Back to Journals » Neuropsychiatric Disease and Treatment » Volume 14
Hyperbaric oxygen therapy to improve cognitive dysfunction and encephalatrophy induced by N2O for recreational use: a case report
Authors Luo D, Xu JJ , Hu L, Yu LM, Xie LL, Li J
Received 3 April 2018
Accepted for publication 29 May 2018
Published 3 August 2018 Volume 2018:14 Pages 1963—1967
DOI https://doi.org/10.2147/NDT.S170037
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Wai Kwong Tang
Dan Luo,* Jiajun Xu,* Li Hu, Liangming Yu, Leling Xie, Jing Li
Department of Psychiatry, Mental Health Center, West China Hospital of Sichuan University, Chengdu, Sichuan, People’s Republic of China
*These authors contributed equally to this work
Abstract: N2O, or laughing gas, is generally used for anesthesia, especially in stomatology and pediatrics but is also commonly used recreationally. Cognitive dysfunction induced by the recreational use of N2O is rare. Here, we present the case of an 18-year-old female with a history of having used N2O recreationally for 5 months who suffered from encephalatrophy and severe cognitive dysfunction. All of the symptoms gradually subsided with ~20 days of treatment by hyperbaric oxygenation. We hypothesize that the long-term use of N2O may have induced a chronic state of systemic hypoxia that further induced cerebral atrophy with impaired cognitive function. Hyperbaric oxygen therapy (HBOT) is reported here for the first time as an important therapeutic element for treating N2O toxicity due to recreational use.
Keywords: N2O, recreational use, cognitive dysfunction, encephalatrophy, hyperbaric oxygen therapy
Introduction
Nitrous oxide (N2O) is a colorless, nonflammable, inorganic volatile gas with psychedelic effects that is commonly referred to as laughing gas.1 It is widely used for anesthesia and as an analgesic; it is also commonly used recreationally. The effects of N2O were first reported in 1799 as consisting of a brief but vivid intoxication, accompanied by a powerful euphoria that may distort sensation, as well as temporal and spatial perceptions.2 During the 19th century, N2O was a popular recreational drug used in theater halls to relieve pain during performances. The recreational use of N2O re-emerged during the 1960s, and it is now widely used and available via a variety of different manners of administration, including inhalation via canisters, balloons, respirators, and airtight bags.2 The side effects of N2O include transient dizziness, dissociation, disorientation, loss of balance, impaired memory and cognition, and weakness in the legs.3 It was previously reported that N2O might induce cognitive impairment when used as an anesthetic.4,5 We present a case report in which a patient presented with encephalatrophy with cognitive dysfunction caused by the recreational use of N2O. Encephalatrophy with impaired cognitive function caused by N2O recreational use has not been reported previously. This case report is the first to report encephalatrophy accompanied by altered cognitive functioning apart from peripheral neuropathy following intense N2O abuse. The benefits of hyperbaric oxygen therapy (HBOT) are extensive, and chief among them is the capacity to improve cognitive functioning with delayed encephalopathy after acute carbon monoxide poisoning.6 Additionally, we tried to use HBOT to relieve the symptoms N2O toxicity caused by recreational use.
Case report
An 18-year-old female who studied abroad in Australia and recently returned presented at our inpatient department with numbness and weakness in all four limbs, disturbance of orientation, and memory impairment for 5 days. She also presented with abnormal sensation in the lower limbs, difficulty walking, trouble speaking, and irritation. She was found lying on the ground in her house in Sydney and was unable to identify her brother. They also found thousands of steel bulbs3 (each of which contained 10 mL of pressurized N2O) in the house. She admitted that she had used N2O bulbs recreationally for >5 months. She used at least 50 bulbs during the past 5 months, one bulb every other day. During the last 4 days, she used more frequently than before, but she could not remember the exact number of bulbs she used.
Vital signs (temperature [T] 36.6°C, pulse [P] 80/min, respiratory rate [R] 20/min, and blood pressure [BP] 112/62 mmHg) were normal, and the physical examination was notable for a weakly positive Babinski sign, enhanced sensation in all four limbs, and ataxia. The upper limbs exhibited stage 4 muscle strength, while the lower limbs exhibited stage 3 muscle strength. The patient’s past history of medical and psychiatric diseases were unremarkable, and there was no family history of psychiatric disorders. A full blood examination showed hemoglobin (Hb) 112 g/L, platelet count 174×109/L, white cell count 7.95×109/L, and mean corpuscular volume (MCV) 93.2 fL. Vitamin B12 was <1,500 pmol/L. The results of a urine toxicology screen were all negative, including methylamphetamine, heroin, morphine, ketamine, and methylenedioxyphenethylamine. Blood gas (arterial blood) analysis showed partial pressure of oxygen in the alveolar (PAO2) 97.5 mmHg, partial pressure of oxygen in the artery (PaO2) 80.0 mmHg, PCO2 37.0 mmHg, pH 7.48, and base excess (BE) 3.4 mmol/L. Liver function, renal function, and electrolyte tests showed total bilirubin (TBIL) 4.2 μmol/L, direct bilirubin (DBIL) 2.4 μmol/L, indirect bilirubin (IBIL) 1.8 μmol/L, alanine aminotransferase (ALT) 21 IU/L, aspartate aminotransferase (AST) 20 IU/L, AST/ALT 0.95, total protein (TP) 66.0 g/L, albumin (ALB) 41.8 g/L, glucose (GLU) 4.96 mmol/L, alkaline phosphatase (ALP) 40 IU/L, lactate dehydrogenase (LDH) 380 IU/L, amylase (AMY) 83 IU/L, lipase (LIP) 36 IU/L, creatinine (CREA) 46.0 μmol/L, sodium (Na) 142.7 mmol/L, potassium (K) 4.43 mmol/L, and chlorine (Cl) 104.0 mmol/L. Due to concerns regarding a potential spinal cord compromise and cerebral disease, an enhanced spinal and cerebral magnetic resonance imaging (MRI) was implemented. Figure 1A shows gyrus atrophy and broadened anfractuosity compared to the normal state on a T1-weighted image. Table 1 (before the treatment) shows the results of motor nerve conduction velocity (MCV). The motor conduction amplitude of bilateral tibial nerves, peroneal nerves, median nerves, and ulnar nerves decreased, and motor conduction velocity of bilateral median nerves and ulnar nerves similarly slowed down. Table 2 (before the treatment) shows the result of sensory nerve conduction velocity (SCV). Sensory conduction amplitude of bilateral peroneal nerves decreased, and sensory conduction velocity of bilateral median nerves, ulnar nerves, and peroneal nerves slowed down. Bilateral anterior tibial muscles showed a large amount of spontaneous potential (positive sharp wave and fibrillation wave), and the right abductor hallucis brevis showed multiple spontaneous potential (positive sharp wave and fibrillation wave), small contraction, and huge motor unit action potentials (MUAPs) in a quiet state. The patient was unable to complete the Montreal Cognitive Assessment (MoCA) and the Mini-Mental State Examination (MMSE) due to the poor medical condition.
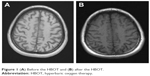  | Figure 1 (A) Before the HBOT and (B) after the HBOT. |
The patient did not use N2O recreationally again, and we prescribed vitamin B12 to improve neurological symptoms and an atypical antipsychotic drug, quetiapine, to help control irritation. The patient also received HBOT with a treatment pressure of 2 atm in an air-pressurized chamber and was given 100% oxygen for a period of 90–120 minutes at a time, a procedure that was repeated three times a session for 20 sessions. The dysfunction in sensory ataxia, numbness, and impaired cognitive functioning gradually improved with treatment by hyperbaric oxygenation. The MoCA and MMSE scores after treatment were in the normal range. A subsequent MRI to re-examine the cranium showed (Figure 1B) improvement in the encephalatrophy compared with the previous MRI. The results of motor (Table 1, after the treatment) and sensory (Table 2, after the treatment) NCV showed peripheral nerve impairment improved, as indicated by increased NCV values.
Ethics statement
Written informed consent for the publication of her clinical details and clinical images was obtained from the patient. A copy of the consent from is available for review from the editor of this journal.
Discussion
The patient met the diagnostic criteria for N2O abuse-induced encephalatrophy and cognitive impairment due to the history of N2O abuse and MRI-determined encephalatrophy accompanied by impaired cognitive functioning and peripheral nerve conduction abnormalities. The patient recovered fully as a result of HBOT.
There were several reported cases of the recreational use of N2O resulting in myelopathy and polyneuropathy.7–10 The neurological symptoms of these patients were commonly associated with the deficiency of vitamin B12.11 In this case, the reason why the level of vitamin B12 was high was that the patient received vitamin B12 treatment after she was found and sent to the Emergency Department of the local hospital in Sydney. However, there were no cases in which cognitive impairment was induced by N2O and the mechanism by which N2O induces encephalatrophy is not yet completely understood. As an anesthetic, N2O can affect cognitive functioning after surgery by influencing brain activity12 and the depth of the anesthesia is also related to cognitive functioning.13 The dose of N2O that the patient used recreationally is much greater than that used in anesthesia, so it is not too difficult to understand why the patient exhibited cognitive dysfunction. However, we cannot determine if the cognitive dysfunction induced by the N2O was acute or chronic. One study demonstrated that the most important safety consideration in the use of N2O as an anesthetic is the prevention of hypoxia.14 A related study suggested that hypoxia may damage brain cells,15 and other studies also found that N2O increases brain injury after ischemia or hypoxia in surgery.16–20 In this case, the cerebral atrophy induced by N2O may be the result of chronic hypoxia because of the long-term recreational use of N2O and the associated cognitive impairment.
Hyperbaric oxygen (HBO) provides 100% oxygen under high pressure, which significantly increases oxygen delivery to the mitochondria at the cellular level, reduces intracranial pressure, and has both anti-inflammatory and neuroplasticity effects in different types of brain injuries. According to the Henry law, maximizing tissue oxygenation, HBO increases the amount of oxygen carried in solutions and tissue by raising the external pressure, which is sufficient to support resting tissues without a contribution from the Hb, and induces rapid and significant vasoconstriction.21,22 Generation of oxygen-derived free radicals increased as a result of HBO, destroying DNA and inhibiting bacterial metabolic functions.21 HBOT not only accelerates collateral circulation to protect neurons from ischemic death but also repairs the damaged microvessels, thereby stimulating angiogenesis and neurogenesis.22,23 Additionally, HBOT can effectively counter ischemia and hypoxia, so all instances of hypoxia, ischemic diseases, or a series of diseases caused by hypoxia and ischemia can be resolved successfully. There were a number of reports about the use of HBOT to treat hypoxia and ischemic diseases as a result of all kinds of causes, such as brain injury, cerebral palsy, stroke, and others,24,25 especially to improve cognitive dysfunction after brain injury.26 In particular, HBOT can ameliorate cognitive functioning in someone suffering from anoxic brain damage.27 In this case, we speculate that the high dosage of N2O induced hypoxia and hypoxia induced cerebral atrophy and cognitive impairment. We tried to use HBOT to relieve the patient’s cerebral atrophy and cognitive impairment, which we did successfully. After the HBOT treatment, the patient’s cerebral atrophy and cognitive impairment improved, a strong confirmation of our initial hypothesis regarding pathophysiology.
There are important points to consider regarding this case report; for example, we could not conclusively determine whether the patient’s cognitive impairment and cerebral atrophy caused by the N2O was acute or chronic. Additionally, although we affirmed the effectiveness of HBOT for cognitive dysfunction caused by N2O, there were no previous reports of the use of HBOT to treat cognitive impairment and brain atrophy caused by N2O. As such, the use of HBOT as the standard therapy for ailments associated with laughing gas abuse still warrants further research.
Conclusion
This case report is the first to present encephalatrophy with severe cognitive impairment as a side effect of recreational abuse of N2O. Symptoms such as numbness and weakness in all four limbs, disturbance of orientation, memory impairment, abnormal sensation in the lower limbs, and difficulty walking and speaking were relieved by the HBOT treatment, providing an important clue regarding the mechanism behind N2O-induced encephalatrophy and the role of HBOT as a new treatment for this pathophysiology.
Disclosure
The authors report no conflicts of interest in this work.
References
Wong SL, Harrison R, Mattman A, Hsiung GY. Nitrous oxide (N2O)-induced acute psychosis. Can J Neurol Sci. 2014;41(5):672–674. | ||
Mike J. Nitrous oxide: recreational use, regulation and harm reduction. Drugs Alcohol Today. 2008;8(3):22–25. | ||
van Amsterdam J, Nabben T, van den Brink W. Recreational nitrous oxide use: prevalence and risks. Regul Toxicol Pharmacol. 2015;73(3):790–796. | ||
Zou YQ, Li XB, Yang ZX, et al. Impact of inhalational anesthetics on postoperative cognitive function: study protocol of a systematic review and meta-analysis. Medicine (Baltimore). 2018;97(1):e9316. | ||
Samur Ergüven S, Delilbaşi EA, Işik B, Öktem F. The effects of conscious sedation with nitrous oxide/oxygen on cognitive functions. Turk J Med Sci. 2016;46(4):997–1003. | ||
Xiang W, Xue H, Wang B, et al. Efficacy of N-butylphthalide and hyperbaric oxygen therapy on cognitive dysfunction in patients with delayed encephalopathy after acute carbon monoxide poisoning. Med Sci Monit. 2017;23:1501–1506. | ||
Stockton L, Simonsen C, Seago S. Nitrous oxide-induced vitamin B12 deficiency. Proc (Bayl Univ Med Cent). 2017;30(2):171–172. | ||
Richardson PG. Peripheral neuropathy following nitrous oxide abuse. Emerg Med Australas. 2010;22(1):88–90. | ||
Hsu CK, Chen YQ, Lung VZ, His SC, Lo HC, Shyu HY. Myelopathy and polyneuropathy caused by nitrous oxide toxicity: a case report. Am J Emerg Med. 2012;30(6):e3–e6. | ||
Rheinboldt M, Harper D, Parrish D, Francis K, Blase J. Nitrous oxide induced myeloneuropathy: a case report. Emerg Radiol. 2014;21(1):85–88. | ||
Hathout L, El-Saden S. Nitrous oxide-induced B12 deficiency myelopathy: perspectives on the clinical biochemistry of vitamin B12. J Neurol Sci. 2011;301(1–2):1–8. | ||
Mandal PK, Schifilliti D, Mafrica F, Fodale V. Inhaled anesthesia and cognitive performance. Drugs Today (Barc). 2009;45(1):47–54. | ||
Farag E, Chelune GJ, Schubert A, Mascha EJ. Is depth of anesthesia, as assessed by the Bispectral Index, related to postoperative cognitive dysfunction and recovery? Anesth Analg. 2006;103(3):633–640. | ||
Donaldson M, Donaldson D, Quarnstrom FC. Nitrous oxide-oxygen administration: when safety features no longer are safe. J Am Dent Assoc. 2012;143(2):134–143. | ||
Geddes R, Vannucci RC, Vannucci SJ. Delayed cerebral atrophy following moderate hypoxia-ischemia in the immature rat. Dev Neurosci. 2001;23(3):180–185. | ||
Baughman VL, Hoffman WE, Thomas C, Albrecht RF, Miletich DJ. The interaction of nitrous oxide and isoflurane with incomplete cerebral ischemia in the rat. Anesthesiology. 1989;70(5):767–774. | ||
Matta BF, Lam AM. Nitrous oxide increases cerebral blood flow velocity during pharmacologically induced EEG silence in humans. J Neurosurg Anesthesiol. 1995;7(2):89–93. | ||
Baughman VL, Hoffman WE, Miletich DJ, Albrecht RF, Thomas C. Neurologic outcome in rats following incomplete cerebral ischemia during halothane, isoflurane, or N2O. Anesthesiology. 1988;69(2):192–198. | ||
Sakabe T, Kuramoto T, Inoue S, Takeshita H. Cerebral effects of nitrous oxide in the dog. Anesthesiology. 1978;48(3):195–200. | ||
Pelligrino DA, Miletich DJ, Hoffman WE, Albrecht RF. Nitrous oxide markedly increases cerebral cortical metabolic rate and blood flow in the goat. Anesthesiology. 1984;60(5):405–412. | ||
Gill AL, Bell CN. Hyperbaric oxygen: its uses, mechanisms of action and outcomes. QJM. 2004;97(7):385–395. | ||
Sánchez EC. Mechanisms of action of hyperbaric oxygenation in stroke: a review. Crit Care Nurs Q. 2013;36(3):290–298. | ||
Wang Y, Chen D, Chen G. Hyperbaric oxygen therapy applied research in traumatic brain injury: from mechanisms to clinical investigation. Med Gas Res. 2014;4:18. | ||
McDonagh M, Carson S, Ash J, et al. Hyperbaric oxygen therapy for brain injury, cerebral palsy, and stroke. Evid Rep Technol Assess (Summ). 2003;(85):1–6. | ||
Michalski D, Härtig W, Schneider D, Hobohm C. Use of normobaric and hyperbaric oxygen in acute focal cerebral ischemia – a preclinical and clinical review. Acta Neurol Scand. 2011;123(2):85–97. | ||
Liu S, Shen G, Deng S, Wang X, Wu Q, Guo A. Hyperbaric oxygen therapy improves cognitive functioning after brain injury. Neural Regen Res. 2013;8(35):3334–3343. | ||
Hadanny A, Golan H, Fishlev G, et al. Hyperbaric oxygen can induce neuroplasticity and improve cognitive functions of patients suffering from anoxic brain damage. Restor Neurol Neurosci. 2015;33(4):471–486. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


