Back to Journals » OncoTargets and Therapy » Volume 13
HMGA1 Regulates the Stem Cell-Like Properties of Circulating Tumor Cells from GIST Patients via Wnt/β-Catenin Pathway
Authors Chen M, Xu K, Li B, Wang N , Zhang Q, Chen L, Zhang D, Yang L, Xu Z, Xu H
Received 10 February 2020
Accepted for publication 6 April 2020
Published 11 June 2020 Volume 2020:13 Pages 4943—4956
DOI https://doi.org/10.2147/OTT.S249063
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
Ming Chen,1,2,* Kangjing Xu,1,2,* Bowen Li,1,2,* Nuofan Wang,1,2 Qiang Zhang,3 Liang Chen,4 Diancai Zhang,1,2 Li Yang,1,2 Zekuan Xu,1,2,* Hao Xu1,2,*
1Department of General Surgery, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu 210029, People’s Republic of China; 2Jiangsu Key Laboratory of Cancer Biomarkers, Prevention and Treatment, Jiangsu Collaborative Innovation Center for Cancer Personalized Medical University, Nanjing, Jiangsu 211166, People’s Republic of China; 3Department of Gastrointestinal Surgery, The Second People’s Hospital of Lianyungang, Lianyungang, Jiangsu 222000, People’s Republic of China; 4Department of General Surgery, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, The Affiliated Cancer Hospital of Nanjing Medical University, Nanjing, Jiangsu 210009, People’s Republic of China
*These authors contributed equally to this work.
Correspondence: Hao Xu; Zekuan Xu
Department of General Surgery, The First Affiliated Hospital of Nanjing Medical University, 300 Guangzhou Road, Nanjing, Jiangsu Province 210029, People’s Republic of China
Tel +86 25 68306863
Fax +86 25 83781992
Email [email protected]; [email protected]
Background: Gastrointestinal stromal tumor (GIST) is the most common sarcoma of the digestive system. Circulating tumor cells (CTCs) have been proven to be critical in the recurrence and metastasis of diseases; however, the characteristics of CTCs of GIST are still unclear.
Methods: We sorted out and verified the validity of CTCs from peripheral blood of gastrointestinal stromal tumor (GIST) patients with or without heterochronous liver metastasis using flow cytometry (FCM). Differential genes were analyzed between the GIST patients with and without liver metastasis using next-generation sequencing (NGS).
Results: The preliminary study on the characteristics of CTCs revealed that CTCs of GIST patients with heterochronous liver metastasis had stronger stem cell-like properties (SC-like properties) than CTCs of those without liver metastasis. Furthermore, NGS followed with a series of assays revealed that HMGA1 played a critical role in regulating the SC-like properties of CTCs. Mechanistically, HMGA1 could activate Wnt/β-catenin pathway in vitro and vivo. Moreover, we found that the expression level of HMGA1 in CTCs was an independent risk factor probably influencing the prognosis of GIST patients.
Conclusion: Our findings indicate the significant role of HMGA1 in SC-like properties, IM resistance and eventually hepatic metastasis formation of CTCs. Targeting HMGA1 in CTCs may be a therapeutic strategy for GIST patients with hepatic metastasis.
Keywords: circulating tumor cells, gastrointestinal stromal tumor, stem cell-like properties, heterochronous liver metastasis, HMGA1, Wnt/β-catenin
Introduction
Gastrointestinal stromal tumor (GIST), originating from the Cajal cells in the gastrointestinal tract, is the most common sarcoma of the digestive system.1 While IM has revolutionized the treatment of GIST, IM resistance is inevitable.2 Increasing evidence indicates that most GIST patients gradually become IM resistance, and eventually suffer from recurrence or metastasis 2–3 years later even though they have undergone complete gross resection.3 Tumor recurrence tends to be intraabdominal including the gastric, the intestine, the liver and the peritoneum, of which liver is the most common location of metastasis in GIST due to the gastrointestinal venous blood flowing back from the portal system.4
Many studies show that liquid biopsy via CTC enumeration is highly correlated with disease status in patients and plays an efficient role in monitoring the progression of tumor patients.5–7 Meanwhile, CTCs are considered to be associated with an increased risk of local recurrence as well as the development of distant metastasis.8 Tang et al reported that mCTCs (mesenchymal CTCs) are independent risk factors in metastasis of hepatocellular carcinoma (HCC) and demonstrate the mechanism of axis promoting organ colonization by HCC CTCs.9 In GIST, although most tumor cells perish after the tumor resection and IM administration, a small proportion of tumor cells falling from primary tumor, which could be considered as CTCs, manages to survive and infiltrate into distant organs as disseminated seeds for eventual relapse.10
The CTCs for epithelial tumors were enumerated by utilizing the epithelial cell adhesion molecule (EpCAM) biomarker and the Cell Search-based CTC capturing system.11–13 However, the EpCAM biomarker cannot be used to capture CTCs from mesenchymal tumor patients.14 At present, there is no unified system for the isolation and cultivation of CTC in stromal tumors. This is a novel use of DOG-1 as a specific biomarker for capturing CTCs of GIST patients and cultivate CTCs in vivo and vitro successfully.
The mechanism of heterochronous liver metastasis in GIST is still little known for now. Shen et al demonstrated that SPARCL1 had a certain degree of malignancy-suppressing potential through inhibiting the metastasis of gastric GIST.15 It was reported by Wang that KIT exon 11557–558 deletion upregulated CXCR4 through increased binding of ETV1 to the CXCR4 promoter in GIST cells, thus promoting liver metastasis.16
HMGA1 (high mobility group protein 1) is a member of the high mobility group (HMG) superfamily of proteins comprised of diverse, low molecular weight proteins named by their rapid migration or high mobility in polyacrylamide gel.17 HMGA proteins are among the most abundant, nonhistone chromatin-binding proteins found in the nucleus of cancer cells, sharing an acidic, carboxyl terminus and associate with chromatin, but distinguished by unique functional domains that confer distinct DNA binding motifs and biologic activities.18 To date, HMGA1 proteins are known to participate in a myriad of cellular processes including transcriptional regulation, neoplastic transformation, embryogenesis, anoikis, metastatic progression, cell cycle regulation, repair of DNA damage, cellular senescence, mitochondrial function, and retroviral integration.19 HMGA1, which is a core transcription factor in embryonic stem cells, is enriched in human and murine embryonic stem cells as well as induced pluripotent stem cells and adult stem cells, such as CD34+ hematopoietic stem cells and intestinal stem cells, suggesting that HMGA1 orchestrates transcriptional networks that maintain a primitive, poorly differentiated, stem-like state, both in cancer and normal development.20 Despite its having been characterized as a tumor promotor gene for liver cancer and colorectal cancer, the biological functions of HMGA1 gene and its molecular mechanisms in GIST remain unknown.21 In this study, we discovered that HMGA1 was upregulated in CTCs of GIST patients with heterochronous liver metastasis and that HMGA1 overexpression activated the stem cell-like properties of GIST CTCs via Wnt/β-catenin pathway both in vitro and in vivo.
Herein, we isolated and cultivated CTCs of GIST patients with heterochronous liver metastasis and non-liver metastatic patients. We confirmed that CTCs of patients with heterochronous liver metastasis had stronger stem cell-like properties, which is at least partly caused by HMGA1 overexpression followed with activation of Wnt/β-catenin pathway.
Methods
Patient Recruitment and Sample Collection
The study retrospectively enrolled a total of 73 patients accepting complete resection of primary tumor from September 2014 to December 2018, including 26 GIST patients with heterochronous liver metastasis and 47 without progression. This study was approved by the Clinical Ethics Committee of Nanjing Medical University. Each patient has signed the written informed consent before the enrollment. A 10mL peripheral blood samples from 73 patients were collected and processed. Primary tumor tissues were obtained from patients who underwent radical resection for GIST at the First Affiliated Hospital of Nanjing Medical University China. As for the liver metastatic samples, core needle biopsy (CNB) was performed as percutaneous under the guidance of CT22 and the first 2cm was taken for research use. All tissues were put into liquid nitrogen immediately after isolation.
Sample Processing
A 10mL peripheral blood from patients were collected in 5-mL EDTA tubes (BD Diagnostics) which would be processed as soon as possible within 12 hours to ensure the reliability of results. After that, the blood was layered slowly over equal volume of Ficoll medium (Solarbio, Beijing) in a 50-mL centrifuge tube (Corning, USA) and then density gradient centrifugated at 500 g for 25 minutes at 4°Cwithout brake. Peripheral blood mononuclear cells (PBMCs) and CTCs were harvested by pipetting the mid-layer, transferred into a 15-mL clean tube, and washed twice with phosphate buffered saline (PBS). Finally, cells were resuspended with PBS for further experiments.
CTCs Isolation
CTCs were sorted by FCM (BD, USA) after staining with anti-DOG-1 (ab64085, Abcam, USA) and anti-CD45 (ab214501, Abcam, USA). Alexa Fluor® 488 Goat Anti-Rabbit IgG H&L (ab150077, Abcam, USA) were used to conjugate with anti-DOG-1 antibody. We considered the DOG-1+/CD45- cells as CTCs; meanwhile, the number and percentage of cells were recorded according to the same gating strategy.
CTCs Culture and Viral Infection
CTCs were grown in ultralow attachment plates (Corning, USA) containing tumor sphere medium consisting of RPMI-1640 medium supplemented with EGF (20ng/mL), basic FGF (20ng/mL) in a humidified 37 °C incubator with 5% CO2 and 4% O2.10 Culture medium was changed every 3–4 days and cell clusters were observed under a microscope. For viral infection, Lenti Luc-GFP (GenePharma, Shanghai) was transfected into CTC lines with high titer to make sure that Luc-GFP-CTC lines express both luciferase and GFP stably.
Immunofluorescence Assay
CTC lines were spun down onto poly-L-lysine-functionalized glass slides with Spintrap,23 and postfixed with 4% paraformaldehyde for 20 min followed by washing with PBS. Then, cells were permeabilized with 0.1% Triton X-100 for 10 min, blocked with 1% BSA, and immunostained with the primary antibodies at 4°C overnight. The relevant primary antibodies were anti-DOG-1 (ab64085, Abcam, USA) and anti-CD45 (ab8216, Abcam, USA).The secondary antibodies were Alexa Fluor® 488 goat anti-rabbit IgG (ab150077, Abcam, USA) and Alexa Fluor® 647 goat anti-mouse IgG (ab150115, Abcam, USA). Finally, nuclei were stained with DAPI (Beyotime, Shanghai) for 15min.
Sphere Formation Assay
CTCs were trypsinized (Gibco, USA) and resuspended in RPMI-1640 medium as single-cell suspensions consisting of EGF and basic FGF, and approximately 5000 cells were seeded into 6-well plates with ultra-low attachment surface. After 14 days of culture with replenishment of the medium every 3–4 days, then spheres (diameter ≥ 50μm) were imaged and counted under a microscope. Based on the ratio of sphere number to the total cell count, the efficiency of sphere formation was calculated.
RNA Extraction and qRT-PCR
Total RNA were extracted from CTCs and tumor tissues with TRIzol (Takara, Japan). NanoDrop spectrophotometer (ND-100, Thermo, USA) was used to detect the quality and concentration of RNA. mRNA reverse transcription and qRT-PCR were conducted with PrimeScript RT Master Mix Kit (Takara, Japan) and ChamQ Universal SYBR qPCR Master Mix (Vazyme, Nanjing), respectively. The primers we used were purchased from Realgene (Nanjing). The specific primer sequences are listed in Supplementary Table 1.
IC 50 and Resistance Index (RI) Calculation
The CCK-8 assay (CK04, Dojindo, Japan) was performed to determine the IC50 and RI of CTCs. Briefly, 5000 cells were seeded in 96-well plate and cultured in 100 μL complete medium. After washed with PBS carefully, CTCs were exposed to fresh RPMI-1640 medium with 2.5, 5, 10, 15, 20, 25, 30, 35, 40, 50, 62.5, 75 and 80 μM IM for 48h in humidified chamber. For each concentration, a blank well for each cell line was set up as well as three duplicate wells. Next, each well was added 10μL CCK-8 reagent and incubated for 2 h. Then, automated microplate reader was used to measure the OD value of each well at 450 nm.
Western Blot
CTCs cells were harvested in RIPA buffer with protease and phosphatase inhibitor (KeyGen, Nanjing). Lysates were collected after centrifugation and proteins were quantified with the BCA assay kit (Beyotime, Shanghai). Followed by the electrophoresis, proteins were transferred to nitrocellulose membranes. After blocked with 5% BSA, the membranes were probed with the primary antibodies against with HMGA1 (86846S, Cell Signaling Technology, USA), β-catenin (8480S, Cell Signaling Technology, USA), Actin (AA128, Beyotime, Shanghai), CD44 (ab157107, Abcam, USA) overnight at 4°C.The next day, membranes were incubated with secondary antibody for 2h at room temperature. Protein bands were detected with HRP substrate.
RNA-Seq Analysis
Total RNA was isolated with TRIzol reagent (Takara, Japan). Ribominus Eukaryote Kit (Qiagen, Valencia, CA) was used to remove ribosomal RNA. Then, prepare the strand-specific RNA-seq libraries with the NEBNext Ultra Directional RNA Library Prep Kit for Illumina (NEB, USA).Approximately 50 ng of ribosome-depleted RNA was fragmented and then used for reverse transcription with random hexamer primers. The purified first-strand cDNA was amplified by 13–15 PCR cycles, and then the library was analyzed by Bioanalyser 2100 (Agilent, USA). Finally, the cDNA was sequenced in the Hiseq 2000 system (Illumina, USA) with a 100 bp pairing terminal.
ALDH Activity Analysis
A 1× 106 CTCs were stained with Aldeflour assay Kit (Stemcell technology, Canada) following the manufacturer’s protocol. ALDH inhibitor diethylaminobenzaldehyde (DEAB) was used to set the ALDEFLUOR-positive FACS gate. Percentage of cells with high ALDH-activity in CTC was quantified by flow cytometry (FACS Calibur, BD, USA).
Cell Apoptosis Assay
Cell apoptosis was analyzed using flow cytometric methods. Suspended CTCs were centrifuged at 1200 rpm for 5 min. Before FCM detection, the cells were washed three times with PBS. Afterwards, apoptotic cells were stained with PI (BD, USA) and Annexin V-FITC (BD, USA) in a dark room for 20 min according to the manufacturer’s instructions. Finally, CTCs were subjected to flow cytometry (FACS Calibur, BD, USA).
Animal Studies
For tumor growth assay, 4 × 106 logarithmically growing CTCs of different groups (N= 3 per group) in 100 μL PBS was injected into the flanks subcutaneously of female nude mice which were 4-week-old. The IVIS Imaging system (Caliper life Sciences, USA) was used to observe the tumor growth. Care of experimental animals was in accordance with Nanjing Medical University Institutional Animal Care and Use Committee. And this study was approved by the Clinical Ethics Committee of Nanjing Medical University.
Immunohistochemistry Staining
The xenograft tumor specimens were fixed with 4% paraformaldehyde solution. Specimens were sectioned onto 4-μm slices after embedding with paraffin. Then, the slides were incubated with the primary antibody, including CD117 (ab32363, Abcam, USA), CD44 (37259, Cell Signaling Technology, USA), vimentin (5741, Cell Signaling Technology, USA), Ki67 (ab156956, Abcam, USA) at 4°C overnight and then washed three times with PBS. After incubation with the HRP polymer-conjugated secondary antibody for 1 h at room temperature, it was stained with a diaminobenzidine solution (DAB) for 3min and then stained with hematoxylin as a counterstain. Three random fields were imaged for each sample with a microscope.
TUNEL Assay
Cell Death Detection Kits (Roche, USA) were used according to the manufacturer’s protocol. The sections were rehydrated in gradient concentration of ethanol (100% for 5 min, 100% for 3 min, 95% for 3 min, 85% for 3 min, 70% for 3 min and 50% for 3 min) following by fixing in 4% paraformaldehyde. Cells were counted under the microscope and photographed in five random fields of each group.
Statistical Analysis
All statistical analyses were conducted using the SPSS 22.0 software and P<0.05 was considered statistically significant. The data was expressed as mean ± standard deviation and are representative of at least three independent experiments. The relationship between the HMGA1 expression and PFS of GIST patients was evaluated by drawing the receiver operating characteristic (AUROC) curve. The association of two different groups with clinicopathological variables was evaluated using the Chi-square test. The progression-free survival (PFS) rates of the patients were calculated using the Kaplan– Meier method. Univariate and multivariate logistic regression analyses were performed to identify risk factors for survival after complete surgery. For the multivariable analysis, Cox’s regression hazard model was used. We included only those variables exhibiting P values <0.05 on univariate analysis in multivariable analysis.
Results
CTCs Detection, Validation and Cultivation
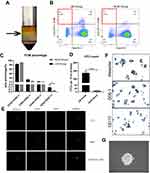
Density gradient centrifugation was carried on to isolate mononuclear cells (Figure 1A). After separation of possible cell debris, further analysis of blood cells was performed by FCM, with cells in the DOG-1+/CD45- region considered to be CTCs (Figure 1B). FCM parameters of GIST patients with heterochronous liver metastasis (LM group) including the percentage and enumeration (CTCs per 10 mL of blood) were both significantly higher than patients without progression (NLM group) (Figure 1C and D).
In order to verify that cells in DOG-1+/CD45- region separated by FCM were indeed CTCs of GIST patients, cells in different regions were identified by laser confocal microscopy (Figure 1E). Compared to DOG-1-/CD45+ white blood cells (WBCs), cells in DOG-1/CD45- region exhibited the classical characteristics of CTCs. DOG-1/+CD45+ cells can be recognized as the combination of CTCs and WBCs or some other cell type that cannot be verified. Immunohistochemistry of DOG-1, CD117 and vimentin antibody of CTCs was also performed, demonstrating its origin of GIST (Figure 1F). Next-generation sequencing showed that DOG1+/CD45- cells had same mutations (KIT exon 11) as the primary tumors, strongly confirming the reliability and authenticity of our isolating process and DOG-1+/CD45- cells were indeed CTCs of GIST patients (Supplementary Table 2).
After separating CTCs from blood of GIST patients successfully, we tried to cultivate them in vitro. Finally, after testing several culture conditions,24–26 we found that CTCs proliferated best as tumor spheres when cultured in media without serum and supplemented with EGF and basic FGF under hypoxic conditions (4% O2)10 (Figure 1G). Nonadherent culture conditions were critical to maintain the undifferentiated phenotype of CTCs. We established CTC lines (sustained in vitro for >5 months) isolated from six patients, three of which were with heterochronous liver metastasis (CTC-1, CTC-2, CTC-3) while three without progression after surgery (CTC-4, CTC-5, CTC-6) (Supplementary Table 2).
CTC Lines are Endowed with Strong Metastatic Potential in vitro
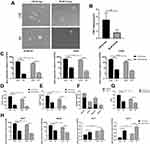
In the process of culturing CTC lines, we found that in LM group, the sphere-forming capability of CTCs was much stronger than those in NLM group and the primary cells extracted from GIST tumors shown lower sphere-forming efficiency compared with CTCs (Figure 2A). Based on that CTC lines had the ability to self-renew over 20 passages and form sphere, we speculated that CTCs had the characteristics of stem cells. Using extreme limiting dilution analysis27 on spheres at least over 3 passages, we quantified CSC frequency, finding that CTCs of metastatic patients had more self-renewing cells than those of non-metastatic patients (Figure 2B).
To validate the strong CSC phenotype of our CTC lines, we focused on aldehyde dehydrogenase (ALDH1A1) expression and enzymatic activity, which has been suggested as a specific marker for the CSC/progenitor population.28 Our results showed that ALDH1A1 mRNA expression and ALDH activity were higher in CTC lines compared with primary tumors. Moreover, CTCs of metastatic patients expressed increased ALDH1A1 and had higher ALDH activity than those of non-metastatic patients (Figure 2C and D). We then quantified the expression of two markers specifically associated with CSCs with metastatic potential: CD26 and CD44v6.29,30 mRNA and protein levels of CD26 were both highly expressed in CTC lines (Figure 2C and E) and particularly higher in metastatic CTC lines. CD44 mRNA was enriched in CTCs, especially increased in metastatic CTC lines (Figure 2C). What is more, CTC lines had strong CD44v6 expression, which was barely detected in primary tumor tissues (Figure 2F). In addition, metastatic CTC lines had much more CD44v6 expression than non-metastatic CTC lines.
It is known to us that drug resistance contributes to recurrence in many tumors, so we quantified the sensitivity of our GIST CTC lines to IM. Overall, we found that CTC lines were significantly more resistant to IM than primary tumor-derived cells grown under the same conditions, and LM group CTC lines had larger resistant capability than NLM group CTC lines as well (Figure 2G). We then confirmed by qRT-PCR that CTC lines displayed higher expression of genes associated with IM resistance, including MRP1, ABCG2, OCT-1 and OCT-2, in comparison with primary tumor tissues. At the same time, the expression of those transporters associated with drug resistance was higher in LM group CTC lines than NLM group CTC lines (Figure 2H). These results strongly suggest that CTCs, especially from metastatic patients, had stronger IM tolerance leading to heterochronous liver metastasis.
HMGA1 Upregulates the Stem Cell-Like Properties and Resistance of CTCs
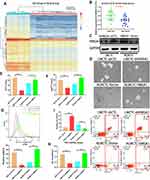
For further clarification the cause of higher stem cell-like properties in CTCs from LM group, we performed next-generation sequencing of CTCs in two groups. As shown in Figure 3A, HMGA1, PINK1, MRVI1, DAPK2 and MBOAT2 had the top five most significant differences. We confirmed the differential HGMA1 expression between two groups by qRT-PCR (Figure 3B). Based on the HMGA1 expression levels in CTCs, LMCTC-shHMGA1, LMCTC-shCTL, NLMCTC-HMGA1, NLMCTC-HMGA1-Vector were constructed using lentivirus. The efficiency of transfection was evaluated by Western blots (Figure 3C). It was observed that the sphere-forming capability of CTCs was positively related to the expression of HMGA1 (Figure 3D). We then qualified the expression of three CSC markers (ALDH, CD26, CD44) using flow cytometry and verified that HMGA1 could induce the stem cell-like properties of CTCs (Figure 3E–G). CCK-8 assays were performed to find that NLMCTC-HMGA1 and LMCTC-shCTL were both more resistant to IM compared with LMCTC-shHMGA1 and NLMCTC-Vector (Figure 3H). Moreover, HMGA1 overexpression in CTCs significantly decreased the rate of apoptosis (Figure 3I).
HMGA1 Upregulated the Stem Cell-Like Properties and Resistance of CTCs via Wnt/β-Catenin Pathway
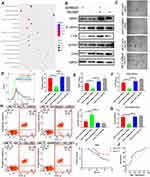
KEGG pathway analysis revealed that Wnt/β-catenin signal pathway, a well-studied pathway associated with tumor stem cell-like properties, to be a potential target of HMGA1 (Figure 4A). Meanwhile, it has been reported that HMGA1 positively regulates Wnt/β-catenin signaling through interacting with β-catenin, which leads to increase the β-catenin–TCF4 complex formation.31 In this study, Western Blot was performed to address this hypothesis and the downregulated expression of β-catenin, c-myc, cyclinD1, CD44 was detected in LMCTC-shHMGA1 compared with LMCTC-shCTL (Figure 4B). As shown in Figure 4B, the inactivation of Wnt /β-catenin signal pathway in LMCTC-shHMGA1 was rescued by SLK2001 (an agonist of the Wnt/β-catenin pathway). Similarly, the downregulated SFE, expression of stem cell-like properties markers, proliferation and evaluated cell apoptosis rate induced by HMGA1 knockdown were all counteracted by SLK2001 (Figure 4C–H). Therefore, we supposed that HGMA1 was of great significance for CTCs to have the potential of recurrence and metastasis. To further investigate the correlation between HMGA1 expression of CTCs and prognosis of patients, we divided those CTC lines into two groups according to the median of HMGA1 expression level—low HMGA1 and high HMGA1. Figure 4I indicates that patients with low HMGA1 had longer PFS than those with high HMGA1. ROC curve was drawn to assess HMGA1 expression performance in the prognosis of patients. The area under the curve (AUC) was 0.704 (95% CI, 0.574–0.834), associated with a sensitivity of 76.6% and a specificity of 61.54% (Figure 4J). Of factors possibly affecting survival, HMGA1 expression was significant both in univariate and multivariate analysis (Table 1). These results suggest that HMGA1 might be an important factor inducing the stemness of CTCs.
 |
Table 1 Clinicopathological Characteristics of 73 Patients with GIST |
CTC Lines are Endowed with Strong Metastatic Potential in vivo
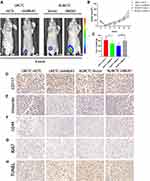
To better investigate the tolerance of CTCs in vivo, cells were injected into the flanks of nude mice to generate tumors ectopically. As shown in Figure 5A and B, the tumor progression was faster in LMCTC-shCTL and NLMCTC-HMGA1 group compared with LMCTC-shHMGA1 and NLMCTC-HMGA1-Vector group. It is accordant in Figure 5C that the tumor weight in LMCTC-shHMGA1 and NLMCTC-HMGA1-Vector group CTCs was lighter.
Furthermore, immunohistochemical analyses of CD117 and vimentin demonstrating the mesenchyme origin of those cells, which were both significantly upregulated in LMCTC-shCTL and NLMCTC-HMGA1 group compared with that in LMCTC-shHMGA1 and NLMCTC-HMGA1-Vector group (Figure 5D and E). The distribution of CD44 revealed reduced stem cell-like properties in those LMCTC-shHMGA1 and NLMCTC-HMGA1-Vector groupscompared with that of LMCTC-shCTL and NLMCTC-HMGA1 groups (Figure 5F). Subsequently, Ki-67 staining and TUNEL assays were used to further verify that LMCTC-shCTL and NLMCTC-HMGA1 group had improved progression and suppressed apoptosis (Figure 5G and H). These in vivo data strongly indicated LMCTC-shCTL and NLMCTC-HMGA1 group CTCs had larger IM tolerance and stem cell-like properties capability, which may be of crucial causes of heterochronous liver metastasis.
Discussion
It has been demonstrated that CTCs are the main causes of cancer metastasis and recurrence and considered as promising prognostic factors for tumors.32–34 However, CTCs isolation and culture are key problems bothering us so far and limiting further experiments of CTCs mechanism. The different isolation methods were invented on the basis of specific CTC characteristics such as physical properties, biological characteristics and the expression of tumor-specific surface proteins. Isolation by size of epithelial tumor cells (ISET) using size to select for CTCs provides efficient and high throughput of CTCs, but at the same time having limitation of applicability in clinical settings due to heterogeneity of CTCs in term of their size.35–37 Currently, CellSearch, the exclusively US-FDA (Food and Drug Administration) cleared device for CTC detection is a prognostic indicator for breast, prostate and colorectal cancer, which also has disadvantages of high-cost, invasion, and complicated process.38–40 So An efficient, safe, and accurate method is urgent to be found. It is universally known that DOG-1 is a specific expressed biomarker in GIST, and CD45 is a common leukocyte antigen used during the cell sorting assay to remove leukocytes via CD45 depletion.41 The innovative application of FCM with CD45 and DOG-1 by us could isolate hundreds of CTCs in GIST patients with heterochronous liver metastasis while Heming Li reported they used microbeads with CSV (cell surface vimentin) antibody to isolate dozens of CTCs in GIST patients with metastasis, which indicating that this credible, feasible and noninvasive approach could discover much more CTCs to help create good conditions for cultivating CTCs in vitro.42
Stem cell-like properties have been reported to be associated with increased metastatic and recurrent potential in various tumors.43–45 Cells enriched with stem cell-like properties share these acquired capabilities, including self-sufficiency in growth signals, evasion of antigrowth signals, resistance to cell death, limitless replicative potential, sustained angiogenesis, tissue invasion and metastasis.46 In addition, CTC is closely related to stem cell-like properties, because CTC is a special tumor cell subpopulation that separates from primary tumor cells, thus is considered to have stem cell-like characteristics. They are much more tolerant to chemotherapy and targeted drugs, defected in apoptosis mechanism, having infinite proliferation and self-renewal capability, making it to be the originator of tumor metastasis. A series of functional experiments between LMCTC-shCTL and NLMCTC-HMGA1 group and LMCTC-shHMGA1 and NLMCTC-HMGA1-Vector group including CD44-positive levels and sphere formation assay was carried on, illustrating that CTCs were enriched with stem cell-like properties which could contribute to tumor relapse and poor prognosis.
Many studies have uncovered that high levels of HMGA1 expression at the mRNA or protein level were related to cancer stem cells (CSC) in human cancer including, lung, breast, prostate, colon and liver.47 Many pathways have been reported to be regulated by HMGA1 including Wnt pathway, NF-κB pathway, IFN-β pathway and JAK-STAT signaling pathway, of which Wnt pathway is the most directly related to stem cell-like properties. Although HMGA1 has been identified as a tumor promotor and found to be upregulated in certain malignancies,11–14 the expression pattern and functions of HMGA1 in tumors remain to be fully elucidated. Therefore, further investigation of the role of HMGA1 in the tumor is warranted. In this study, we found that HMGA1 expression was dramatically higher in LM CTCs of GIST patients compared with NLM CTCs of GIST patients via NGS. We explored the role of HMGA1 in stem cell-like properties and IM resistance both in vitro and in vivo. Our results showed that HMGA1 overexpression promoted stem cell-like properties and IM resistance and suppressed apoptosis both in vitro and in vivo, while the converse effects were observed following HMGA1 knockdown. These results indicated the importance of HMGA1 upregulation in the progression and recurrence of GIST.
To clarify the mechanisms underlying the effects of HMGA1 on stem cell-like properties and apoptosis, we predicted putative targets of HMGA1 in GIST by KEGG analysis. Among the candidate target pathway, we concentrated on Wnt/β-catenin pathway, which is correlated with stem cell-like properties. It has been recognized as a key regulator of a variety of human tumors, infectious diseases, neurological diseases, and aging. Accumulating evidence has demonstrated the involvement of Wnt/β-catenin pathway in the regulation of various biological processes, such as EMT, apoptosis, drug resistance, as well as stem cell-like properties. Studies have also revealed that Wnt/β-catenin pathway plays vital roles in the initiation, development, progression, and prognosis of numerous malignancies, including prostate cancer, breast carcinoma, hepatocellular carcinoma and GC 0.25–28 Nowadays, there are more than two hundred inhibitors of Wnt signaling pathway in the initial stage of biological activity testing, providing materials for the research and development of antitumor drugs. In the present study, stepwise investigations revealed high expression of HMGA1 was associated with activation of Wnt/β-catenin pathway, eventually enhanced the stem cell-like properties and IM tolerance. In addition, agonist of Wnt/β-catenin pathway rescued the downregulation of stem cell-like properties and upregulation of apoptosis caused by HMGA1 knockdown. Taken together, the results of our study suggest that the stem cell-like properties of CTC in GIST patients mediated by overexpression of HMGA1/ Wnt/β-catenin pathway may be one of the causes of IM resistance. HMGA1 may be a promising candidate gene for imatinib-resistance so it is necessary to perform more studies to discover novel targets in imatinib-resistant GIST. And whether there is an intersection between IM/kit pathway and HMGA1/Wnt pathway and whether there are other mechanisms of IM resistance in HMGA1 overexpression are still needed us to explore in the future.
In conclusion, we constructed a set of systematic methods for CTC extraction and long-term culture for functional experiments. Our study also demonstrates that HMGA1 is associated with stem cell-like properties of CTCs and that HMGA1 enhances GIST progression and recurrence in a Wnt/β-catenin -dependent manner. It may help provide a practical method to better study CTCs and suggest the potential of HMGA1 as a novel therapeutic target for GIST.
Date Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Ethics Approval and Consent to Participate
All the patients have signed the informed consents before the enrollment. This study was approved by the Clinical Ethics Committee of Nanjing Medical University (Ethical number: 2013-SR-142).
Funding
Our work was sponsored by funds from Natural Science Foundation of Province (BK20191495, to H. Xu), Natural Science Foundation of Province (BK20141493, to H. Xu), Jiangsu Key Medical Discipline (General Surgery; ZDXKA2016005), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD, JX10231801), Jiangsu Key Lab of Cancer for Cancer Personalized Medicine, Nanjing Medical University (all to Z. Xu), and Construction Program of Jiangsu Provincial Clinical Research Center Support System (BL2014084).
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Ravegnini G, Sammarini G, Nannini M, et al. Gastrointestinal stromal tumors (GIST): facing cell death between autophagy and apoptosis. Autophagy. 2017;13(3):452–463. doi:10.1080/15548627.2016.1256522
2. Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11(12):865–878. doi:10.1038/nrc3143
3. Antonescu CR, Besmer P, Guo T, et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res. 2005;11(11):4182–4190. doi:10.1158/1078-0432.CCR-04-2245
4. DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231(1):51–58. doi:10.1097/00000658-200001000-00008
5. Grillet F, Bayet E, Villeronce O, et al. Circulating tumour cells from patients with colorectal cancer have cancer stem cell hallmarks in ex vivo culture. Gut. 2017;66(10):1802–1810. doi:10.1136/gutjnl-2016-311447
6. Palmirotta R, Lovero D, Silvestris E, et al. Next-generation sequencing (NGS) analysis on single circulating tumor cells (CTCs) with no need of whole-genome amplification (WGA). Cancer Genomics Proteomics. 2017;14(3):173–179. doi:10.21873/cgp.20029
7. Manier S, Park J, Capelletti M, et al. Whole-exome sequencing of cell-free DNA and circulating tumor cells in multiple myeloma. Nat Commun. 2018;9(1):1691. doi:10.1038/s41467-018-04001-5
8. Fabisiewicz A, Grzybowska E. CTC clusters in cancer progression and metastasis. Med Oncol. 2017;34(1):12. doi:10.1007/s12032-016-0875-0
9. Tang Y, Lu Y, Chen Y, et al. Pre-metastatic niche triggers SDF-1/CXCR4 axis and promotes organ colonisation by hepatocellular circulating tumour cells via downregulation of Prrx1. J Exp Clin Cancer Res. 2019;38(1):473. doi:10.1186/s13046-019-1475-6
10. Aceto N, Bardia A, Miyamoto DT, et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell. 2014;158(5):1110–1122. doi:10.1016/j.cell.2014.07.013
11. Cristofanilli M, Budd GT, Ellis MJ, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351(8):781–791. doi:10.1056/NEJMoa040766
12. Cristofanilli M. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. Semin Oncol. 2006;33:S9–S14. doi:10.1053/j.seminoncol.2006.03.016
13. Bauernhofer T, Zenahlik S, Hofmann G, et al. Association of disease progression and poor overall survival with detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer. Oncol Rep. 2005;13(2):179–184.
14. Armstrong AJ, Marengo MS, Oltean S, et al. Circulating tumor cells from patients with advanced prostate and breast cancer display both epithelial and mesenchymal markers. Mol Cancer Res. 2011;9(8):997–1007. doi:10.1158/1541-7786.MCR-10-0490
15. Shen C, Yin Y, Chen H, et al. Secreted protein acidic and rich in cysteine-like 1 suppresses metastasis in gastric stromal tumors. BMC Gastroenterol. 2018;18(1):105. doi:10.1186/s12876-018-0833-8
16. Wang HC, Li TY, Chao YJ, et al. KIT exon 11 codons 557–558 deletion mutation promotes liver metastasis through the CXCL12/CXCR4 axis in gastrointestinal stromal tumors. Clin Cancer Res. 2016;22(14):3477–3487. doi:10.1158/1078-0432.CCR-15-2748
17. Lund T, Holtlund J, Fredriksen M, Laland SG. On the presence of two new high mobility group-like proteins in HeLa S3 cells. FEBS Lett. 1983;152(2):163–167. doi:10.1016/0014-5793(83)80370-6
18. Hock R, Furusawa T, Ueda T, Bustin M. HMG chromosomal proteins in development and disease. Trends Cell Biol. 2007;17(2):72–79. doi:10.1016/j.tcb.2006.12.001
19. Farnet CM, Bushman FD. HIV-1 cDNA integration: requirement of HMG I(Y) protein for function of preintegration complexes in vitro. Cell. 1997;88(4):483–492. doi:10.1016/S0092-8674(00)81888-7
20. Munoz J, Stange DE, Schepers AG, et al. The Lgr5 intestinal stem cell signature: robust expression of proposed quiescent ‘+4’ cell markers. EMBO J. 2012;31(14):3079–3091. doi:10.1038/emboj.2012.166
21. Han X, Cao Y, Wang K, Zhu G. HMGA1 facilitates tumor progression through regulating Wnt/beta-catenin pathway in endometrial cancer. Biomed Pharmacother. 2016;82:312–318. doi:10.1016/j.biopha.2016.05.004
22. Li J, Ye Y, Wang J, et al. Chinese society of clinical oncology CSCO expert committee on gastrointestinal stromal, Chinese consensus guidelines for diagnosis and management of gastrointestinal stromal tumor. Chin J Cancer Res. 2017;29(4):281–293. doi:10.21147/j.issn.1000-9604.2017.04.01
23. Karabacak NM, Spuhler PS, Fachin F, et al. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. Nat Protoc. 2014;9(3):694–710. doi:10.1038/nprot.2014.044
24. Liu X, Ory V, Chapman S, et al. ROCK inhibitor and feeder cells induce the conditional reprogramming of epithelial cells. Am J Pathol. 2012;180(2):599–607. doi:10.1016/j.ajpath.2011.10.036
25. Sato T, Clevers H. Primary mouse small intestinal epithelial cell cultures. Methods Mol Biol. 2013;945:319–328.
26. Ince TA, Richardson AL, Bell GW, et al. Transformation of different human breast epithelial cell types leads to distinct tumor phenotypes. Cancer Cell. 2007;12(2):160–170. doi:10.1016/j.ccr.2007.06.013
27. Hu Y, Smyth GK. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J Immunol Methods. 2009;347(1–2):70–78. doi:10.1016/j.jim.2009.06.008
28. Huang EH, Hynes MJ, Zhang T, et al. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res. 2009;69(8):3382–3389. doi:10.1158/0008-5472.CAN-08-4418
29. Pang R, Law WL, Chu AC, et al. A subpopulation of CD26+ cancer stem cells with metastatic capacity in human colorectal cancer. Cell Stem Cell. 2010;6(6):603–615. doi:10.1016/j.stem.2010.04.001
30. Todaro M, Gaggianesi M, Catalano V, et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell. 2014;14(3):342–356. doi:10.1016/j.stem.2014.01.009
31. Xing J, Cao G, Fu C. HMGA1 interacts with beta-catenin to positively regulate Wnt/beta-catenin signaling in colorectal cancer cells. Pathol Oncol Res. 2014;20(4):847–851. doi:10.1007/s12253-014-9763-0
32. Paoletti C, Cani AK, Larios JM, et al. Comprehensive mutation and copy number profiling in archived circulating breast cancer tumor cells documents heterogeneous resistance mechanisms. Cancer Res. 2018;78(4):1110–1122. doi:10.1158/0008-5472.CAN-17-2686
33. Xu H, Chen L, Shao Y, et al. Clinical application of circulating tumor DNA in the genetic analysis of patients with advanced GIST. Mol Cancer Ther. 2018;17(1):290–296. doi:10.1158/1535-7163.MCT-17-0436
34. Miyamoto DT, Zheng Y, Wittner BS, et al. RNA-Seq of single prostate CTCs implicates noncanonical Wnt signaling in antiandrogen resistance. Science. 2015;349(6254):1351–1356. doi:10.1126/science.aab0917
35. Dolfus C, Piton N, Toure E, Sabourin JC. Circulating tumor cell isolation: the assets of filtration methods with polycarbonate track-etched filters. Chin J Cancer Res. 2015;27(5):479–487. doi:10.3978/j.issn.1000-9604.2015.09.01
36. Vona G, Estepa L, Beroud C, et al. Impact of cytomorphological detection of circulating tumor cells in patients with liver cancer. Hepatology. 2004;39(3):792–797. doi:10.1002/hep.20091
37. Wang L, Asghar W, Demirci U, Wan Y. Nanostructured substrates for isolation of circulating tumor cells. Nano Today. 2013;8(4):347–387. doi:10.1016/j.nantod.2013.07.001
38. Krebs MG, Sloane R, Priest L, et al. Evaluation and prognostic significance of circulating tumor cells in patients with non-small-cell lung cancer. J Clin Oncol. 2011;29(12):1556–1563. doi:10.1200/JCO.2010.28.7045
39. Sieuwerts AM, Kraan J, Bolt J, et al. Anti-epithelial cell adhesion molecule antibodies and the detection of circulating normal-like breast tumor cells. J Natl Cancer Inst. 2009;101(1):61–66. doi:10.1093/jnci/djn419
40. Kim MS, Sim TS, Kim YJ, et al. SSA-MOA: a novel CTC isolation platform using selective size amplification (SSA) and a multi-obstacle architecture (MOA) filter. Lab Chip. 2012;12(16):2874–2880. doi:10.1039/c2lc40065k
41. Wu S, Liu Z, Liu S, Lin L, Yang W, Xu J. Enrichment and enumeration of circulating tumor cells by efficient depletion of leukocyte fractions. Clin Chem Lab Med. 2014;52(2):243–251. doi:10.1515/cclm-2013-0558
42. Li H, Meng QH, Noh H, et al. Cell-surface vimentin-positive macrophage-like circulating tumor cells as a novel biomarker of metastatic gastrointestinal stromal tumors. Oncoimmunology. 2018;7(5):e1420450. doi:10.1080/2162402X.2017.1420450
43. Micalizzi DS, Maheswaran S, Haber DA. A conduit to metastasis: circulating tumor cell biology. Genes Dev. 2017;31(18):1827–1840. doi:10.1101/gad.305805.117
44. Ling S, Shan Q, Zhan Q, et al. USP22 promotes hypoxia-induced hepatocellular carcinoma stemness by a HIF1alpha/USP22 positive feedback loop upon TP53 inactivation. Gut. 2019;
45. Elcheva IA, Wood T, Chiarolanzio K, et al. RNA-binding protein IGF2BP1 maintains leukemia stem cell properties by regulating HOXB4, MYB, and ALDH1A1. Leukemia. 2019:1–10.
46. Su M, Xiao Y, Ma J, et al. Circular RNAs in Cancer: emerging functions in hallmarks, stemness, resistance and roles as potential biomarkers. Mol Cancer. 2019;18(1):90. doi:10.1186/s12943-019-1002-6
47. Takeuchi I, Takaha N, Nakamura T, et al. High mobility group protein AT-hook 1 (HMGA1) is associated with the development of androgen independence in prostate cancer cells. Prostate. 2012;72(10):1124–1132. doi:10.1002/pros.22460
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.