter procedure time. It may also be more efficient than saline HyCoSy at diagnosing tubal patency. More research is needed to ascertain if it is a cost-effective replacement to saline HyCoSy, determine the incidence of procedural complications, and explore the effects of 3D technology on its accuracy.
Disclosure
The authors report no conflicts of interest in this work.
References
Serafini P, Batzofin J. Diagnosis of female infertility. A comprehensive approach. J Reprod Med. 1989;34(1):29–40. | ||
Patil M. Assessing tubal damage. J Hum Reprod Sci. 2009;2(1):2. | ||
Dun EC, Nezhat CH. Tubal factor infertility. Obstet Gynecol Clin North Am. 2012;39(4):551–566. | ||
Optimal evaluation of the infertile female. Fertil Steril. 2006;86(5):S264–S267. | ||
Campbell S, Bourne TH, Tan SL, Collins WP. Hysterosalpingo contrast sonography (HyCoSy) and its future role within the investigation of infertility in Europe. Ultrasound Obstet Gynecol. 1994;4(3):245–253. | ||
Randolph JR, Ying YK, Maier DB, Schmidt CL, Riddick DH. Comparison of real-time ultrasonography, hysterosalpingography, and laparoscopy/hysteroscopy in the evaluation of uterine abnormalities and tubal patency. Fertil Steril. 1986;46(5):828–832. | ||
Tüfekçi EC, Girit S, Bayirli E, Durmuşuoğlu F, Yalti S. Evaluation of tubal patency by transvaginal sonosalpingography. Fertil Steril. 1992;57(2):336–340. | ||
Inki P, Palo P, Anttila L. Vaginal sonosalpingography in the evaluation of tubal patency. Acta Obstet Gynecol Scand. 1998;77(10):978–982. | ||
Volpi E, Zuccaro G, Patriarca A, Rustichelli S, Sismondi P. Transvaginal sonographic tubal patency testing using air and saline solution as contrast media in a routine infertility clinic setting. Ultrasound Obstet Gynecol. 1996;7(1):43–48. | ||
Emanuel MH, Exalto N. Hysterosalpingo-foam sonography (HyFoSy): a new technique to visualize tubal patency. Ultrasound Obstet Gynecol. 2011;37(4):498–499. | ||
Frequently asked questions for Exem foam kit. de Smit Medical. Available from: http://www.exemfoamkit.co.uk/faq.php. Accessed July 1, 2016. | ||
Good Clinical Treatment in Assisted Reproduction. European Society of Human Reproduction and Embryology. Available from: https://www.eshre.eu/Guidelines-and-Legal/ESHRE-Position-Papers.aspx. Updated June 2008. Accessed July 1, 2016. | ||
Lim SL, Jung JJ, Yu SL, Rajesh H. A comparison of hysterosalpingo-foam sonography (HyFoSy) and hysterosalpingo-contrast sonography with saline medium (HyCoSy) in the assessment of tubal patency. Eur J Obstet Gynecol Reprod Biol. 2015;195:168–172. | ||
Exalto N, Stassen M, Emanuel MH. Safety aspects and side-effects of ExEm-gel and foam for uterine cavity distension and tubal patency testing. Reprod Biomed Online. 2014;29(5):534–540. | ||
Van den Bosch T, Betsas G, Van Schoubroeck D, et al. Gel infusion sonography in the evaluation of the uterine cavity. Ultrasound Obstet Gynecol. 2009;34(6):711–714. | ||
Exalto N, Stappers C, van Raamsdonk L, Emanuel M. Gel instillation sonohysterography: first experience with a new technique. Fertil Steril. 2007;87(1):152–155. | ||
Werbrouck E, Veldman J, Luts J, et al. Detection of endometrial pathology using saline infusion sonography versus gel instillation sonography: a prospective cohort study. Fertil Steril. 2011;95(1):285–288. | ||
Van Schoubroeck D, Van den Bosch T, Meuleman C, Tomassetti C, D’Hooghe T, Timmerman D. The use of a new gel foam for the evaluation of Tubal Patency. Gynecol Obstet Invest. 2013;75(3):152–156. | ||
Emanuel MH, Exalto N. First experiences with Hysterosalpingo-Foam Sonography (HyFoSy) for office Tubal Patency testing. Hum Reprod 2012;27(1):114–117. | ||
Van Schoubroeck D, Van den Bosch T, Ameye L, Boes AS, D’Hooghe T, Timmerman D. Pain during Fallopian-tube patency testing by hysterosalpingo-foam sonography. Ultrasound Obstet Gynecol. 2015;45(3):346–350. | ||
Holz K, Becker R, Schurmann R. Ultrasound in the investigation of tubal patency. A meta-analysis of three comparative studies of Echovist-200 including 1,007 women. Zentralbl Gynakol. 1997;119(8):366–373. | ||
Watrelot A, Hamilton J, Grudzinskas JG. Advances in the assessment of the uterus and fallopian tube function. Best Pract Res Clin Obstet Gynaecol. 2003;17(2):187–209. | ||
Sladkevicius P, Ojha K, Campbell S, Nargund G. Three-dimensional power Doppler imaging in the assessment of Fallopian tube patency. Ultrasound Obstet Gynecol. 2000;16(7):644–647. | ||
Exacoustos C, Di Giovanni A, Szabolcs B, Binder-Reisinger H, Gabardi C, Arduini D. Automated sonographic tubal patency evaluation with three-dimensional coded contrast imaging (CCI) during hysterosalpingo-contrast sonography (HyCoSy). Ultrasound Obstet Gynecol. 2009;34(5):609–612. | ||
Exacoustos C, Zupi E, Szabolcs B, et al. Contrast-tuned imaging and Second-Generation contrast agent SonoVue: a new ultrasound approach to evaluation of Tubal Patency. J Minim Invasive Gynecol. 2009;16(4):437–444. | ||
Lanzani C, Savasi V, Leone FP, Ratti M, Ferrazzi E. Two-dimensional HyCoSy with contrast tuned imaging technology and a second-generation contrast media for the assessment of tubal patency in an infertility program. Fertil Steril. 2009;92(3):1158–1161. | ||
Boudghene FP, Bazot M, Robert Y, et al. Assessment of Fallopian tube patency by HyCoSy: comparison of a positive contrast agent with saline solution. Ultrasound Obstet Gynecol. 2001;18(5):525–530. | ||
Hamilton JA, Larson AJ, Lower AM, Hasnain S, Grudzinskas JG. Evaluation of the performance of hysterosalpingo contrast sonography in 500 consecutive, unselected, infertile women. Hum Reprod. 1998;13(6):1519–1526. | ||
Maheux-Lacroix S, Boutin A, Moore L, et al. Hysterosalpingosonography for diagnosing tubal occlusion in subfertile women: a systematic review with meta-analysis. Hum Reprod. 2014;29(5):953–963. | ||
Lo Monte G, Capobianco G, Piva I, Caserta D, Dessole S, Marci R. Hysterosalpingo contrast sonography (HyCoSy): let’s make the point! Arch Gynecol Obstet. 2015;291(1):19–30. | ||
Luciano DE, Exacoustos C, Johns DA, Luciano AA. Can hysterosalpingo-contrast sonography replace hysterosalpingography in confirming tubal blockage after hysteroscopic sterilization and in the evaluation of the uterus and tubes in infertile patients? Am J Obstet Gynecol. 2011;204(1):79.e1–e5. | ||
Dreyer K, Hompes PGA, Mijatovic V. Diagnostic accuracy of hysterosalpingo-foam-sonography to confirm tubal occlusion after Essure® placement as treatment for hydrosalpinges. Reprod Biomed Online. 2015;30(4):421–425. | ||
Dessole S. Side effects and complications of sonohysterosalpingography. Fertil Steril. 2003;80(3):620–624. | ||
Saunders RD, Shwayder JM, Nakajima ST. Current methods of tubal patency assessment. Fertil Steril. 2011;95(7):2171–2179. | ||
American College of Obstetrics and Gynecology. ACOG practice bulletin No. 104: antibiotic prophylaxis for gynecologic procedures. Obstet Gynecol. 2009;113(5):1180–1189. | ||
Dreyer K, Out R, Hompes PG, Mijatovic V. Hysterosalpingo-foam sonography, a less painful procedure for tubal patency testing during fertility workup compared with (serial) hysterosalpingography: a randomized controlled trial. Fertil Steril. 2014;102(3):821–825. | ||
National Institute for Health and Clinical Excellence (NICE) Fertility: Assessment and treatment of people with fertility problems. London, UK: RCOG Press; 2013. | ||
Wang Y, Qian L. Three- or four-dimensional hysterosalpingo contrast sonography for diagnosing tubal patency in infertile females: a systematic review with meta-analysis. Br J Radiol. 2016;89(1063):20151013. | ||
Alcázar JL, Martinez-Astorquiza Corral T, Orozco R, Dominguez-Piriz J, Juez L, Errasti T. Three-Dimensional Hysterosalpingo-Contrast-Sonography for the assessment of Tubal Patency in women with infertility: a systematic review with Meta-Analysis. Gynecol Obstet Invest. 2016;81(4):289–295. | ||
Badawy SZA, Spirt BA. Ultrasound-guided fallopian tube catheterization. In: Rizk B, Puscheck EE, editors. Ultrasonography in gynecology. 1st ed. Cambridge, UK: Cambridge University Press; 2014:285–286. | ||
Session DR, Lerner JP, Tchen CK, Kelly AC. Ultrasound-guided fallopian tube cannulation using Albunex. Fertil Steril. 1997;67(5):972–974. | ||
Lisse K, Sydow P. Fallopian tube catheterization and recanalization under ultrasonic observation: a simplified technique to evaluate tubal patency and open proximally obstructed tubes. Fertil Steril. 1991;56(2):198–201. | ||
Mohiyiddeen L, Hardiman A, Fitzgerald C, et al. Tubal flushing for subfertility. May 2015. Cochrane Database Syst Rev. 2015;(5):CD003718. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.