Back to Journals » Cancer Management and Research » Volume 13
Hsa-miR-149-5p Suppresses Prostate Carcinoma Malignancy by Suppressing RGS17
Authors Ma J, Wei H, Li X, Qu X
Received 14 September 2020
Accepted for publication 19 February 2021
Published 25 March 2021 Volume 2021:13 Pages 2773—2783
DOI https://doi.org/10.2147/CMAR.S281968
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Eileen O'Reilly
Jinhua Ma, Hongbing Wei, Xianlin Li, Xi Qu
Department of Urinary Surgery, The Third People’s Hospital of Hubei Province, Wuhan, Hubei, 430030, People’s Republic of China
Correspondence: Xi Qu
Department of Urinary Surgery, The Third People’s Hospital of Hubei Province, No. 26 Zhongshan Road, Wuhan, Hubei, 430030, People’s Republic of China
Tel +86 13476046068
Email [email protected]
Background: MicroRNAs (miRNAs) are key players in the progression of human cancers. While several miRNAs have been reported to regulate the development of tumors, the molecular mechanisms and roles of miR-149-5p in prostate carcinoma (PCa) remain unclear. Our aim was to investigate the interaction and functions of miR-149-5p and RGS17 in PCa.
Methods: Microarray analysis was performed to identify the key miRNA and gene involved in PCa progression. The expression levels of miRNA and mRNA in PCa tissues and cells were verified by qRT-PCR. MTT assay, BrdU proliferation assay and wound-healing assay were applied to assess the effect of miR-149-5p and RGS17 on PCa cells’ viability, proliferation, and migration ability. The association between RGS17 and miR-149-5p was identify using dual-luciferase reporter assay and Western blot assay.
Results: Data analysis indicated the reduction of miR-149-5p expression in PCa tissues and cells. Experimental investigations also showed that this miRNA suppressed the viability, proliferation and migration ability of PCa cells. RGS17 was found to be the target of miR-149-5p, and the low expression of miR-149-5p upregulated RGS17 in PCa tissues and cells. The results of the cell-function assays showed that RGS17 acted as an oncogene in PCa even though its promotive effect could be reversed by miR-149-5p.
Conclusion: This research confirmed that by targeting and inhibiting RGS17, miR-149-5p could suppress PCa development.
Keywords: miR-149-5p, prostate carcinoma, PCa, RGS17, malignancy
Introduction
Prostate carcinoma (PCa) is touted as the most common malignancy among males. This tumor has affected the reproductive systems of men and resulted in their untimely death.1 The incidence of PCa has increased gradually in developing countries such as China, and the risk factors for PCa include family history, genetics, diets, obesity and diabetes.2 Although much progress has been made to improve the survival rate of patients with this cancer, existing treatment methods such as surgery, chemotherapy, and radiotherapy have several limitations. To improve the survival rate of PCa patients, especially advanced patients, diagnostic and treatment methods need to be further improved by understanding the underlying mechanisms of this tumor.
MicroRNAs(miRNAs) represent a group of non-coding RNAs characterized by a length of t 20–24 nucleotides.3 Even though they cannot be translated into protein, miRNAs can regulate gene expression after transcription.4 By binding to the 3ʹ-untranslated region (UTR) of corresponding protein-coding genes, miRNAs participate in multiple tumor-formation processes.5–7 Recent research demonstrated the low expression of miR-149 in many cancer types, including lung cancer, osteosarcoma, and bladder cancer.8–10 However, no studies have systematically explored the relationship between miR-149-5p and PCa or the roles played by miR-149-5p in PCa.
Located on chromosome 6q25.3, RGS17 (Regulator of G Protein Signaling 17) can encode multiple proteins to regulate the G-protein signaling family.11,12 This protein-coding gene contains a conserved structure domain, named the RGS domain; it also has a region rich in cysteine that contains 120 amino acid motifs. RGS17 can influence the activity of G-proteins and serve as a GTPase activating protein (GAP), thereby enhancing the conversion rate of GTP to GDP.13–15 It is reported that the conversion of GTP to GDP facilitates tumor angiogenesis/growth and the intrusion/metastasis of cancer cells.16 Therefore, RGS17 might have the capability to influence the development of malignancies. One research reported that by targeting RGS17, miR-199 suppressed cell intrusion, migration and proliferation in hepatocellular carcinoma.17 However, the impact of RGS17 on prostate cancer is still unknown.
This research aimed to investigate the effect of miR-149-5p and RGS17 on PCa using microarray analysis and cell-function experiments. Based on previous studies, we hypothesized that miR-149-5p might function as a tumor suppressor in PCa cells by directly targeting RGS17. Regardless of the outcome of the research, our findings could provide a therapeutic solution for patients with PCa.
Methods
Microarray Analysis
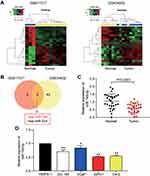
GSE17317 and GSE34932 downloaded from the GEO DataSets were the mRNA profile, while GSE69223 downloaded from the GEO DataSets was the mRNA profile. GSE17317 included four cell lines including two late prostate cancer cell lines (PC3 and DU 145), early prostate cancer cell line LNCaP and prostate epithelial cell line RWPE-1, GSE34932 consisted eight normal frozen prostate tissue samples and eight frozen prostate cancer tissue samples, and GSE69223 was comprised of 15 paired normal prostate samples and prostate cancer tissue samples. The limma package was employed to select the differentially expressed miRNAs or differentially expressed genes (DEGs). Also, Venny 2.1.0 was utilized to select the overlapping miRNAs or DEGs.
Clinical Tissues and Cell Clines
A total of 30 paired PCa tissues and adjacent normal tissues were provided by The Third People ‘s Hospital of Hubei Province. Prostate tissues were stored in a liquid nitrogen tank. This research was performed according to the recommendations enshrined in the Declaration of Helsinki and was approved by the Ethics Committee of the Third People ‘s Hospital of Hubei Province. The patients completed their consent forms to participate in this study. The clinical characteristics of the patients are shown in Table 1. Four human prostate cancer cell lines (DU 145, VCaP, 22Rv1 and C4-2) and one human normal prostatic cell line (RWPE-1) were purchased from the American Type Culture Collection (ATCC, USA).
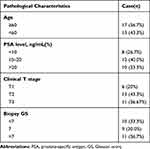 |
Table 1 Clinical Parameters of Patients with Prostate Carcinoma in This Study |
Cell Culture
DU 145, RWPE-1 and VCaP cells were kept in DMEM (Cat#:11320033, Invitrogen, USA), which contained 10% fetal bovine serum (FBS, Cat#:16140071, Gibco, USA), 100U/mL penicillin (Cat#:61305, Sigma, China) and 100μg/mL streptomycin (Cat#:85886, Sigma, China). Next, 22RV1 cells were cultured in RPMI-1640 Medium (Cat#:30–2001, ATCC, USA), which contained 10% FBS. Then, C4-2 cells were cultured in DMEM and F12 Medium (DMEM/F12, 4: 1 mixing ratio), which was supplemented with 10% FBS. The cells were subsequently incubated at 37°C in an atmosphere that contained 5% CO2. They were finally removed and used for subsequent experiments.
Cell Transfection
The miR-149-5p mimics, RGS17 overexpression vectors, as well as their corresponding negative control (NC), were purchased from RiboBio (Guangzhou, China). To overexpress miR-149-5p or RGS17, C4-2 and 22Rv1 cells were first seeded in 96-well plates at a concentration of 1×106 cells/well. After the cells were added to 180 μL DMEM or RPMI-1640 Medium, they were incubated at 37°C in air containing 5% CO2 for 24 h. Lipofectamine 2000 Reagent (Cat#:11668019, Invitrogen, USA) was used according to the manufacturer’s instructions to induce the miR-149-5p mimic, NC, or RGS17 over-expression vectors transfected into 22Rv1 and C4-2 cells. After being transfected for two days, the cells were collected, and the transfection efficiency was verified using qRT-PCR.
Identification of miR-149-5p and RGS17 Expression
To quantify the expression of miR-149-5p and RGS17, the total RNA was first extracted from different prostate tissues and cells. This extraction was done with TRIzol Reagent (Cat#:1555960226, Invitrogen, USA) according to the manufacturer’s guidelines. The total RNAs were reverse-transcribed to cDNA with the cDNA synthesis kit (Cat#:11483188001, Sigma, USA). The reaction was performed in the following order: pre-degeneration at 95°C continued for 10 min, denaturation at 95°C continued for 30 s, renaturation at 55°C continued for 30 s, and extension at 72°C lasted for 2 min for a total of 35 cycles. Finally, the 2−ΔΔCt analysis was applied to calculate the expression of miR-149-5p and RGS17. The primer sequences used are listed in Table 2.
 |
Table 2 The Sequences of the Primers in This Study |
Cell Viability Detection
The cells in the log phase were first seeded into 96-well plates at a concentration of 2 × 103 cells/well. They were then incubated for 24 h. The MTT assay was later performed to detect the viability of the cells at 0 h, 24 h, 48 h, and 72 h after transfection. Following this, the MTT solution (5mg/mL) was added to the cells, and the mixture was incubated for 4 h. Finally, the absorbance at 570 nm was detected using a microplate reader.
Cell Proliferation Detection
The Cell Proliferation ELISA BrdU Kit (Roche, Germany) was used to detect the proliferation ability of the transfected 22Rv1 and C4-2 cells. Briefly, the cells (6000 cells/well) were seeded in the 96-well plate and cultured for 72 h. Then, the BrdU solution (10 μL/well) was added to cells, and the mixture was incubated for 4 h. The absorbance at 450 nm was detected using a microplate reader.
Cell Migration Assessment
The wound-healing assay was performed to identify the migration ability of C4-2 and 22Rv1 cells. Briefly, the 2×105/well transfected cells were cultured in a 6-well plate and incubated until the cell confluence was over 90%. Then, a clean pipette tip (200μL) was applied to create a scratch from the cell monolayer, and the fallen cells were washed gently with PBS. After incubating the cells with the fresh serum-free medium for 24 h, the wound was photographed and measured with an inverted microscope. The migration rate was eventually calculated with the following formula: (wound width at 0 h-wound width at 24 h)/(wound width at 0 h).18
Relationship Between miR-149-5p and RGS17
TargetScan was employed to predict the binding site of the RGS17 3ʹUTR for miR-149-5p, and the relationship between miR-149-5p and RGS17 was confirmed using luciferase assay. The wild-type RGS17 (RGS17-WT) and mutated binding sites of the RGS17 3ʹUTR (RGS17-MUT) were cloned to the pmirGLO Dual-Luciferase miRNA Target Expression Vector (Promega, USA). After the 22Rv1 and C4-2 cells were cultured in 24-well plates, they were co-transfected with 25 ng NC/miR-149-5p mimic and 100 ng RGS17-WT/RGS17-MUT vectors containing firefly luciferase. After transfecting the cells for 48 h, the luciferase activities of firefly and renilla were detected with a Dual-Luciferase Reporter Assay System (Promega, USA).
Detection of RGS17 Protein Expression
The Western blot assay was performed to examine the expression level of RGS17 in C4-2 and 22Rv1 cells. Briefly, proteins were extracted from the transfected cells using RIPA lysis buffer (Cat#:C500005, Sangon, China). After quantifying the concentration of extracted protein using the bicinchoninic acid (Pierce, Waltham, MA, USA), 30 μg protein was detached with 12% SDS-PAGE gel and transferred to the PVDF membrane. After being sealed with 5% skimmed milk for 1 h, the membrane was incubated overnight at 4°C with the primary antibodies of rabbit anti-human RGS17 (Cat#: ab96675, Abcam, UK) and GAPDH (Cat#: ab205718, Abcam, UK). Next, the membranes were incubated with the goat anti-rabbit IgG antibody (Cat#: ab6721, Abcam, UK) for 1 h at 25°C. After the protein signal was enhanced with the ECL system (Thermo Fisher Scientific, USA), the expression level of RGS17 was calculated using ImageJ software.
Statistical Analysis
Statistical analysis of the data collected was performed with SPSS 23.0 software (IBM SPSS, USA). All collected data were presented in this format: mean ± standard deviation (SD). The statistical difference between two groups was analyzed using the Student’s t-test, whereas the statistical difference between three or more groups was analyzed using the one-way or two-way ANOVA. If the P-value was below 0.05, the data were assumed to be statistically significant.
Results
miR-149-5p Expression is Downregulated in PCa Samples
A total of 44 differentially expressed miRNAs and 88 differentially expressed miRNAs were screened out from GSE17317 and GSE34932, respectively, with P-value less than 0.05 and |log2FC| more than 0.02 (Figure 1A). After performing Venny analysis, hsa-miR-149 and hsa-miR-224 were the overlapping differentially expressed miRNAs for GSE17317 (P-value <0.001, |log2FC| >1) and GSE34932 (P-value <0.05, |log2FC| >1), respectively (Figure 1B). After literature review, we finally selected miR-149-5p as our miRNA of interest. After qRT-PCR was used to analyze the expression of miR-149-5p in PCa tissues, results showed that the expression level of miR-149-5p in prostate tissues (n=30) was reduced by 42.6% compared with the paired adjacent normal tissues (Figure 1C). The expression level of miR-149-5p was also downregulated in the PCa cell lines (VCaP, DU 145, 22Rv1 and C4-2) compared with the normal prostatic cell line (RWPE-1) (Figure 1D). Among the PCa cell lines, the C4-2 and 22Rv1 cells had the lowest relative expression level. For this reason,the C4-2 and 22Rv1 cells were used as the cell model in subsequent experiments.
miR-149-5p Suppresses PCa Cells
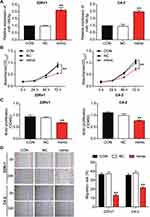
The 22Rv1 and C4-2 cells were transfected with the miR-149-5p mimic to identify the function of miR-149-5p in PCa cells. The qRT-PCR results showed that miR-149-5p expression increased by 2.12-fold in 22Rv1 and 1.99-fold in C4-2 cells (Figure 2A). An MTT assay was carried out to assess cell viability at 0, 24, 48 and 72 h. The MTT assay results indicated that overexpressed miR-149-5p significantly inhibited the viability of the 22Rv1 and C4-2 cells (Figure 2B). The BrdU proliferation assay results indicated that miR-149-5p inhibited the proliferation ability of the 22Rv1 and C4-2 cells (Figure 2C). Besides, the wound-healing assay data demonstrated miR-149-5p mimic decreased the migration rate of the 22Rv1 cells by 28.4% and that of C4-2 cells by 62.1% (Figure 2D). Overall, these results suggested that miR-149-5p could suppress 22Rv1 and C4-2 cells.
RGS17 is the Target of miR-149-5p
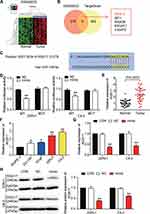
GSE69223 included 1730 DEGs with the adj.P-value less than 0.05 and |log2FC| more than 1 (Figure 3A). TargetScan predicted 497 genes to which miR-149-5p could bind, and 223 upregulated genes were screened out from GSE69233 with |log2FC| more than 1.5. Finally, the five overlapping genes (RGS17, WT1, RAB3B, B3GAT1, C2orf72) were identified from GSE69233 and TargetScan prediction (Figure 3B). RGS17 was selected as our gene of interest to investigate after literature review. The binding site between RGS17 and miR-149-5p was predicted by TargetScan (Figure 3C). The relative luciferase activity was reduced when the 22Rv1 and C4-2 cells were transfected with the RGS17-WT and miR-149-5p mimic, a result that indicated that RGS17 was the target of miR-149-5p (Figure 3D). The expression of RGS17 mRNA was also detected in tumor and non-tumor prostate tissues, and the result showed that the RGS17 expression was upregulated by 2.20-fold in tumor tissues (Figure 3E). The expression of RGS17 mRNA in PCa cells and normal prostate cells displayed the same result as that in tissues (Figure 3F). Besides, when the 22Rv1 and C4-2 cells were transfected with the miR-149-5p mimic, the expression of both the RGS17 mRNA and the protein was reduced, meaning RGS17 expression could be suppressed due to the direct targeting of miR-149-5p (Figure 3G-H). In short, these data showed that miR-149-5p could restrain RGS17 expression by directly targeting the RGS17 3ʹUTR.
RGS17 Overexpression Reverses the Inhibitory Effect of miR-149-5p on PCa Cells
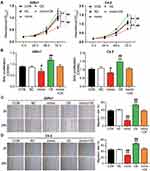
To verify whether RGS17 was related to the regulatory mechanism of miR-149-5p in PCa cells, RGS17 overexpression vectors were transfected or co-transfected with the miR-149-5p mimic in 22Rv1 and C4-2 cells. The MTT assay data revealed that RGS17 overexpression enhanced cell viability in both 22Rv1 and C4-2 cells; however, the co-transfection of RGS17 with the miR-149-5p mimic overturned the influence of RGS17 or miR-149-5p on cell viability (Figure 4A). The BrdU proliferation assay results showed that RGS17 overexpression increased the cell proliferation ability by 1.32-fold and 1.42-fold in 22Rv1 and C4-2 cells, respectively; nonetheless, the co-transfection of RGS17 with the miR-149-5p mimic did not show any difference compared with the control group (Figure 4B). In 22Rv1 cells, the migration rate rose by 1.60-fold in the RGS17 overexpression group; however, the migration rate did not change in the co-transfection group compared with the control group (Figure 4C). The result in C4-2 cells was similar to that in 22Rv1 cells (Figure 4D). Overall, these results indicated that RGS17 could promote cell viability, proliferation, and migration in C4-2 and 22Rv1 cells; however, these effects could be reversed by upregulating miR-149-5p.
Discussion
PCa, the most common cancer of males, threats to the health of males. In our research, we found that the expression of miR-149-5p was downregulated in PCa, and the miR-149-5p overexpression impaired the malignancy of PCa cells. Meanwhile, we found that the RGS17 targeted by miR-149-5p was upregulated in PCa, and its expression was inhibited by miR-149-5p overexpression. RGS17 enhanced the ability of cell viability, proliferation and migration in C4-2 and 22Rv1 cells. What’s more, RGS17 could reverse the inhibitory function of miR-149-5p in malignancy of PCa cells.
Increasing evidence suggests that miRNAs influence the progression of cancer. For instance, miR-149, miR-149-5p and miR-149-3p have been found to be crucial in regulating cancerous processes. They can also play dual roles in the pathogenesis of different cancer types. For instance, one study demonstrated that miR-149-5p suppressed acute myeloid leukemia cells’ apoptosis, thus exacerbating myeloid leukemia progression.19 Nevertheless, the vast majority of studies revealed that miR-149-5p could downregulate malignant tissues and cells and hinder the progression of tumor types such as renal cancer,20 melanoma,21 thyroid carcinoma22 and colorectal carcinoma.23 Furthermore, miR-149-3p could serve as an oncogene, thereby promoting the proliferation of tumor cells and limiting the apoptosis of tumor cells.24,25 In another study, the suppressive effect of tumors was documented in other cancer types, such as bladder cancer,9 colorectal cancer26 and oral squamous cell carcinoma.27 It thus appears that miR-149-5p and miR-149-3p may exert neither antergic or synergetic function in the progression of a cancer. In PCa, miR-149 was found to promote cell proliferation in PCa cell lines.28 Moreover, miR-149-3p has been reported to promote the cell growth of prostate tumors and inhibit the apoptosis of prostate tumors.29 However, the significance of miR-149-5p in PCa has not been investigated. In our study, we identified miR-149-5p with aberrantly low expression in PCa tumor tissues and cell lines through bioinformatical analysis and qRT-PCR, which is consistent with the data published by Schaefer et al.30 Furthermore, our study revealed that miR-149-5p overexpression impaired the malignancy of PCa cells, thereby acting as a tumor suppressor in PCa cells. What’s more, we also found that miR-149-5p could target RGS17 to reverse the positive effect of RGS17 on PCa cells.
RGS17 belongs to the regulator of G protein signaling family which is identified as GTPase-activators to negatively regulate the GPCR signaling.31 Although, the function of RGS17 in biological events has little been explored, it is worthy of highlighting its significance due to the crucial and extensive role of GPCR signally in physiological and pathological processes carcinogenesis.32 Several researches have demonstrated RGS17 was overexpressed in colorectal and lung carcinoma tissues and could facilitate the tumor development and progression in vitro.33,34 In addition, RGS17 was reported to serve as the downstream target of miR-203 in PCa cells, and knockdown of RGS17 inhibited the tumorigenicity of PCa cells.35 Consistently, in this study, we found that RGS17 targeted by miR-149-5p was upregulated in PCa, and its expression was inhibited by miR-149-5p overexpression. More specifically, RGS17 enhanced cell viability, proliferation and migration in C4-2 and 22Rv1 cells. What’s more, RGS17 could reverse the inhibitory function of miR-149-5p in PCa cells. In this sense, we explored a novel miRNA that could target and negatively regulate RGS17 in PCa, which extend the complex pathogenesis of PCa.
In spite of our findings, several limitations can be found in this study. Only in vitro experiments were performed. In the future, experiments should be conducted in vivo to confirm the impact of miR-149-5p and RGS17 on PCa. More specifically, in future work, a mouse model should be constructed by transplanting PCa cell lines transfected with miR-149-5p mimics and/or RGS17 overexpression vector into nude mice to observe the effect of miR-149-5p and RGS17 on tumor formation. In addition, specific signaling mechanisms of the miR-149-5p/RGS17 axis in PCa cells should be further explored. A study reported that RGS17 overexpression induced PCa cells’ proliferation via cyclic AMP-PKA-CREB pathways,34 and PKA and CREB have been verified to play a key role in the tumorigenesis of endocrine tissues.36 However, given that RGS17 could inhibit GPCR, we speculated many other signaling pathways might be instrumental in the progression of PCa. Therefore, an in-depth transcriptome sequencing study should be conducted to exploit novel downstream pathways. Finally, there are lack of the integrated analysis of the express and functional mode of miR-149-5p with many other aberrantly expressed miRNAs reported previously in PCa tissues. This research inquiry might provide invaluable and practical information for PCa treatments.
Conclusion
This research suggested that by targeting and inhibiting RGS17, miR-149-5p could suppress PCa progression. The knowledge of this research has expanded our understanding of PCa pathogenesis and provided more insights into how to treat and diagnose patients with this malignancy.
Abbreviations
PCa, Prostate carcinoma; miRNA, MicroRNA; UTR, untranslated region ; RGS17, The Regulator of G Protein Signaling 17; GAP, GTPase activating protein; DEGs, differentially expressed genes; NC, negative control.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
The present study was approved by the Ethics Committee of the Third People’s Hospital of Hubei Province (Wuhan, China).
Consent for Publication
Consent for publication was obtained from the participants.
Funding
No funding was received for this work.
Disclosure
The authors declare that they have no conflict of interests for this work.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108.
2. Kimura T, Egawa S. Epidemiology of prostate cancer in Asian countries. Int J Urol. 2018;25(6):524–531.
3. Jeon YJ, Kim OJ, Kim SY, et al. Association of the miR-146a, miR-149, miR-196a2, and miR-499 polymorphisms with ischemic stroke and silent brain infarction risk. Arterioscler Thromb Vasc Biol. 2013;33(2):420–430.
4. Lee KT, Nam JW. Post-transcriptional and translational regulation of mRNA-like long non-coding RNAs by microRNAs in early developmental stages of zebrafish embryos. BMB Rep. 2017;50(4):226–231.
5. Matsushita R, Seki N, Chiyomaru T, et al. Tumour-suppressive microRNA-144-5p directly targets CCNE1/2 as potential prognostic markers in bladder cancer. Br J Cancer. 2015;113(2):282–289.
6. Tsai MM, Wang CS, Tsai CY, et al. MicroRNA-196a/-196b promote cell metastasis via negative regulation of radixin in human gastric cancer. Cancer Lett. 2014;351(2):222–231.
7. Tian X, Wei Z, Wang J, Liu P, Qin Y, Zhong M. MicroRNA-429 inhibits the migration and invasion of colon cancer cells by targeting PAK6/cofilin signaling. Oncol Rep. 2015;34(2):707–714.
8. Xu RD, Feng F, Yu XS, Liu ZD, Lao LF. miR-149-5p inhibits cell growth by regulating TWEAK/Fn14/PI3K/AKT pathway and predicts favorable survival in human osteosarcoma. Int J Immunopathol Pharmacol. 2018;32:2058738418786656.
9. Yang D, Du G, Xu A, Xi X, Li D. Expression of miR-149-3p inhibits proliferation, migration, and invasion of bladder cancer by targeting S100A4. Am J Cancer Res. 2017;7(11):2209–2219.
10. Zhao L, Liu L, Dong Z, Xiong J. miR-149 suppresses human non-small cell lung cancer growth and metastasis by inhibiting the FOXM1/cyclin D1/MMP2 axis. Oncol Rep. 2017;38(6):3522–3530.
11. Rangel J, Nosrati M, Leong SP, et al. Novel role for RGS1 in melanoma progression. Am J Surg Pathol. 2008;32(8):1207–1212.
12. Sierra DA, Gilbert DJ, Householder D, et al. Evolution of the regulators of G-protein signaling multigene family in mouse and human. Genomics. 2002;79(2):177–185.
13. Bodle CR, Mackie DI, Roman DL. RGS17: an emerging therapeutic target for lung and prostate cancers. Future Med Chem. 2013;5(9):995–1007.
14. Sternweis PC, Robishaw JD. Isolation of two proteins with high affinity for guanine nucleotides from membranes of bovine brain. J Biol Chem. 1984;259(22):13806–13813.
15. Roman DL, Traynor JR. Regulators of G protein signaling (RGS) proteins as drug targets: modulating G-protein-coupled receptor (GPCR) signal transduction. J Med Chem. 2011;54(21):7433–7440.
16. Yamauchi Y, Miura Y, Kanaho Y. Machineries regulating the activity of the small GTPase Arf6 in cancer cells are potential targets for developing innovative anti-cancer drugs. Adv Biol Regul. 2017;63:115–121.
17. Zhang W, Qian S, Yang G, et al. MicroRNA-199 suppresses cell proliferation, migration and invasion by downregulating RGS17 in hepatocellular carcinoma. Gene. 2018;659:22–28.
18. Zhu M, Guo J, Xia H, et al. Alpha-fetoprotein activates AKT/mTOR signaling to promote CXCR4 expression and migration of hepatoma cells. Oncoscience. 2015;2(1):59–70.
19. Tian P, Yan L. Inhibition of MicroRNA-149-5p induces apoptosis of acute myeloid leukemia cell line THP-1 by Targeting Fas Ligand (FASLG). Med Sci Monit. 2016;22:5116–5123.
20. Jin L, Li Y, Liu J, et al. Tumor suppressor miR-149-5p is associated with cellular migration, proliferation and apoptosis in renal cell carcinoma. Mol Med Rep. 2016;13(6):5386–5392.
21. Chen W, Zhang J, Xu H, Dai J, Zhang X. The negative regulation of miR-149-5p in melanoma cell survival and apoptosis by targeting LRIG2. Am J Transl Res. 2017;9(9):4331–4340.
22. Ye X, Chen X. miR-149-5p inhibits cell proliferation and invasion through targeting GIT1 in medullary thyroid carcinoma. Oncol Lett. 2019;17(1):372–378.
23. Wang AH, Fan WJ, Fu L, Wang XT. LncRNA PCAT-1 regulated cell proliferation, invasion, migration and apoptosis in colorectal cancer through targeting miR-149-5p. Eur Rev Med Pharmacol Sci. 2019;23(19):8310–8320.
24. Jin L, Hu WL, Jiang CC, et al. MicroRNA-149*, a p53-responsive microRNA, functions as an oncogenic regulator in human melanoma. Proc Natl Acad Sci U S A. 2011;108(38):15840–15845.
25. Fan SJ, Li HB, Cui G, et al. miRNA-149* promotes cell proliferation and suppresses apoptosis by mediating JunB in T-cell acute lymphoblastic leukemia. Leuk Res. 2016;41:62–70.
26. Chen D, Zhang M, Ruan J, et al. The long non-coding RNA HOXA11-AS promotes epithelial mesenchymal transition by sponging miR-149-3p in Colorectal Cancer. J Cancer. 2020;11(20):6050–6058.
27. Shen Q, Zhu H, Lei Q, Chen L, Yang D, Sui W. MicroRNA‑149‑3p inhibits cell proliferation by targeting AKT2 in oral squamous cell carcinoma. Mol Med Rep. 2021;23:3.
28. Fujii T, Shimada K, Tatsumi Y, Fujimoto K, Konishi N. Syndecan-1 responsive microRNA-126 and 149 regulate cell proliferation in prostate cancer. Biochem Biophys Res Commun. 2015;456(1):183–189.
29. Chen Y, Zhao J, Luo Y, Wang Y, Jiang Y. Downregulated expression of miRNA-149 promotes apoptosis in side population cells sorted from the TSU prostate cancer cell line. Oncol Rep. 2016;36(5):2587–2600.
30. Schaefer A, Jung M, Mollenkopf HJ, et al. Diagnostic and prognostic implications of microRNA profiling in prostate carcinoma. Int J Cancer. 2010;126(5):1166–1176.
31. Mao H, Zhao Q, Daigle M, Ghahremani MH, Chidiac P, Albert PR. RGS17/RGSZ2, a novel regulator of Gi/o, Gz, and Gq signaling. J Biol Chem. 2004;279(25):26314–26322.
32. Bar-Shavit R, Maoz M, Kancharla A, et al. G protein-coupled receptors in Cancer. Int J Mol Sci. 2016;17:8.
33. Li L, Luo HS. G-Protein Signaling Protein-17 (RGS17) is upregulated and promotes tumor growth and migration in human colorectal carcinoma. Oncol Res. 2018;26(1):27–35.
34. James MA, Lu Y, Liu Y, Vikis HG, You M. RGS17, an overexpressed gene in human lung and prostate cancer, induces tumor cell proliferation through the cyclic AMP-PKA-CREB pathway. Cancer Res. 2009;69(5):2108–2116.
35. Zhang LS, Ma HG, Sun FH, Zhao WC, Li G. MiR-203 inhibits the malignant behavior of prostate cancer cells by targeting RGS17. Eur Rev Med Pharmacol Sci. 2019;23(13):5667–5674.
36. Rosenberg D, Groussin L, Jullian E, Perlemoine K, Bertagna X, Bertherat J. Role of the PKA-regulated transcription factor CREB in development and tumorigenesis of endocrine tissues. Ann N Y Acad Sci. 2002;968:65–74.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.