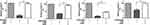Back to Journals » Journal of Inflammation Research » Volume 13
Human Neutrophil Elastase Proteolytic Activity in Ulcerative Colitis Favors the Loss of Function of Therapeutic Monoclonal Antibodies
Authors Curciarello R , Sobande T, Jones S, Giuffrida P, Di Sabatino A, Docena GH, MacDonald TT, Kok K
Received 15 October 2019
Accepted for publication 15 January 2020
Published 22 May 2020 Volume 2020:13 Pages 233—243
DOI https://doi.org/10.2147/JIR.S234710
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ning Quan
Renata Curciarello,1,2 Toni Sobande,2 Samantha Jones,2 Paolo Giuffrida,2,3 Antonio Di Sabatino,3 Guillermo H Docena,1 Thomas T MacDonald,2 Klaartje Kok2,4
1Instituto de Estudios Inmunológicos y Fisiopatológicos IIFP-CONICET, Universidad Nacional de La Plata, La Plata, Buenos Aires, Argentina; 2Centre for Immunobiology, The Blizard Institute, Queen Mary University of London, London, UK; 3First Department of Internal Medicine, University of Pavia and Fondazione IRCCS Policlinico San Matteo, Pavia, Italy; 4Barts Health NHS Trust, Royal London Hospital, London, UK
Correspondence: Klaartje Kok 4 Newark Street, London E1 2AT, UK
Tel +44207882 2321
Fax +44 207882 2194
Email [email protected]
Purpose: Proteases play an essential role in the pathophysiology of inflammatory bowel disease (IBD), contributing to the intestinal mucosal lesions through the degradation of the extracellular matrix and alteration of the barrier function. Ulcerative colitis (UC) is characterized by an extensive infiltrate of neutrophils into the mucosa and hence, increased proteolytic activity. Human neutrophil elastase (HNE) is a serine protease that has been reported to be increased in UC patients’ intestinal mucosa. Based on our previous studies, we hypothesized that HNE might induce proteolytic degradation and loss of function of therapeutic monoclonal antibodies in IBD patients.
Patients and Methods: Elastase expression and elastinolytic activity were determined in mucosal explants from ulcerative colitis patients (n=6) and cultured ex vivo in the presence or absence of recombinant elafin. Enzymatic digestions of therapeutic monoclonal antibodies were performed using recombinant HNE and elafin. The integrity of the therapeutic antibodies was evaluated by immunoblotting and protein G binding assay, whereas their TNF-neutralizing activity was assessed with a reporter cell line.
Results: We found that HNE and its elastinolytic activity were increased in the gut mucosa of UC patients. We also demonstrated that HNE cleaved biological drugs, impairing the TNF-α neutralizing capacity of anti-TNF monoclonal antibodies. This proteolytic degradation was inhibited by the addition of the specific inhibitor, elafin.
Conclusion: Our results suggest that the high level of proteolytic degradation by mucosal neutrophil elastase, along with a potential imbalance with elafin, contributes to the loss of function of biologic agents, which are currently used in patients with IBD. These findings might explain the non-responsiveness of UC patients to therapeutic monoclonal antibodies and suggest the potential beneficial concomitant use of elafin in this treatment.
Keywords: elastinolytic activity, elafin, anti-TNF, inflammatory bowel disease, biological drugs
Introduction
Inflammatory bowel disease (IBD), comprising the two major disorders Crohn’s Disease (CD) and Ulcerative colitis (UC), is a chronic relapsing condition characterized by intestinal inflammation and epithelial injury. IBD is thought to be triggered by inappropriate activation of the intestinal immune system against the microbiota in genetically susceptible individuals. However, CD and UC represent independent clinical entities.1,2 The main difference between CD and UC is that inflammation in UC is continuous and marked by an extensive infiltrate of neutrophils. The primary granules of neutrophils contain elastase and cathepsin G, the secondary granules contain collagenase and the tertiary granules contain metalloproteinases (MMP)-2 and MMP-9.3,4 These proteases contribute to mucosal lesions through the digestion of the extracellular matrix and alteration of the barrier function.5
Human neutrophil elastase (HNE) is a serine protease that cleaves the extracellular matrix protein elastin. There is evidence that it also up-regulates a number of pro-inflammatory cytokines, as HNE-deficient mice have been shown to be protected against dextran sodium sulfate (DSS)-induced colitis.6 HNE is naturally inhibited by elafin or trappin-2, a serine protease inhibitor mostly released by epithelial cells throughout the gastrointestinal tract.7 It has been reported that transgenic mice over-expressing elafin do not develop experimental colitis, whereas oral administration of elafin-expressing lactic acid bacteria diminishes proteolytic activity in the gut mucosa and, thus, inflammation in both T cell transfer- and DSS-induced colitis mouse models.6 Additionally, HNE proteolytic activity has been described to be increased in the mucosa of UC patients.8,9
Anti-tumor necrosis factor (TNF)-α therapy can result in marked clinical improvement and macroscopic healing of the inflamed IBD mucosa. However, a substantial proportion of patients do not respond to these biological agents. A rate of 20–40% of anti-TNFs’ primary non-response has been reported in clinical trials, while 10–20% in real-life cohorts.10 Our group has previously demonstrated that this non-responsiveness is due in part to the highly proteolytic mucosal microenvironment in IBD, and that particularly MMP-3 and MMP-12 degrade therapeutic antibodies.11
We have therefore hypothesized that HNE may be part of the proteolytic mucosal environment in UC, contributing to the functional degradation of anti-TNF agents. On this basis, here we explore the presence of HNE in the intestinal mucosa of UC patients and its effect on the integrity and function of anti-TNF therapeutic drugs.
Patients and Methods
Patients and Tissue
Colonic biopsies were taken from macroscopically and microscopically inflamed or uninflamed mucosa of adult patients (mean age 35.3 years, range 25–62) affected by CD (n=6) or UC (n=12). The diagnosis was made according to clinical and histological criteria, and the site and extent of the disease were confirmed by endoscopy. Endoscopic disease activity in UC patients was assessed according to the Mayo score (n=1 Score 0, n=1 Score 1, n=4 Score 2 and n=6 Score 3).12 In addition, mucosal samples were collected endoscopically from the colon of a total of 10 adult subjects who were not diagnosed with IBD nor any other inflammatory condition of the gut, and constituted the “healthy control patient” group (HC). Peripheral venous blood from UC patients and healthy volunteers was also collected. The study was carried out in accordance with the Declaration of Helsinki. Each patient who took part in the study was recruited after appropriate local Ethics Committee approval (National Research Ethics Service, East London Research Ethics Committee 2; REC study number 10/H0704/74) and informed written consent was obtained in all cases.
Organ Culture
Endoscopic mucosal biopsies from UC and CD patients and control subjects were weighed and then placed (one biopsy per well) in 24-well plates (VWR International, Lutterworth, UK). Individual biopsies were cultured in 300 μL of serum-free HL-1 medium (Lonza, Slough, UK), supplemented with 100 U/mL penicillin and 100 μg/mL streptomycin, and cultured at 37ºC, 5% CO2, with or without 5 µg/mL of human recombinant elafin (Sigma-Aldrich, St.Luis, USA). After 24 h of ex vivo culture, supernatants of mucosal biopsies were collected and stored at −70º C until used. All biopsies were lysed in ice-cold RIPA buffer (Sigma-Aldrich, St.Luis, USA). The protein concentration was determined using the Bradford Protein Assay (Bio-Rad Laboratories, Hemel Hempstead, UK).
Elastin and Elafin Specific Assays
Elastin was detected and quantified in mucosal homogenates and supernatants of the organ cultures described above, using a commercial colorimetric assay (FastinTM-Elastin; Biocolor-Ltd, UK) according to the manufacturer’s instructions. Elafin was detected in mucosal homogenates by Western blot. Briefly, 20 µg of total protein from homogenates were run on 10% SDS-PAGE gels under reducing conditions and subsequently transferred to a nitrocellulose membrane (Bio-Rad Laboratories, Hemel Hempsted, UK). Mouse anti-elafin antibody (1:10 dilution, ab81681 Abcam, UK) was used as the primary antibody, followed by a horseradish peroxidase-conjugated goat anti-mouse Ig secondary antibody (1:1000 dilution, Dako, Ely, UK). The elafin content was quantified in the mucosal homogenates using a specific ELISA kit (R&D Systems, Abingdon, UK), according to the manufacturer’s instructions.
Elastase Activity Assay
Elastinolytic activity was measured in mucosal homogenates after ex vivo culture in HL-1 medium for 24h with or without 5 µg/mL recombinant elafin (Sigma-Aldrich, St Luis, USA). Protein homogenates were obtained, and the elastase activity was measured at different time points using the EnzCheck Elastase Assay kit (Invitrogen, Paisley, UK), following the manufacturer’s instructions.
Neutrophil Isolation from the Blood
Polymorphonuclear granulocytes were separated from whole blood using the commercial mixture of Sodium diatrizoate and Dextran 500 solution Lympholyte®-poly (Cedarlane Corporation, Burlington, Canada) according to the manufacturer´s instructions. Isolated cells were then lysed in ice-cold RIPA buffer (Sigma-Aldrich, St.Luis, USA). The protein concentration was determined using the Bradford Protein Assay (Bio-Rad Laboratories, Hemel Hempstead, UK).
Cleavage Reactions
Recombinant human neutrophil elastase (HNE) (Athens Research & Technology, USA) was incubated with different biological agents to assess its effect on drug integrity and function. Infliximab (Remicade; Merck Sharp & Dohme Limited, Hoddesdon, UK and Remsima, Napp Pharmaceuticals Ltd., Cambridge, UK), adalimumab (Humira; Abbott Laboratories, Chicago, IL, USA), etanercept (Enbrel; Wyeth, Maidenhead, UK) or vedolizumab (Entyvio, Takeda Pharmaceuticals, Deerfield, IL, USA) (all at a concentration of 0.1 µg/mL) were incubated for 24 h at 37°C with increasing concentrations of recombinant HNE (0 to 10 µg/mL) in HNE buffer (100 mM Tris-HCl, pH 7.5), with or without 5 µg/mL elafin (Sigma-Aldrich, USA). Similarly, 1 µg/mL infliximab was co-incubated with increasing concentrations of blood neutrophil protein extracts from UC and HC donors (0–5 µg/mL), with or without 5 µg/mL elafin. The protease activity was stopped by snap freezing.
Immunoblotting
The effect of HNE on the integrity of the biologic drugs was assessed by immunoblotting. Cleavage reaction products were run on 10% SDS-PAGE gels under denaturing conditions and then transferred onto a nitrocellulose membrane (Bio-Rad Laboratories, Hemel Hempsted, UK). Rabbit anti-human immunoglobulin Fc antibody (1:2000 dilution, #31142 Thermo Fisher Scientific Massachusetts, USA) was used as the primary antibody, followed by a horseradish peroxidase-conjugated goat anti-rabbit immunoglobulins secondary antibody (1:3000 dilution, Dako, Ely, UK). Band density was quantified using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Protein-G Binding Assay
The effect of recombinant HNE on the Fc region of the biological agents was assessed by protein-G binding assay. Untreated or HNE-treated drugs were added onto a protein-G-coated 96-well plate (Fisher Scientific Loughborough, UK). Because chicken antibodies are unable to bind to protein-G, chicken anti-human p75 TNF-receptor antibody (Abcam, Cambridge, UK), previously conjugated with horseradish peroxidase using the Easy link HRP conjugation kit (Abcam, Cambridge, UK), was added in order to detect etanercept bound to the coated plate. In parallel, horseradish peroxidase-conjugated chicken anti-human F(ab’)2 (Fisher Scientific Loughborough, UK) was added to detect plate-bound infliximab or adalimumab. Tetramethyl benzidine (TMB) was used as the substrate, and the optical density of the color reaction was measured on a plate reader (BioTek, Swindon, UK).
Luciferase Reporter Assay
The effect of recombinant HNE on the integrity and function of anti-TNF agents was assessed using a HeLa reporter cell line, stably transfected with the luciferase reporter gene under the control of nuclear factor-κB enhancer elements.13 HeLa cells were cultured for 6 h with 10 ng/mL recombinant human TNF-α (R&D Systems, Abingdon, UK), which was pre-incubated for one hour with the HNE-cleaved infliximab, adalimumab or etanercept. Untreated anti-TNF agents, HNE and DMEM were also pre-incubated with human recombinant TNF-α and included as controls. After culture, HeLa cells were lysed, and luciferase activity was detected with a luminescence microplate reader using Bright-Glo™ Luciferase Assay System substrate (Promega UK, Southampton, UK).
Statistical Analysis
Data were analyzed with GraphPad Prism software (San Diego, CA, USA) and using the unpaired or paired two-tailed t-test and the Mann–Whitney U-test, depending on the assay. A p-value <0.05 was considered statistically significant.
Results
Human Neutrophil Elastase Activity Is Increased in UC Patients’ Mucosa Along with Lower Content of Elastin
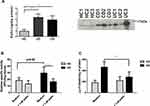
In order to evaluate specific proteolytic activity in IBD mucosa, HNE enzymatic activity was measured in mucosal explants homogenates from UC (n=6) and HC (n=6) patients. UC colonic homogenates showed a significant increasing enzymatic activity over time compared with samples from HC (Figure 1A). As HNE is a specific protease for the extracellular matrix component elastin, we quantified the content of elastin in biological samples. We found that biopsy homogenates from UC patients contained lower amounts of elastin than those from HC mucosa (p<0.01) (Figure 1B).
Elafin Reduces Elastase Activity ex vivo
Considering that elafin is the natural inhibitor of elastase, we first evaluated whether this inhibitor was present in IBD mucosa. Elafin was detected in mucosal homogenates from three representative IBD and three representative healthy control patients by Western blot, and then by ELISA (n=6 UC, n=6 CD and n=6 HC; Figure 2A). We found significantly higher amounts of elafin in IBD patient samples compared to HC. After the addition of human recombinant elafin to the ex vivo culture for 24 h, elastase enzymatic activity was diminished in mucosal homogenates (Figure 2B); mainly in biopsies from UC patients (n=11, p<0.05). Additionally, the presence of soluble elastin fragments was assessed in supernatants from UC (n=11) and HC (n=6) cultured samples (Figure 2C). We observed that UC biopsies cultured ex vivo released more soluble elastin fragments than HC biopsies, and the addition of elafin significantly reduced the elastin fragment release in UC mucosa organ culture.
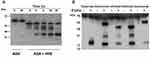
Recombinant HNE Degrades Biological Therapeutic Drugs
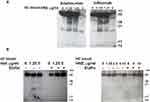
The effect of HNE on the integrity of TNF-α neutralizing agents and the anti-integrin antibody vedolizumab was evaluated by immunoblotting. First, we optimized the digestion time (0 to 24 hours) with the highest concentration of HNE (10 µg/mL) on the different drugs (Figure 3A). Then we tested 24 hours of digestion with 10 µg/mL HNE (Figure 3B). Figure 3 shows the dose-dependent enzymatic effect of increasing amounts of HNE on infliximab, adalimumab, etanercept, and vedolizumab (0.1 µg/mL) during 24 hours at 37°C. The proteolytic products were analyzed by immunoblotting under reducing conditions using an anti-human IgG Fc primary antibody. As shown in Figure 4, the heavy chains of infliximab, adalimumab, and vedolizumab appeared as a single band of approximately 55 kilodaltons (kDa) that remained intact after 24 hours of co-incubation with HNE buffer (0 µg/mL HNE). Adalimumab and vedolizumab were partially cleaved by different concentrations of HNE, resulting in 38 and 17 kDa-fragments, and 25 kDa-fragments, respectively, which likely correspond to Fc monomers, Fab monomers and a result of non-enzymatic fragmentation under the denaturing conditions of IgG1, respectively.14 Infliximab’s heavy chain was completely cleaved by the highest concentrations of HNE tested. Fragments of 45, 38, 25, and 12 kDa were observed in the blots. Etanercept was entirely degraded by the highest concentration of HNE, producing fragments of approximately 32 and 12 kDa. Densitometry analysis of bands revealed that increasing concentrations of HNE degraded the biologics from 6% to 99.9%. However, when recombinant elafin was added, cleavage reactions were inhibited. Infliximab and adalimumab were also degraded after 24 h co-incubation with increasing concentrations of blood neutrophil protein homogenates from UC or HC subjects. Multiple bands were observed by Western blot, including the IgG Fc monomers of approximately 30 kDa (Figure 5A). In addition, recombinant elafin specifically inhibited the proteolysis, as bands corresponding to low molecular weight fragments were not detected (Figure 5B).
Loss of Fc fraction of the therapeutic monoclonal antibodies was also evaluated by ELISA performed on protein-G-coated plates. Infliximab, adalimumab, etanercept, or vedolizumab treated with HNE (for 24 h) or HNE plus elafin were incubated in the protein G-coated wells. We found that almost 80% of infliximab and etanercept binding, and 40 to 50% of adalimumab and vedolizumab binding to protein G was impaired by HNE digestion. The binding capacity of the four drugs to protein G was significantly rescued by elafin (Figure 6).
Cleavage of Anti-TNF Agents by HNE Impairs Their Functional Capacity in vitro
Finally, we investigated whether HNE degradation also affected the ability of anti-TNF biologics to neutralize soluble TNF-α using a TNFR-HeLa reporter cell line. Intact infliximab, adalimumab, and etanercept effectively inhibited TNF-α-induced luciferase activity of the reporter cell line. Neutralization of TNF-α with HNE-treated infliximab, adalimumab, or etanercept was significantly reduced compared to the untreated biologics, an effect that was reversed by the addition of elafin (Figure 7A). Soluble TNF-α is not degraded by any components of the cell culture medium nor by HNE (Figure 7B).
Discussion
Proteases play an essential role in gastrointestinal homeostasis and barrier function, and their activity is tightly regulated by natural endogenous inhibitors.15–17 However, there is increasing evidence suggesting that a deregulation of the protease/anti-protease balance in the gut contributes to gastrointestinal diseases such as colorectal cancer, inflammatory bowel disease, and irritable bowel syndrome.18 In the inflamed gut, cell infiltration and activation take place in the mucosa, and this is a source of proteases, which are released to degrade extracellular tissue. Neutrophils in particular release large amounts of elastase, proteinase-3, and cathepsin G.19,20 Increased systemic and intestinal neutrophil activity has been found in inflamed UC mucosa, rather than in CD, driving tissue damage (ulceration and crypt abscesses).21,22 While several studies have shown that the expression of a very large number of proteases is up-regulated in inflamed mucosa from patients with IBD, reports considering their enzymatic activity are scarce.15,23
In this study, we have investigated the enzymatic activity of neutrophil elastase in the mucosa of UC patients, along with its specific inhibition by elafin ex vivo. It has been reported that elastase and elastinolytic activity are increased in IBD, contributing to ECM remodelling.24,25 Elastase degrades elastin, which is a crucial component of the extracellular matrix.26,27 Here, we have shown that there is a higher and increasing elastinolytic activity in UC mucosa compared to uninflamed healthy control mucosa, leading to decrease in elastin content within the tissue. However, the elastase specific inhibitor elafin has been found surprisingly increased in mucosal homogenates from IBD patients, reflecting the usual unbalanced relationship proteases/anti-proteases that contributes to gut diseases. Even though elafin is increased in these mucosal explants, it is not enough to compensate for excessive elastinolytic activity. Our data suggest that the addition of exogenous elafin ex vivo to organ cultures can redress this imbalance as we have found that elastase activity in UC mucosa was inhibited by the effect of exogenous elafin, preventing elastin degradation. Our data demonstrate that elastase in inflamed mucosa is sensitive to the suppressive effect of elafin.
Apart from protease-specific substrates, we studied the effect on therapeutic monoclonal antibodies used in UC and CD patients. Immunoglobulins are sensitive to proteases, and various studies have demonstrated IgG degradation. In addition, immunoglobulin cleavage sites have been identified for several endogenous and also bacterial proteases.28–30 The degradation of intestinal immunoglobulins is not only relevant for the loss of function of IgG or IgA exerted by the microbiota, but also for the cleavage of exogenous immunoglobulins used as therapeutic biologic drugs. Our group has previously demonstrated that MMP-3 and -12 in the inflamed mucosa of CD and UC patients contribute to the degradation and the loss of function of infliximab, adalimumab, and etanercept, which are Fcγ1-containing TNF-blocking agents.11 Infliximab and adalimumab are monoclonal IgG1 antibodies specific for TNF-α, etanercept is a dimeric p75 TNF receptor–IgG Fc fusion protein and vedolizumab is a monoclonal IgG1 specific for α4β7 integrin.31,32 Based on the deregulated elastinolytic activity found in the mucosa of UC patients, we further investigated the effect of human recombinant neutrophil elastase on different biologic anti-TNF drugs and the anti-integrin vedolizumab. We observed a dose-dependent degradation of infliximab, adalimumab, and vedolizumab, resulting in Fc monomers and small size fragments of human IgG1. The addition of elafin inhibited the production of the small fragments, reflecting that the therapeutic monoclonal antibodies remained intact after incubation with an inhibited elastase. The integrity of the biologic agents was confirmed by protein G capture ELISA.11 Cleaved antibodies were not detected using the ELISA for Fab or TNFRp75 detection, demonstrating the proteolytic degradation of these drugs by HNE. However, in the presence of elafin, intact infliximab, adalimumab and etanercept could be detected in this assay. Interestingly, neutrophil protein extracts containing native proteases, isolated from human blood, similarly affected the integrity of anti-TNF drugs, which was also rescued by elafin. Overall, we found that HNE cleaved all biologics tested, and elafin prevented such degradation.
Moreover, all these biologicals were affected by the in vitro HNE treatment, impairing not only protein integrity but their neutralizing capacity. We used a TNFR-reporter cell line and found that infliximab, adalimumab, and etanercept lost their ability to bind soluble TNF-α after elastase proteolytic degradation. Elafin inhibited the elastinolytic degradation with the concomitant restoration of the TNF-α-neutralization effect. As the proteolytic site for cleavage is the threonine 223 residue in the upper-hinge site of IgG1, the treatment of the TNF-blocking agents rendered Fab monomers and the Fcγ1 fraction.14 The absence of binding of Fab to soluble TNF might be explained by a rearrangement of the Fab fragment protein conformation, thus the Fc fraction being necessary for the spatial and bivalent capacity of biologics to recognize and block a cytokine. To reach a similar conclusion for vedolizumab, one should perform a similar study in a cell line sensitive to α4β7 binding.
In summary, we showed in this study that neutrophil elastase activity was increased in the mucosa of patients with UC, contributing to extracellular matrix degradation, although neutrophil elastase specific inhibitor elafin was highly expressed. We also demonstrated that elastase affected the integrity of therapeutic monoclonal antibodies and the fusion protein etanercept. Specifically, the TNF-α-neutralizing capacity of anti-TNF biologics was compromised by HNE. Our results suggest that the high level of neutrophil elastase and the imbalance with the specific inhibitor, elafin, in active UC mucosa reflect the deregulated protease activity occurring in the inflamed intestinal mucosa and contribute to the loss of function of different biologic agents which are extensively used in these patients. Consequently, this might at least partially explain why many UC patients are non-responsive to biologic treatment.33 Notwithstanding, we could inhibit mucosal elastynolitic activity by adding exogenous elafin to mucosa ex vivo, and elafin protected the biologics from elastase degradation in vitro. Taking into account that the administration of protease inhibitors has been proposed as a potential treatment for IBD, and the recent advances in the development of a personalized treatment that targets an underlying genetic or microbial origin of the disease, complementation of anti-inflammatory biologic therapy with exogenous elafin should be considered as an option for UC patients.34,35
Acknowledgments
Renata Curciarello and Guillermo H. Docena are researchers from CONICET (Argentina) and received funding from Agencia Nacional de Promoción Científica (PICT2016-2387 and PICT2015-1648, respectively). RC received a special travel grant for Assistant Researchers from CONICET and performed part of this research study at Centre for Immunobiology-QMUL under Prof. MacDonald’s supervision (2017). We acknowledge Dr. Anna Vossenkaemper, from Prof. MacDonald’s group, for her support and advice. We are very grateful to the staff at the endoscopy unit at the Royal London Hospital, as well as to all patients who kindly contributed to this study.
Disclosure
Dr Klaartje Kok reports she has had consultancies from janssen and takeda outside the submitted work (these companies have no financial interest in this article). The authors report no other possible conflicts of interest in this work.
References
1. Torres J, Mehandru S, Colombel J-F, Peyrin-Biroulet L. Crohn’s disease. Lancet. 2017;389(10080):1741–1755. doi:10.1016/S0140-6736(16)31711-1
2. Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel J-F. Ulcerative colitis. Lancet. 2017;389(10080):1756–1770. doi:10.1016/S0140-6736(16)32126-2
3. Fournier BM, Parkos CA. The role of neutrophils during intestinal inflammation. Mucosal Immunol. 2012;5(4):354–366. doi:10.1038/mi.2012.24
4. de Bruyn M, Vandooren J, Ugarte-Berzal E, Arijs I, Vermeire S, Opdenakker G. The molecular biology of matrix metalloproteinases and tissue inhibitors of metalloproteinases in inflammatory bowel diseases. Crit Rev Biochem Mol Biol. 2016;1–64. doi:10.1080/10409238.2016.1199535
5. Giuffrida P, Biancheri P, MacDonald TT. Proteases and small intestinal barrier function in health and disease. Curr Opin Gastroenterol. 2014;30(2):147–153. doi:10.1097/MOG.0000000000000042
6. Motta J-P, Magne L, Descamps D, et al. Modifying the protease, antiprotease pattern by elafin overexpression protects mice from colitis. Gastroenterology. 2011;140(4):1272–1282. doi:10.1053/j.gastro.2010.12.050
7. Pfundt R, van Ruissen F, van Vlijmen-willems IM, et al. Constitutive and inducible expression of SKALP/elafin provides anti-elastase defense in human epithelia. J Clin Invest. 1996;98(6):1389–1399. doi:10.1172/JCI118926
8. Motta J-P, Bermúdez-Humarán LG, Deraison C, et al. Food-grade bacteria expressing elafin protect against inflammation and restore colon homeostasis. Sci Transl Med. 2012;4(158):158ra144. doi:10.1126/scitranslmed.3004212
9. Morohoshi Y, Matsuoka K, Chinen H, et al. Inhibition of neutrophil elastase prevents the development of murine dextran sulfate sodium-induced colitis. J Gastroenterol. 2006;41(4):318–324. doi:10.1007/s00535-005-1768-8
10. Ben-Horin S, Kopylov U, Chowers Y. Optimizing anti-TNF treatments in inflammatory bowel disease. Autoimmun Rev. 2014;13(1):24–30. doi:10.1016/j.autrev.2013.06.002
11. Biancheri P, Brezski RJ, Di Sabatino A, et al. Proteolytic cleavage and loss of function of biologic agents that neutralize tumor necrosis factor in the mucosa of patients with inflammatory bowel disease. Gastroenterology. 2015;149(6):1564–1574.e3. doi:10.1053/j.gastro.2015.07.002
12. Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N Engl J Med. 1987;317(26):1625–1629. doi:10.1056/NEJM198712243172603
13. Kabouridis PS, Hasan M, Newson J, Gilroy DW, Lawrence T. Inhibition of NF-kappa B activity by a membrane-transducing mutant of I kappa B alpha. J Immunol Baltim Md 1950. 2002;169(5):2587–2593.
14. Ryan MH, Petrone D, Nemeth JF, Barnathan E, Björck L, Jordan RE. Proteolysis of purified IgGs by human and bacterial enzymes in vitro and the detection of specific proteolytic fragments of endogenous IgG in rheumatoid synovial fluid. Mol Immunol. 2008;45(7):1837–1846. doi:10.1016/j.molimm.2007.10.043
15. Sengupta N, MacDonald TT. The role of matrix metalloproteinases in stromal/epithelial interactions in the gut. Physiol Bethesda Md. 2007;22:401–409. doi:10.1152/physiol.00027.2007
16. Motta J-P, Martin Laurence VN. Proteases/antiproteases in inflammatory bowel diseases. In: Vergnolle N, Chingnard M (Editors). Proteases and Their Receptors in Inflammation. Springer; 2011:173–215. Available from: https://www.springer.com/gp/book/9783034801560.
17. Van Spaendonk H, Ceuleers H, Witters L, et al. Regulation of intestinal permeability: the role of proteases. World J Gastroenterol. 2017;23(12):2106–2123. doi:10.3748/wjg.v23.i12.2106
18. Edgington-Mitchell LE. Pathophysiological roles of proteases in gastrointestinal disease. Am J Physiol Gastrointest Liver Physiol. 2016;310(4):G234–G239. doi:10.1152/ajpgi.00393.2015
19. Pederoli-Ribeil M, Gabillet J, Witko-Sarsat V. Proteases from inflammatory cells: regulation of inflammatory response. In: Vergnolle N, Chingnard M (Editors). Proteases and Their Receptors in Inflammation. Springer; 2011:73–100. Available from: https://www.springer.com/gp/book/9783034801560.
20. Segel GB, Halterman MW, Lichtman MA. The paradox of the neutrophil’s role in tissue injury. J Leukoc Biol. 2011;89(3):359–372. doi:10.1189/jlb.0910538
21. Bennike TB, Carlsen TG, Ellingsen T, et al. Neutrophil extracellular traps in ulcerative colitis: a proteome analysis of intestinal biopsies. Inflamm Bowel Dis. 2015;21(9):2052–2067. doi:10.1097/MIB.0000000000000460
22. Muthas D, Reznichenko A, Balendran CA, et al. Neutrophils in ulcerative colitis: a review of selected biomarkers and their potential therapeutic implications. Scand J Gastroenterol. 2017;52(2):125–135. doi:10.1080/00365521.2016.1235224
23. Denadai-Souza A, Bonnart C, Tapias NS, et al. Functional proteomic profiling of secreted serine proteases in health and inflammatory bowel disease. Sci Rep. 2018;8(1):7834. doi:10.1038/s41598-018-26282-y
24. Gottlieb Y, Elhasid R, Berger-Achituv S, Brazowski E, Yerushalmy-Feler A, Cohen S. Neutrophil extracellular traps in pediatric inflammatory bowel disease. Pathol Int. 2018;68(9):517–523. doi:10.1111/pin.12715
25. Langhorst J, Elsenbruch S, Koelzer J, Rueffer A, Michalsen A, Dobos GJ. Noninvasive markers in the assessment of intestinal inflammation in inflammatory bowel diseases: performance of fecal lactoferrin, calprotectin, and PMN-elastase, CRP, and clinical indices. Am J Gastroenterol. 2008;103(1):162–169. doi:10.1111/j.1572-0241.2007.01556.x
26. Theocharis AD, Manou D, Karamanos NK. The extracellular matrix as a multitasking player in disease. FEBS J. 2019;286(15):2830–2869. doi:10.1111/febs.14818
27. Antonicelli F, Bellon G, Debelle L, Hornebeck W. Elastin-elastases and inflamm-aging. Curr Top Dev Biol. 2007;79:99–155. doi:10.1016/S0070-2153(06)79005-6
28. Brezski RJ, Vafa O, Petrone D, et al. Tumor-associated and microbial proteases compromise host IgG effector functions by a single cleavage proximal to the hinge. Proc Natl Acad Sci. 2009;106(42):17864–17869. doi:10.1073/pnas.0904174106
29. Guentsch A, Hirsch C, Pfister W, et al. Cleavage of IgG1 in gingival crevicular fluid is associated with the presence of Porphyromonas gingivalis. J Periodontal Res. 2013;48(4):458–465. doi:10.1111/jre.12027
30. Brezski RJ, Jordan RE. Cleavage of IgGs by proteases associated with invasive diseases: an evasion tactic against host immunity? mAbs. 2010;2(3):212–220. doi:10.4161/mabs.2.3.11780
31. Billmeier U, Dieterich W, Neurath MF, Atreya R. Molecular mechanism of action of anti-tumor necrosis factor antibodies in inflammatory bowel diseases. World J Gastroenterol. 2016;22(42):9300–9313. doi:10.3748/wjg.v22.i42.9300
32. Bye WA, Jairath V, Travis SPL. Systematic review: the safety of vedolizumab for the treatment of inflammatory bowel disease. Aliment Pharmacol Ther. 2017;46(1):3–15. doi:10.1111/apt.14075
33. Naviglio S, Giuffrida P, Stocco G, et al. How to predict response to anti-tumour necrosis factor agents in inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2018;12(8):797–810. doi:10.1080/17474124.2018.1494573
34. Bermúdez-Humarán LG, Motta J-P, Aubry C, et al. Serine protease inhibitors protect better than IL-10 and TGF-β anti-inflammatory cytokines against mouse colitis when delivered by recombinant lactococci. Microb Cell Fact. 2015;14:26. doi:10.1186/s12934-015-0198-4
35. Ashton JJ, Mossotto E, Ennis S, Beattie RM. Personalising medicine in inflammatory bowel disease-current and future perspectives. Transl Pediatr. 2019;8(1):56–69. doi:10.21037/tp.2018.12.03
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.